PART 2: Cardinal Manifestations and Presentation of Diseases
SECTION 1 PAIN
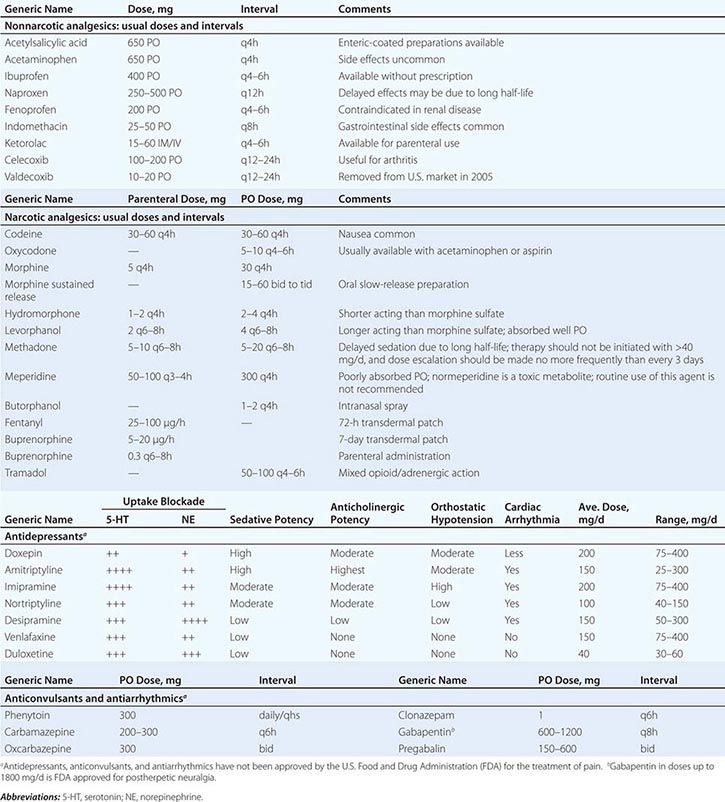
18 | Pain: Pathophysiology and Management |
The province of medicine is to preserve and restore health and to relieve suffering. Understanding pain is essential to both of these goals. Because pain is universally understood as a signal of disease, it is the most common symptom that brings a patient to a physician’s attention. The function of the pain sensory system is to protect the body and maintain homeostasis. It does this by detecting, localizing, and identifying potential or actual tissue-damaging processes. Because different diseases produce characteristic patterns of tissue damage, the quality, time course, and location of a patient’s pain lend important diagnostic clues. It is the physician’s responsibility to provide rapid and effective pain relief.
THE PAIN SENSORY SYSTEM
Pain is an unpleasant sensation localized to a part of the body. It is often described in terms of a penetrating or tissue-destructive process (e.g., stabbing, burning, twisting, tearing, squeezing) and/or of a bodily or emotional reaction (e.g., terrifying, nauseating, sickening). Furthermore, any pain of moderate or higher intensity is accompanied by anxiety and the urge to escape or terminate the feeling. These properties illustrate the duality of pain: it is both sensation and emotion. When it is acute, pain is characteristically associated with behavioral arousal and a stress response consisting of increased blood pressure, heart rate, pupil diameter, and plasma cortisol levels. In addition, local muscle contraction (e.g., limb flexion, abdominal wall rigidity) is often present.
PERIPHERAL MECHANISMS
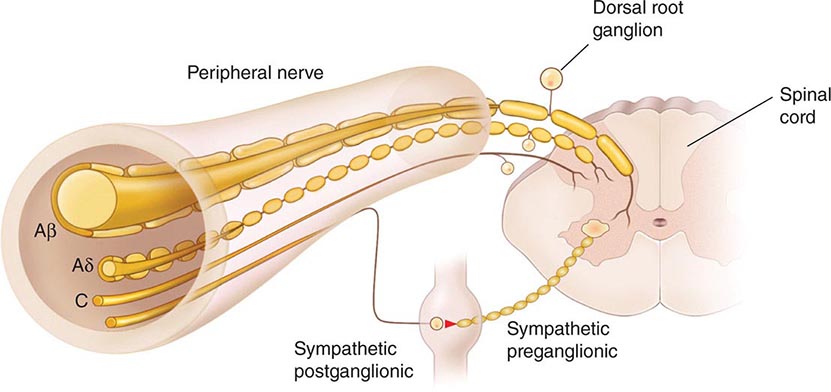
The Primary Afferent Nociceptor A peripheral nerve consists of the axons of three different types of neurons: primary sensory afferents, motor neurons, and sympathetic postganglionic neurons (Fig. 18-1). The cell bodies of primary sensory afferents are located in the dorsal root ganglia within the vertebral foramina. The primary afferent axon has two branches: one projects centrally into the spinal cord and the other projects peripherally to innervate tissues. Primary afferents are classified by their diameter, degree of myelination, and conduction velocity. The largest diameter afferent fibers, A-beta (Aβ), respond maximally to light touch and/or moving stimuli; they are present primarily in nerves that innervate the skin. In normal individuals, the activity of these fibers does not produce pain. There are two other classes of primary afferent nerve fibers: the small diameter myelinated A-delta (Aδ) and the unmyelinated (C) axons (Fig. 18-1). These fibers are present in nerves to the skin and to deep somatic and visceral structures. Some tissues, such as the cornea, are innervated only by Aδ and C fiber afferents. Most Aδ and C fiber afferents respond maximally only to intense (painful) stimuli and produce the subjective experience of pain when they are electrically stimulated; this defines them as primary afferent nociceptors (pain receptors). The ability to detect painful stimuli is completely abolished when conduction in Aδ and C fiber axons is blocked.
FIGURE 18-1 Components of a typical cutaneous nerve. There are two distinct functional categories of axons: primary afferents with cell bodies in the dorsal root ganglion, and sympathetic postganglionic fibers with cell bodies in the sympathetic ganglion. Primary afferents include those with large-diameter myelinated (Aβ), small-diameter myelinated (Aδ), and unmyelinated (C) axons. All sympathetic postganglionic fibers are unmyelinated.
Individual primary afferent nociceptors can respond to several different types of noxious stimuli. For example, most nociceptors respond to heat; intense cold; intense mechanical distortion, such as a pinch; changes in pH, particularly an acidic environment; and application of chemical irritants including adenosine triphosphate (ATP), serotonin, bradykinin, and histamine.
