
Figure 107-1. Examples of skin lesions. A: Giant congenital nevus. B: Lentigo maligna (Hutchinson freckle). C: Melanoma arising in a lentigo maligna (lentigo maligna melanoma). D: Superficial spreading melanoma. E: Nodular melanoma. F: Acral lentiginous melanoma (ulcerated nodular plantar melanoma with satellite lesions). G: Subungual melanoma. H: Pigmented basal cell carcinoma.
SSM accounts for approximately 70% of cutaneous melanomas. This pattern is the most common in melanomas arising in a pre-existing nevus. SSM is typically characterized by variation in color, irregular borders, and irregular surface and often exhibits the classic melanoma clinical features.
NM occurs in approximately 15% to 30% of patients with cutaneous melanoma and is often the most aggressive of the four types of melanoma because of later detection and rapid growth. In general, NM may be more uniform in coloration than the other types, have regular borders with size less than 6 mm in early lesions, and lack the classic melanoma features initially. A change in a lesion is important for early detection.
ALM is a distinct clinicopathologic variant of melanoma that most commonly occurs on the hands, feet, fingers, and toes as well as in subungual locations (Fig. 107-1). ALM represents only 2% to 8% of melanoma in whites but 35% to 60% in people of color. Later detection frequently occurs because of location that is not easily or routinely examined and a low index of suspicion. When corrected for Breslow depth, the overall prognosis for ALM appears similar to the other subtype categories. In subungual locations, ALM can appear as an irregular, tan-brown streak in the nail that originates from the base of the nail bed. More than three fourths of subungual melanomas involve the great toe or thumb, and they can be confused with subungual hematoma.
Other rare melanoma subtypes deserve brief mention. Mucosal, anal, and vulvovaginal melanomas are associated with a poorer prognosis, possibly related to advanced disease at presentation with late detection and high vascularity at the lesion site. Desmoplastic melanoma is associated with a higher rate of neurotropism and lower rate of lymph node metastases.13–15 Small cell or nevoid melanoma often lacks any of the classic features, is difficult to accurately diagnose without expert dermatopathology interpretation, and thus may be initially more frequently misdiagnosed. And, amelanotic melanoma, occurring in 3% to 4% of cases, is often associated with late detection due to lack of pigment and failure to diagnose early.
Staging and Prognostic Factors
4 A great deal of information is available regarding various factors that correlate with the clinical outcome of patients with melanoma. Some of these prognostic factors, such as microstaging and nodal status, are of sufficient independent significance to be incorporated into staging systems with known survival rates. Other prognostic factors such as tumor ulceration and microscopic versus macroscopic nodal disease have also been found to be significant variables that influence survival and have been incorporated into the staging system.
Microstaging
One of the most important prognostic features of cutaneous melanoma is the stage of development of the primary tumor. The microstaging method that is used routinely was originally described by Breslow.16 This method classifies the primary tumor according to its thickness in millimeters, as measured with an ocular micrometer, from the top of the granular layer to the base of the tumor. Many investigators have documented an inverse correlation between tumor thickness and survival. The largest series of 17,600 patients reported by Balch et al.17 is summarized in Table 107-1. The ulceration status and mitotic rate are also independent factors that are used to determine microstaging of the primary lesion.17,18 Prior to the use of the Breslow microstaging method, melanomas were staged according to the level of invasion into the histologic layer of skin. This was known as the Clark level of invasion and comprised five levels (I, in situ lesions; V, subcutaneous involvement). Several studies have confirmed that Breslow thickness conveys more accurate prognostic information than does the determination of Clark level.17,19
The presence of regional lymph node metastases is associated with a worsening prognosis. The tumor burden (microscopic vs. macroscopic disease) of involved lymph nodes has an inverse correlation with long-term survival.17,19 The 5-year survival rate for patients with involved lymph nodes ranges from 70% to 25%, based primarily on tumor burden and ulceration status of the primary lesion (Table 107-1). The use of sentinel lymph node biopsy (SLNB) has identified a subgroup of patients with micrometastatic nodal disease who have a favorable prognosis compared with patients with macroscopic nodal involvement. This information has been incorporated in the staging system as well.
