KEY CONCEPTS
![]() Advancing age, inherited and acquired genetic susceptibilities, lifestyle choices, inflammatory bowel disease, type 2 diabetes mellitus, and environmental factors are associated with colorectal cancer risk.
Advancing age, inherited and acquired genetic susceptibilities, lifestyle choices, inflammatory bowel disease, type 2 diabetes mellitus, and environmental factors are associated with colorectal cancer risk.
![]() Regular use of aspirin and other nonsteroidal antiinflammatory drugs, calcium intake, and higher blood vitamin D levels may reduce risk of colorectal cancer, but they are not currently recommended for routine cancer prevention.
Regular use of aspirin and other nonsteroidal antiinflammatory drugs, calcium intake, and higher blood vitamin D levels may reduce risk of colorectal cancer, but they are not currently recommended for routine cancer prevention.
![]() Effective colorectal cancer screening programs incorporate regular examination of the entire colon starting at age 50 years for average-risk individuals. Colorectal adenomas can progress to cancer and should be removed.
Effective colorectal cancer screening programs incorporate regular examination of the entire colon starting at age 50 years for average-risk individuals. Colorectal adenomas can progress to cancer and should be removed.
![]() The histologic stage of colorectal cancer upon diagnosis—determined by depth of bowel invasion, lymph node involvement, and presence of metastases—is the most important prognostic factor for disease recurrence and survival.
The histologic stage of colorectal cancer upon diagnosis—determined by depth of bowel invasion, lymph node involvement, and presence of metastases—is the most important prognostic factor for disease recurrence and survival.
![]() The treatment goal for stages I, II, and III colon cancer is cure; surgery should be offered to all eligible patients for this purpose. Six months of fluoropyrimidine-based adjuvant systemic therapy reduces the risk of cancer recurrence and overall mortality in patients with stage III and select populations with stage II colon cancer. An oxaliplatin-containing regimen further reduces risk as compared with fluoropyrimidine alone.
The treatment goal for stages I, II, and III colon cancer is cure; surgery should be offered to all eligible patients for this purpose. Six months of fluoropyrimidine-based adjuvant systemic therapy reduces the risk of cancer recurrence and overall mortality in patients with stage III and select populations with stage II colon cancer. An oxaliplatin-containing regimen further reduces risk as compared with fluoropyrimidine alone.
![]() Combined modality neoadjuvant therapy consists of fluoropyrimidine-based chemosensitized radiation therapy and surgery for patients with stage II or III cancer of the rectum and is considered standard of care to decrease risk of local and distant disease recurrence.
Combined modality neoadjuvant therapy consists of fluoropyrimidine-based chemosensitized radiation therapy and surgery for patients with stage II or III cancer of the rectum and is considered standard of care to decrease risk of local and distant disease recurrence.
![]() Preoperative chemotherapy may reduce tumor size and convert unresectable disease to resectable disease in selected patients with metastatic colorectal cancer. This strategy offers the potential for prolonging overall survival and cure for metastatic disease.
Preoperative chemotherapy may reduce tumor size and convert unresectable disease to resectable disease in selected patients with metastatic colorectal cancer. This strategy offers the potential for prolonging overall survival and cure for metastatic disease.
![]() Chemotherapy is palliative for metastatic disease. A fluoropyrimidine with oxaliplatin or irinotecan improves survival compared to fluoropyrimidine monotherapy and should be offered to patients who are candidates for aggressive treatment. The ability for patients to receive all active cytotoxic agents (e.g., fluoropyrimidine, oxaliplatin, irinotecan) during the course of their disease improves their overall survival.
Chemotherapy is palliative for metastatic disease. A fluoropyrimidine with oxaliplatin or irinotecan improves survival compared to fluoropyrimidine monotherapy and should be offered to patients who are candidates for aggressive treatment. The ability for patients to receive all active cytotoxic agents (e.g., fluoropyrimidine, oxaliplatin, irinotecan) during the course of their disease improves their overall survival.
![]() Bevacizumab plus fluoropyrimidine-based chemotherapy as initial therapy for metastatic disease is considered standard of care and provides a survival benefit as compared with combination chemotherapy alone.
Bevacizumab plus fluoropyrimidine-based chemotherapy as initial therapy for metastatic disease is considered standard of care and provides a survival benefit as compared with combination chemotherapy alone.
![]() The addition of cetuximab or panitumumab to initial treatment for KRAS wild-type advanced or metastatic disease may improve tumor response rates and survival. Individuals who have disease progression after initial therapy not containing an epidermal growth factor receptor (EGFR) inhibitor may benefit from cetuximab or panitumumab, either alone as a single agent or combined with other drugs. However, patients with codon 12 or 13 KRAS gene mutations should not receive cetuximab or panitumumab as these tumor mutations predict lack of treatment response.
The addition of cetuximab or panitumumab to initial treatment for KRAS wild-type advanced or metastatic disease may improve tumor response rates and survival. Individuals who have disease progression after initial therapy not containing an epidermal growth factor receptor (EGFR) inhibitor may benefit from cetuximab or panitumumab, either alone as a single agent or combined with other drugs. However, patients with codon 12 or 13 KRAS gene mutations should not receive cetuximab or panitumumab as these tumor mutations predict lack of treatment response.
Colorectal cancer involves the colon, rectum, and anal canal. It is one of the three most common cancers occurring in adult men and women in the United States and accounts for about one in nine cancer diagnoses. In 2013, an estimated 142,820 new cases will be diagnosed, of which 102,480 will involve the colon and 40,340 the rectum.1 An additional 7,060 new cases of cancer involve the anus, anal canal, or anorectum.1
For both adult men and women, colorectal cancer is the third leading cause of cancer-related deaths in the United States. An estimated 50,830 deaths will occur during 2013.1
Mortality and incidence rates associated with colorectal cancer in the United States have decreased steadily over the past two decades. Incidence rates vary worldwide, with the highest incidence rates in economically developed countries in North America, Europe, New Zealand, and Australia, whereas lowest rates are found in Central America, Africa, and South-Central Asia.2 Colorectal cancer mortality rates have been decreasing in and are comparable between the United States and several Western countries; mortality rates continue to increase in less developed countries in eastern Europe and Central and South America.2
Multiple factors are associated with the development of colorectal cancer, including inherited susceptibility, environmental and lifestyle factors, and certain disease states. Overall, about 39% of affected individuals undergo a surgical procedure alone intended for cure. An additional 37% of individuals can potentially be cured with surgery followed by adjuvant radiation therapy (XRT), chemotherapy, or both. Curability is influenced primarily by the depth of tumor penetration, involvement of lymph nodes, and presence of metastatic disease. Five-year survival rates are about 91% and 88% for persons with early stages of colon and rectal cancer, respectively.3 After the tumor has spread regionally to adjacent lymph nodes or tissues, 5-year survival rates drop to about 70% for both colon and rectal cancer; 5-year survival for individuals with metastatic disease is about 12%.
Treatment modalities for colorectal cancer include surgery, XRT, chemotherapy, and targeted molecular therapies (e.g., angiogenesis inhibitors, epidermal growth factor receptor inhibitors). Surgery is the important and definitive procedure associated with cure. XRT can improve curability following surgical resection in rectal cancer and may reduce symptoms and complications associated with advanced disease. Chemotherapy is used in the adjuvant setting to increase cure rates and in treatment for advanced stages of disease to prolong survival. Selected patients with advanced disease who receive aggressive preoperative chemotherapy and targeted therapies experience higher resection rates and can be potentially cured. Much progress has been made in the treatment of advanced disease, the ability to identify candidates for potentially curative surgical procedures, and the availability of active drug regimens that improve patients’ survival.
EPIDEMIOLOGY
Colorectal cancer is the third most common malignancy worldwide, accounting for more than 1.2 million new cases annually.2 The variation in colorectal cancer occurrence worldwide is at least 20-fold.2 The highest incidence rates are found in Australia and New Zealand, Europe, and North America. The lowest incidence rates are seen in less-developed areas such as Africa, South Central Asia, and Central America. Most recently, incidence rates have rapidly increased in newer economically developed countries in eastern Europe and in Japan, Korea, and China.2 The influence of environmental factors (e.g., increased intake of caloric-dense foods and physical inactivity) on colorectal cancer risk has become evident through studies of migrants, where the incidence of colorectal cancer increases rapidly within first-generation immigrants who migrate from low- to high-risk areas.2 However, colorectal cancers are known to develop more frequently in certain families, and genetic predisposition to this disease is also well recognized.
The incidence of invasive colon cancer is greatest among males, who have an age-adjusted incidence rate of 37.4 per 100,000, as compared with females for whom the rate is 29.9 per 100,000.3 Invasive cancer of the rectum occurs less frequently; the incidence rate is 16.5 and 10.3 per 100,000 for males and females, respectively. Differences in colorectal cancer incidence exist among ethnic groups in the United States, where incidence is highest among African Americans compared to white, American Indian/Alaska Native, Hispanic/Latino, and Asian American/Pacific Islander males and females.1 Cultural and genetic factors, as well as disparities in access to healthcare services, may influence risk among population groups.1
The overall incidence of colon and rectal cancers in the United States continues to decline, with an annual percent decrease of 2.5% from 1975 to 2009.3 Cancer incidence rates have declined in every major ethnic group since 1975, although less among American Indian/Alaska Natives. Most recent rapid declines in incidence rates are attributed to screening and polyp removal.1 Figure 107-1 displays trends for incidence and mortality rates among white and African American males and females in the United States.