Sensitization When intense, repeated, or prolonged stimuli are applied to damaged or inflamed tissues, the threshold for activating primary afferent nociceptors is lowered, and the frequency of firing is higher for all stimulus intensities. Inflammatory mediators such as bradykinin, nerve-growth factor, some prostaglandins, and leukotrienes contribute to this process, which is called sensitization. Sensitization occurs at the level of the peripheral nerve terminal (peripheral sensitization) as well as at the level of the dorsal horn of the spinal cord (central sensitization). Peripheral sensitization occurs in damaged or inflamed tissues, when inflammatory mediators activate intracellular signal transduction in nociceptors, prompting an increase in the production, transport, and membrane insertion of chemically gated and voltage-gated ion channels. These changes increase the excitability of nociceptor terminals and lower their threshold for activation by mechanical, thermal, and chemical stimuli. Central sensitization occurs when activity, generated by nociceptors during inflammation, enhances the excitability of nerve cells in the dorsal horn of the spinal cord. Following injury and resultant sensitization, normally innocuous stimuli can produce pain (termed allodynia). Sensitization is a clinically important process that contributes to tenderness, soreness, and hyperalgesia (increased pain intensity in response to the same noxious stimulus; e.g., moderate pressure causes severe pain). A striking example of sensitization is sunburned skin, in which severe pain can be produced by a gentle slap on the back or a warm shower.
Sensitization is of particular importance for pain and tenderness in deep tissues. Viscera are normally relatively insensitive to noxious mechanical and thermal stimuli, although hollow viscera do generate significant discomfort when distended. In contrast, when affected by a disease process with an inflammatory component, deep structures such as joints or hollow viscera characteristically become exquisitely sensitive to mechanical stimulation.
A large proportion of Aδ and C fiber afferents innervating viscera are completely insensitive in normal noninjured, noninflamed tissue. That is, they cannot be activated by known mechanical or thermal stimuli and are not spontaneously active. However, in the presence of inflammatory mediators, these afferents become sensitive to mechanical stimuli. Such afferents have been termed silent nociceptors, and their characteristic properties may explain how, under pathologic conditions, the relatively insensitive deep structures can become the source of severe and debilitating pain and tenderness. Low pH, prostaglandins, leukotrienes, and other inflammatory mediators such as bradykinin play a significant role in sensitization.
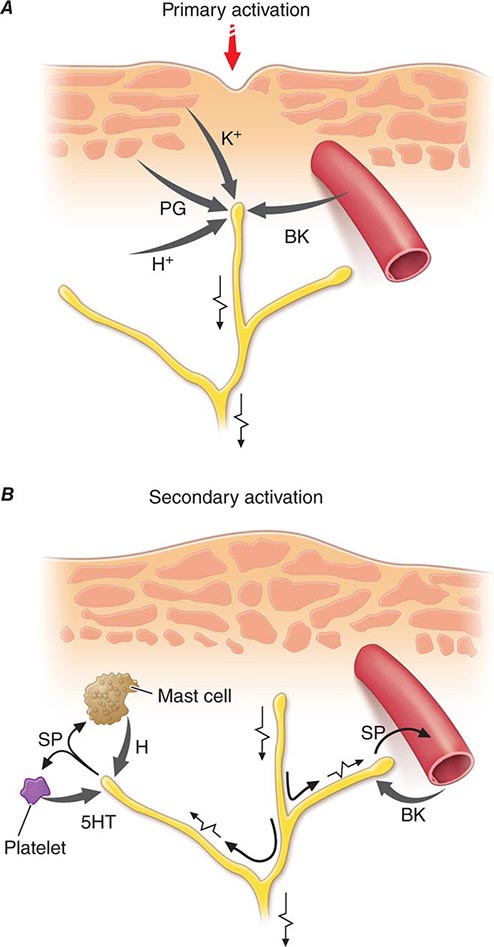
Nociceptor-Induced Inflammation Primary afferent nociceptors also have a neuroeffector function. Most nociceptors contain polypeptide mediators that are released from their peripheral terminals when they are activated (Fig. 18-2). An example is substance P, an 11-amino-acid peptide. Substance P is released from primary afferent nociceptors and has multiple biologic activities. It is a potent vasodilator, degranulates mast cells, is a chemoattractant for leukocytes, and increases the production and release of inflammatory mediators. Interestingly, depletion of substance P from joints reduces the severity of experimental arthritis. Primary afferent nociceptors are not simply passive messengers of threats to tissue injury but also play an active role in tissue protection through these neuroeffector functions.
FIGURE 18-2 Events leading to activation, sensitization, and spread of sensitization of primary afferent nociceptor terminals. A. Direct activation by intense pressure and consequent cell damage. Cell damage induces lower pH (H+) and leads to release of potassium (K+) and to synthesis of prostaglandins (PG) and bradykinin (BK). Prostaglandins increase the sensitivity of the terminal to bradykinin and other pain-producing substances. B. Secondary activation. Impulses generated in the stimulated terminal propagate not only to the spinal cord but also into other terminal branches where they induce the release of peptides, including substance P (SP). Substance P causes vasodilation and neurogenic edema with further accumulation of bradykinin (BK). Substance P also causes the release of histamine (H) from mast cells and serotonin (5HT) from platelets.
CENTRAL MECHANISMS
The Spinal Cord and Referred Pain The axons of primary afferent nociceptors enter the spinal cord via the dorsal root. They terminate in the dorsal horn of the spinal gray matter (Fig. 18-3). The terminals of primary afferent axons contact spinal neurons that transmit the pain signal to brain sites involved in pain perception. When primary afferents are activated by noxious stimuli, they release neurotransmitters from their terminals that excite the spinal cord neurons. The major neurotransmitter released is glutamate, which rapidly excites dorsal horn neurons. Primary afferent nociceptor terminals also release peptides, including substance P and calcitonin gene-related peptide, which produce a slower and longer-lasting excitation of the dorsal horn neurons. The axon of each primary afferent contacts many spinal neurons, and each spinal neuron receives convergent inputs from many primary afferents.
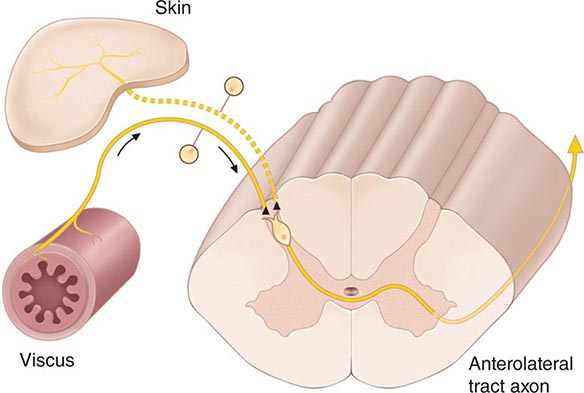
FIGURE 18-3 The convergence-projection hypothesis of referred pain. According to this hypothesis, visceral afferent nociceptors converge on the same pain-projection neurons as the afferents from the somatic structures in which the pain is perceived. The brain has no way of knowing the actual source of input and mistakenly “projects” the sensation to the somatic structure.
