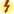http://evolve.elsevier.com/McCuistion/pharmacology/
Inflammation is a response to tissue injury and infection. When the inflammatory process occurs, a vascular reaction takes place in which fluid, elements of blood, leukocytes (white blood cells [WBCs]), and chemical mediators accumulate at the injured tissue or infection site. The process of inflammation is a protective mechanism in which the body attempts to neutralize and destroy harmful agents at the site of injury and to establish conditions for tissue repair.
Although a relationship exists between inflammation and infection, these terms should not be used interchangeably. Infection is caused by microorganisms and results in inflammation, but not all inflammations are caused by infections.
Pathophysiology
The five characteristics of inflammation, called the cardinal signs of inflammation, are (1) redness, (2) swelling (edema), (3) heat, (4) pain, and (5) loss of function. Table 24.1 gives the description and explanation of the cardinal signs of inflammation. Inflammation also has two phases: the vascular phase, which occurs 10 to 15 minutes after an injury, and the delayed phase. The vascular phase is associated with vasodilation and increased capillary permeability, during which blood substances and fluid leave the plasma and go to the injured site. The delayed phase occurs when leukocytes infiltrate the inflamed tissue.
TABLE 24.1
Cardinal Signs of Inflammation
| Signs | Description and Explanation |
| Erythema (redness) | Redness occurs in the first phase of inflammation. Blood accumulates in the area of tissue injury because of the release of the body’s chemical mediators (kinins, prostaglandins, and histamine). These chemical mediators dilate the arterioles. |
| Edema (swelling) | Swelling is the second phase of inflammation. Plasma leaks into the interstitial tissue at the injury site. Kinins and histamine increase capillary permeability. |
| Heat | Heat at the inflammatory site can be caused by increased blood accumulation and may result from pyrogens, substances that produce fever, which interfere with the temperature-regulating center in the hypothalamus. |
| Pain | Pain is caused by tissue swelling and release of chemical mediators. |
| Loss of function | Function is lost because of the accumulation of fluid at the tissue injury site and because of pain, which decreases mobility at the affected area. |
Various chemical mediators are released during the inflammation process. Among these are prostaglandins, chemical mediators that have been isolated from the exudate at inflammatory sites. Prostaglandins have many effects that include vasodilation, relaxation of smooth muscle, increased capillary permeability, and sensitization of nerve cells to pain.
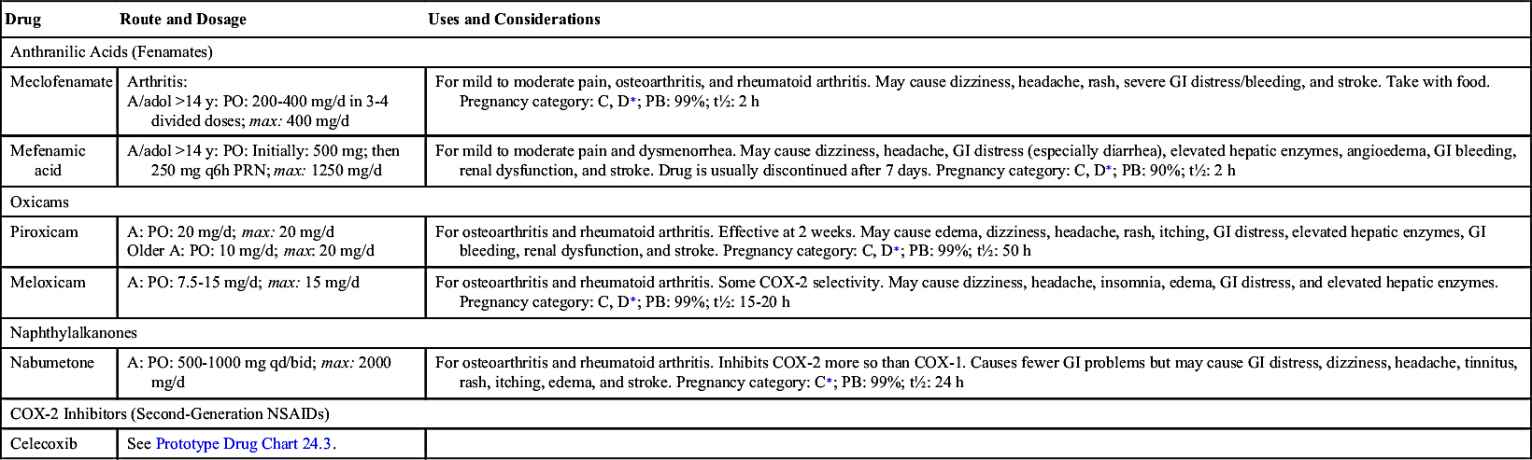
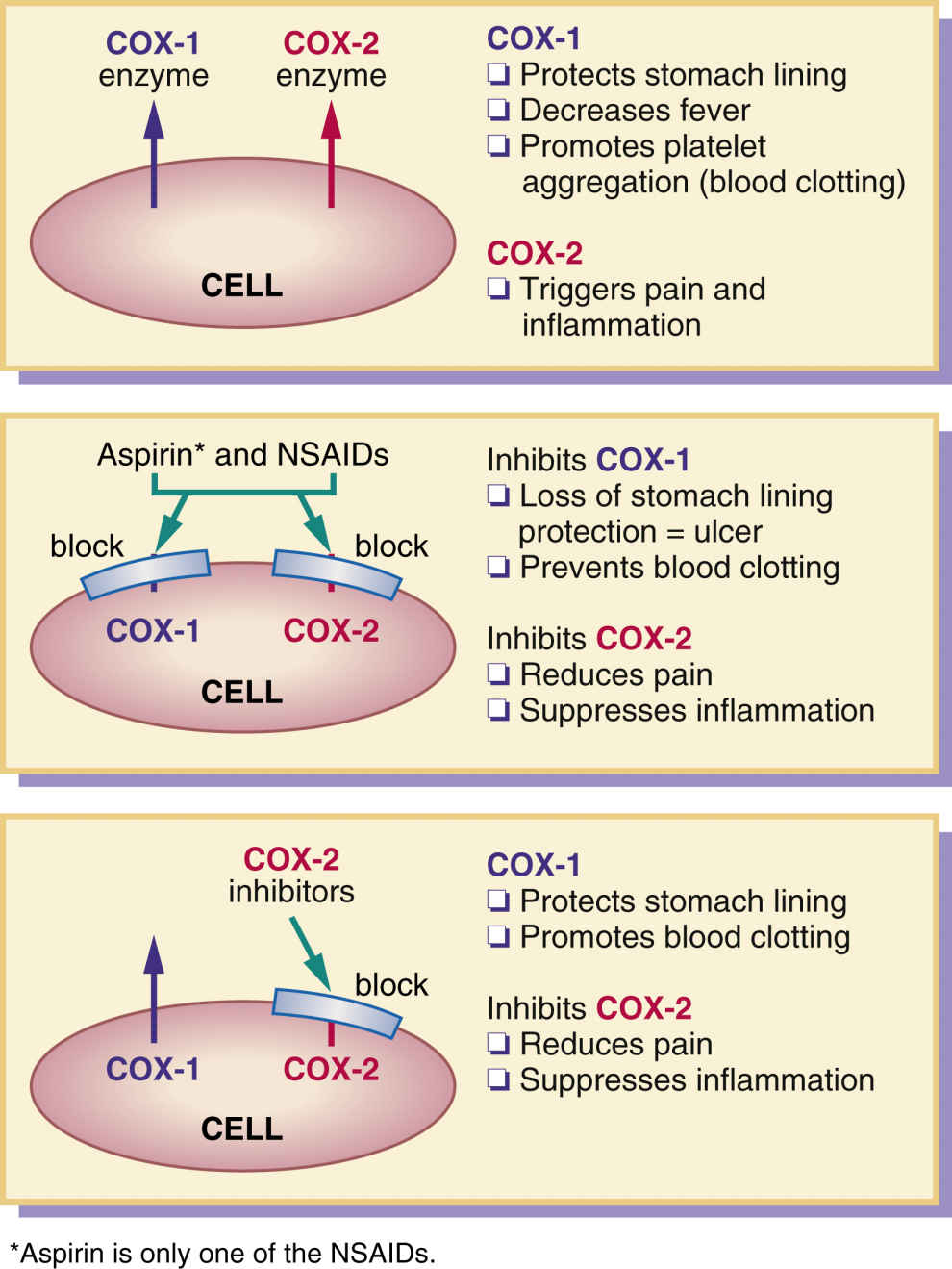
Cyclooxygenase (COX) is the enzyme responsible for converting arachidonic acid into prostaglandins and their products. This synthesis of prostaglandins causes inflammation and pain at a tissue injury site. Cyclooxygenase has two enzyme forms, COX-1 and COX-2. COX-1 protects the stomach lining and regulates blood platelets, and COX-2 triggers inflammation and pain.
