http://evolve.elsevier.com/McCuistion/pharmacology/
This chapter covers antituberculars, antifungals, and antivirals. Although these drug categories differ from one another, they each contain drugs that inhibit or kill organisms that cause disease.
Tuberculosis
Tuberculosis (TB) is caused by the acid-fast bacillus Mycobacterium tuberculosis. The number of TB cases had declined until the mid-1980s, when the number of cases started to increase. The increase in cases of TB was attributed to multiple factors, such as human immunodeficiency virus (HIV), increased immigration, and the spread of multidrug-resistant TB (MDR TB). According to the U.S. Department of Health and Human Services (DHHS), tuberculosis is one of the world’s leading causes of death due to infectious diseases in persons older than 5 years of age. Each year, about 9 million people develop TB, and 2 million die because of it. With increased funding, prompt identification of persons with TB, appropriate initial treatment, and completion of treatments, the number of TB cases started declining again in the mid-1990s. However, MDR TB continues to be a serious health concern. Patients started on therapy for TB who do not finish the prescribed therapy can develop and spread resistant strains of M. tuberculosis.
Pathophysiology
TB is transmitted from one person to another by droplets dispersed in the air through coughing, sneezing, and speaking. TB microorganisms can be inhaled into the lungs, therefore persons in close contact with the infected patient are at highest risk of becoming infected. Others at high risk for contracting the disease include the immunocompromised (e.g., patients with HIV, diabetes, and renal failure and those taking certain medications, such as cortisol), people living or working in high-risk residential settings (e.g., nursing homes, shelters, correctional facilities), those who inject illegal drugs, and health care workers who serve high-risk patients.
Not everyone infected with TB will develop clinical manifestations, rather some will harbor the microorganisms and will have what is called latent tuberculosis infection; these persons are at risk of developing TB disease later, and only those with TB disease can infect others. Symptoms of TB disease include anorexia, cough with sputum production or blood, chest pain, fever, night sweats, weight loss, and positive acid-fast bacilli in the sputum. Isolating infectious persons and initiating treatment for TB disease as soon as possible is the best way to decrease transmission.
Nurses should be aware that persons coming to the United States from high-risk countries where TB disease is common may have been vaccinated with bacille Calmette-Guérin (BCG) as a child. This vaccine is seldom used in the United States. Previous vaccination with BCG may cause a positive reaction to skin testing; however, it does not affect interferon-gamma release assay (IGRA) blood testing.
Antitubercular Drugs
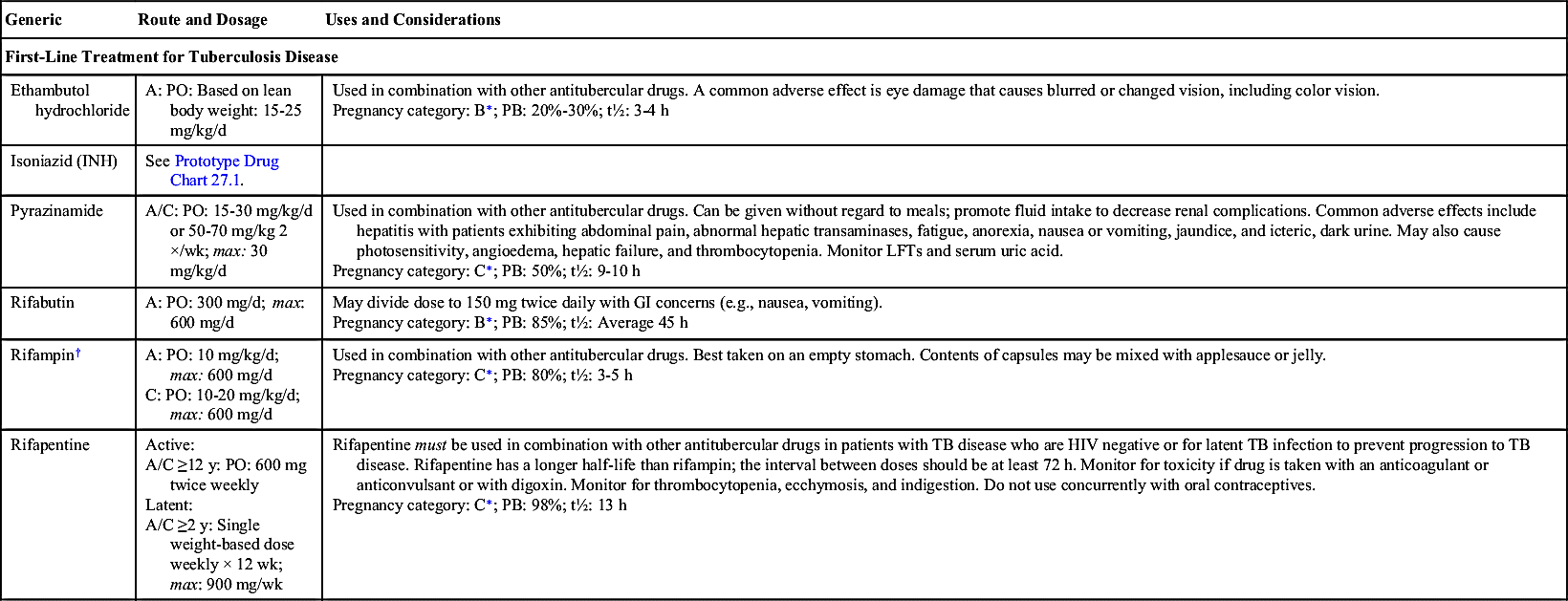
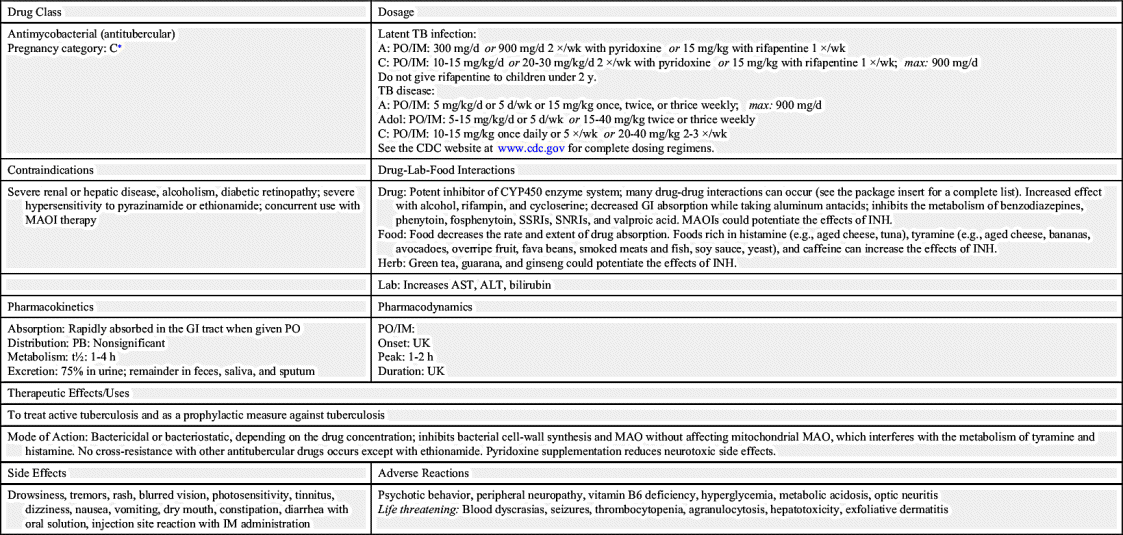
Antitubercular drugs (Table 27.1), which include antimycobacterials, treat persons with TB disease and those exposed to TB disease. Streptomycin was the first drug used in the treatment of TB disease in 1943. However, it was noted that patients began to deteriorate after 3 months of therapy due to drug resistance. In 1952, isoniazid (INH) began to see widespread use in the treatment of TB disease and was felt to be a “wonder drug.” To this day, INH remains the first-line treatment for TB disease. INH is a bactericidal drug that inhibits tubercle cell-wall synthesis and blocks pyridoxine (vitamin B6), which is used for intracellular enzyme production. When INH is prescribed, pyridoxine may also be prescribed to avoid vitamin B deficiency and to minimize peripheral neuropathy. INH is administered orally. Prototype Drug Chart 27.1 lists the data for INH.
Prophylactic antituberculars are drugs to prevent TB disease in individuals with latent TB infection. Prophylaxis is recommended for those who have a clinically significant result on tuberculin skin testing (≥5 mm for immunocompromised individuals or ≥10 mm for high-risk groups; Box 27.1) or a positive IGRA result. Patients who have converted from a negative to a positive TB skin test (TST) or from a negative to positive IGRA should be considered candidates for prophylactic therapy as well. Prototype Drug Chart 27.1 shows guidelines for latent TB infection treatment with INH.
Prophylactic therapy is contraindicated for persons with liver disease because INH is the primary antitubercular drug used, and it may cause INH-induced liver damage. Other antitubercular drugs may also cause liver damage if given in high doses over an extended period.
Single-drug therapy is ineffective in the treatment of TB disease due to drug resistance. Using a combination of antitubercular drugs has been shown to decrease bacterial resistance. Additionally, using combination therapy has decreased the duration of treatment from 2 years to 6 to 9 months. Different combinations of drugs can be used with INH, rifampin, ethambutol, and pyrazinamide.
Antitubercular drugs are divided into two categories: first-line drugs that form the core of treatment regimens and drugs used in the treatment of drug-resistant TB. First-line drugs—those drugs chosen first, such as INH, rifampin, pyrazinamide, and ethambutol—are considered more effective and less toxic than drugs used in the treatment of drug-resistant TB. Drugs used in the treatment of drug-resistant TB, in which M. tuberculosis is resistant to at least one first-line drug, or MDR TB, in which M. tuberculosis is resistant to INH and rifampin plus one other first-line drug, are used in combination with first-line drugs to treat drug-resistant M. tuberculosis. See Table 27.1 for drugs used in the treatment of drug-resistant TB.
Combination therapy against TB disease is more effective in eradicating infection than any single drug. The treatment regimen is divided into two phases: the initial phase lasts 2 months, and the continuation phase consists of the next 4 or 7 months. The total treatment plan can be up to 9 months or longer, depending on response to drug therapy. If drug resistance develops, other antibacterial drugs such as aminoglycosides (streptomycin, kanamycin, amikacin) or fluoroquinolones (levofloxacin, ciprofloxacin, or ofloxacin) may be given as part of combination therapy. Combination therapy for drug-resistant TB disease consists of a minimum of three drugs, but preferably four to five drugs, administered as part of directly observed therapy to ensure adherence. Drug therapy should be managed by an expert in the disease, and susceptibility testing to determine drug resistance should be performed before drug therapy; however, treatment with first-line therapy should not be delayed if TB disease is suspected. Aminoglycoside antibiotics should not be taken if kidney dysfunction is present. When antibacterial agents are used continuously or at high doses, serum drug levels should be closely monitored to avoid drug toxicity.
Many drug-drug interactions and side effects occur with antituberculars. To increase adherence to drug therapy, direct observation therapy (DOT) is recommended.
Side Effects and Adverse Reactions
Side effects and adverse reactions to antituberculars differ according to the drug prescribed. For INH, peripheral neuropathy can be a problem, especially for those who are malnourished, have diabetes mellitus, or are alcoholics. This condition can be prevented if pyridoxine (vitamin B6) is administered. Hepatotoxicity (liver toxicity) is an adverse reaction to many antituberculars. Patients with moderate to severe liver dysfunction should not take these drugs. Patients with liver disease should have hepatic transaminases monitored closely. Patients may also develop headaches, blood dyscrasias, paresthesias, gastrointestinal (GI) distress (e.g., nausea, vomiting, diarrhea, dyspepsia), and ocular toxicity. An ophthalmic exam before and during treatment is warranted. INH may cause hyperglycemia, hyperkalemia, hypophosphatemia, and hypocalcemia. Rifampin turns body fluids orange, and soft contact lenses may be permanently discolored. It may also cause thrombocytopenia and GI intolerance. The patient taking ethambutol may develop dizziness, confusion, hallucinations, and joint pain. Streptomycin may lead to many adverse effects such as ototoxicity, optic nerve toxicity, encephalopathy, angioedema, central nervous system (CNS) and respiratory depression, nephrotoxicity, and hepatotoxicity.
Special Populations
Pregnancy and Tuberculosis
The benefits of treating a pregnant woman with TB disease outweigh the risks of treatment. Women with untreated TB disease are at risk for passing the disease to their fetus and delivering an infant with low birthweight. The drugs used in initial treatment of TB disease do cross the placenta but do not appear to harm the fetus.
Treatment of latent TB infection in the pregnant woman includes INH daily or twice weekly for 9 months with pyridoxine supplementation. Three-month combination therapy of INH with rifapentinem, referred to as 3HP, is not recommended for pregnant women or those planning to become pregnant within 3 months.
The pregnant woman with TB disease should be treated with INH, rifampin, and ethambutol daily for 2 months followed by INH and rifampin daily or twice weekly for 7 months for a total of 9 months. Streptomycin should not be used due to potential harmful effects on the fetus. Pyrazinamide is also not recommended due to unknown effects on the fetus.
Tuberculosis and HIV Coinfection
HIV is a risk factor for the development of TB, and TB disease is one of the leading causes of death for people living with HIV. Left untreated, latent TB infection can quickly develop into TB disease. The recommended treatment for an adult with latent TB infection and HIV is INH daily for 9 months.
Adults with HIV and TB disease should be treated for 6 months with INH, rifabutin, pyrazinamide, and ethambutol during the initial phase. The 4-month continuation phase should consist of INH and rifabutin.
A pregnant woman with HIV and TB disease should be treated the same as a nonpregnant woman but with concern for the fetus when choosing drug therapy.
Pediatric Tuberculosis
Children are more likely than adults to be sickened more quickly by TB. Because of this, children with latent TB infection should be treated to prevent development of TB disease. The recommended treatment is INH for 9 months. Treatment of TB disease should be managed by a pediatric TB expert in conjunction with drug susceptibility studies. It is very important for the nurse to make sure parents understand that if a child stops taking the drugs before therapy is finished, the child can become sick again. Additionally, nurses should stress to parents that if drugs are not taken correctly, the bacteria may become drug resistant. Drug-resistant TB is harder and more expensive to treat.