The convergence of sensory inputs to a single spinal pain-transmission neuron is of great importance because it underlies the phenomenon of referred pain. All spinal neurons that receive input from the viscera and deep musculoskeletal structures also receive input from the skin. The convergence patterns are determined by the spinal segment of the dorsal root ganglion that supplies the afferent innervation of a structure. For example, the afferents that supply the central diaphragm are derived from the third and fourth cervical dorsal root ganglia. Primary afferents with cell bodies in these same ganglia supply the skin of the shoulder and lower neck. Thus, sensory inputs from both the shoulder skin and the central diaphragm converge on pain-transmission neurons in the third and fourth cervical spinal segments. Because of this convergence and the fact that the spinal neurons are most often activated by inputs from the skin, activity evoked in spinal neurons by input from deep structures is mislocalized by the patient to a place that roughly corresponds with the region of skin innervated by the same spinal segment. Thus, inflammation near the central diaphragm is often reported as shoulder discomfort. This spatial displacement of pain sensation from the site of the injury that produces it is known as referred pain.
Ascending Pathways for Pain A majority of spinal neurons contacted by primary afferent nociceptors send their axons to the contralateral thalamus. These axons form the contralateral spinothalamic tract, which lies in the anterolateral white matter of the spinal cord, the lateral edge of the medulla, and the lateral pons and midbrain. The spinothalamic pathway is crucial for pain sensation in humans. Interruption of this pathway produces permanent deficits in pain and temperature discrimination.
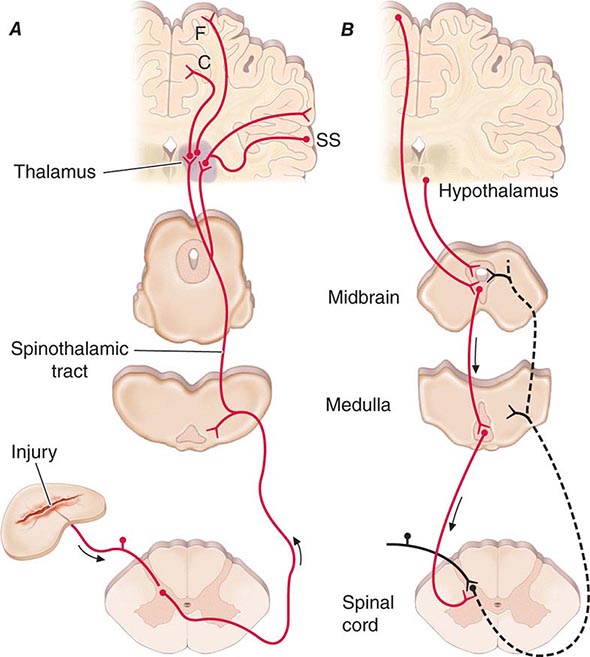
Spinothalamic tract axons ascend to several regions of the thalamus. There is tremendous divergence of the pain signal from these thalamic sites to several distinct areas of the cerebral cortex that subserve different aspects of the pain experience (Fig. 18-4). One of the thalamic projections is to the somatosensory cortex. This projection mediates the purely sensory aspects of pain, i.e., its location, intensity, and quality. Other thalamic neurons project to cortical regions that are linked to emotional responses, such as the cingulate gyrus and other areas of the frontal lobes, including the insular cortex. These pathways to the frontal cortex subserve the affective or unpleasant emotional dimension of pain. This affective dimension of pain produces suffering and exerts potent control of behavior. Because of this dimension, fear is a constant companion of pain. As a consequence, injury or surgical lesions to areas of the frontal cortex activated by painful stimuli can diminish the emotional impact of pain while largely preserving the individual’s ability to recognize noxious stimuli as painful.
FIGURE 18-4 Pain transmission and modulatory pathways. A. Transmission system for nociceptive messages. Noxious stimuli activate the sensitive peripheral ending of the primary afferent nociceptor by the process of transduction. The message is then transmitted over the peripheral nerve to the spinal cord, where it synapses with cells of origin of the major ascending pain pathway, the spinothalamic tract. The message is relayed in the thalamus to the anterior cingulate (C), frontal insular (F), and somatosensory cortex (SS). B. Pain-modulation network. Inputs from frontal cortex and hypothalamus activate cells in the midbrain that control spinal pain-transmission cells via cells in the medulla.
PAIN MODULATION
The pain produced by injuries of similar magnitude is remarkably variable in different situations and in different individuals. For example, athletes have been known to sustain serious fractures with only minor pain, and Beecher’s classic World War II survey revealed that many soldiers in battle were unbothered by injuries that would have produced agonizing pain in civilian patients. Furthermore, even the suggestion that a treatment will relieve pain can have a significant analgesic effect (the placebo effect). On the other hand, many patients find even minor injuries (such as venipuncture) frightening and unbearable, and the expectation of pain can induce pain even without a noxious stimulus. The suggestion that pain will worsen following administration of an inert substance can increase its perceived intensity (the nocebo effect).
The powerful effect of expectation and other psychological variables on the perceived intensity of pain is explained by brain circuits that modulate the activity of the pain-transmission pathways. One of these circuits has links to the hypothalamus, midbrain, and medulla, and it selectively controls spinal pain-transmission neurons through a descending pathway (Fig. 18-4).
Human brain–imaging studies have implicated this pain-modulating circuit in the pain-relieving effect of attention, suggestion, and opioid analgesic medications (Fig. 18-5). Furthermore, each of the component structures of the pathway contains opioid receptors and is sensitive to the direct application of opioid drugs. In animals, lesions of this descending modulatory system reduce the analgesic effect of systemically administered opioids such as morphine. Along with the opioid receptor, the component nuclei of this pain-modulating circuit contain endogenous opioid peptides such as the enkephalins and β-endorphin.