RESULTS
Table 107-1 Survival Rates for Melanoma TNM and Staging Categories

Clinical and Pathologic Staging
The American Joint Committee on Cancer (AJCC) developed a five-stage system that divides melanomas according to tumor thickness (T), nodal status (N), and metastatic disease (M).17,19 The current system was updated in 2009 and is summarized in Tables 107-2 and 107-3. There are five stages based on prognosis: stage 0 (in situ melanoma), stage I (local disease), stage II (local disease), stage III (regional nodal, in-transit, or satellite metastases), and stage IV (distant metastases).
Other Prognostic Factors
The major prognostic factors that predict survival in melanoma patients have been accounted for in the AJCC staging system – namely, tumor microstaging, ulceration, nodal status, and distant metastases.17,19 The presence of ulceration in a melanoma appears to be associated with a poorer prognosis. Men have a higher proportion of ulcerated lesions than women (27% vs. 19%, respectively). Although ulceration appears to correlate with thickness of the melanoma, the presence of ulceration is an independent prognostic factor and has been included in the staging system.17,19 In addition, a higher mitotic rate of 1.0 mm2 or greater is an independent prognostic factor of both nodal involvement and survival.18,20–22 The presence of angiolymphatic invasion is also a poor prognostic sign.
Treatment of Primary Melanoma
Biopsy
For localized melanoma, the tumor thickness (Breslow depth of invasion) is the single variable that most accurately determines therapy and prognosis. The ulceration status and mitotic rate also represent independent prognostic variables. A full-thickness biopsy with 1- to 2-mm margins only, to the adipose tissue, is preferred for any lesion highly suspect for melanoma.23 If the melanoma is transected with a partial-thickness shave biopsy, the ability to obtain an accurate measurement of tumor thickness is lost. Therefore, a superficial shave biopsy is never recommended for a suspect pigmented lesion. Saucerization, which uses a curved blade to perform a deeper shave biopsy down to the subcutaneous fat, is acceptable.
Excisional biopsy with 1- to 2-mm margins is the preferred method for suspect lesions to provide the pathologist a total specimen for histologic interpretation and accurate microstaging. Performing a wide excision as the first step, especially on the trunk or head and neck, is not recommended, as several benign and malignant lesions can mimic melanoma and because doing so may result in the inability to accurately perform SLNB. Formalin-fixed, paraffin-embedded, permanent sections should be used for biopsy diagnosis of primary cutaneous melanoma to accurately determine tumor thickness and other histopathologic prognostic variables. Frozen sections have no role in the diagnosis or microstaging of primary melanoma. If the lesion is a melanoma, the excisional biopsy represents the first stage of a two-stage procedure. The second stage is reexcision with margins generally ranging from 0.5 to 2.0 cm, with or without SLNB, depending primarily on the tumor thickness. When biopsying a suspected melanoma on the extremities, it is critical that the biopsy be oriented on the long axis of the extremity, parallel to the lymphatics. Performing a biopsy oriented transversely on the extremity may not only compromise the accuracy of lymphatic mapping but may also obligate the patient to a skin graft to close the subsequent wide excision defect.
For suspicious lesions that are too large for complete excision and those that are located where the amount of skin is critical in terms of functional or cosmetic results, an incisional biopsy may be performed with an elliptical incision, deep saucerization shave to adipose or deep dermis in thick skin areas, or a punch biopsy. Incisional biopsies for melanoma do not increase the risk of local recurrence and distant metastasis or affect patient survival. They should generally be performed on the most raised or most pigmented area of the lesion to maximize the obtainable diagnostic and prognostic information. The most raised area usually corresponds to the maximal thickness of the lesion, but not always. Several incisional biopsies can be obtained from different areas for large lesions with multiple morphologic features. In the scenario of incisional biopsy with significant remaining lesion, complete excision for accurate microstaging should be considered prior to definitive treatment, unless the lesion is already 1 mm or greater in Breslow depth.