Antiinflammatory Agents
Drugs such as aspirin inhibit the biosynthesis of prostaglandin and are therefore called prostaglandin inhibitors. Because prostaglandin inhibitors affect the inflammatory process, they are more commonly called antiinflammatories or antiinflammatory drugs.
Antiinflammatories also relieve pain (analgesic), reduce elevated body temperature (antipyretic), and inhibit platelet aggregation (anticoagulant). Aspirin is the oldest antiinflammatory drug, but it was first used for its analgesic and antipyretic properties. As a result of searching for a more effective drug with fewer side effects, many other antiinflammatories or prostaglandin inhibitors have been discovered. Although these drugs have potent antiinflammatory effects that mimic the effects of corticosteroids (cortisone), they are not chemically related to corticosteroids and therefore are called nonsteroidal antiinflammatory drugs (NSAIDs). Most NSAIDs are used to decrease inflammation and pain for patients who have some type of musculoskeletal condition.
Nonsteroidal Antiinflammatory Drugs
NSAIDs include aspirin and aspirin-like drugs that inhibit the enzyme COX, which is needed for the biosynthesis of prostaglandins. Chapters 25 and 40 present expanded discussions of NSAIDs in their roles as analgesics and anticoagulants, respectively. These drugs may be called prostaglandin inhibitors with varying degrees of analgesic and antipyretic effects, but they are used primarily as antiinflammatories to relieve inflammation and pain. Their antipyretic effect is less than their antiinflammatory effect. With several exceptions, NSAID preparations are not suggested for use in alleviating mild headaches and mildly elevated temperature. Preferred drugs for headaches and fever are aspirin (adults only for fever), acetaminophen, and ibuprofen. NSAIDs are more appropriate for reducing swelling, pain, and stiffness in joints.
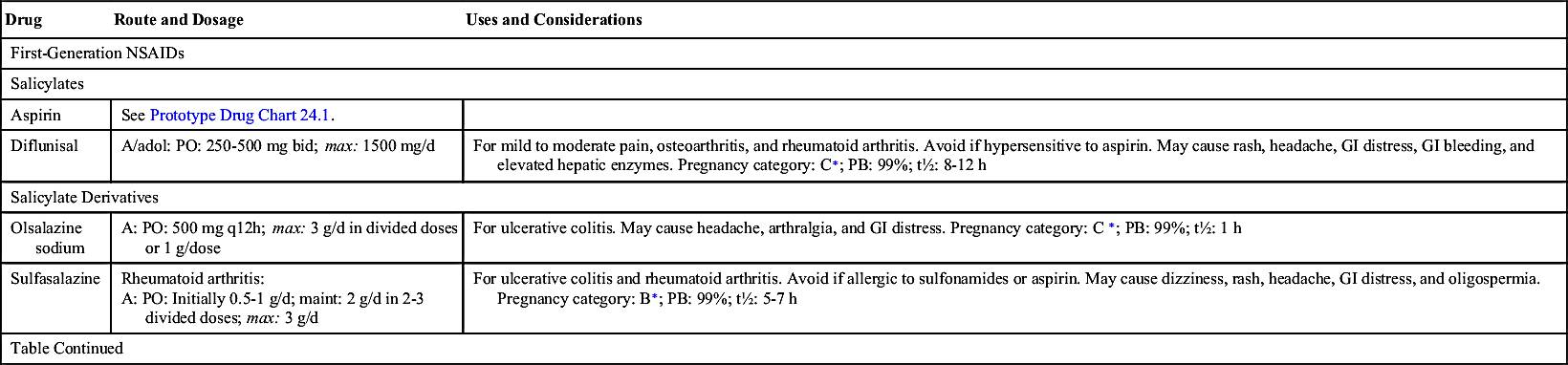
Most NSAIDs cost more than aspirin. Other than aspirin, the only NSAIDs that can be purchased over the counter (OTC) are ibuprofen and naproxen. All other NSAIDs require a prescription. Examples of prescription products on the market that contain NSAID contents alone or in combination include celecoxib, meloxicam, oxaprozin, nabumetone, sulindac, and ketorolac. If a patient can take aspirin for the inflammatory process without gastrointestinal (GI) upset, salicylate products are usually recommended.