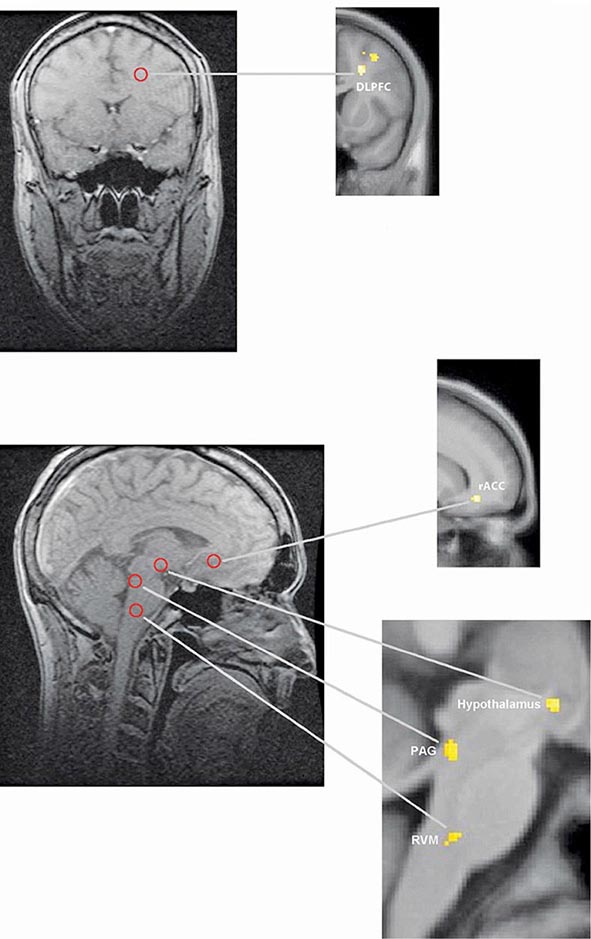
FIGURE 18-5 Functional magnetic resonance imaging (fMRI) demonstrates placebo-enhanced brain activity in anatomic regions correlating with the opioidergic descending pain control system. Top panel: Frontal fMRI image shows placebo-enhanced brain activity in the dorsal lateral prefrontal cortex (DLPFC). Bottom panel: Sagittal fMRI images show placebo-enhanced responses in the rostral anterior cingulate cortex (rACC), the rostral ventral medullae (RVM), the periaqueductal gray (PAG) area, and the hypothalamus. The placebo-enhanced activity in all areas was reduced by naloxone, demonstrating the link between the descending opioidergic system and the placebo analgesic response. (Adapted with permission from F Eippert et al: Neuron 63:533, 2009.)
The most reliable way to activate this endogenous opioid-mediated modulating system is by suggestion of pain relief or by intense emotion directed away from the pain-causing injury (e.g., during severe threat or an athletic competition). In fact, pain-relieving endogenous opioids are released following surgical procedures and in patients given a placebo for pain relief.
Pain-modulating circuits can enhance as well as suppress pain. Both pain-inhibiting and pain-facilitating neurons in the medulla project to and control spinal pain-transmission neurons. Because pain-transmission neurons can be activated by modulatory neurons, it is theoretically possible to generate a pain signal with no peripheral noxious stimulus. In fact, human functional imaging studies have demonstrated increased activity in this circuit during migraine headaches. A central circuit that facilitates pain could account for the finding that pain can be induced by suggestion or enhanced by expectation and provides a framework for understanding how psychological factors can contribute to chronic pain.
NEUROPATHIC PAIN
Lesions of the peripheral or central nociceptive pathways typically result in a loss or impairment of pain sensation. Paradoxically, damage to or dysfunction of these pathways can also produce pain. For example, damage to peripheral nerves, as occurs in diabetic neuropathy, or to primary afferents, as in herpes zoster infection, can result in pain that is referred to the body region innervated by the damaged nerves. Pain may also be produced by damage to the central nervous system (CNS), for example, in some patients following trauma or vascular injury to the spinal cord, brainstem, or thalamic areas that contain central nociceptive pathways. Such neuropathic pains are often severe and are typically resistant to standard treatments for pain.
Neuropathic pain typically has an unusual burning, tingling, or electric shock–like quality and may be triggered by very light touch. These features are rare in other types of pain. On examination, a sensory deficit is characteristically present in the area of the patient’s pain. Hyperpathia, a greatly exaggerated pain sensation to innocuous or mild nociceptive stimuli, is also characteristic of neuropathic pain; patients often complain that the very lightest moving stimulus evokes exquisite pain (allodynia). In this regard, it is of clinical interest that a topical preparation of 5% lidocaine in patch form is effective for patients with postherpetic neuralgia who have prominent allodynia.
A variety of mechanisms contribute to neuropathic pain. As with sensitized primary afferent nociceptors, damaged primary afferents, including nociceptors, become highly sensitive to mechanical stimulation and may generate impulses in the absence of stimulation. Increased sensitivity and spontaneous activity are due, in part, to an increased concentration of sodium channels in the damaged nerve fiber. Damaged primary afferents may also develop sensitivity to norepinephrine. Interestingly, spinal cord pain-transmission neurons cut off from their normal input may also become spontaneously active. Thus, both CNS and peripheral nervous system hyperactivity contribute to neuropathic pain.
Sympathetically Maintained Pain Patients with peripheral nerve injury occasionally develop spontaneous pain in the region innervated by the nerve. This pain is often described as having a burning quality. The pain typically begins after a delay of hours to days or even weeks and is accompanied by swelling of the extremity, periarticular bone loss, and arthritic changes in the distal joints. The pain may be relieved by a local anesthetic block of the sympathetic innervation to the affected extremity. Damaged primary afferent nociceptors acquire adrenergic sensitivity and can be activated by stimulation of the sympathetic outflow. This constellation of spontaneous pain and signs of sympathetic dysfunction following injury has been termed complex regional pain syndrome (CRPS). When this occurs after an identifiable nerve injury, it is termed CRPS type II (also known as posttraumatic neuralgia or, if severe, causalgia). When a similar clinical picture appears without obvious nerve injury, it is termed CRPS type I (also known as reflex sympathetic dystrophy). CRPS can be produced by a variety of injuries, including fractures of bone, soft tissue trauma, myocardial infarction, and stroke (Chap. 446). CRPS type I typically resolves with symptomatic treatment; however, when it persists, detailed examination often reveals evidence of peripheral nerve injury. Although the pathophysiology of CRPS is poorly understood, the pain and the signs of inflammation, when acute, can be rapidly relieved by blocking the sympathetic nervous system. This implies that sympathetic activity can activate undamaged nociceptors when inflammation is present. Signs of sympathetic hyperactivity should be sought in patients with posttraumatic pain and inflammation and no other obvious explanation.
CHRONIC PAIN
Managing patients with chronic pain is intellectually and emotionally challenging. The patient’s problem is often difficult or impossible to diagnose with certainty; such patients are demanding of the physician’s time and often appear emotionally distraught. The traditional medical approach of seeking an obscure organic pathology is usually unhelpful. On the other hand, psychological evaluation and behaviorally based treatment paradigms are frequently helpful, particularly in the setting of a multidisciplinary pain-management center. Unfortunately, this approach, while effective, remains largely underused in current medical practice.
