Collagenous Spherulosis
Key Facts
Terminology
Collagenous (mucinous) spherulosis (CS)
Incidental microscopic finding in 1-2% of biopsies that contain hyperplastic ductal lesions
Important to recognize CS because lesion may superficially resemble cribriform DCIS
Clinical Issues
Typically seen in breast tissue containing sclerosing lesions
Calcification of CS can lead to mammographic detection and diagnosis by needle core biopsy
Benign process; no evidence that CS is directly associated with precancerous lesions or is risk factor for breast cancer
Microscopic Pathology
CS is characterized by eosinophilic fibrillar &/or hyaline spherules consisting of basement membrane-like material (polysaccharides, laminin, and type IV collagen) within areas of epithelial hyperplasia
In foci of CS, myoepithelial cells are present around periphery and also surround inner spherules
Immunohistochemical studies to demonstrate presence of myoepithelial cells around cribriform spaces and associated with spherules can be helpful
Top Differential Diagnoses
Low-grade cribriform DCIS
CS involved by LCIS can be difficult to distinguish from cribriform DCIS
Low-grade adenoid cystic carcinoma
TERMINOLOGY
Abbreviations
Collagenous spherulosis (CS)
Synonyms
Mucinous spherulosis
Definitions
Incidental microscopic finding in 1-2% of biopsies that contain hyperplastic ductal lesions
Important to recognize CS because lesion may superficially resemble cribriform DCIS
CS may also superficially resemble low-grade adenoid cystic carcinoma
CLINICAL ISSUES
Presentation
Incidental finding
Typically seen in breast tissue containing other sclerosing benign proliferative lesions
Includes sclerosing adenosis, papillomas, ductal hyperplasia, and atypical ductal hyperplasia
Calcification of CS can lead to mammographic detection and diagnosis by needle core biopsy
Treatment
CS may be seen associated with atypical ductal hyperplasia, LCIS, or DCIS
Appropriate treatment is related to other lesions present
Prognosis
Benign process
No evidence that CS is directly associated with precancerous lesions or predisposes to future development of breast cancer
IMAGE FINDINGS
Mammographic Findings
CS may be detected mammographically for those lesions with calcifications
MICROSCOPIC PATHOLOGY
Histologic Features
CS characterized by lobulocentric aggregates of eosinophilic fibrillar &/or hyaline spherules of extracellular material
Spherules composed of varying amounts of basement membrane-like material, including polysaccharides, laminin, and type IV collagen
Positive staining for PAS and Alcian blue by histochemistry
In some cases, spherules contain mucoid-like material (“mucinous spherulosis”)
Spherules surrounded by inner myoepithelial layer and outer luminal layer of cells
Myoepithelial cells may become attenuated and difficult to appreciate in H&E sections
Immunohistochemical stains for myoepithelial cells (calponin, p63, myosin heavy chain) to highlight myoepithelial cells may be helpful
Spherules and cellular arrangement give rise to appearance of cribriform or fenestrated proliferation when viewed at low power
DIFFERENTIAL DIAGNOSIS
Low-Grade Cribriform DCIS
Microlumens formed and surrounded by single monotonous population of neoplastic epithelial cells
Nuclei of cells tend to stand apart and appear polarized around microlumens
Gives rise to a rigid or “punched-out” appearance due to microlumens with round or smooth internal contours
Microluminal spaces may be empty, contain secretions, necrotic cells, or calcifications
Stains to highlight myoepithelial component of CS may be helpful in difficult cases
Low-Grade Adenoid Cystic Carcinoma
Typically demonstrates invasive growth pattern at periphery with tumor infiltrating beyond central gross nodule
Tumor is composed of 2 different components