Metastatic Workup
In an attempt to standardize staging workup for melanoma, the National Comprehensive Cancer Network (NCCN) has published guidelines.24 There are three basic reasons to perform a metastatic workup following the diagnosis of primary cutaneous melanoma: (a) for staging and prognosis, (b) to detect an early metastasis with potential survival benefit, and (c) to avoid morbidity of an extensive surgical procedure by detection of a distant metastasis.24 The best test for the staging workup still starts with a history (a focused review concentrating on constitutional, respiratory, neurologic, hepatic, musculoskeletal, gastrointestinal, skin, and lymphatic systems) and physical examination (total body skin examination, palpation of lymph nodes). Routine imaging (chest x-ray, computed tomography [CT] scans, positron emission tomography [PET] scans) and blood studies (complete blood cell count, comprehensive metabolic profile, liver function tests, serum lactate dehydrogenase [LDH] levels) in asymptomatic, clinically node-negative patients are low in both sensitivity and specificity and are not necessary.25–28 False-positive staging tests are common and lead to more tests and patient distress. SLNB represents the best baseline staging test, with both relatively high sensitivity and specificity in patients at significant risk for metastasis. The ability to detect stage IV disease with routine studies is small if the SLNB is negative. Both ultrasound and PET scanning have been examined as methods of identifying nodal metastases prior to surgery. While they detect disease in a minority of patients, the sizes of most metastases to the regional nodes are below the threshold of ultrasound or PET to detect, so this is not a cost-effective strategy.29–31 If a thorough history and physical and detailed review of systems reveal no signs or symptoms suspicious for regional or distant disease, then no further staging is necessary. However, a high index of suspicion should be maintained and a thorough symptom-directed workup should be initiated for any worrisome finding on the history and physical.
STAGING
Table 107-2 American Joint Commission on Cancer Melanoma Staging System, TNM Definitions

Approximately 5% to 10% of newly diagnosed melanoma patients present with clinically involved lymph nodes. Any clinically suspicious node should be confirmed by fine needle aspiration (FNA) biopsy, as enlarged lymph nodes may be reactive (particularly after a biopsy). Ultrasound-guided FNA biopsy can be useful in cases where the suspicious node is difficult to biopsy by hand. Patients documented to have macroscopic regional disease should undergo complete staging prior to definitive surgery, as a significant percentage may have more advanced regional or distant disease that may alter surgical therapy.32,33 Staging should include a serum LDH level; MRI of the head and either CT scan of the chest, abdomen, and pelvis; PET scan; or combined CT/PET scan. For patients with involved groin nodes, CT scan is recommended to visualize the pelvic nodes, as this may help determine the extent of inguinal node dissection.