There are several factors that can cause, perpetuate, or exacerbate chronic pain. First, of course, the patient may simply have a disease that is characteristically painful for which there is presently no cure. Arthritis, cancer, chronic daily headaches, fibromyalgia, and diabetic neuropathy are examples of this. Second, there may be secondary perpetuating factors that are initiated by disease and persist after that disease has resolved. Examples include damaged sensory nerves, sympathetic efferent activity, and painful reflex muscle contraction (spasm). Finally, a variety of psychological conditions can exacerbate or even cause pain.
There are certain areas to which special attention should be paid in a patient’s medical history. Because depression is the most common emotional disturbance in patients with chronic pain, patients should be questioned about their mood, appetite, sleep patterns, and daily activity. A simple standardized questionnaire, such as the Beck Depression Inventory, can be a useful screening device. It is important to remember that major depression is a common, treatable, and potentially fatal illness.
Other clues that a significant emotional disturbance is contributing to a patient’s chronic pain complaint include pain that occurs in multiple, unrelated sites; a pattern of recurrent, but separate, pain problems beginning in childhood or adolescence; pain beginning at a time of emotional trauma, such as the loss of a parent or spouse; a history of physical or sexual abuse; and past or present substance abuse.
On examination, special attention should be paid to whether the patient guards the painful area and whether certain movements or postures are avoided because of pain. Discovering a mechanical component to the pain can be useful both diagnostically and therapeutically. Painful areas should be examined for deep tenderness, noting whether this is localized to muscle, ligamentous structures, or joints. Chronic myofascial pain is very common, and, in these patients, deep palpation may reveal highly localized trigger points that are firm bands or knots in muscle. Relief of the pain following injection of local anesthetic into these trigger points supports the diagnosis. A neuropathic component to the pain is indicated by evidence of nerve damage, such as sensory impairment, exquisitely sensitive skin (allodynia), weakness, and muscle atrophy, or loss of deep tendon reflexes. Evidence suggesting sympathetic nervous system involvement includes the presence of diffuse swelling, changes in skin color and temperature, and hypersensitive skin and joint tenderness compared with the normal side. Relief of the pain with a sympathetic block supports the diagnosis, but once the condition becomes chronic, the response to sympathetic blockade is of variable magnitude and duration; the role for repeated sympathetic blocks in the overall management of CRPS is not established.
A guiding principle in evaluating patients with chronic pain is to assess both emotional and organic factors before initiating therapy. Addressing these issues together, rather than waiting to address emotional issues after organic causes of pain have been ruled out, improves compliance in part because it assures patients that a psychological evaluation does not mean that the physician is questioning the validity of their complaint. Even when an organic cause for a patient’s pain can be found, it is still wise to look for other factors. For example, a cancer patient with painful bony metastases may have additional pain due to nerve damage and may also be depressed. Optimal therapy requires that each of these factors be looked for and treated.
19 | Chest Discomfort |
Chest discomfort is among the most common reasons for which patients present for medical attention at either an emergency department (ED) or an outpatient clinic. The evaluation of nontraumatic chest discomfort is inherently challenging owing to the broad variety of possible causes, a minority of which are life-threatening conditions that should not be missed. It is helpful to frame the initial diagnostic assessment and triage of patients with acute chest discomfort around three categories: (1) myocardial ischemia; (2) other cardiopulmonary causes (pericardial disease, aortic emergencies, and pulmonary conditions); and (3) non-cardiopulmonary causes. Although rapid identification of high-risk conditions is a priority of the initial assessment, strategies that incorporate routine liberal use of testing carry the potential for adverse effects of unnecessary investigations.
EPIDEMIOLOGY AND NATURAL HISTORY
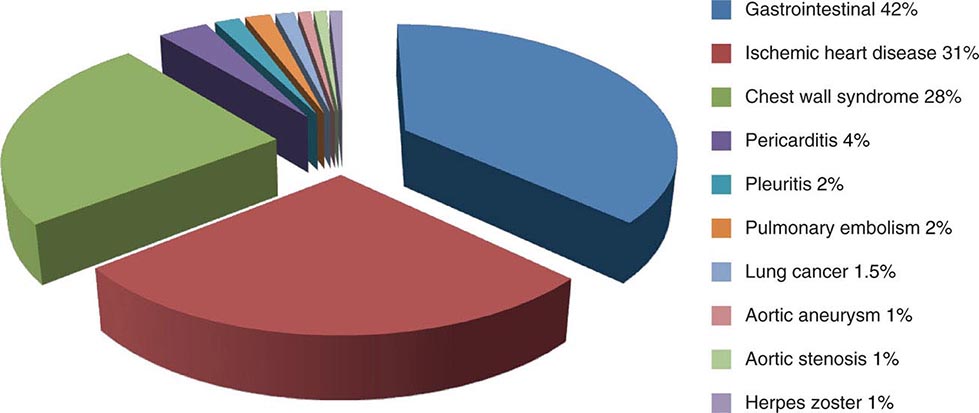
Chest discomfort is the third most common reason for visits to the ED in the United States, resulting in 6 to 7 million emergency visits each year. More than 60% of patients with this presentation are hospitalized for further testing, and the rest undergo additional investigation in the ED. Fewer than 25% of evaluated patients are eventually diagnosed with acute coronary syndrome (ACS), with rates of 5–15% in most series of unselected populations. In the remainder, the most common diagnoses are gastrointestinal causes (Fig. 19-1), and fewer than 10% are other life-threatening cardiopulmonary conditions. In a large proportion of patients with transient acute chest discomfort, ACS or another acute cardiopulmonary cause is excluded but the cause is not determined. Therefore, the resources and time devoted to the evaluation of chest discomfort in the absence of a severe cause are substantial. Nevertheless, a disconcerting 2–6% of patients with chest discomfort of presumed non-ischemic etiology who are discharged from the ED are later deemed to have had a missed myocardial infarction (MI). Patients with a missed diagnosis of MI have a 30-day risk of death that is double that of their counterparts who are hospitalized.
FIGURE 19-1 Distribution of final discharge diagnoses in patients with nontraumatic acute chest pain. (Figure prepared from data in P Fruergaard et al: Eur Heart J 17:1028, 1996.)