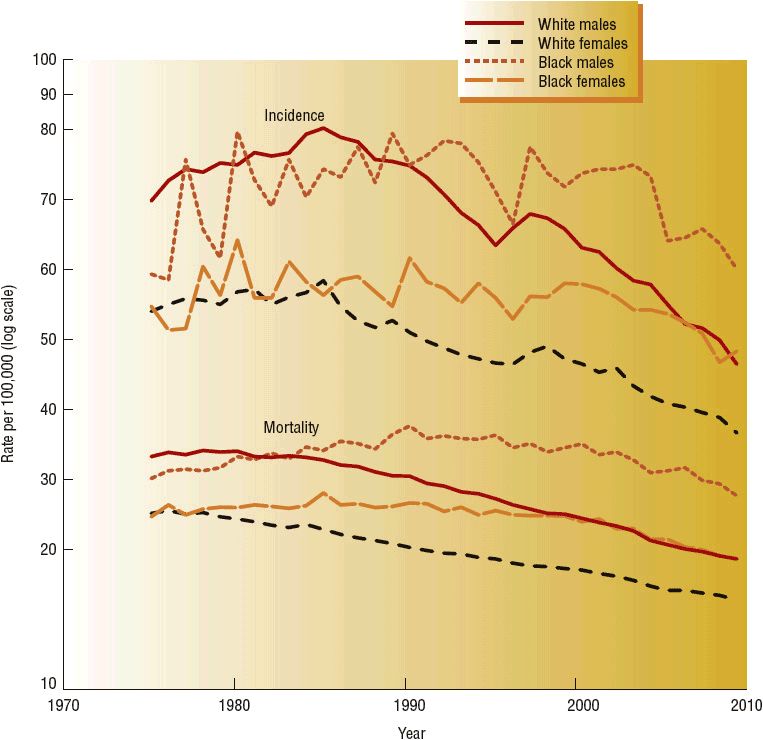
FIGURE 107-1 National Cancer Institute, Surveillance Epidemiology and End Results (SEER) incidence and mortality rates for invasive colon and rectum cancer, 1975–2009. SEER 9 areas and US Mortality Files (National Center for Health Statistics, CDC). Rates are age-adjusted to the 2000 U.S. standard population (19 age groups—Census P25-1103). (From reference 3.)
Cancer of the colon and rectum accounts for about 9% of all cancer deaths in the United States. The median age for death from cancer of the colon or rectum is 74 years.3 It is estimated that 50,830 individuals will die of colorectal cancer in the United States in 2013, which represents a continued decline in overall combined mortality for both colon and rectal cancer by more than 30% observed during the last 20 years.1 Overall mortality rates are highest among African American males and females, although a steep rate of decline began in the late 1990s.3 Colorectal cancer death rates are decreasing among all ethnic groups; however, mortality rates are not statistically lower in American Indian/Alaska Natives.3 Factors contributing to the overall decline in colorectal cancer mortality include decreasing incidence rates, screening programs with early polyp removal, and more effective and better tolerated treatments. Differences among different world geographic regions, and in population groups in the United States, may also reflect variations in underlying tumor biology, stage at diagnosis, access to screening programs, and availability of effective treatments.1–3
ETIOLOGY AND RISK FACTORS
Numerous studies suggest that the development of colorectal cancer is related to both uncontrollable and modifiable risk factors. Age, family history, clinical and genetic susceptibilities cannot be controlled by individuals. However, lifestyle choices, dietary and environmental factors that affect the bowel may influence an individual’s risk of developing colorectal cancer.
Personal Medical History
Age
An individual’s risk of developing cancer of the colon or rectum increases with advancing age, with the likelihood of cancer diagnosis increasing after 40 years of age and rising progressively after age 50.3 The median age at diagnosis is 69 years.3 Although fewer than 20% of patients are less than 50 years of age at the time of diagnosis, the incidence of colorectal cancer is increasing in this age group, in contrast to overall rates of decline among adults age 50 years and older. The reasons for this pattern are unclear, but may reflect increasing trends in obesity and detrimental dietary factors among younger people.4
Adenomatous Polyps or Colorectal Cancer
A prior history of high-risk adenomatous polyps, particularly multiple adenomas or size ≥10 mm, is associated with increased risk of colorectal cancer.5 Individuals with a prior diagnosis of colon or rectal cancer have a greater risk of developing a new malignancy at another area in their colon or rectum as compared to individuals without a prior history of colorectal cancer.
Inflammatory Bowel Disease
![]() Chronic ulcerative colitis, particularly when it involves the entire large intestine, predisposes individuals to colorectal cancer at a rate that is 5- to 10-fold greater than average.6 The risk is even greater for young individuals and increases for all affected individuals with increasing extent of bowel involvement and disease duration. The cumulative risk of colorectal cancer is low early in life, but increases from 2% at 10 years after diagnosis to 8% and 18% at 20 and 30 years, respectively.6 Chronic underlying inflammation, oxidative stress, and release of various cytokines, including nuclear factor-kappa B (NF-κB) and tumor necrosis factor-alpha (TNF-α), appear to promote tumorigenesis.7 The progressive dysplastic changes that bowel mucosa undergo are similar to those observed in adenomatous polyps. Similarly, patients with Crohn’s disease are also at increased risk, and the risk is believed to be about that of patients with ulcerative colitis.6 As compared with sporadic colon cancer or cancer associated with ulcerative colitis, colon cancer in patients with Crohn’s disease tends to arise in the proximal colon.6 This is most likely related to the area of bowel affected by the chronic inflammatory process in individuals with Crohn’s disease. Overall, persons diagnosed with either disease constitute about 1% to 2% of all new cases of colorectal cancer each year.
Chronic ulcerative colitis, particularly when it involves the entire large intestine, predisposes individuals to colorectal cancer at a rate that is 5- to 10-fold greater than average.6 The risk is even greater for young individuals and increases for all affected individuals with increasing extent of bowel involvement and disease duration. The cumulative risk of colorectal cancer is low early in life, but increases from 2% at 10 years after diagnosis to 8% and 18% at 20 and 30 years, respectively.6 Chronic underlying inflammation, oxidative stress, and release of various cytokines, including nuclear factor-kappa B (NF-κB) and tumor necrosis factor-alpha (TNF-α), appear to promote tumorigenesis.7 The progressive dysplastic changes that bowel mucosa undergo are similar to those observed in adenomatous polyps. Similarly, patients with Crohn’s disease are also at increased risk, and the risk is believed to be about that of patients with ulcerative colitis.6 As compared with sporadic colon cancer or cancer associated with ulcerative colitis, colon cancer in patients with Crohn’s disease tends to arise in the proximal colon.6 This is most likely related to the area of bowel affected by the chronic inflammatory process in individuals with Crohn’s disease. Overall, persons diagnosed with either disease constitute about 1% to 2% of all new cases of colorectal cancer each year.
Type 2 Diabetes Mellitus
![]() Type 2 diabetes mellitus, independent of body mass size and physical activity level, is associated with increased colorectal cancer risk, although glycosylated hemoglobin (HbA1c) alone as an indicator of hyperglycemia and association with colorectal cancer is inconsistent.8 Metabolic syndrome is associated with an elevated risk of colorectal cancer.8 In a meta-analysis of 15 studies, diabetes was associated with a 30% increase in risk of colorectal cancer and increased risk of colorectal cancer mortality.9 Features associated with type 2 diabetes, such as hyperinsulinemia and elevated levels of free insulin-like growth factor-1 (IGF-1), promote tumor cell proliferation.8,10 Individuals diagnosed with colorectal cancer and type 2 diabetes have a higher risk of all-cause mortality compared to individuals without diabetes.10 Risk of death from cardiovascular disease was higher among patients receiving insulin whereas colorectal cancer related mortality was lower with insulin use. Individuals with type 2 diabetes mellitus treated for colorectal cancer also have decreased disease-free survival and overall survival and experience a higher incidence of treatment-related diarrhea and risk of death.
Type 2 diabetes mellitus, independent of body mass size and physical activity level, is associated with increased colorectal cancer risk, although glycosylated hemoglobin (HbA1c) alone as an indicator of hyperglycemia and association with colorectal cancer is inconsistent.8 Metabolic syndrome is associated with an elevated risk of colorectal cancer.8 In a meta-analysis of 15 studies, diabetes was associated with a 30% increase in risk of colorectal cancer and increased risk of colorectal cancer mortality.9 Features associated with type 2 diabetes, such as hyperinsulinemia and elevated levels of free insulin-like growth factor-1 (IGF-1), promote tumor cell proliferation.8,10 Individuals diagnosed with colorectal cancer and type 2 diabetes have a higher risk of all-cause mortality compared to individuals without diabetes.10 Risk of death from cardiovascular disease was higher among patients receiving insulin whereas colorectal cancer related mortality was lower with insulin use. Individuals with type 2 diabetes mellitus treated for colorectal cancer also have decreased disease-free survival and overall survival and experience a higher incidence of treatment-related diarrhea and risk of death.