There are seven groups of NSAIDs:
1. Salicylates
2. Para-chlorobenzoic acid derivatives, or indoles
3. Phenylacetic acids
4. Propionic acid derivatives
5. Fenamates
6. Oxicams
7. Selective COX-2 inhibitors
The first six NSAID groups on the list are known as first-generation NSAIDs, and the COX-2 inhibitors are called second-generation NSAIDs.
Table 24.2 provides dosage information and considerations for use for the most commonly used NSAIDs. The half-lives of NSAIDs differ greatly; some have a short half-life of 2 hours, and others have a moderate to long half-life of 6 to 24 hours. Aspirin should not be taken with an NSAID because of the potentiation of side effects. In addition, combined therapy does not increase effectiveness.
Salicylates
Aspirin comes from the family of salicylates derived from salicylic acid. Aspirin is also called acetylsalicylic acid (ASA) after the acetyl group used in its composition. The abbreviation frequently used for aspirin is ASA.
Aspirin was developed in 1899 by Adolph Bayer, making it the oldest antiinflammatory agent. It was the most frequently used antiinflammatory agent before the introduction of ibuprofen. Aspirin is a prostaglandin inhibitor that decreases the inflammatory process. It is also considered an antiplatelet drug for patients with cardiac or cerebrovascular disorders; aspirin decreases platelet aggregation, and thus blood clotting is decreased. Because high doses of aspirin are usually needed to relieve inflammation, gastric distress—which includes anorexia, dyspepsia, nausea, vomiting, diarrhea, constipation, abdominal pain, heartburn, and flatulence—is a common problem. In such cases, enteric-coated (EC) tablets may be used. Aspirin should not be taken with other NSAIDs because it decreases the blood level and effectiveness of NSAIDs.
Aspirin and other NSAIDs relieve pain by inhibiting the COX enzyme, which is needed for the biosynthesis of prostaglandins. As mentioned earlier, the two enzyme forms of cyclooxygenase, COX-1 and COX-2 (Fig. 24.1), serve different purposes: COX-1 protects the stomach lining and regulates blood platelets, promoting blood clotting, whereas COX-2 triggers pain and inflammation at the injured site. NSAIDs usually inhibit or block both COX-1 and COX-2. Inhibition of COX-1 produces the desirable effect of decreasing platelet aggregation, but it has the undesirable effect of decreasing protection to the stomach lining; therefore stomach bleeding and ulcers may occur with aspirin and NSAID agents. When COX-2 is inhibited, pain and fever are reduced and inflammation is suppressed, but COX-2 inhibitors do not cause gastric ulceration, and they have no effect on platelet function.
Newer NSAIDs, called COX-2 inhibitors, block only COX-2 and not COX-1. These drugs leave protection for the stomach lining intact, so no gastric bleeding or ulcers result, but they still deliver relief for pain and inflammation.
A COX-2 inhibitor approved by the U.S. Food and Drug Administration (FDA) is celecoxib. Drugs similar to COX-2 inhibitors include meloxicam and nabumetone, which allow some stomach protection. Patients at risk for stroke or heart attack who take aspirin to prevent blood clotting (decreased platelet aggregation) would not benefit from COX-2 inhibitors. If the COX-1 enzyme was not blocked, increased blood clotting would remain even though the stomach lining is protected.