The natural histories of ACS, acute pericardial diseases, pulmonary embolism, and aortic emergencies are discussed in Chaps. 288, 294 and 295, 300, and 301, respectively. In a study of more than 350,000 patients with unspecified presumed non-cardiopulmonary chest discomfort, the mortality rate 1 year after discharge was <2% and did not differ significantly from age-adjusted mortality in the general population. The estimated rate of major cardiovascular events through 30 days in patients with acute chest pain who had been stratified as low risk was 2.5% in a large population-based study that excluded patients with ST-segment elevation or definite noncardiac chest pain.
CAUSES OF CHEST DISCOMFORT
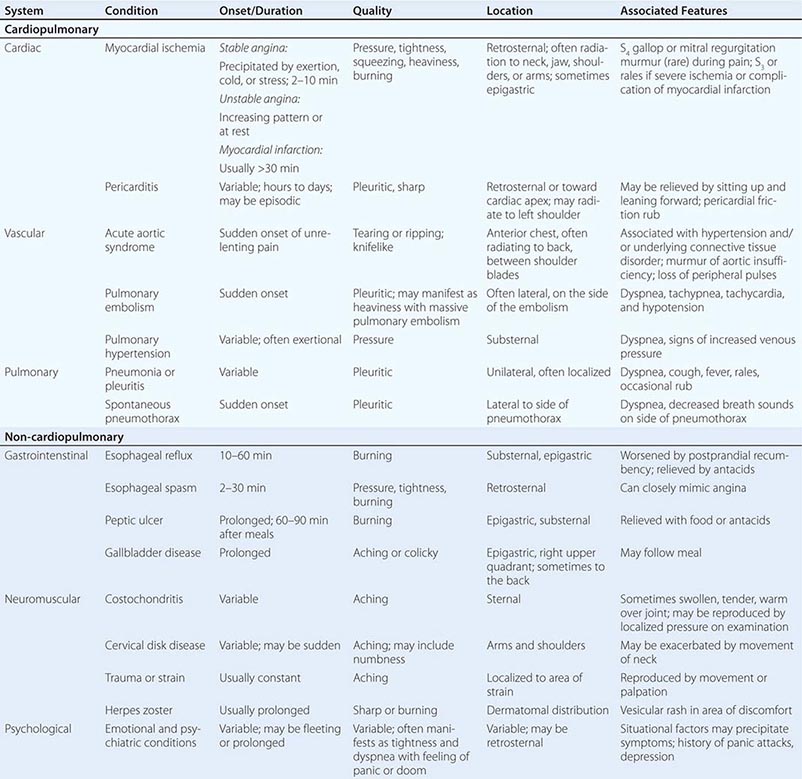
The major etiologies of chest discomfort are discussed in this section and summarized in Table 19-1. Additional elements of the history, physical examination, and diagnostic testing that aid in distinguishing these causes are discussed in a later section (see “Approach to the Patient”).
TYPICAL CLINICAL FEATURES OF MAJOR CAUSES OF ACUTE CHEST DISCOMFORT |
MYOCARDIAL ISCHEMIA/INJURY
Myocardial ischemia causing chest discomfort, termed angina pectoris, is a primary clinical concern in patients presenting with chest symptoms. Myocardial ischemia is precipitated by an imbalance between myocardial oxygen requirements and myocardial oxygen supply, resulting in insufficient delivery of oxygen to meet the heart’s metabolic demands. Myocardial oxygen consumption may be elevated by increases in heart rate, ventricular wall stress, and myocardial contractility, whereas myocardial oxygen supply is determined by coronary blood flow and coronary arterial oxygen content. When myocardial ischemia is sufficiently severe and prolonged in duration (as little as 20 min), irreversible cellular injury occurs, resulting in MI.
Ischemic heart disease is most commonly caused by atheromatous plaque that obstructs one or more of the epicardial coronary arteries. Stable ischemic heart disease (Chap. 293) usually results from the gradual atherosclerotic narrowing of the coronary arteries. Stable angina is characterized by ischemic episodes that are typically precipitated by a superimposed increase in oxygen demand during physical exertion and relieved upon resting. Ischemic heart disease becomes unstable most commonly when rupture or erosion of one or more atherosclerotic lesions triggers coronary thrombosis (Chap. 291e). Unstable ischemic heart disease is classified clinically by the presence or absence of detectable myocardial injury and the presence or absence of ST-segment elevation on the patient’s electrocardiogram (ECG). When acute coronary atherothrombosis occurs, the intracoronary thrombus may be partially obstructive, generally leading to myocardial ischemia in the absence of ST-segment elevation. Marked by ischemic symptoms at rest, with minimal activity, or in an accelerating pattern, unstable ischemic heart disease is classified as unstable angina when there is no detectable myocardial injury and as non–ST elevation MI (NSTEMI) when there is evidence of myocardial necrosis (Chap. 294). When the coronary thrombus is acutely and completely occlusive, transmural myocardial ischemia usually ensues, with ST-segment elevation on the ECG and myocardial necrosis leading to a diagnosis of ST elevation MI (STEMI, see Chap. 295).
Clinicians should be aware that unstable ischemic symptoms may also occur predominantly because of increased myocardial oxygen demand (e.g., during intense psychological stress or fever) or because of decreased oxygen delivery due to anemia, hypoxia, or hypotension. However, the term acute coronary syndrome, which encompasses unstable angina, NSTEMI, and STEMI, is in general reserved for ischemia precipitated by acute coronary atherothrombosis. In order to guide therapeutic strategies, a standardized system for classification of MI has been expanded to discriminate MI resulting from acute coronary thrombosis (type 1) from MI occurring secondary to other imbalances of myocardial oxygen supply and demand (type 2; see Chap. 294).
Other contributors to stable and unstable ischemic heart disease, such as endothelial dysfunction, microvascular disease, and vasospasm, may exist alone or in combination with coronary atherosclerosis and may be the dominant cause of myocardial ischemia in some patients. Moreover, non-atherosclerotic processes, including congenital abnormalities of the coronary vessels, myocardial bridging, coronary arteritis, and radiation-induced coronary disease, can lead to coronary obstruction. In addition, conditions associated with extreme myocardial oxygen demand and impaired endocardial blood flow, such as aortic valve disease (Chap. 301), hypertrophic cardiomyopathy, or idiopathic dilated cardiomyopathy (Chap. 287), can precipitate myocardial ischemia in patients with or without underlying obstructive atherosclerosis.