Family History and Inherited Genetic Risk
Colorectal Cancer or Adenomatous Polyps
![]() Three specific patterns of colon cancer occurrence are generally observed: sporadic, familial, and recognized hereditary syndromes. Although most cases of colon cancer are sporadic in nature, about 20% of patients who develop colorectal cancer will have a family history of colorectal cancer.11,12 In these families, the frequency of colorectal cancer is too high to be considered sporadic, but the pattern is not consistent with an inherited syndrome. First-degree relatives of patients diagnosed with colorectal cancer have an increased risk of the disease, particularly if the relative was diagnosed at age 60 or younger.12 Similarly, parents and siblings of relatives diagnosed with adenomatous polyps are at increased risk for developing colorectal cancer. The reasons for these associations are not established, but may be related to a combination of inherited genes and environmental factors.12
Three specific patterns of colon cancer occurrence are generally observed: sporadic, familial, and recognized hereditary syndromes. Although most cases of colon cancer are sporadic in nature, about 20% of patients who develop colorectal cancer will have a family history of colorectal cancer.11,12 In these families, the frequency of colorectal cancer is too high to be considered sporadic, but the pattern is not consistent with an inherited syndrome. First-degree relatives of patients diagnosed with colorectal cancer have an increased risk of the disease, particularly if the relative was diagnosed at age 60 or younger.12 Similarly, parents and siblings of relatives diagnosed with adenomatous polyps are at increased risk for developing colorectal cancer. The reasons for these associations are not established, but may be related to a combination of inherited genes and environmental factors.12
Hereditary Syndromes
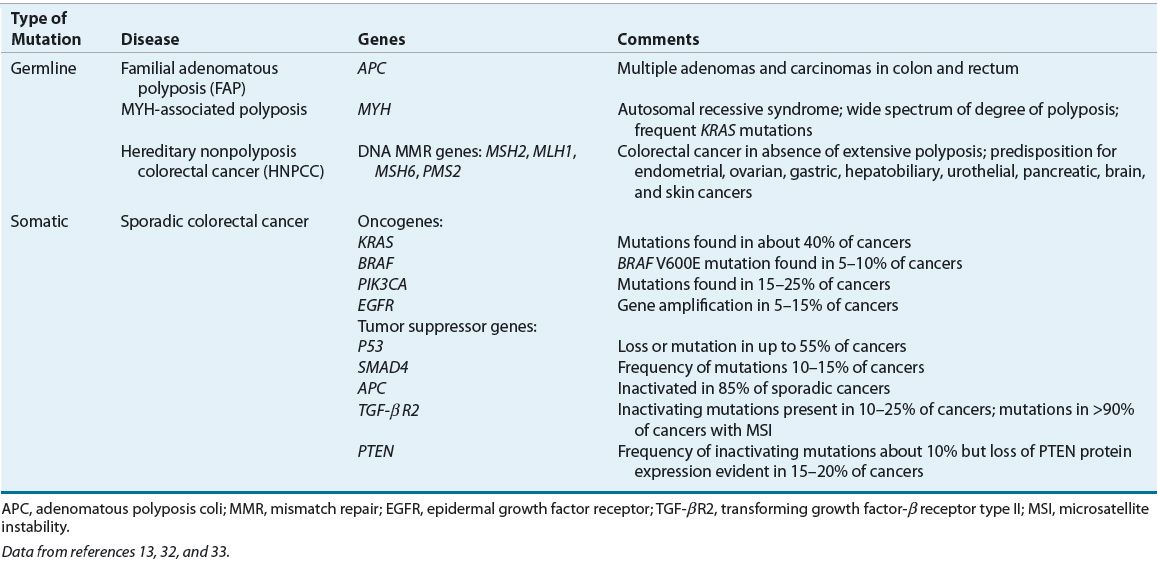
![]() Colorectal cancer is a consequence of several well-defined genetic syndromes.11–15 The two most common forms of hereditary colon cancer are familial adenomatous polyposis (FAP) and hereditary nonpolyposis colorectal cancer (HNPCC). Both forms result from a specific germline mutation. FAP is a rare autosomal dominant trait caused by inactivating mutations of the adenomatous polyposis coli (APC) gene and accounts for 0.2% to 1% of all colorectal cancers. The disease is manifested by hundreds to thousands of tiny sessile adenomatous polyps that carpet the colon and rectum, typically arising during adolescence.13 The polyps continue to proliferate throughout the colon, with eventual transformation to malignancy. The risk of developing colorectal cancer for individuals with untreated FAP is virtually 100%; most will develop colorectal cancer by the fourth and fifth decades of life.12,13 Several variants of FAP exist and are associated with different extracolonic manifestations.13
Colorectal cancer is a consequence of several well-defined genetic syndromes.11–15 The two most common forms of hereditary colon cancer are familial adenomatous polyposis (FAP) and hereditary nonpolyposis colorectal cancer (HNPCC). Both forms result from a specific germline mutation. FAP is a rare autosomal dominant trait caused by inactivating mutations of the adenomatous polyposis coli (APC) gene and accounts for 0.2% to 1% of all colorectal cancers. The disease is manifested by hundreds to thousands of tiny sessile adenomatous polyps that carpet the colon and rectum, typically arising during adolescence.13 The polyps continue to proliferate throughout the colon, with eventual transformation to malignancy. The risk of developing colorectal cancer for individuals with untreated FAP is virtually 100%; most will develop colorectal cancer by the fourth and fifth decades of life.12,13 Several variants of FAP exist and are associated with different extracolonic manifestations.13
HNPCC, also referred to as Lynch syndrome, is an autosomal dominant inherited syndrome and is the most common hereditary predisposition for colorectal cancer.11 Germline mutations in one of the DNA mismatch-repair (MMR) genes, most commonly MLH1, MSH2, MSH6, or PMS2, are responsible for HNPCC, which accounts for 2% to 4% of overall colorectal cancer cases.11 The estimated lifetime risk of developing colorectal cancer by age 70 years is about 66% and 43% for male and female carriers of germline MMR mutations, respectively.14 Multiple generations within a family are affected, and colorectal cancer develops early in life, with a mean age at time of diagnosis of about 45 years of age.13–15 About one-third of individuals with HNPCC develop another HNPCC-related extracolonic malignancy within the following 10 years.15 In contrast to FAP, adenomatous polyps are not a primary manifestation of the HNPCC. Polyps that do form tend to be located primarily in the right-sided, or proximal colon. If HNPCC is suspected in a patient diagnosed with colorectal cancer, typically due to early age at diagnosis or family cancer history, the tumor is examined for evidence of deficient MMR to distinguish between sporadic or germline genetic mutations. Criteria for diagnosis of HNPCC have been established, and it is important to identify carriers of these MMR mutations so that they can be counseled and followed appropriately.13–15
Enzyme Polymorphisms
![]() Increasing evidence suggests that genetic polymorphisms in drug-metabolizing enzymes, such as N-acetyltransferases (NAT1 and NAT2), cytochrome P450 (CYP) isoenzymes, glutathione-S-transferase (GST) enzymes, methylenetetrahydrofolate reductase (MTHFR), and hemochromatosis gene mutations, may confer genetic susceptibility to colorectal cancer.16 Individuals with certain variations in NAT1, NAT2, CYP1A2, CYP1A1, and CYP2E1 enzyme genotypes may be particularly susceptible to carcinogenic effects of a high dietary intake of meat, tobacco smoke, or other environmental factors.16,17
Increasing evidence suggests that genetic polymorphisms in drug-metabolizing enzymes, such as N-acetyltransferases (NAT1 and NAT2), cytochrome P450 (CYP) isoenzymes, glutathione-S-transferase (GST) enzymes, methylenetetrahydrofolate reductase (MTHFR), and hemochromatosis gene mutations, may confer genetic susceptibility to colorectal cancer.16 Individuals with certain variations in NAT1, NAT2, CYP1A2, CYP1A1, and CYP2E1 enzyme genotypes may be particularly susceptible to carcinogenic effects of a high dietary intake of meat, tobacco smoke, or other environmental factors.16,17
Lifestyle Factors
Nonsteroidal Antiinflammatory Drug and Aspirin Use
![]() Several lifestyle factors are known to affect colorectal cancer risk (Table 107-1). Observational studies have reported that regular (at least two doses per week) nonsteroidal antiinflammatory drug (NSAID) and aspirin use is associated with a reduced risk of colorectal cancer. In an average-risk individual, regular aspirin use is associated with a 13% to 28% reduction in the risk of colorectal adenoma, and the risk of colorectal cancer and mortality is reduced by 30% to 40%.18,19 Regular daily aspirin use reduces colorectal adenoma recurrence, and colorectal cancer incidence and mortality in patients with prior adenomas or diagnosis of colorectal cancer.8,18,19
Several lifestyle factors are known to affect colorectal cancer risk (Table 107-1). Observational studies have reported that regular (at least two doses per week) nonsteroidal antiinflammatory drug (NSAID) and aspirin use is associated with a reduced risk of colorectal cancer. In an average-risk individual, regular aspirin use is associated with a 13% to 28% reduction in the risk of colorectal adenoma, and the risk of colorectal cancer and mortality is reduced by 30% to 40%.18,19 Regular daily aspirin use reduces colorectal adenoma recurrence, and colorectal cancer incidence and mortality in patients with prior adenomas or diagnosis of colorectal cancer.8,18,19
TABLE 107-1 Lifestyle Factors Associated with Colorectal Cancer Risk
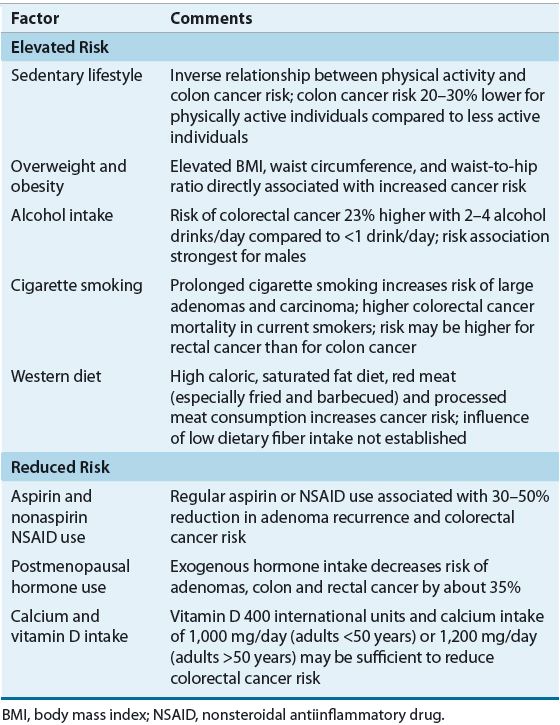
Benefit has also been seen with NSAID and cyclooxygenase-2 inhibitor (COX-2) use. NSAID use over a 10- to 15-year period is associated with protection against adenomas and colorectal cancer, with a 30% to 50% reduction in the risk of colorectal cancer.19,20 The protective effects of these agents appear to be related to their inhibition of COX-2 and free radical formation. COX-2 overexpression is seen in precancerous and cancerous lesions in the colon and is associated with decreased colon cancer cell apoptosis and increased production of angiogenesis-promoting factors.18,19 Up to 50% of colorectal adenomas and 85% of sporadic colon carcinomas have elevated levels of COX-2 and COX-2 overexpression in colorectal cancer is associated with a worse survival. COX-2 appears to play a role in polyp formation, and COX-2 inhibition suppresses polyp growth, restores apoptosis, and decreases expression of proangiogenic factors. Inhibition of COX-2 also downregulates the phosphatidylinositol 3-kinase (PI3K) signaling pathway, which plays an important role in carcinogenesis and cancer cell resistance to apoptosis.21
Postmenopausal Hormone Replacement Therapy
Exogenous postmenopausal oral hormone replacement therapy is associated with a significant reduction in colorectal cancer risk.22 Risk reduction is seen in postmenopausal women receiving both estrogen only and combined estrogen and progestin therapy, and persists for about 10 years after therapy is discontinued.
Several mechanisms for a protective effect of estrogens on the bowel have been identified.8 Age-related declines in estrogen levels are associated with estrogen receptor hypermethylation, which is associated with reduced expression of the estrogen receptor gene and dysregulated colonic mucosal cell growth. Estrogen may also interact with bile acids, or alter levels of insulin and IGF-1, an important mitogen that influences cell-cycle progression in certain cells. However, because postmenopausal hormone replacement therapy increases breast cancer risk and harmful cardiovascular effects, its use is not recommended to prevent colorectal cancer.