True glandular component (formed by luminal-type cells)
“Pseudoglandular” component consisting of basement membrane deposition surrounded by myoepithelial-like cells
Different components typically show heterogeneity in distribution throughout tumor
Higher grade lesions typically show areas with solid growth pattern
Adenoid cystic carcinomas may have luminal cells positive for C-Kit (CD117) whereas cells of CS are negative for this marker
DIAGNOSTIC CHECKLIST
Clinically Relevant Pathologic Features
Tissue distribution
Ductal or lobular process only
May involve intraductal papillomas
Most often seen in association with sclerosing adenosis, ductal hyperplasia, and papillomas
Usually incidental finding
Important to recognize because it can mimic other more serious processes
Rarely CS can be involved by lobular neoplasia
Neoplastic lobular cells displace indigenous luminal cells, leaving myoepithelium and spherules in place
May be difficult to distinguish from cribriform DCIS
Presence of basement membrane material and dyshesive quality of lobular cells may be helpful
Immunostains for E-cadherin and myoepithelial cells can identify 2 cell types
Pathologic Interpretation Pearls
Cribriform or fenestrated proliferation
Luminal spaces tend to have irregular or angulated shapes
Look for acellular spherules of basement membrane-like material within luminal spaces
Look for attenuated myoepithelial cells surrounding luminal spaces admixed with luminal cells
Immunostains can help to highlight different cell types
CS typically associated with other sclerosing and benign proliferative lesions
SELECTED REFERENCES
1. Hill P et al: Collagenous spherulosis with lobular carcinoma in situ: a potential diagnostic pitfall. Pathology. 39(3):361-3, 2007
2. Rabban JT et al: Immunophenotypic overlap between adenoid cystic carcinoma and collagenous spherulosis of the breast: potential diagnostic pitfalls using myoepithelial markers. Mod Pathol. 19(10):1351-7, 2006
3. Resetkova E et al: Collagenous spherulosis of breast: morphologic study of 59 cases and review of the literature. Am J Surg Pathol. 30(1):20-7, 2006
4. Mooney EE et al: Spherulosis of the breast. A spectrum of municous and collagenous lesions. Arch Pathol Lab Med. 123(7):626-30, 1999
5. Sgroi D et al: Involvement of collagenous spherulosis by lobular carcinoma in situ. Potential confusion with cribriform ductal carcinoma in situ. Am J Surg Pathol. 19(12):1366-70, 1995
6. Stephenson TJ et al: Nodular basement membrane deposits in breast carcinoma and atypical ductal hyperplasia: mimics of collagenous spherulosis. Pathologica. 86(3):234-9, 1994
7. Clement PB et al: Collagenous spherulosis of the breast. Am J Surg Pathol. 11(6):411-7, 1987
Image Gallery
Microscopic Features
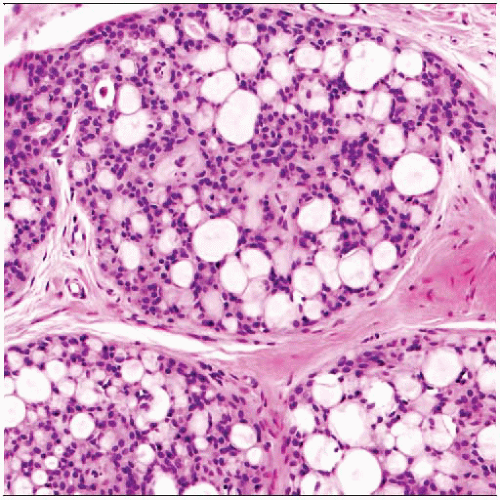
 (Left) Collagenous spherulosis
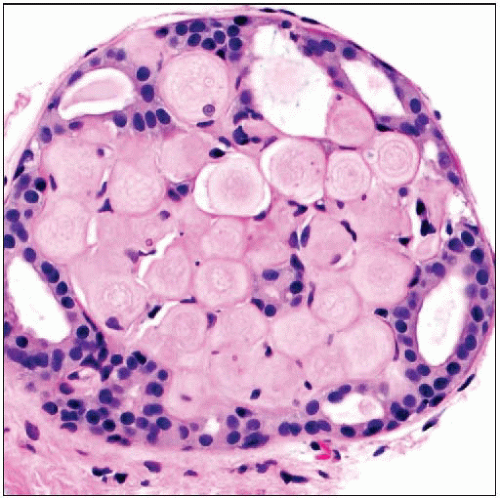
 is a benign sclerosing process, typically seen with other benign sclerosing lesions. This example is associated with florid sclerosing adenosis is a benign sclerosing process, typically seen with other benign sclerosing lesions. This example is associated with florid sclerosing adenosis  . The lesion gives the appearance of an intraductal proliferation; however, studies of the earliest phases suggest that the process usually affects lobules. (Right) A higher power view shows the typical fenestrated or cribriform appearance of the epithelium surrounding eosinophilic hyaline material . The lesion gives the appearance of an intraductal proliferation; however, studies of the earliest phases suggest that the process usually affects lobules. (Right) A higher power view shows the typical fenestrated or cribriform appearance of the epithelium surrounding eosinophilic hyaline material  . .Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|