Surgical Excision of Primary Melanoma
5 The primary purpose of a melanoma excision is to prevent local recurrence due to persistent disease. For melanoma in situ, excision margins of 0.5 to 1.0 cm are indicated. For invasive melanoma, wide excision of the primary tumor, with margins generally ranging from 1 to 2 cm, is indicated for local control. The optimal margin width remains somewhat controversial. The historical approach of excising all primary melanomas with a 3- to 5-cm margin is extinct. At least five randomized controlled trials failed to demonstrate a difference in overall survival or local recurrence with narrow (1- to 2-cm) versus wide (3- to 5-cm) margins (Table 107-4). Of note, one trial demonstrated an increased risk of locoregional recurrence with 1-cm versus 3-cm margins for melanoma more than 2 mm thick.34 Based on these trials, current consensus guidelines recommend margins of 1 cm for melanomas 1 mm or less thick, 2 cm for melanomas greater than 2 mm thick, and 1 to 2 cm for melanomas 1 to 2 mm thick. For this last group, the size of the margins should be based on the anatomic location of the melanoma and the morbidity of excising 2 cm versus 1 cm, including the need for an advancement flap or skin graft (Table 107-5). For several locations, such as melanomas on the head and neck, the maximum margins attainable are dictated by anatomic limitations. Most subungual invasive melanomas require amputation of the digit. Perianal melanomas should be treated by wide excision, as these achieve similar long-term results compared with more radical surgery.35,36 Abdominoperineal resection (APR) may be necessary for locally advanced disease or melanoma of the anal mucosa. A complete staging workup should be performed before proceeding with APR, as these patients have a very high rate of regional and distant disease.37
RESULTS
Table 107-4 Randomized Controlled Trials: Narrow Versus Wide Excision Margins

TREATMENT
Table 107-5 Recommended Surgical Margins for Melanoma Excision

For the clinically ill-defined LMM, histologic confirmation of negative margins is important, and it should be emphasized that these margin recommendations may not apply to in situ LM and invasive LMM on the head and neck.38,39 LM and LMM are often associated with extensive subclinical involvement to as much as several centimeters beyond the clinical component. Careful and complete margin control, including excision of the lesional trailing edge of atypical melanocytic hyperplasia, often beyond the standard margins, may be necessary for complete surgical resection of LM/LMM on the head and neck.
Treatment of Regional Metastatic Melanoma
Lymphadenectomy Results and Indications
6 Surgical excision of metastases to regional lymph nodes is potentially curative therapy. The 5-year survival rate for patients who undergo lymphadenectomy for clinically positive involved nodes (AJCC stage III) ranges from 25% to 70%. In addition, for those patients not cured by lymphadenectomy, resection can avoid potential pain associated with tumor enlargement, skin breakdown, and tumor necrosis (Fig. 107-2). Only 5% to 10% of patients who first present with the diagnosis of melanoma have clinical evidence of nodal metastases, approximately 85% to 90% have localized disease, and the remaining 5% have distant metastases. In less than 3% of patients, a diagnosis of melanoma is made in the absence of a definable primary lesion.40 When patients present with isolated nodal disease from an unknown primary site (an occult primary melanoma), the results of lymphadenectomy are similar to or better than those for patients with known primary tumors.41 For patients with melanoma 1 mm thick or greater who present with clinically negative nodes, SLNB should be considered to determine whether therapeutic lymphadenectomy is indicated.42
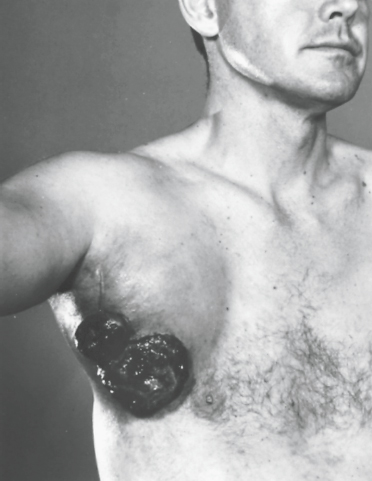
Figure 107-2. A patient with large melanoma axillary involvement with skin ulceration.