Obesity and Physical Inactivity
![]() Physical inactivity and elevated body mass index (BMI), independent of level of physical activity, are associated with an elevated risk of colon adenoma, colon cancer, and rectal cancer.8,12,23,24 Individuals with a higher level of activity throughout life have the lowest risk, which may be up to 50% lower than that of physically inactive individuals. Possible hypotheses are that physical activity stimulates bowel peristalsis, resulting in decreased bowel transit time; or that exercise-induced alterations in body glucose, insulin resistance, hyperinsulinemia, and possibly other hormones reduce tumor cell growth.23
Physical inactivity and elevated body mass index (BMI), independent of level of physical activity, are associated with an elevated risk of colon adenoma, colon cancer, and rectal cancer.8,12,23,24 Individuals with a higher level of activity throughout life have the lowest risk, which may be up to 50% lower than that of physically inactive individuals. Possible hypotheses are that physical activity stimulates bowel peristalsis, resulting in decreased bowel transit time; or that exercise-induced alterations in body glucose, insulin resistance, hyperinsulinemia, and possibly other hormones reduce tumor cell growth.23
In most studies, a 5-unit increase above a healthy BMI was associated with increased risk of colorectal cancer in men, but the relationship is weaker and less consistent for women, possibly because of interactions with age or hormone replacement therapy.23,24 Differences in body composition and distribution of fat weight among men and women could contribute to this discrepancy.8,22 Several mechanisms have been proposed to explain the association between body size and colorectal cancer risk, including insulin resistance, chronic inflammation, and alterations in growth factors or steroid hormones.8
Alcohol and Tobacco Use
![]() Alcohol consumption increases the risk of colorectal cancer, but stronger associations have been observed for men than for women, possibly because alcohol consumption is generally greater in men than in women.8 Lifetime and baseline alcohol consumption increase risk of cancer of the colon and rectum, and an alcohol intake greater than 30 g/day (about two drinks/day) affects risk.8,12 Proposed mechanisms include impaired folate metabolism, abnormal DNA methylation, suppressed tumor immune surveillance, and other procarcinogenic effects related to alcohol intake.8
Alcohol consumption increases the risk of colorectal cancer, but stronger associations have been observed for men than for women, possibly because alcohol consumption is generally greater in men than in women.8 Lifetime and baseline alcohol consumption increase risk of cancer of the colon and rectum, and an alcohol intake greater than 30 g/day (about two drinks/day) affects risk.8,12 Proposed mechanisms include impaired folate metabolism, abnormal DNA methylation, suppressed tumor immune surveillance, and other procarcinogenic effects related to alcohol intake.8
Cigarette smoking is associated with an increased risk of colorectal cancer and mortality, with a stronger association for cancer of the rectum than for cancer of the colon.8,12,25 A dose relationship with increasing number of pack-years and cigarettes smoked per day was also statistically significant but only among patients who had smoked for at least 30 years. As compared to never-smokers, the risks of colorectal cancer and mortality in smokers were 18% and 25% higher, respectively.25 Early tobacco use may also influence risk of cancer recurrence and mortality among colon cancer survivors, possibly due to an increase in genetic alterations that influence tumor behavior.26
Dietary Intake and Nutrients
![]() Epidemiologic studies of worldwide incidence of colorectal cancer suggest that economic development and dietary habits strongly influence its development. However, findings based on epidemiologic data are subject to potential biases and inconsistencies in how dietary factors are categorized and measured, and numerous studies have been able to clearly establish only a few specific dietary habits as independent risk factors for colorectal cancer development.
Epidemiologic studies of worldwide incidence of colorectal cancer suggest that economic development and dietary habits strongly influence its development. However, findings based on epidemiologic data are subject to potential biases and inconsistencies in how dietary factors are categorized and measured, and numerous studies have been able to clearly establish only a few specific dietary habits as independent risk factors for colorectal cancer development.
Fiber, Fruit, and Vegetables
![]() Worldwide, high-fiber dietary patterns have been associated with a low incidence of colorectal cancer.8,27,28 Dietary fiber is composed of both water-soluble and insoluble remnants of plant cells that are not processed by normal human digestive enzymes. Foods that are high in fiber include vegetables, fruits, grains, and cereals. Dietary fiber is postulated to reduce colonic mucosal cell exposure to carcinogens through the dilution or reduced absorption of carcinogens in the bowel, reduced fecal pH, reduced bowel transit time, alterations in bile acid metabolism, or increased production of short-chain fatty acids.8 At present, the role of dietary fiber with regard to amount, source, and type and colorectal cancer risk requires further study.
Worldwide, high-fiber dietary patterns have been associated with a low incidence of colorectal cancer.8,27,28 Dietary fiber is composed of both water-soluble and insoluble remnants of plant cells that are not processed by normal human digestive enzymes. Foods that are high in fiber include vegetables, fruits, grains, and cereals. Dietary fiber is postulated to reduce colonic mucosal cell exposure to carcinogens through the dilution or reduced absorption of carcinogens in the bowel, reduced fecal pH, reduced bowel transit time, alterations in bile acid metabolism, or increased production of short-chain fatty acids.8 At present, the role of dietary fiber with regard to amount, source, and type and colorectal cancer risk requires further study.
Red Meat, Processed Meat, and Fat
![]() Studies suggest that dietary fat intake may be associated with colorectal cancer risk.8,27 This may have resulted from the use of dietary evaluations that focused on the quantity, origin, or type (saturated, monounsaturated, and polyunsaturated) of fat rather than on the source of dietary fat ingested. Dietary fat may promote cancer development as a result of its effect on fecal bile acid concentrations. Dietary fat ingestion stimulates the release of bile acids that are converted by colonic flora to secondary bile acids, which are associated with bowel mucosal irritation and cell proliferation responses and may promote tumor growth.27
Studies suggest that dietary fat intake may be associated with colorectal cancer risk.8,27 This may have resulted from the use of dietary evaluations that focused on the quantity, origin, or type (saturated, monounsaturated, and polyunsaturated) of fat rather than on the source of dietary fat ingested. Dietary fat may promote cancer development as a result of its effect on fecal bile acid concentrations. Dietary fat ingestion stimulates the release of bile acids that are converted by colonic flora to secondary bile acids, which are associated with bowel mucosal irritation and cell proliferation responses and may promote tumor growth.27
The association between red, but not white, meat consumption and colorectal cancer is strongest, which may be related to the heterocyclic amines and polycyclic aromatic hydrocarbons formed during the cooking process, or the presence of specific fatty acids in red meat such as arachidonic acid.8,27 Processed meat products containing certain preservatives may increase exogenous exposure to carcinogenic N-nitroso compounds.27 Although red and processed meat and high saturated fat intake has been associated with increased risk of colorectal cancer, the exact nature and magnitude of these risks have not been determined.
Calcium and Vitamin D
![]() Inverse associations between dietary calcium, vitamin D intake, and serum 25-hydroxyvitamin D3 levels, and colorectal cancer risk have been reported in several observational studies.8,27,29 Calcium may exert antiproliferative effects by binding to bile and fatty acids in the small intestine, thereby reducing colonic epithelial cell exposure to mutagens.8 In addition, calcium induces differentiating, pro-apoptotic, and direct growth-restraining activities on both normal and tumor cells in the gastrointestinal tract.8,27 Vitamin D has antiproliferative and differentiation and pro-apoptotic effects on colonic epithelial cells and on a variety of tumor cells.8,29,30 Most of its actions are mediated through a high-affinity nuclear vitamin D receptor (VDR), and the expression of this receptor is altered during different phases of colon cancer development.30 Other genes involved in key signaling pathways that influence colorectal cancer development, such as Wnt/β-catenin, are also regulated by the VDR transcription factor.30 Thus, cellular responsiveness to vitamin D and associated cancer risk is unlikely limited to dietary intake alone. Vitamin D and calcium appear to interact synergistically to protect against adenoma recurrence and colorectal cancer.8
Inverse associations between dietary calcium, vitamin D intake, and serum 25-hydroxyvitamin D3 levels, and colorectal cancer risk have been reported in several observational studies.8,27,29 Calcium may exert antiproliferative effects by binding to bile and fatty acids in the small intestine, thereby reducing colonic epithelial cell exposure to mutagens.8 In addition, calcium induces differentiating, pro-apoptotic, and direct growth-restraining activities on both normal and tumor cells in the gastrointestinal tract.8,27 Vitamin D has antiproliferative and differentiation and pro-apoptotic effects on colonic epithelial cells and on a variety of tumor cells.8,29,30 Most of its actions are mediated through a high-affinity nuclear vitamin D receptor (VDR), and the expression of this receptor is altered during different phases of colon cancer development.30 Other genes involved in key signaling pathways that influence colorectal cancer development, such as Wnt/β-catenin, are also regulated by the VDR transcription factor.30 Thus, cellular responsiveness to vitamin D and associated cancer risk is unlikely limited to dietary intake alone. Vitamin D and calcium appear to interact synergistically to protect against adenoma recurrence and colorectal cancer.8
Folate and Other Micronutrients
Folate intake has been linked to colorectal cancer risk through epidemiologic and experimental studies in cell lines, animals, and humans.8,31 However, the underlying basis for this is complex, particularly because alcohol use, smoking, genetic variants of the MTHFR gene, and other factors can interfere with folate metabolism.8,31 Cellular folates act to accept and donate methyl groups in cellular processes that influence DNA synthesis and methylation of DNA, RNA, and proteins.31 Variations in DNA methylation of gene promoter regions influence gene expression and DNA stability. Inappropriate hypermethylation leads to inactivation of tumor suppressor gene function and hypomethylation can result in oncogene activation.31
The relationship between the timing of folate exposure to the development of neoplastic foci may influence what appears to be a bimodal impact of folate on tumorigenesis.8,31 Moderate folate supplementation, if initiated prior to the establishment of neoplastic foci, may be protective, whereas excessive or increased intake might enhance growth of established early neoplastic lesions.8,31 Thus, an adequate dietary folate intake may be enough to lower the risk of colorectal cancer, and exceeding normal intake may not be beneficial.
Epidemiologic and animal model data suggest that deficiencies in other dietary micronutrients, including vitamin B6, selenium, vitamin C, vitamin E, and carotenoids, may increase colorectal cancer risk, but there is no convincing evidence that the incidence of colorectal cancer is greater in patients with low serum levels than in patients with adequate levels.8
PATHOPHYSIOLOGY
Anatomy and Bowel Function
The large intestine consists of the cecum; the ascending, transverse, descending, and sigmoid colon; and the rectum (Fig. 107-2). In adults, it extends about 1.5 m and has a diameter ranging from 8 cm in the cecum to 2 cm in the sigmoid colon. The function of the large intestine is to receive 500 to 2,000 mL of ileal contents per day. Absorption of fluid and solutes occurs in the right colon or the segments proximal to the middle of the transverse colon, with movement and storage of fecal material in the left colon and distal segments of the colon. Mucus secretion from goblet cells into the intestinal lumen lubricates the mucosal surface and facilitates movement of the dehydrated feces. It also serves to protect the luminal wall from bacteria and colonic irritants such as bile acids.