Many researchers believe that COX-2 inhibitors may prevent some types of cancer, such as colon cancer. Fruits and vegetables block the COX-2 enzyme naturally, protecting the colon from malignant growths.
Pharmacokinetics
Aspirin is well absorbed from the GI tract (Prototype Drug Chart 24.1). It can cause GI distress, which includes anorexia, nausea, vomiting, diarrhea, and abdominal pain, so it should be taken with water, milk, or food. The EC or buffered form can decrease gastric distress. EC tablets should not be crushed or broken.
Aspirin has a short half-life. It should not be taken during the last trimester of pregnancy because it could cause premature closure of the ductus arteriosus in the fetus. Aspirin should not be taken by children with flu symptoms because it may cause the potentially fatal Reye syndrome.
Pharmacodynamics
Like other NSAIDs, aspirin inhibits prostaglandin synthesis by inhibiting COX-1 and COX-2, thus it decreases inflammation and pain. The onset of action for aspirin is within 30 minutes. It peaks in 1 to 2 hours, and the duration of action is an average of 4 to 6 hours. The action for the rectal preparation of aspirin can be erratic because of blood supply and fecal material in the rectum; it may take a week or longer for a therapeutic antiinflammatory effect.
Hypersensitivity to Salicylate Products
Patients may be hypersensitive to aspirin. Tinnitus (ringing in the ears), vertigo (dizziness), and bronchospasm—especially in asthmatic patients—are symptoms of aspirin overdose or hypersensitivity to aspirin.
Salicylates are present in numerous foods such as prunes, raisins, and licorice and in spices such as curry and paprika.
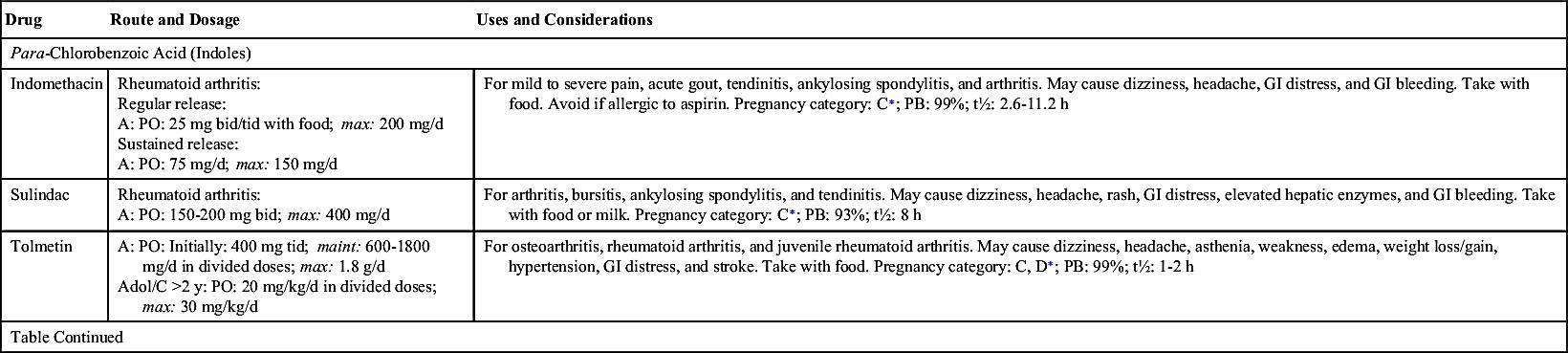
Para-Chlorobenzoic Acid
One of the first NSAIDs introduced was indomethacin, a para-chlorobenzoic acid. It is used for rheumatoid arthritis (RA), gouty arthritis, and osteoarthritis and is a potent prostaglandin inhibitor. It is highly protein bound (99%) and displaces other protein-bound drugs, resulting in potential toxicity. It has a moderate half-life (2.6 to 11.2 hours). Indomethacin is very irritating to the stomach and should be taken with food.