Sentinel Lymph Node Biopsy
Morton et al.43 were the first to develop the concept of “sentinel lymph nodes” in the nodal basins draining cutaneous melanomas. They hypothesized that melanoma involvement of a nodal basin develops in an orderly fashion with metastasis to the sentinel lymph node as the first step in that process. With the intradermal injection of a blue dye, these investigators were able to identify sentinel lymph nodes 90% of the time. If the sentinel node was negative for melanoma, the remaining lymph nodes were also free of involvement in at least 96% of cases. These results have been confirmed by multiple other groups.44,45
Two tracers are typically used for identifying the sentinel lymph nodes: a radiolabeled colloid solution, and a blue dye – either methylene blue or 1% isosulfan blue (Lymphazurin). Lymphazurin carries a risk of allergic reaction in 0.5% to 2.0% of patients, which can be life-threatening.45 Methylene blue dye has an extremely low risk of allergic reaction, although this should be diluted as it can cause skin necrosis if not completely resected. One to 4 hours before surgery, a radiolabeled colloid solution is injected intradermally around the primary tumor site or excision site, taking care to avoid injecting directly into any residual tumor. Lymphoscintigraphy imaging is then performed, which is critical in that it can identify sentinel nodes outside the traditional nodal basin. The blue dye is injected intradermally in the operating room a few minutes before the procedure. Using a handheld gamma detector probe, the surgeon is able to identify the sentinel lymph node location through the skin, thereby limiting the incision necessary to find the node. Blue-stained lymphatics and lymph nodes can be directly visualized (Fig. 107-3). The combined use of the blue dye plus the radiolabeled colloid enables the detection of the sentinel node in 97.3% to 98.6%.46 The technique of SLNB is most often performed in conjunction with the wide excision of the primary tumor. It can routinely be performed as an outpatient procedure.
Once the sentinel lymph node is removed, it is processed by the pathologist by step sectioning the entire node into multiple sections for routine hematoxylin and eosin staining. This is done to examine for micrometastases that could be missed by bivalving the node and performing an examination of only one section. If serial sectioning and staining are negative for metastasis, then immunohistochemical staining for melanoma markers such as S-100, Melan-A, and HMB-45 is performed.47 These stains can identify microscopic clusters of tumor cells that are hard or impossible to identify by hematoxylin and eosin staining. The thoroughness of assessing the sentinel lymph nodes for metastatic disease has resulted in much more accurate staging of patients. Micrometastases to lymph nodes have been incorporated in the current staging system as described earlier.
If the sentinel lymph node is positive for melanoma, then complete lymph node dissection (CLND) (Fig. 107-4) is indicated on the basis of potential survival and clinical benefit. The final results of the Multicenter Selective Lymphadenectomy Trial I (MSLT-I), which randomized patients to wide local excision (WLE) alone or WLE in combination with SLNB and CLND for a positive SLN, demonstrated that disease-free survival rates were significantly improved in the SLN group, compared with the observation group, for both patients with intermediate-thickness melanoma (71.3 ± 1.8% vs. 64.7 ± 2.3%; HR = 0.76, P = 0.01) and thick melanoma (50.7 ± 4.0% vs. 40.5 ± 4.7%; HR = 0.70, P = 0.03).48 Although MSLT-I showed no benefit on overall survival, subset analysis showed that node positive patients discovered on SLN biopsy had a 10-year melanoma-specific survival of 62.1 ± 4.8% compared with 41.5 ± 5.6% for patients who recurred during observation. Even after factoring for false-negative sentinel nodes, this remained significant (HR = 0.67; 95% CI, 0.46 to 0.97; P = 0.04). Although this subset analysis has been questioned as being valid,49,50 latent-subgroup analysis showed a clear, significant benefit of sentinel node biopsy.48
An unresolved question is whether every patient with a positive sentinel lymph node requires CLND. Several attempts to retrospectively identify clinical or histologic factors that may predict the absence or presence of disease within the nonsentinel nodes have not identified any absolute way to determine who requires additional surgery, although a combination of the Breslow depth and burden of disease within the sentinel lymph node seems promising.51–54 In addition, retrospective studies of SLN-positive patients who did not undergo CLND appear to have similar outcomes to those who did.55–57 The Multicenter Selective Lymphadenectomy Trial-II (MSLT-2), which randomized patients with a positive sentinel lymph node to CLND or serial ultrasonography of the regional basin, has completed accrual and will provide more information regarding the benefit of CLND for sentinel lymph node–positive patients. Until that time, CLND for a positive sentinel lymph node remains the standard of care in the treatment of melanoma.

Figure 107-4. Technique of groin dissection.