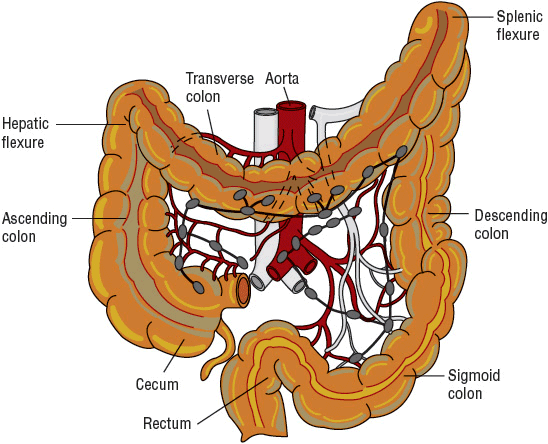
FIGURE 107-2 Colon and rectum anatomy.
Four major tissue layers, from the lumen outward, form the large intestine: the mucosa, submucosa, muscularis propria, and serosa (Fig. 107-3). Embedded in the submucosa and muscularis propria is a rich lymphatic capillary system. Lymphatic channels do not extend into the mucosa. The muscularis propria consists of circular smooth muscle and outer longitudinal smooth muscle bands. Contraction of these muscle groups moves colonic material toward the anal canal. The outermost layer of the colon, the serosa, secretes a fluid that allows the colon to slide easily over nearby structures within the peritoneum. The serosa covers only the anterior and lateral aspects of the upper third of the rectum. The lower third lies completely extraperitoneal and is surrounded by fibrofatty tissue as well as adjacent organs and structures.
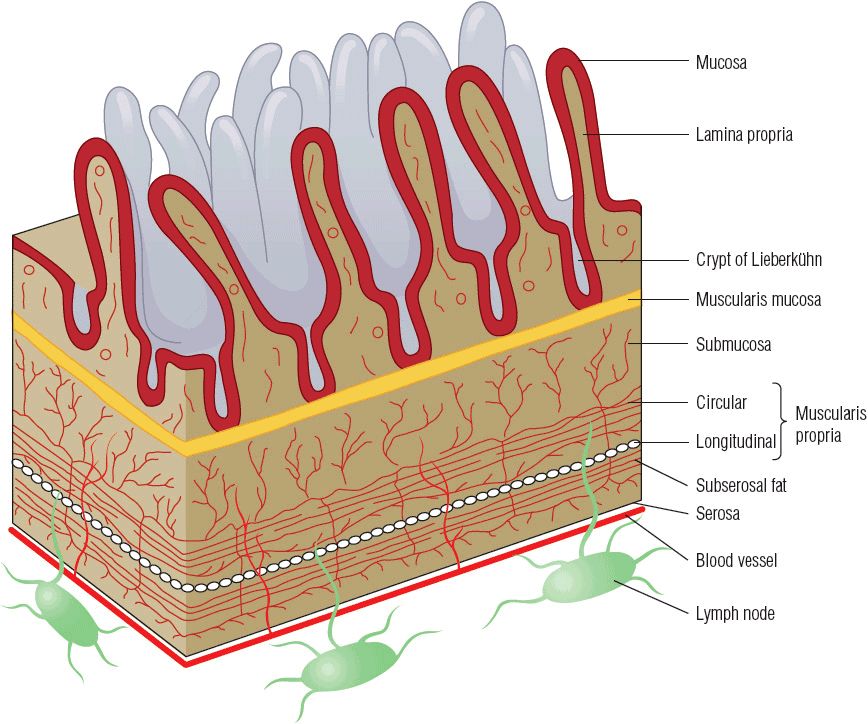
FIGURE 107-3 Cross-section of bowel wall.
The surface epithelium of the colonic mucosa undergoes continual renewal, and complete replacement of epithelial cells occurs every 4 to 8 days. Cell replication normally takes place within the lower third of the crypts, the tubular glands located within the intestinal mucosa. The cells then mature and differentiate to either goblet or absorptive cells as they migrate toward the bowel lumen. The total number of epithelial cells remains relatively constant as the number of cells migrating from the crypts is balanced by the rate of exfoliation of cells from the mucosal surface. This two-phase process is critical to the malignant transformation of the epithelial cells. The number of dysplastic and hyperplastic aberrant crypt foci increases with increasing age; as the mass of abnormal cells accumulates at the top of the crypt and starts to protrude into the stream of fecal matter, their contact with fecal mutagens can lead to further cell mutations and eventual adenoma formation.
Colorectal Tumorigenesis
The development of a colorectal neoplasm is a multistep process involving several genetic and phenotypic alterations of normal bowel epithelium structure and function, leading to dysregulated cell growth, proliferation, and tumor development. Because most colorectal cancers develop sporadically, with no inherited or familial disposition, efforts have been directed toward identifying these alterations and learning whether detection of such changes may lead to improved cancer detection or treatment outcomes.
Features of colorectal tumorigenesis include genomic instability, activation of oncogene pathways, mutational inactivation or silencing of tumor-suppressor genes, and activation of growth factor pathways.32,33 A genetic model has been proposed for colorectal tumorigenesis that describes a process of transformation from adenoma to carcinoma (Fig. 107-4). The adenoma to carcinoma sequence of tumor development reflects an accumulation of mutations within colonic epithelium that confers a selective growth advantage to the affected cells. Key elements of this process include hyperproliferation of epithelial cells to form a small benign neoplasm or adenoma in conjunction with acquisition of various genetic mutations.32 These mutations occur early and frequently in sporadic cases of both adenomas and colorectal cancer. Somatic mutations must occur in multiple genes to produce the malignant transformation. Table 107-2 lists important genetic mutations that are associated with colorectal cancers.
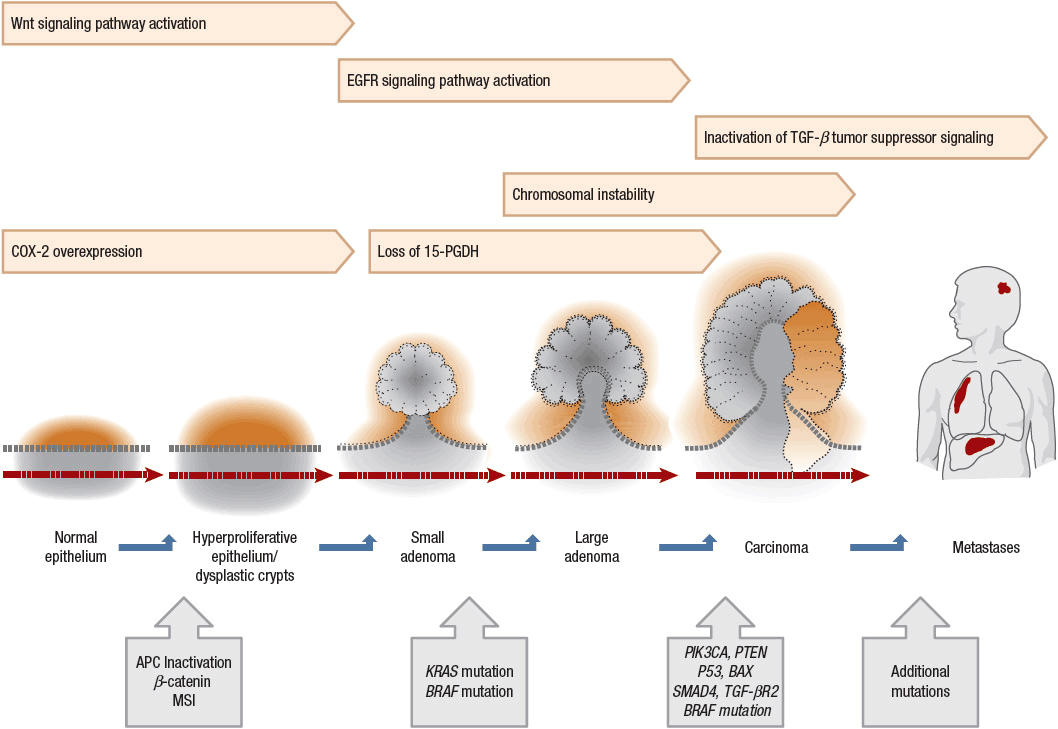
FIGURE 107-4 Genetic changes associated with the adenoma–carcinoma sequence in colorectal cancer. The accumulation of genetic changes in the pathogenesis of colorectal cancer includes microsatellite instability (MSI) initiated by aberrant DNA methylation or mismatch repair (MMR) gene mutation with subsequent disruption in transforming growth factor-β receptor type II (TGF- β2R) and BAX signaling; mutation in the adenomatous polyposis coli (APC) gene or abnormalities in β-catenin leading to inappropriate activation of the Wnt signaling pathway; mutational activation of cyclooxygenase-2 (COX-2) and impaired prostaglandin degradation from loss of 15-prostaglandin dehydrogenase (15-PGDH); KRAS, PIK3CA, or BRAF oncogene activation; increased epidermal growth factor receptor (EGFR) signaling; and deletions or mutations of tumor suppressor genes SMAD4, PTEN, P53. Chromosomal instability (CIN) is a common feature of sporadic disease, but causative factors are not defined. The sequence of molecular events may differ between somatic and inherited genetic alterations. (Data from references 13, 32, 33, and 34.)
TABLE 107-2 Genetic Mutations Associated with Colorectal Cancer
Genomic Instability
Genomic instability plays an integral role in normal colonic or rectal mucosal transformation to carcinoma.34 Three molecular pathways that lead to genomic instability are the microsatellite instability (MSI), CpG island methylator phenotype (CIMP), and chromosomal instability (CIN) pathways. The most common type is CIN, which leads to alterations in chromosomal structure and copy number.32,34 Important consequences of CIN include imbalanced chromosome number (aneuploidy), chromosomal gene amplifications, and loss of a wild-type allele of a tumor-suppressor gene, also referred to as loss of heterozygosity (LOH). Up to 85% of sporadic colorectal cancers exhibit CIN and the tumor suppressor genes APC, P53, and SMAD4 are commonly affected.32–34
Microsatellites are series of repeat nucleotide sequences that are spread out across the entire genome. Microsatellite replication errors within tumor DNA occur frequently, and mutations of the MMR genes that recognize and regulate DNA mismatch-repair errors contribute to MSI and colorectal tumorigenesis.32,34 Germline mutation of MMR genes is an important characteristic of HNPCC but somatic mutations are also present in about 15% of sporadic colorectal cancers.34
Alterations in gene expression or function in the absence of DNA sequence alterations are referred to as epigenetic changes, and these are usually due to methylation of DNA gene promotor regions or histone modifications.34 CIMP is characterized by hypermethylation of a panel of multiple genes that are associated with gene silencing and subsequent loss of tumor suppressor gene function.34 About 15% of sporadic colorectal cancers arise as a consequence of CIMP.