Two other para-chlorobenzoic acid derivatives—sulindac and tolmetin—produce less severe adverse reactions than indomethacin. Tolmetin is not as highly protein bound as indomethacin and sulindac, and it has a short half-life. This group of NSAIDs may cause sodium and water retention and increased blood pressure.
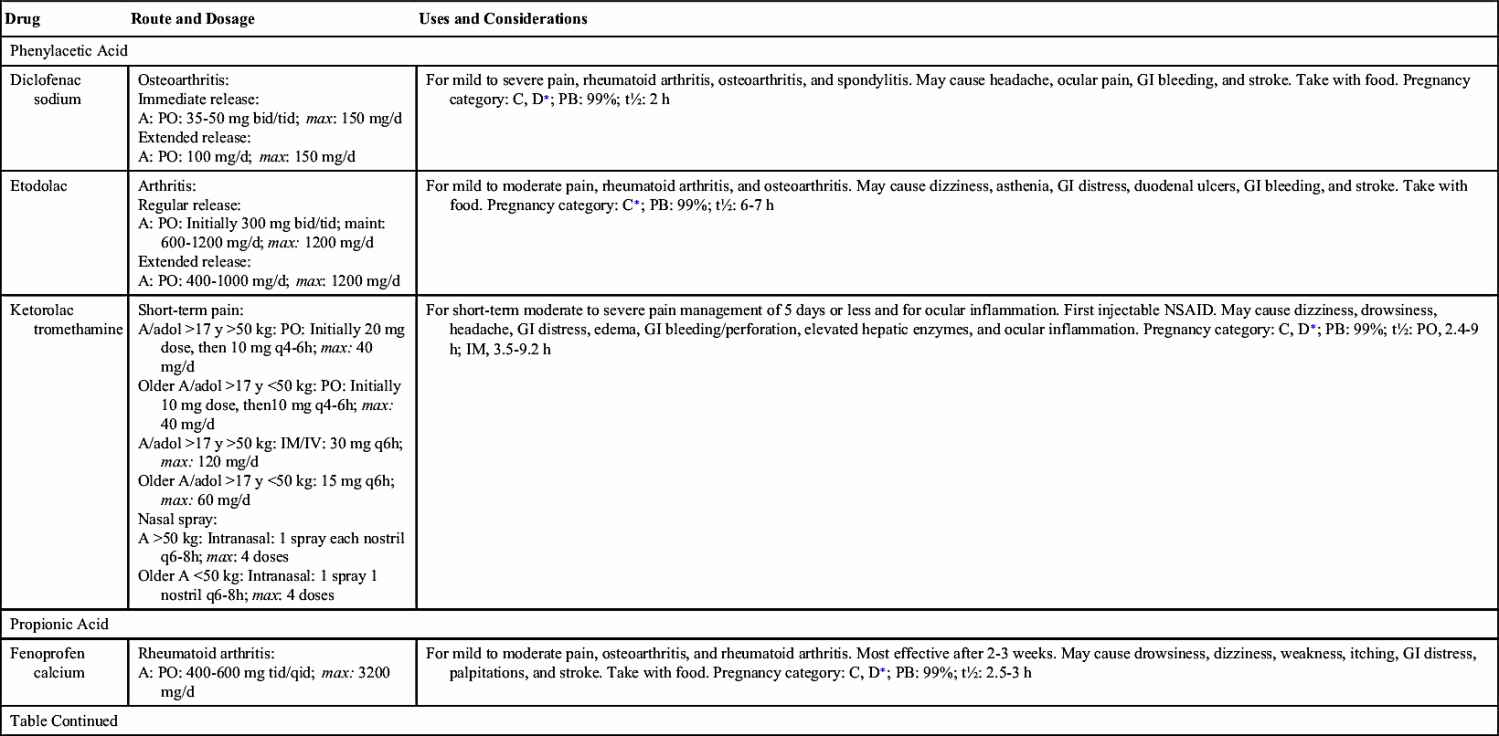
Phenylacetic Acid Derivatives
Diclofenac sodium, a phenylacetic acid derivative, has a plasma half-life of 2 hours. Its analgesic and antiinflammatory effects are similar to those of aspirin, but it has minimal to no antipyretic effects. It is indicated for RA, osteoarthritis, and ankylosing spondylitis. Diclofenac is available in oral, extended-release, and topical 1% gel preparations. Side effects and adverse reactions are similar to those of other NSAIDs, with far less detrimental reactions when using a topical preparation.
Ketorolac, another phenylacetic acid derivative, is the first injectable NSAID. Like other NSAIDs, it inhibits prostaglandin synthesis, but it has greater analgesic properties than other antiinflammatory agents. Ketorolac is recommended for short-term management of pain. For postsurgical pain, it has shown analgesic efficacy equal or superior to that of opioid analgesics. It is administered intramuscularly in doses of 30 to 60 mg every 6 hours for adults. Ketorolac is also available in oral, intravenous (IV), and intranasal preparations.
Propionic Acid Derivatives
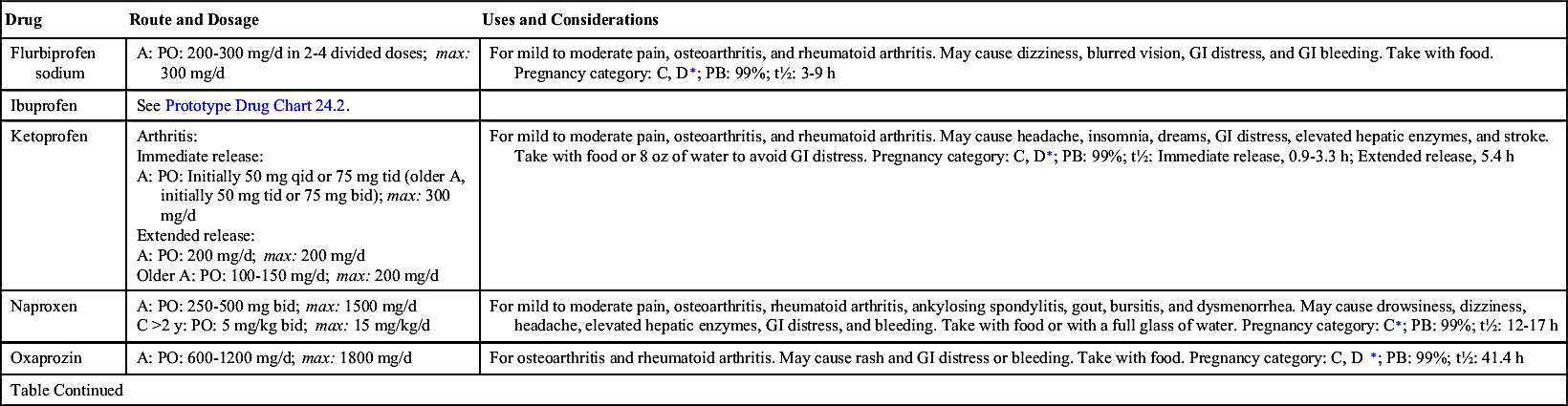
The propionic acid group is a relatively new group of NSAIDs. These drugs are aspirin-like but have stronger effects and create less GI irritation. Drugs in this group are highly protein bound, so drug interactions might occur, especially when given with another highly protein-bound drug. Propionic acid derivatives are better tolerated than other NSAIDs. Gastric upset occurs, but it is not as severe as with aspirin and indomethacin. Severe adverse reactions such as blood dyscrasias are not frequently seen. Ibuprofen is the most widely used propionic acid NSAID, and it may be purchased OTC in lower doses (200 mg). Prototype Drug Chart 24.2 details the pharmacologic behavior of ibuprofen. Five other propionic acid agents are fenoprofen calcium, naproxen, ketoprofen, flurbiprofen, and oxaprozin.
Pharmacokinetics
Ibuprofens are well absorbed from the GI tract. These drugs have a short half-life but are highly protein bound. If ibuprofen is taken with another highly protein-bound drug, severe side effects may occur. The drug is metabolized in the liver to inactivate metabolites and is excreted as inactive metabolites in the urine.