For patients with primary melanoma equal to or greater than 1-mm thickness with clinically negative nodes, SLN biopsy is recommended.42 For patients with thin melanoma, SLN should not be performed routinely but can be considered for patients with melanoma Brelsow depth of 0.76 to 0.99, particularly if they have other risk factors for regional micrometastases, including a mitotic rate of 1 mitosis/mm2 or greater, extensive dermal regression >1 mm, ulceration, angiolymphatic invasion, younger age, or a broadly transected or positive deep margin on shave biopsy.20,58,59 Reasons not to perform SLNB include significant medical comorbidities or the patient does not desire SLNB after an informed discussion.
Radiation therapy has also been utilized as an adjuvant to the resection of high-risk nodal metastases (extranodal extension, nodes greater than 3 to 4 cm in size, or multiple nodes). Several retrospective studies had suggested improved regional control rates when radiation was combined with surgery, particularly for cervical metastases. A large, prospective randomized trial from the Tasman Radiation Oncology Group randomized patients at high-risk of nodal relapse to adjuvant radiation versus observation. With a median follow-up of 40 months, the risk of relapse in the lymph-node field was reduced with radiation (HR = 0.56, 95% CI: 0.32 to 0.98; P = 0.041) but there was no significant difference in either relapse-free or overall survival.60 Adjuvant radiation after lymph node dissection will increase the overall morbidity, but this depends on the basin being irradiated, with ilioinguinal Radiation therapy (RT) being more problematic than cervical or axillary RT.61,62 The decision to consider adjuvant radiation after completion node dissection should be individualized on the basis of the predicted risk of regional recurrence, expected survival, and associated toxicities.
Adjuvant Therapy
Although the prognosis for patients with early-stage cutaneous melanoma is quite good, fewer than 50% of patients with thick primaries (i.e., >4 mm) or regional node involvement are cured by surgery alone. The development of effective adjuvant therapy capable of increasing postsurgical survival in high-risk groups of patients has been a long-standing goal of clinicians. Unfortunately, numerous randomized trials evaluating systemic chemotherapeutic agents (most notably dacarbazine), nonspecific immunotherapy (such as Bacille Calmette–Guérin), and tumor-specific vaccines in the adjuvant setting have failed to demonstrate any impact on survival.63–67
Interferon-α2b (IFN-α2b) has a variety of modulatory effects on the immune system that could enhance antitumor reactivity. In 1996, Kirkwood et al.68 reported the results of the Eastern Cooperative Oncology Group E1684 trial, which studied the effectiveness of high-dose IFN-α2b to improve disease-free and overall survival rates of patients with stage IIb (>4 mm) or III disease after surgery. High-dose IFN-a2b includes a 1-month induction phase (IV 20 MU/m2 M-F) followed by 11 months of maintenance (SC 10 MU/m2 3 days/week). In this multi-institutional prospective randomized trial, 287 high-risk patients with deep primary (>4-mm) tumors or positive nodes were randomly assigned after surgery to either postoperative adjuvant treatment with IFN-α2b or observation. IFN-α2b therapy significantly increased median overall survival by 1 year and produced a 24% improvement in the 5-year overall survival rate (46% for IFN-α2b patients vs. 37% for observation patients). On the basis of these results, the Food and Drug Administration (FDA) approved the use of adjuvant IFN-α2b after surgical resection of stage IIb or III melanoma. Two large, prospective randomized trials followed. E1690 compared high-dose interferon with both observation and low-dose interferon. The effect of high-dose interferon on 5-year relapse-free survival was significant (44% vs. 35%, P = 0.03) but neither high-dose nor low-dose interferon had any significant impact on overall survival.69 Elective node dissection was not mandated in E1690 (nor E1684), and many patients who had regional recurrences on observation subsequently received IFN-α salvage therapy, which may have impacted the overall survival analysis. The third trial, E1694, compared high-dose IFN to the GMK vaccine; the ganglioside GM2 coupled to kehole limpet hemocyanin and combined with the QS-21 adjuvant. The trial was stopped early when an advantage was seen for interferon in both relapse-free (HR = 1.47; P = 0.001) and overall (HR = 1.52; P = 0.009) survival.