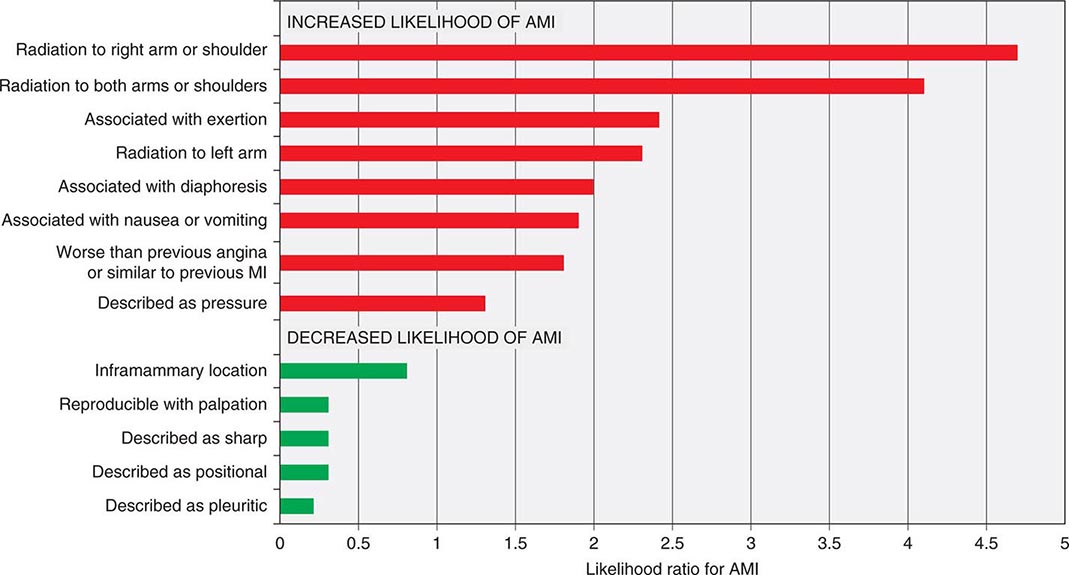
Characteristics of Ischemic Chest Discomfort The clinical characteristics of angina pectoris, often referred to simply as “angina,” are highly similar whether the ischemic discomfort is a manifestation of stable ischemic heart disease, unstable angina, or MI; the exceptions are differences in the pattern and duration of symptoms associated with these syndromes (Table 19-1). Heberden initially described angina as a sense of “strangling and anxiety.” Chest discomfort characteristic of myocardial ischemia is typically described as aching, heavy, squeezing, crushing, or constricting. However, in a substantial minority of patients, the quality of discomfort is extremely vague and may be described as a mild tightness, or merely an uncomfortable feeling, that sometimes is experienced as numbness or a burning sensation. The site of the discomfort is usually retrosternal, but radiation is common and generally occurs down the ulnar surface of the left arm; the right arm, both arms, neck, jaw, or shoulders may also be involved. These and other characteristics of ischemic chest discomfort pertinent to discrimination from other causes of chest pain are discussed later in this chapter (see “Approach to the Patient”).
Stable angina usually begins gradually and reaches its maximal intensity over a period of minutes before dissipating within several minutes with rest or with nitroglycerin. The discomfort typically occurs predictably at a characteristic level of exertion or psychological stress. By definition, unstable angina is manifest by self-limited anginal chest discomfort that is exertional but occurs at increased frequency with progressively lower intensity of physical activity or even at rest. Chest discomfort associated with MI is typically more severe, is prolonged (usually lasting ≥30 min), and is not relieved by rest.
Mechanisms of Cardiac Pain The neural pathways involved in ischemic cardiac pain are poorly understood. Ischemic episodes are thought to excite local chemosensitive and mechanoreceptive receptors that, in turn, stimulate release of adenosine, bradykinin, and other substances that activate the sensory ends of sympathetic and vagal afferent fibers. The afferent fibers traverse the nerves that connect to the upper five thoracic sympathetic ganglia and upper five distal thoracic roots of the spinal cord. From there, impulses are transmitted to the thalamus. Within the spinal cord, cardiac sympathetic afferent impulses may converge with impulses from somatic thoracic structures, and this convergence may be the basis for referred cardiac pain. In addition, cardiac vagal afferent fibers synapse in the nucleus tractus solitarius of the medulla and then descend to the upper cervical spinothalamic tract, and this route may contribute to anginal pain experienced in the neck and jaw.
OTHER CARDIOPULMONARY CAUSES
Pericardial and Other Myocardial Diseases (See also Chap. 288) Inflammation of the pericardium due to infectious or noninfectious causes can be responsible for acute or chronic chest discomfort. The visceral surface and most of the parietal surface of the pericardium are insensitive to pain. Therefore, the pain of pericarditis is thought to arise principally from associated pleural inflammation and is more common with infectious causes of pericarditis, which typically involve the pleura. Because of this pleural association, the discomfort of pericarditis is usually pleuritic pain that is exacerbated by breathing, coughing, or changes in position. Moreover, owing to the overlapping sensory supply of the central diaphragm via the phrenic nerve with somatic sensory fibers originating in the third to fifth cervical segments, the pain of pleural pericarditis is often referred to the shoulder and neck. Involvement of the pleural surface of the lateral diaphragm can lead to pain in the upper abdomen.
Acute inflammatory and other non-ischemic myocardial diseases can also produce chest discomfort. The symptoms of Takotsubo (stress-related) cardiomyopathy often start abruptly with chest pain and shortness of breath. This form of cardiomyopathy, in its most recognizable form, is triggered by an emotionally or physically stressful event and may mimic acute MI because of its commonly associated ECG abnormalities, including ST-segment elevation, and elevated biomarkers of myocardial injury. Observational studies support a predilection for women >50 years of age. The symptoms of acute myocarditis are highly varied. Chest discomfort may either originate with inflammatory injury of the myocardium or be due to severe increases in wall stress related to poor ventricular performance.
Diseases of the Aorta (See also Chap. 301) Acute aortic dissection (Fig. 19-1) is a less common cause of chest discomfort but is important because of the catastrophic natural history of certain subsets of cases when recognized late or left untreated. Acute aortic syndromes encompass a spectrum of acute aortic diseases related to disruption of the media of the aortic wall. Aortic dissection involves a tear in the aortic intima, resulting in separation of the media and creation of a separate “false” lumen. A penetrating ulcer has been described as ulceration of an aortic atheromatous plaque that extends through the intima and into the aortic media, with the potential to initiate an intramedial dissection or rupture into the adventitia. Intramural hematoma is an aortic wall hematoma with no demonstrable intimal flap, no radiologically apparent intimal tear, and no false lumen. Intramural hematoma can occur due to either rupture of the vasa vasorum or, less commonly, a penetrating ulcer.
Each of these subtypes of acute aortic syndrome typically presents with chest discomfort that is often severe, sudden in onset, and sometimes described as “tearing” in quality. Acute aortic syndromes involving the ascending aorta tend to cause pain in the midline of the anterior chest, whereas descending aortic syndromes most often present with pain in the back. Therefore, dissections that begin in the ascending aorta and extend to the descending aorta tend to cause pain in the front of the chest that extends toward the back, between the shoulder blades. Proximal aortic dissections that involve the ascending aorta (type A in the Stanford nomenclature) are at high risk for major complications that may influence the clinical presentation, including (1) compromise of the aortic ostia of the coronary arteries, resulting in MI; (2) disruption of the aortic valve, causing acute aortic insufficiency; and (3) rupture of the hematoma into the pericardial space, leading to pericardial tamponade.
Knowledge of the epidemiology of acute aortic syndromes can be helpful in maintaining awareness of this relatively uncommon group of disorders (estimated annual incidence, 3 cases per 100,000 population). Nontraumatic aortic dissections are very rare in the absence of hypertension or conditions associated with deterioration of the elastic or muscular components of the aortic media, including pregnancy, bicuspid aortic disease, or inherited connective tissue diseases, such as Marfan and Ehlers-Danlos syndromes.
Although aortic aneurysms are most often asymptomatic, thoracic aortic aneurysms can cause chest pain and other symptoms by compressing adjacent structures. This pain tends to be steady, deep, and occasionally severe. Aortitis, whether of noninfectious or infectious etiology, in the absence of aortic dissection is a rare cause of chest or back discomfort.
Pulmonary Conditions Pulmonary and pulmonary-vascular conditions that cause chest discomfort usually do so in conjunction with dyspnea and often produce symptoms that have a pleuritic nature.
PULMONARY EMBOLISM (See also Chap. 300) Pulmonary emboli (annual incidence, ~1 per 1000) can produce dyspnea and chest discomfort that is sudden in onset. Typically pleuritic in pattern, the chest discomfort associated with pulmonary embolism may result from (1) involvement of the pleural surface of the lung adjacent to a resultant pulmonary infarction; (2) distention of the pulmonary artery; or (3) possibly, right ventricular wall stress and/or subendocardial ischemia related to acute pulmonary hypertension. The pain associated with small pulmonary emboli is often lateral and pleuritic and is believed to be related to the first of these three possible mechanisms. In contrast, massive pulmonary emboli may cause severe substernal pain that may mimic an MI and that is plausibly attributed to the second and third of these potential mechanisms. Massive or submassive pulmonary embolism may also be associated with syncope, hypotension, and signs of right heart failure. Other typical characteristics that aid in the recognition of pulmonary embolism are discussed later in this chapter (see “Approach to the Patient”).
PNEUMOTHORAX (See also Chap. 317) Primary spontaneous pneumothorax is a rare cause of chest discomfort, with an estimated annual incidence in the United States of 7 per 100,000 among men and <2 per 100,000 among women. Risk factors include male sex, smoking, family history, and Marfan syndrome. The symptoms are usually sudden in onset, and dyspnea may be mild; thus, presentation to medical attention is sometimes delayed. Secondary spontaneous pneumothorax may occur in patients with underlying lung disorders, such as chronic obstructive pulmonary disease, asthma, or cystic fibrosis, and usually produces symptoms that are more severe. Tension pneumothorax is a medical emergency caused by trapped intrathoracic air that precipitates hemodynamic collapse.
Other Pulmonary Parenchymal, Pleural, or Vascular Disease (See also Chaps. 304, 305, and 316) Most pulmonary diseases that produce chest pain, including pneumonia and malignancy, do so because of involvement of the pleura or surrounding structures. Pleurisy is typically described as a knifelike pain that is worsened by inspiration or coughing. In contrast, chronic pulmonary hypertension can manifest as chest pain that may be very similar to angina in its characteristics, suggesting right ventricular myocardial ischemia in some cases. Reactive airways diseases similarly can cause chest tightness associated with breathlessness rather than pleurisy.
NON-CARDIOPULMONARY CAUSES
Gastrointenstinal Conditions (See also Chap. 344) Gastrointestinal disorders are the most common cause of nontraumatic chest discomfort and often produce symptoms that are difficult to discern from more serious causes of chest pain, including myocardial ischemia. Esophageal disorders, in particular, may simulate angina in the character and location of the pain. Gastroesophageal reflux and disorders of esophageal motility are common and should be considered in the differential diagnosis of chest pain (Fig. 19-1 and Table 19-1). Acid reflux often causes a burning discomfort. The pain of esophageal spasm, in contrast, is commonly an intense, squeezing discomfort that is retrosternal in location and, like angina, may be relieved by nitroglycerin or dihydropyridine calcium channel antagonists. Chest pain can also result from injury to the esophagus, such as a Mallory-Weiss tear or even an esophageal rupture (Boerhaave syndrome) caused by severe vomiting. Peptic ulcer disease is most commonly epigastric in location but can radiate into the chest (Table 19-1).
Hepatobiliary disorders, including cholecystitis and biliary colic, may mimic acute cardiopulmonary diseases. Although the pain arising from these disorders usually localizes to the right upper quadrant of the abdomen, it is variable and may be felt in the epigastrium and radiate to the back and lower chest. This discomfort is sometimes referred to the scapula or may in rare cases be felt in the shoulder, suggesting diaphragmatic irritation. The pain is steady, usually lasts several hours, and subsides spontaneously, without symptoms between attacks. Pain resulting from pancreatitis is typically aching epigastric pain that radiates to the back.
Musculoskeletal and Other Causes (See also Chap. 393) Chest discomfort can be produced by any musculoskeletal disorder involving the chest wall or the nerves of the chest wall, neck, or upper limbs. Costochondritis causing tenderness of the costochondral junctions (Tietze’s syndrome) is relatively common. Cervical radiculitis may manifest as a prolonged or constant aching discomfort in the upper chest and limbs. The pain may be exacerbated by motion of the neck. Occasionally, chest pain can be caused by compression of the brachial plexus by the cervical ribs, and tendinitis or bursitis involving the left shoulder may mimic the radiation of angina. Pain in a dermatomal distribution can also be caused by cramping of intercostal muscles or by herpes zoster (Chap. 217).
Emotional and Psychiatric Conditions As many as 10% of patients who present to emergency departments with acute chest discomfort have a panic disorder or related condition (Table 19-1). The symptoms may include chest tightness or aching that is associated with a sense of anxiety and difficulty breathing. The symptoms may be prolonged or fleeting.
CONSIDERATIONS IN THE ASSESSMENT OF THE PATIENT WITH CHEST DISCOMFORT |