Oncogene and Tumor Suppressor Gene Alterations
Mutation or loss of the APC tumor suppressor gene is a key factor involved in tumor formation through activation of the Wnt signaling pathway, a mediator of cell cycle progression, cell proliferation, differentiation, and apoptosis.10,32,34 The APC gene encodes for APC protein that binds to and degrades cytoplasmic β-catenin, a downstream component of the Wnt signaling pathway. In the absence of functional APC, β-catenin accumulates in the cytoplasm, then enters the nucleus and activates transcription of various genes, leading to constitutive activation of the Wnt signaling pathway. Inactivation of the APC gene is the single gene defect responsible for FAP, and is frequently an initiating event in sporadic colorectal cancer.32
Mutational inactivation of P53 represents a frequent and second key step in colorectal tumorigenesis.32 Normal P53 gene expression is important for G1 cell-cycle arrest to facilitate DNA repair during replication and to induce apoptosis. A third step in tumor progression is the mutational inactivation of the transforming growth factor-β (TGF-β) signaling pathway, which facilitates adenoma transition to high-grade dysplasia or carcinoma and also inactivates SMAD4.32 In normal epithelium, TGF-β has an antiproliferative role and induces growth arrest and apoptosis. Alterations in SMAD4 or TGF-β receptors lead to a loss of the normal growth inhibitory response to TGF-β.
Several oncogene activating mutations play an important role in promoting colorectal cancer.32 Mutations in members of the Ras gene family—KRAS, HRAS, and NRAS—in addition to BRAF, activate the mitogen-activated protein kinase (MAPK) signaling pathway, which stimulates cell proliferation and other activities that promote carcinogenesis. Mutations of PIK3CA, which encodes the catalytic subunit of a PI3K survival pathway, increase production of phosphatidylinositol-3,4,5-triphosphate (PIP3), which influences cell growth, proliferation, and survival.34 Mutation or loss of PTEN, a tumor suppressor gene that antagonizes PI3K signaling, produces similar effects.32,34 Multiple additional genetic alterations contribute to carcinoma formation and metastases by altering cellular growth, metabolism, migration and invasive capabilities, and angiogenesis.34
Growth Factor Signaling Pathways
Aberrant signaling of growth factor pathways plays an important role in colorectal tumorigenesis. Activation of prostaglandin signaling is an early step in the adenoma to carcinoma transformation process and is induced by upregulated expression of COX-2 and inflammation.32 COX-2 mediates the synthesis of prostaglandin E2, which stimulates cancer growth.32 Furthermore, 80% of colorectal cancers have loss of 15-prostaglandin dehydrogenase (15-PGDH), the rate-limiting enzyme responsible for prostaglandin degradation. Gene amplification of the epidermal growth factor receptor (EGFR) gene that encodes for a transmembrane glycoprotein involved in signaling pathways that affect cell growth, differentiation, proliferation, and angiogenesis, is present in 5% to 15% of colorectal cancers.33 EGFR activation enables downstream signaling of the MAPK, PI3K, and Akt pathways that influence colorectal tumorigenesis. EGFR is over-expressed in up to 75% of colorectal cancers and high tumor EGFR overexpression is associated with worse prognosis.35 These mechanisms are relevant because of the availability of pharmacologic agents that can influence these signaling pathways and affect cell growth.
Histology
Adenocarcinomas account for about 94% of tumors of the large intestine.3 Other histologic types such as mucinous adenocarcinoma, mucin-producing adenocarcinoma, signet-ring adenocarcinoma, and neuroendocrine carcinomas occur less frequently. Adenocarcinomas are assigned one of three tumor grade designations based on the degree of cellular differentiation, the degree to which the tumor resembles the structure, and function of its cell of origin. The most differentiated adenocarcinomas are grade I tumors, whereas grade III tumors are considered “high grade,” the most undifferentiated, and have frequently lost the characteristics of mature normal cells. Poorly differentiated tumors are associated with a worse prognosis than those that are relatively better differentiated.36
Mucinous adenocarcinomas possess the same basic structure as adenocarcinomas but differ in that they secrete an abundant quantity of extracellular mucus. They account for only about 10% of colorectal carcinomas but tend to be frequent in patients with MMR mutations.36 Signet-ring adenocarcinomas also have a characteristic appearance but are uncommon. Signet-ring histology occurs more frequently in individuals younger than 50 years of age, patients with ulcerative colitis, and tends to present at a more advanced stage of disease at diagnosis.36 Both mucinous and signet-ring adenocarcinoma histologies confer a poor prognosis.36
PREVENTION AND SCREENING
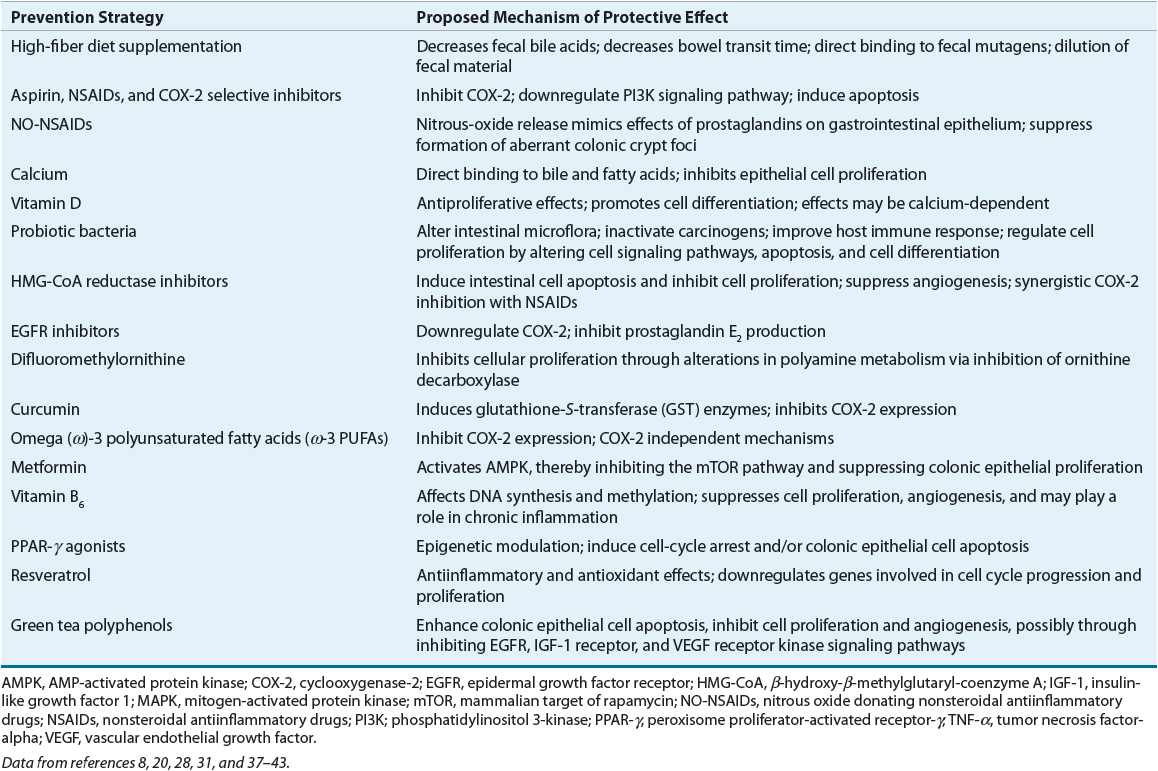
Cancer prevention efforts can be considered as either primary or secondary. Primary prevention strategies aim to prevent the development of colorectal cancer in a population at risk. Secondary prevention approaches are undertaken to prevent malignancy in a population that has already manifested an initial disease process. Several promising primary and secondary prevention strategies are currently undergoing study (Table 107-3).8,18,20,28,37–43
TABLE 107-3 Prevention Strategies for Colorectal Cancer
Diet
![]() Although early studies suggest that a substantial increase in daily dietary fiber or decrease in dietary fat intake might significantly reduce colorectal cancer risk, results from prospective, controlled trials show no protective effects of fiber intake on colorectal adenoma or carcinoma risk. However, a recent meta-analysis suggests a 10% reduction in colorectal cancer risk with 10 g daily intake of total dietary and cereal fiber and up to a 20% risk reduction with three servings of whole grains daily.27,28 There is insufficient evidence to support the use of fiber supplementation as a colorectal cancer prevention strategy at this time.
Although early studies suggest that a substantial increase in daily dietary fiber or decrease in dietary fat intake might significantly reduce colorectal cancer risk, results from prospective, controlled trials show no protective effects of fiber intake on colorectal adenoma or carcinoma risk. However, a recent meta-analysis suggests a 10% reduction in colorectal cancer risk with 10 g daily intake of total dietary and cereal fiber and up to a 20% risk reduction with three servings of whole grains daily.27,28 There is insufficient evidence to support the use of fiber supplementation as a colorectal cancer prevention strategy at this time.
Chemoprevention
![]() The most widely studied agents for the chemoprevention of colorectal cancer are aspirin, nonaspirin NSAIDs, and COX-2 selective inhibitors, but current guidelines do not recommend their use as chemopreventive agents.8,18,20,40 The effectiveness of these agents has been studied in high-risk individuals and within the general population.
The most widely studied agents for the chemoprevention of colorectal cancer are aspirin, nonaspirin NSAIDs, and COX-2 selective inhibitors, but current guidelines do not recommend their use as chemopreventive agents.8,18,20,40 The effectiveness of these agents has been studied in high-risk individuals and within the general population.
In individuals with FAP, celecoxib, NSAIDs, and aspirin have been studied to delay development of adenomatous polyps and to reduce polyp recurrence following colectomy with a retained rectum, but they are not viewed as alternatives to surgery.20 In randomized, controlled trials, celecoxib 400 mg orally twice daily as an adjunct to usual care significantly reduced the mean size and number of colorectal polyps after 6 to 9 months of treatment. However, FDA approval for celecoxib was withdrawn because of lack of data showing long-term benefit. Sulindac has been shown to induce adenoma regression, but does not appear to delay or prevent malignancy. The benefits of these agents are transient, because patients experience an increase in size and number of polyps within a few months after discontinuing treatment. Sulindac is not recommended as chemoprevention for individuals with FAP. These agents may be useful to reduce adenoma recurrence following surgery, but additional data with long-term use are needed.
Nonaspirin NSAIDs and COX-2 inhibitors were associated with reduced risk of sporadic and recurrent colorectal adenomas in cohort and case-control studies, and COX-2 inhibitors were also effective in controlled trials.8 Celecoxib was associated with a 34% relative risk reduction in adenoma recurrence and 55% risk reduction in the incidence of advanced adenomas.20 Optimal dosing, agents, and duration of treatment remain to be determined, and potential cardiovascular events in addition to risk of gastric ulceration and bleeding with these agents are of concern. Although NSAIDs may be appropriate for selected individuals at high risk for colorectal cancer but low risk for cardiovascular disorders, the United States Preventive Services Task Force has concluded that potential harms associated with their use outweigh benefits for prevention of colorectal cancer in the general population.41
Clinical Controversy…
The use of aspirin as both a primary and a secondary chemopreventive agent remains controversial. Aspirin reduces of risk of sporadic and recurrent adenomas by about 17% and advanced adenomas by 28%.20,42 Higher aspirin doses reduced the incidence of colorectal cancer over a 23-year follow-up period by 26% among the general population, but lower doses (75 to 300 mg) of daily aspirin for 5 years was also associated with a risk reduction in colorectal cancer incidence and in 20-year mortality from colorectal cancer by 34%.20,40,42 Individuals with Lynch syndrome who received aspirin 600 mg daily for at least 2 years experienced a 59% reduction in colorectal cancer risk that became evident 5 years after the aspirin was first started and had been discontinued.42 Although the optimal aspirin dose and treatment durations are unknown, increasing evidence supports a chemoprotective effect of aspirin in select high-risk individuals and in the general population. The extent of risk reduction appears to be inversely related to duration of therapy and the chemopreventive effects of aspirin may be delayed by several years. However, the balance of risks and benefits with long-term aspirin use is currently unclear, and aspirin is not recommended for colorectal cancer chemoprevention. PIK3CA mutations, which are present in up to 20% of colorectal cancers, may serve as a biomarker to identify patients diagnosed with colorectal cancer who may benefit from adjuvant aspirin therapy.21
Randomized controlled trials of calcium, vitamin D, and folate supplementation as chemoprevention have also been conducted, but findings do not support their use at this time.8,20,43 Individuals at high risk of colorectal cancer may experience a moderate reduction in risk of recurrent colorectal adenomas with 5 years of calcium supplementation.20 However, individuals with adequate vitamin D levels and no known increased risk of colorectal cancer do not appear to benefit from calcium or vitamin D supplementation. In two trials, folate supplementation was associated with a nonsignificant increase in adenoma recurrence. Based on these results, the use of folate supplementation to reduce colorectal cancer risk is not recommended at this time.8 Several trials of difluoromethylornithine (DFMO), an irreversible inhibitor of the polyamine synthetic pathway, show promising activity as a chemopreventive agent, particular in combinations.8 Additional intervention trials of various micronutrients, epigenetic modulators, and other chemopreventive agents have been completed or are ongoing.8,18,20,28,29,31,37–39
Surgical Resection
Surgical resection remains an option to prevent colon cancer in individuals at extremely high risk for its development. Despite the effects of NSAIDs and COX-2 selective inhibitors on adenoma development and recurrence in individuals with FAP, their effects are incomplete and surgical resection is necessary for cancer prevention for these high-risk individuals. Individuals with FAP who are found to have polyposis on lower endoscopy screening examinations should undergo total proctocolectomy and ileal pouch–anal anastomosis or subtotal colectomy with an ileorectal anastomosis, typically starting around age 20 years.13 Because of the high incidence of metachronous cancers (45%) in patients with HNPCC, prophylactic subtotal colectomy with an ileorectal anastomosis is recommended for those individuals.13 Colonoscopic polypectomy, removal of polyps detected during screening colonoscopy, is considered the standard of care for all individuals to prevent the progression of premalignant adenomatous polyps to adenocarcinomas.
Screening
![]() Colorectal cancer screening decreases mortality by detecting cancers at an early, curable stage, and by detecting and removing adenomatous polyps. Multiple screening recommendations for early detection of colorectal cancer have been established; differences exist in specific screening guidelines published by various organizations.5,44–49 Structural tests detect colorectal polyps and cancer whereas fecal-based tests detect early cancer. This section reviews available screening techniques for colon and rectal cancer.
Colorectal cancer screening decreases mortality by detecting cancers at an early, curable stage, and by detecting and removing adenomatous polyps. Multiple screening recommendations for early detection of colorectal cancer have been established; differences exist in specific screening guidelines published by various organizations.5,44–49 Structural tests detect colorectal polyps and cancer whereas fecal-based tests detect early cancer. This section reviews available screening techniques for colon and rectal cancer.
Colonoscopy
![]() Colonoscopy facilitates examination of the entire large bowel to the cecum in most patients, and allows for simultaneous removal of premalignant lesions. Although no randomized trials show that colonoscopy decreases colorectal cancer mortality, cohort and case control trials demonstrate a 56% to 77% decrease in the incidence in colorectal cancer with colonoscopy and polyp removal and about a 50% reduction in colorectal mortality.46 Although it allows for greater visualization of the colon, colonoscopy involves sedation, complete bowel preparation, and is associated with greater risk and inconvenience to patients. However, it is the preferred screening method based on its superior ability to detect and remove lesions in the proximal as well as distal colon and colonoscopy is therefore considered the gold standard for colorectal screening.45,46
Colonoscopy facilitates examination of the entire large bowel to the cecum in most patients, and allows for simultaneous removal of premalignant lesions. Although no randomized trials show that colonoscopy decreases colorectal cancer mortality, cohort and case control trials demonstrate a 56% to 77% decrease in the incidence in colorectal cancer with colonoscopy and polyp removal and about a 50% reduction in colorectal mortality.46 Although it allows for greater visualization of the colon, colonoscopy involves sedation, complete bowel preparation, and is associated with greater risk and inconvenience to patients. However, it is the preferred screening method based on its superior ability to detect and remove lesions in the proximal as well as distal colon and colonoscopy is therefore considered the gold standard for colorectal screening.45,46
Flexible Sigmoidoscopy
![]() Flexible sigmoidoscopy (FSIG) uses a 40 to 60 cm flexible sigmoidoscope to examine the lower half of the bowel to the splenic flexure for most patients, and is thus capable of detecting 50% to 60% of cancers.44–46 Randomized trials show that FSIG decreases colorectal cancer incidence and mortality by 31% and 38%, respectively.44–46 The combination of FSIG and a fecal-based test appears to improve sensitivity for lesions that will be missed by sigmoidoscopy alone, but the true benefit of this approach to general practice has not been established.45 FSIG offers the advantage of not requiring sedation or extensive bowel preparation, but the entire colon cannot be examined with FSIG and suspicious lesions must be evaluated by colonoscopy.
Flexible sigmoidoscopy (FSIG) uses a 40 to 60 cm flexible sigmoidoscope to examine the lower half of the bowel to the splenic flexure for most patients, and is thus capable of detecting 50% to 60% of cancers.44–46 Randomized trials show that FSIG decreases colorectal cancer incidence and mortality by 31% and 38%, respectively.44–46 The combination of FSIG and a fecal-based test appears to improve sensitivity for lesions that will be missed by sigmoidoscopy alone, but the true benefit of this approach to general practice has not been established.45 FSIG offers the advantage of not requiring sedation or extensive bowel preparation, but the entire colon cannot be examined with FSIG and suspicious lesions must be evaluated by colonoscopy.
Computed Tomography Colonography
![]() Computed tomography colonography (CTC), also referred to as virtual colonoscopy, is an imaging procedure that creates two- or three-dimensional images of the colon by combining multiple helical computed tomography (CT) scans. Initial tests show high sensitivity and specificity for detecting adenomas at least 6 mm in size and sedation is not required.46 However, the procedure requires complete bowel preparation, is associated with radiation exposure, and many individuals will still be referred for colonoscopy to remove detected lesions. Individuals who refuse to undergo invasive colonoscopy or FSIG may find this screening method more acceptable.
Computed tomography colonography (CTC), also referred to as virtual colonoscopy, is an imaging procedure that creates two- or three-dimensional images of the colon by combining multiple helical computed tomography (CT) scans. Initial tests show high sensitivity and specificity for detecting adenomas at least 6 mm in size and sedation is not required.46 However, the procedure requires complete bowel preparation, is associated with radiation exposure, and many individuals will still be referred for colonoscopy to remove detected lesions. Individuals who refuse to undergo invasive colonoscopy or FSIG may find this screening method more acceptable.
Double-Contrast Barium Enema
![]() A double-contrast barium enema (DCBE) involves coating the interior bowel with barium and distending it with air to produce an image of the entire colon in most examinations, and the retained barium outlines small polyps and mucosal lesions. This approach is the least expensive method of examining the entire colon, but is considered inferior to colonoscopy for detecting polyps and colorectal cancer.44,46 In addition, DCBE requires bowel preparation cleaning, is associated with radiation exposure, and a supplemental colonoscopy is required if suspicious lesions are identified. However, DCBE is considered an alternative for individuals who do not wish to undergo or are not suitable for colonoscopy.
A double-contrast barium enema (DCBE) involves coating the interior bowel with barium and distending it with air to produce an image of the entire colon in most examinations, and the retained barium outlines small polyps and mucosal lesions. This approach is the least expensive method of examining the entire colon, but is considered inferior to colonoscopy for detecting polyps and colorectal cancer.44,46 In addition, DCBE requires bowel preparation cleaning, is associated with radiation exposure, and a supplemental colonoscopy is required if suspicious lesions are identified. However, DCBE is considered an alternative for individuals who do not wish to undergo or are not suitable for colonoscopy.
Fecal Occult Blood Tests
![]() Fecal occult blood tests (FOBTs) are used to detect occult blood in the stool that may be associated with bleeding adenomas or cancer. Results from randomized, controlled trials of annual FOBT screening show a reduction in colorectal cancer mortality by 33%.44–46 Unlike structural tests, FOBTs are noninvasive and do not require bowel preparation. Two main methods are available to detect occult blood in the feces: guaiac-based FOBT (gFOBT) and fecal immunochemical tests (FITs), that is, the immunochemical fecal occult blood test (iFOBT). Several guaiac-based tests are available that detect peroxidase activity of heme when hemoglobin comes in contact with a guaiac-impregnated paper. When a solution containing hydrogen peroxide is poured over the paper, a blue color appears if the test is positive. The testing process is complex and requires specific patient counseling to avoid inaccurate results (Table 107-4).
Fecal occult blood tests (FOBTs) are used to detect occult blood in the stool that may be associated with bleeding adenomas or cancer. Results from randomized, controlled trials of annual FOBT screening show a reduction in colorectal cancer mortality by 33%.44–46 Unlike structural tests, FOBTs are noninvasive and do not require bowel preparation. Two main methods are available to detect occult blood in the feces: guaiac-based FOBT (gFOBT) and fecal immunochemical tests (FITs), that is, the immunochemical fecal occult blood test (iFOBT). Several guaiac-based tests are available that detect peroxidase activity of heme when hemoglobin comes in contact with a guaiac-impregnated paper. When a solution containing hydrogen peroxide is poured over the paper, a blue color appears if the test is positive. The testing process is complex and requires specific patient counseling to avoid inaccurate results (Table 107-4).
TABLE 107-4 Patient Counseling Points Prior to Guaiac-Based Stool Tests
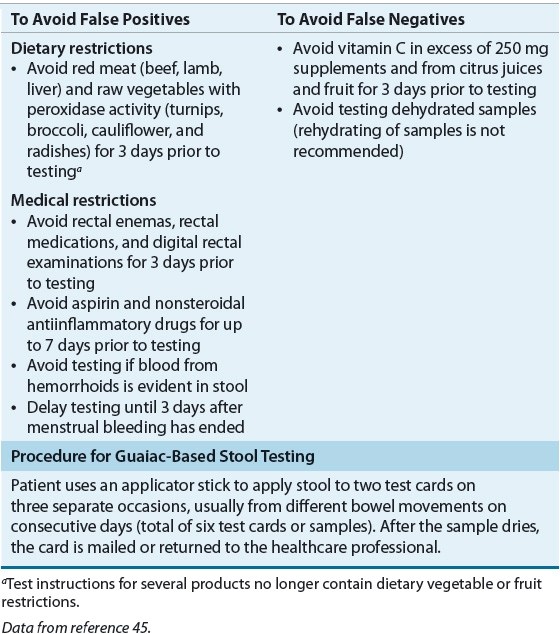
Clinical guidelines have been developed for performing and interpreting results of gFOBT.45 Several limitations associated with FOBT screening are of concern. Many early-stage tumors do not bleed, and therefore the false-negative rates are about 70% for cancer and 90% for polyps. In addition, the test results may not be valid because the test is often poorly performed both in the home and in physician office settings.45,46 However, these concerns are addressed by testing three successive stool samples. False-positive results can prove to be very expensive and inconvenient for a patient because of the follow-up tests required to confirm a positive result. Annual screening, preferably using a high-sensitivity gFOBT (e.g., Hemoccult SENSA), is an acceptable option for individuals at average risk for colorectal cancer. It should be noted that FOBT conducted in conjunction with a digital rectal exam during an office visit is not considered adequate colorectal screening.
FITs (iFOBTs) were developed to reduce false-positive and false-negative test results associated with the gFOBT. FIT uses antibodies to detect the globin protein portion of human hemoglobin. Globin is degraded by enzymes in the upper gastrointestinal tract; therefore, FIT is more specific for lower gastrointestinal bleeding. Also, immunochemical tests do not produce false-negative results in the presence of vitamin C.45 Moreover, testing involves a single stool sample collection annually. Comparative studies report that FIT is more accurate than gFOBT for detecting cancer and advanced adenomas, although colonoscopy identifies more adenomas.50
Stool DNA Screening Tests
Molecular screening strategies analyze stool samples for presence of potential markers of malignancy in cells that are shed from pre-malignant polyps or adenocarcinomas in the bowel.44–46 Adenoma and carcinomas can contain certain DNA mutations and markers of MSI that can be detected using a multiple marker panel for stool DNA (sDNA) testing. However, no FDA-approved sDNA tests are currently commercially available.46
Screening Summary
![]() Table 107-5 outlines current U.S. screening guidelines for early detection of colorectal cancer with the goal of cancer prevention. Men and women who are at average risk for colorectal cancer (their only risk factor is age ≥50 years) should begin regular screening starting at age 50 years with a colonoscopy every 10 years, annually using a sensitive gFOBT or FIT, or undergo FSIG every 5 years, alone or in conjunction with annual FOBT. Several screening methods are available, and because each method is associated with different benefits and potential harms, patient preferences and available resources should be considered for individual patients.45 More rigorous (usually starting at an earlier age) screening recommendations are given for moderate- to high-risk individuals and colonoscopy is generally preferred for initial screening and surveillance following polyp removal in this population.5,45,46,49 Most organizations recommend discontinuing screening and surveillance in populations when risk may outweigh benefit.5 The United States Preventive Services Task Force (USPSTF) recommends routine colorectal cancer screening for individuals age 50 to 75 years with different consideration given to adults 76 to 85 years and recommends against screening for adults older than 85 years.5 The American College of Physicians recommends against screening adults older than age 75 years or with a life expectancy of less than 10 years.44
Table 107-5 outlines current U.S. screening guidelines for early detection of colorectal cancer with the goal of cancer prevention. Men and women who are at average risk for colorectal cancer (their only risk factor is age ≥50 years) should begin regular screening starting at age 50 years with a colonoscopy every 10 years, annually using a sensitive gFOBT or FIT, or undergo FSIG every 5 years, alone or in conjunction with annual FOBT. Several screening methods are available, and because each method is associated with different benefits and potential harms, patient preferences and available resources should be considered for individual patients.45 More rigorous (usually starting at an earlier age) screening recommendations are given for moderate- to high-risk individuals and colonoscopy is generally preferred for initial screening and surveillance following polyp removal in this population.5,45,46,49 Most organizations recommend discontinuing screening and surveillance in populations when risk may outweigh benefit.5 The United States Preventive Services Task Force (USPSTF) recommends routine colorectal cancer screening for individuals age 50 to 75 years with different consideration given to adults 76 to 85 years and recommends against screening for adults older than 85 years.5 The American College of Physicians recommends against screening adults older than age 75 years or with a life expectancy of less than 10 years.44
TABLE 107-5 Guidelines for Colorectal Cancer Screening in the United States for Individuals at Average Risk, 50 Years of Age and Older

DIAGNOSIS
Signs and Symptoms
The signs and symptoms associated with colorectal cancer can be extremely varied and nonspecific. Patients with early-stage colorectal cancer are often asymptomatic, and lesions are usually found as a result of screening studies. Any change in bowel habits (e.g., constipation, diarrhea, or alteration in size or shape of stool), abdominal pain, or distension may all be warning signs of a malignant process. Obstructive symptoms and changes in bowel habits frequently develop with tumors located in the transverse and descending colon. Bleeding is the most common symptom of rectal cancer. Bleeding may be acute or chronic and can appear as bright red blood mixed with stool or melena. Iron-deficiency anemia, presenting as weakness and fatigue, frequently develops as a result of chronic occult blood loss.
About 20% of patients with colorectal cancer present with metastatic disease.3 Metastatic spread occurs as a result of direct tumor invasion of adjacent tissues or by lymphatic or hematogenous spread. The venous drainage of the colon and rectum influences the pattern of metastases most commonly seen. The most common site of metastasis is the liver, often the only site of metastatic disease in 40% of patients, followed by the lungs and then bones, specifically the sacrum, coccyx, pelvis, and lumbar vertebrae. Liver metastases are present in 5% to 10% of patients at presentation.
Workup
When a patient is suspected of having colorectal carcinoma, a complete history and physical examination should be performed. The patient history should include a past medical history and family history, especially noting the presence of inflammatory bowel disease, colorectal cancer, polyps, and familial clustering of cancers to assess risk for an inherited colorectal cancer syndrome. A complete physical examination includes careful abdominal examination for the presence of masses or ascites, a rectal examination, and an assessment for possible hepatomegaly and lymphadenopathy. A breast and pelvic examination is recommended in all women.
An evaluation of the entire large bowel requires a total colonoscopy and allows for tissue collection for a histologic evaluation to provide a preliminary diagnosis following the procedure. Patients with invasive cancer of the colon or rectum require a complete staging workup with laboratory testing and CT scans of the abdomen, pelvis, and chest. Baseline laboratory tests should be obtained and include a complete blood cell count, platelet count, international normalized ratio (INR), prothrombin time, activated partial thromboplastin time, liver chemistries, renal function tests, and carcinoembryonic antigen (CEA) level. Abnormal liver chemistry test results may suggest liver involvement with tumor. However, patients with metastatic disease to the liver may have normal liver chemistries, and abnormal liver test results are not always indicative of metastatic disease. Iron studies (e.g., serum ferritin, serum iron, and total iron-binding capacity) may be useful to identify iron-deficiency in patients with anemia.
CEA belongs to a group of cell-surface glycoproteins termed oncofetal proteins, which are expressed during embryonic development and reexpressed on the cell surfaces of many carcinomas, particularly those originating from the gastrointestinal tract. CEA concentrations can be measured in the blood and can therefore potentially serve as a marker for colorectal cancer. Elevated CEA levels are more frequent in patients with metastatic disease but not all colorectal cancers produce CEA. It is important to recognize, however, that several concomitant disease states are associated with an elevated CEA: liver diseases, gastritis, peptic ulcer disease, diverticulitis, chronic obstructive pulmonary disease, chronic or acute inflammatory conditions, and diabetes.51 Most commercially available assays list a value of less than 5 ng/mL as the upper limit of normal. Although CEA measurement is too insensitive and nonspecific to be used as a screening test for early-stage colorectal cancer, it is the surrogate marker of choice for monitoring colorectal cancer response to treatment, particularly if the pretreatment concentration is elevated.51 The CEA test may have preoperative prognostic implications because it has been shown to correlate with the size and degree of differentiation of the carcinoma. Elevated preoperative CEA levels correlate with a poor survival and may predict likelihood of recurrence, regardless of tumor stage at diagnosis. However, it should not be used as an indication for adjuvant therapy. After a potentially curative resection, CEA levels should return to normal within 4 to 6 weeks. Persistently elevated CEA levels may indicate residual disease, while elevations after normalization may indicate relapsed disease.