Pegylated IFN-α2b (peg-IFN), where the IFN molecule is bound to a polyethylene glycol moiety, allows for sustained absorption and a longer half-life. This regimen has an induction phase of peg-IFN SC 6 μg/kg a week for 8 weeks followed by a maintenance dose of SC 3 μg/kg a week for up to 5 years. In the European Organization for Research and Treatment of Cancer (EORTC) trial 18991 of peg-IFN versus observation in patients with resected stage III melanoma, peg-IFN was associated with an improved RFS (HR = 0.87; 95% CI, 0.76 to 1.00; P = 0.05), with no difference in overall survival.70 In 2011, the FDA approved peg-IFN as an alternative to high-dose IFN in the adjuvant therapy of melanoma.
Biochemotherapy (BCT) combines chemotherapy and immunotherapy and has been examined in stage IV melanoma for many years.71,72 It was studied as a potential adjuvant therapy in the Southwest Oncology Group (SWOG) trial S008, which compared a 9-week BCT regimen composed of cisplatin, vinblastine, dacarbazine, interleukin-2 (IL-2), and IFN-α to high-dose interferon.73 While there was a significant improvement in RFS compared with HDI (4.0 years vs. 1.9 years; P = 0.029), there was a higher rate of grade III/IV toxicity and no difference in overall survival (56% 5-year survival in both groups). The use of BCT may be considered for adjuvant therapy in stage IIIA–IIIC melanoma.
Treatment of In-Transit Disease
In-transit disease and satellitosis develop in 5% to 8% of patients with melanomas thicker than 1.5 mm.74 These are classified as stage III disease but portend not only a poor prognosis but can be a challenge to treat. Treatment involves either local excision or ablation, or regional chemotherapy. Surgical excision is the optimal management when the number of lesions is small and complete excision is possible. Prior to proceeding with surgery, a complete staging workup for metastatic disease is recommended, as these patients often harbor distant disease. When surgery is not feasible, a number of intralesional therapies have been utilized: Bacille Calmette–Guérin, IL-2, granulocyte-macrophage colony stimulating factor (GM-CSF), and rose bengal. A new agent, talimogene laherparepvec (T-VEC), is an oncolytic immunotherapy derived from herpes simplex virus type-1. Injected intralesionally, T-VEC is designed to replicate in tumors and release GM-CSF to enhance systemic antitumor immune responses. In a randomized phase III trial, T-VEC demonstrated an improved durable response rate (16% vs. 2%; P < 0.0001) and a trend toward an improved overall survival.75
When the in-transit disease is unresectable but limited to an extremity, regional administration of high-dose chemotherapy can be effective in controlling disease. Isolated limb perfusion (ILP) uses an extracorporeal membrane oxygenator (as is used with cardiac surgery) to deliver chemotherapy doses 15 to 25 times higher than can be obtained with systemic delivery. The surgeon isolates the vessels and uses a tourniquet to prevent systemic uptake of the agent(s). Using the bypass machine, the limb is perfused with hyperthermic (38°C to 40°C) chemotherapy solutions, most commonly melphalan. After 60 to 90 minutes of treatment, the drug is flushed from the circulation and systemic circulation is restored. Response rates between 80% and 90% are reported, with complete response rates between 40% and 65%.74,76–78 In some cases, these can be quite durable. Toxicities can range from mild erythema and edema to epidermolysis, functional impairment, and in extremely rare cases a need for amputation.
Isolated limb infusion (ILI) is a minimally invasive alternative to ILP. Rather than surgical isolation of the vessels, percutaneous catheters are placed radiologically in the artery and vein and connected to an extracorporeal circuit incorporating a heating coil. A tourniquet is placed on the extremity, but because some systemic uptake is anticipated, lower doses of chemotherapeutic agents are used. Some studies have reported response rates equal to ILP, although others report slightly lower response rates.79–82 However, the role of both ILP and ILI is evolving as new systemic therapeutic options become available.
Treatment of Disseminated Melanoma
Evaluation for Metastatic Disease and Clinical Course
The follow-up evaluation for patients with AJCC stage I, II, or III melanoma who are rendered NED by surgery should include regular histories and physical examinations. The use of extensive and frequent radiographic studies and blood work in asymptomatic, clinically disease-free patients is rarely productive.24 For AJCC stage IA to IIA, the NCCN recommends that patients should undergo a history and physical examination with emphasis on skin and nodal examinations every 6 to 12 months as clinically indicated. For AJCC stage IIB to IV NED, patients should undergo a history and physical examination every 3 to 6 months for 2 years, then every 3 to 12 months for 3 years, and then annually as clinically indicated. Imaging studies (CXR, CT, or PET/CT) can be considered every 3 to 12 months for the first 3 to 5 years on the basis of the conditional probability of recurrence at any point in time after the initial therapy.24
Melanoma can disseminate to any organ. The most common sites of recurrence are skin, subcutaneous tissues, and distant lymph nodes, followed by visceral sites. Common visceral sites of metastasis, in order of decreasing occurrence, are the lung, liver, brain, bone, and gastrointestinal tract. Most patients who die with disseminated disease have multiple organ involvement. Frequently, the cause of death is respiratory failure or brain complications. Cure with any treatment is rare. However, over the past several years, there have been tremendous advances in the systemic therapy of melanoma. Whereas only several years ago there were only two FDA-approved therapies for the treatment of metastatic melanoma (dacarbazine and high-dose IL-2), there are now new targeted therapies and immune checkpoint regulators that have been approved for the treatment of advanced melanoma, with several new therapies and combinations in clinical trial. How to proceed in the management of advanced melanoma should be based on several factors, including the patient’s medical condition and comorbidities, the potential for palliation, and the impact of treatment on quality of life.
Chemotherapy
Melanoma is responsive to few chemotherapeutic drugs. The most widely used single chemotherapeutic agents have been dacarbazine and its oral analogue temozolomide, with objective response rates of 5% to 12%.83 Most responses are transient; only 1% to 2% of patients achieve a durable long-term response.84 Attempts to improve upon these results using combination chemotherapy, or BCT (where chemotherapy is combined with IFN-α2b ± IL-2), failed to significantly improve upon single-agent therapy alone.71,83–85
High-Dose Interleukin-2
IL-2 is a cytokine secreted by antigen-activated helper T cells, initially discovered to be a T-cell growth factor but subsequently found to have many other immunologic effects, including an important role in the enhancement of immune responses. High-dose IL-2 (600,000 IU/kg every 8 hours for up to 14 doses) is associated with an objective response rate of 15% to 20%, with a small percentage of patients (around 5%) experiencing long-term, durable complete responses (Fig. 107-5).86–88 These results may be improved upon with the addition of vaccines.89 However, IL-2 treatment requires inpatient intensive care, as toxicity is severe and can include hypotension, pulmonary edema, decreased renal function, respiratory insufficiency, and neurotoxicity.
BRAF and MEK Inhibitors
The MAP kinase pathway is a key pathway in melanoma pathogenesis, with nearly half of melanomas having constitutive activation due to mutations in the BRAF gene.90 Activation of mitogen-activated protein kinase (MAPK) signaling leads to unregulated cell proliferation. Vemurafenib and dabrafenib are targeted therapies that block the constitutively active mutant BRAF. Both have been associated with significant improvements in both progression-free and overall survival compared with dacarbazine.91,92 Common adverse effects include arthralgia, rash, fatigue, alopecia, and the development of cutaneous keratoacanthomas and SCCs. Patients treated with BRAF inhibition have dramatic responses, but unfortunately the survival benefit is limited because of the eventual development of resistance in most patients.

Figure 107-5. Basal cell carcinoma near the eye.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree




