http://evolve.elsevier.com/McCuistion/pharmacology/
Antidiabetic drugs are used primarily to control diabetes mellitus, a chronic disease that affects carbohydrate metabolism. The two groups of antidiabetic agents are insulin and oral hypoglycemic (antidiabetic) drugs. Insulin, a protein secreted from the beta cells of the pancreas, is necessary for carbohydrate metabolism and plays an important role in protein and fat metabolism. The beta cells make up 75% of the pancreas, and the alpha cells that secrete glucagons—a hyperglycemic substance—occupy approximately 20% of the pancreas. Oral hypoglycemic drugs, also known as oral antidiabetic drugs (to avoid confusion with the term hypoglycemic reaction), are synthetic preparations that stimulate insulin release or otherwise alter the metabolic response to hyperglycemia.
Diabetes Mellitus
Diabetes mellitus, a chronic disease that results from deficient glucose metabolism, is caused by insufficient insulin secretion from the beta cells. This results in high blood glucose (hyperglycemia). Diabetes mellitus is characterized by the three p’s: polyuria (increased urine output), polydipsia (increased thirst), and polyphagia (increased hunger). Diabetes mellitus is a disorder of the pancreas, whereas diabetes insipidus is a disorder of the posterior pituitary gland, discussed in detail in Chapter 46.
The four types of diabetes are presented in Table 47.1. Viral infections, environmental conditions, and genetic factors contribute to the onset of type 1 diabetes mellitus. Type 2 diabetes mellitus is the most common type of diabetes. Some sources suggest that heredity and obesity are the major factors that cause type 2 diabetes. With type 2 diabetes, there is some beta-cell function with varying amounts of insulin secretion. Hyperglycemia may be controlled for some type 2 diabetes patients with oral antidiabetic (hypoglycemic) drugs and a diet prescribed by the American Diabetic Association (ADA); however, about one third of patients with type 2 diabetes need insulin. Patients with type 2 diabetes who use one or two oral antidiabetic drugs may become insulin dependent years later.
TABLE 47.1
Types and Occurrences of Diabetes Mellitus
| Types of Diabetes Mellitus | Percentage of Occurrences |
| Type 1 | 10%-12% |
| Type 2 | 85%-90% |
| Secondary diabetes (medications, hormonal changes) | 2%-3% |
| Gestational diabetes mellitus | 1% (2%-5% of all pregnancies) |
Certain drugs increase blood glucose and can cause hyperglycemia in prediabetic persons. These include glucocorticoids (cortisone, prednisone), thiazide diuretics (hydrochlorothiazide), and epinephrine. Usually the blood glucose level returns to normal after the drug is discontinued.
During the second and third trimesters of pregnancy, the levels of the hormones progesterone, cortisol, and human placental lactogen (hPL) increase. These increased hormone levels can inhibit insulin usage. This is a contributing factor for the occurrence of gestational diabetes mellitus (GDM). Glucose is then mobilized from the tissue and from lipid storage sites. After pregnancy, the blood glucose level may decrease; however, some patients may develop diabetes mellitus, whereas others may develop type 2 diabetes in later years.
Insulin
Insulin is released from the beta cells of the islets of Langerhans in response to an increase in blood glucose. Oral glucose load is more effective in raising the serum insulin level than an intravenous (IV) glucose load. Insulin promotes the uptake of glucose, amino acids, and fatty acids and converts them to substances that are stored in body cells. Glucose is converted to glycogen in the liver and muscle for future glucose needs, thereby lowering the blood glucose level. The normal range for fasting blood glucose is 70 to 99 mg/dL. When the blood glucose level is greater than 180 mg/dL, glycosuria—glucose in the urine—can occur. Increased blood glucose acts as an osmotic diuretic, causing polyuria. When blood glucose remains elevated (>200 mg/dL), diabetes mellitus occurs.
Hemoglobin A1c (HbA1c), a derivative of the interaction of glucose with hemoglobin in red blood cells (RBCs), is used for the diagnosis of diabetes as recommended by the ADA. Because RBCs have a life span of approximately 120 days, the HbA1c level reflects the average glucose level for up to 3 months. In monitoring treatment, the goal is to keep the diabetic patient’s HbA1c below 7%. For diagnostic purposes, an HbA1c level of 5% or less indicates that the patient does not have diabetes, 5.7% to 6.4% indicates prediabetes, and 6.5% or greater indicates a diagnosis of diabetes mellitus.
Beta-Cell Secretion of Insulin
The beta cells in the pancreas secrete approximately 0.2 to 0.5 units/kg/day of insulin. A patient who weighs 70 kg (154 pounds) secretes 14 to 35 units of insulin per day, although more insulin secretion may occur if the person consumes more calories. A patient with diabetes mellitus may require 0.2 to 1 units/kg/day. The higher range may be because of obesity, stress, or tissue insulin resistance.
 Commercially Prepared Insulin
Commercially Prepared Insulin
Early insulin preparations utilized pancreas tissue extracted from animals—either pigs or cattle. Pork insulin is structurally closer to human insulin than beef insulin. Today, insulins are currently manufactured biosynthetically using recombinant DNA technology. Human insulin duplicates insulin produced by the pancreas of the human body; examples of human insulin include Humulin R and Novolin N. The use of human insulin has a low incidence of both allergic effects and insulin resistance. Human insulin analogues are modifications of human insulin with alterations in onset and duration of action. Insulin lispro and insulin aspart are examples of human insulin analogues.
Insulins are usually administered subcutaneously. Abdominal injections of insulin are absorbed faster than those at other body sites and have been found to be more consistent. Newly diagnosed patients with insulin-dependent diabetes are usually prescribed human insulin. In addition, patients in whom hyperglycemia develops during pregnancy or who already have diabetes and become pregnant are usually prescribed human insulin.
The concentration of insulin is 100 units/mL or 500 units/mL (U100/mL or U500/mL, respectively), and the insulin is packaged in a 10-mL vial. Insulin in 500 units/mL is only available in short-acting regular insulin. Insulin in 500 units/mL is seldom used except in emergencies and for patients with serious insulin resistance (>200 U/day). Insulin in 40 units/mL is no longer used in the United States, although it is still used in other countries. Insulin syringes are typically marked in units of 100 U/mL or 50 U/0.5 mL for insulin U100. Insulin syringes must be used for accurate dosing. To prevent dosage errors, the nurse must be certain that there is a match of the insulin concentration with the calibration of units on the insulin syringe. Before use, the patient or nurse must roll—not shake—cloudy insulin bottles to ensure that the insulin and its ingredients are well mixed. Shaking a bottle of insulin can cause bubbles, which can lead to an inaccurate dose. Insulin requirements vary; usually less insulin is needed with increased exercise, and more insulin is needed with infections and high fever.
Administration of Insulin
Insulin is a protein and cannot be administered orally because gastrointestinal (GI) secretions destroy the insulin structure. It is administered subcutaneously at a 45- to 90-degree angle. The 90-degree angle is made by raising the skin and fatty tissue, and the insulin is injected into the pocket between the fat and the muscle. In a thin person with little fatty tissue, the 45- to 60-degree angle is used. Regular insulin is the only type that can be administered intravenously.
The site and depth of insulin injection affect absorption. Insulin absorption is greater when given in the abdomen than when given in the thigh or buttocks. Heat and massage could increase subcutaneous absorption, and cooling the subcutaneous area can decrease absorption.
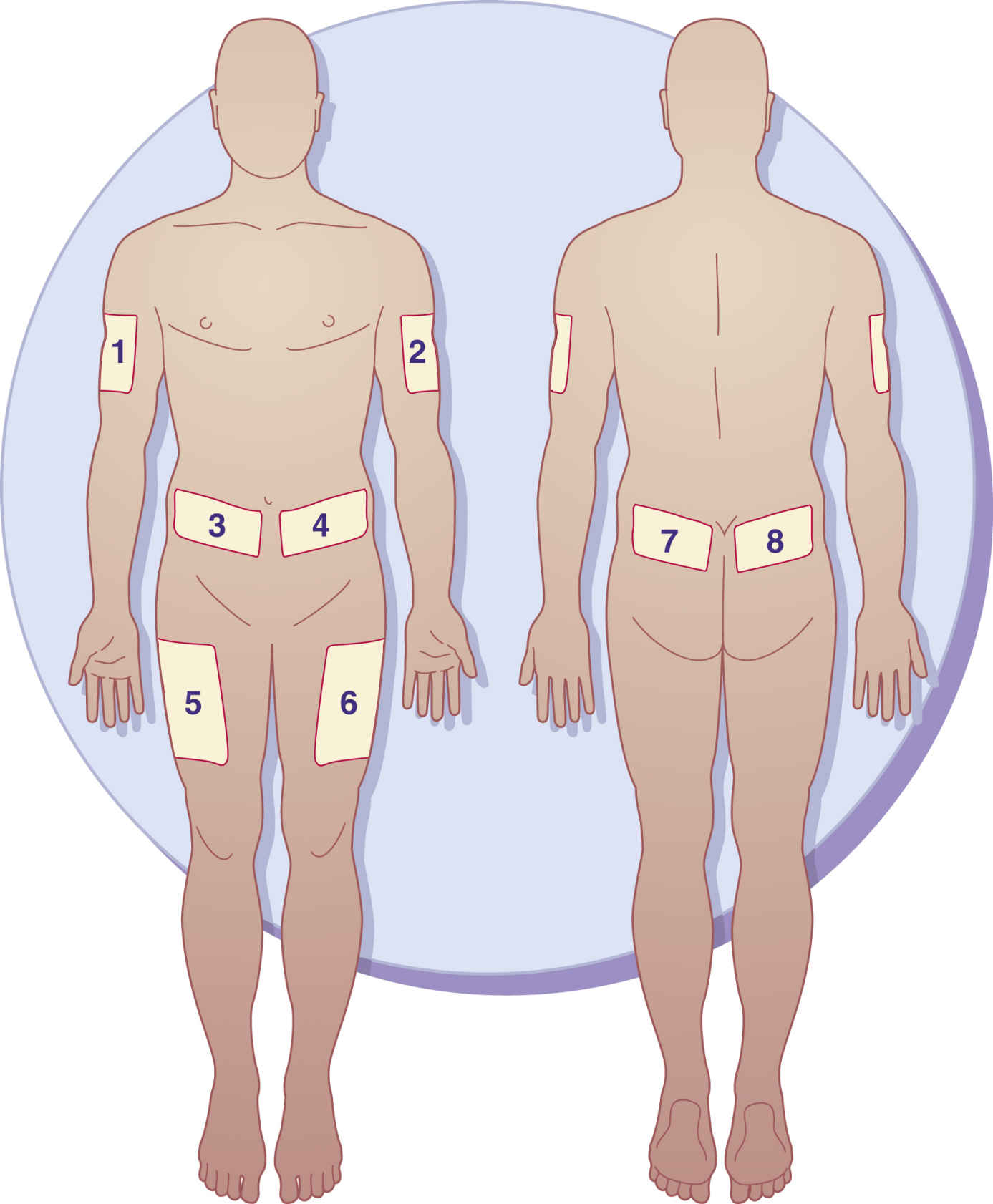
Insulin is usually given in the morning before breakfast, and it can be given several times a day. Insulin injection sites should be rotated to prevent lipodystrophy, tissue atrophy or hypertrophy, which can interfere with insulin absorption. Lipoatrophy, tissue atrophy, is a depression under the skin surface that primarily occurs in women and children; lipohypertrophy, tissue hypertrophy, is a raised lump or knot on the skin surface that is more common in men. It is frequently caused by repeated injections into the same site. The patient needs to develop a “site rotation pattern” to avoid lipodystrophy and to promote insulin absorption. There are various insulin rotation programs, such as an 8-day rotation schedule in which insulin is given at a different site each day. The ADA suggests that insulin be injected daily at a chosen site for 1 week. Injections should be 1.5 inches (a knuckle length) apart each day. When a patient requires two insulin injections a day, morning and evening, one site should be chosen on the right side (morning) and one on the left side (evening). Fig. 47.1 illustrates the sites for insulin injections. A record of injection area sites and dates administered should be kept.
Illness and stress increase the need for insulin, so doses should not be withheld during illness, including infections and stress. Hyperglycemia and ketoacidosis may result from insufficient insulin.
Types of Insulin
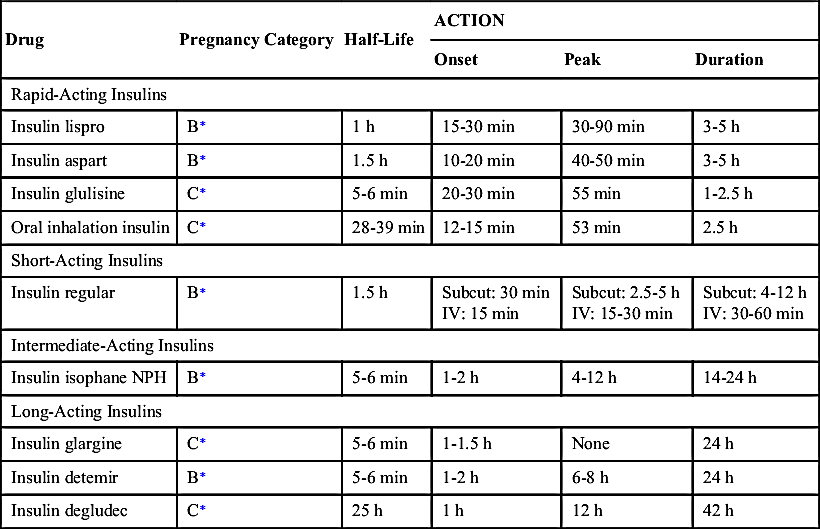
Several standard types of insulin are available that include rapid-, short-, intermediate-, or long-acting types and combinations of these. Rapid- and short-acting insulins are in a clear solution without any added substance to prolong insulin action. Intermediate-acting insulins are cloudy and may contain protamine, a protein that prolongs the action of insulin, or zinc, which also slows the onset of action and prolongs the duration of activity.
Rapid-acting insulins include insulin lispro (human analogue), human insulin aspart (recombinant DNA [rDNA] origin), insulin glulisine, and human oral inhalation insulin. Insulin lispro is formed by reversing two amino acids in human regular insulin. Insulin aspart (rDNA origin) is another human insulin analogue in which a single amino acid (proline) has been substituted with aspartic acid to help prevent the molecules from clumping together to allow quicker entry into blood circulation. Insulin lispro, insulin aspart, insulin glulisine, and human oral inhalation insulin act faster than regular insulin, so they must be administered within 10 to 15 minutes before mealtime (food should be present before administering these insulins). Patients who are insulin dependent and take rapid-acting insulin usually require intermediate-acting insulin as well.
Short-acting insulin has an onset of action of 30 minutes. The peak action occurs in 2.5 to 5 hours, and the duration of action is 4 to 12 hours. Regular (unmodified, crystalline) insulin is short-acting insulin that can be administered intravenously or subcutaneously. Regular insulin is generally given 30 to 60 minutes before meals.
Intermediate-acting insulins include neutral protamine Hagedorn (NPH). Isophane insulins like NPH contain protamine, a protein that prolongs the action of insulin. The onset of intermediate-acting insulin is 1 to 2 hours, peak action occurs in 4 to 12 hours, and the duration of action is 14 to 24 hours.
Insulin glargine is long-acting insulin with an onset of 1 to 1.5 hours. It is evenly distributed over a 24-hour duration of action; thus it is administered once a day, usually at bedtime. The incidence of nocturnal hypoglycemia is not as common as with other insulins because of its continuous sustained release. Insulin detemir is another long-acting insulin that peaks in 6 to 8 hours and lasts for 24 hours, and insulin degludec, a long-acting insulin, has an onset of 1 hour, peaks at 12 hours, and lasts for 42 hours; these insulins are analogues of human insulin. Glargine was the first long-acting rDNA-origin human insulin for patients with types 1 and 2 diabetes. Glargine, detemir, and degludec are available in a prefilled cartridge for the OptiPen insulin pen device. Some patients complain of more pain at the injection site with the administration of glargine than with NPH insulin.
Combination insulins are commercially premixed. These include NPH 70/regular 30 and NPH 50/regular 50. These combinations are widely used. The NPH 70/regular 30 is available in vials or as prefilled disposable pens. The exterior of an insulin pen resembles a fountain pen. It can be stored at room temperature for up to 10 days. With these combinations of insulin, the patient does not have to mix regular and NPH insulins as long as one of these combinations is effective. However, some patients need less than 25% or 30% regular insulin and more intermediate-acting insulin. These patients need to mix the two insulins in the prescribed proportions.
Regular insulin can be mixed with other insulin in the same syringe. However, mixing insulin can alter the absorption rate.
Insulin Resistance
Antibodies develop over time in persons taking animal insulin. This can slow the onset of insulin action and extend its duration of action. Antibody development can cause insulin resistance and insulin allergy, and obesity can also be a causative factor for insulin resistance. Skin tests with different insulin preparations may be performed to determine whether an allergic effect is present. Human and regular insulins produce fewer allergens.
Storage of Insulin
Unopened insulin vials are refrigerated until needed. Once an insulin vial has been opened, it may be kept at room temperature for 1 month or in the refrigerator for 3 months; insulin is less irritating to the tissues when injected at room temperature. Insulin vials should not be put in the freezer, nor should they be placed in direct sunlight or in a high-temperature area. Prefilled syringes should be stored in the refrigerator and should be used within 1 to 2 weeks. Opened insulin vials lose their strength after approximately 3 months.
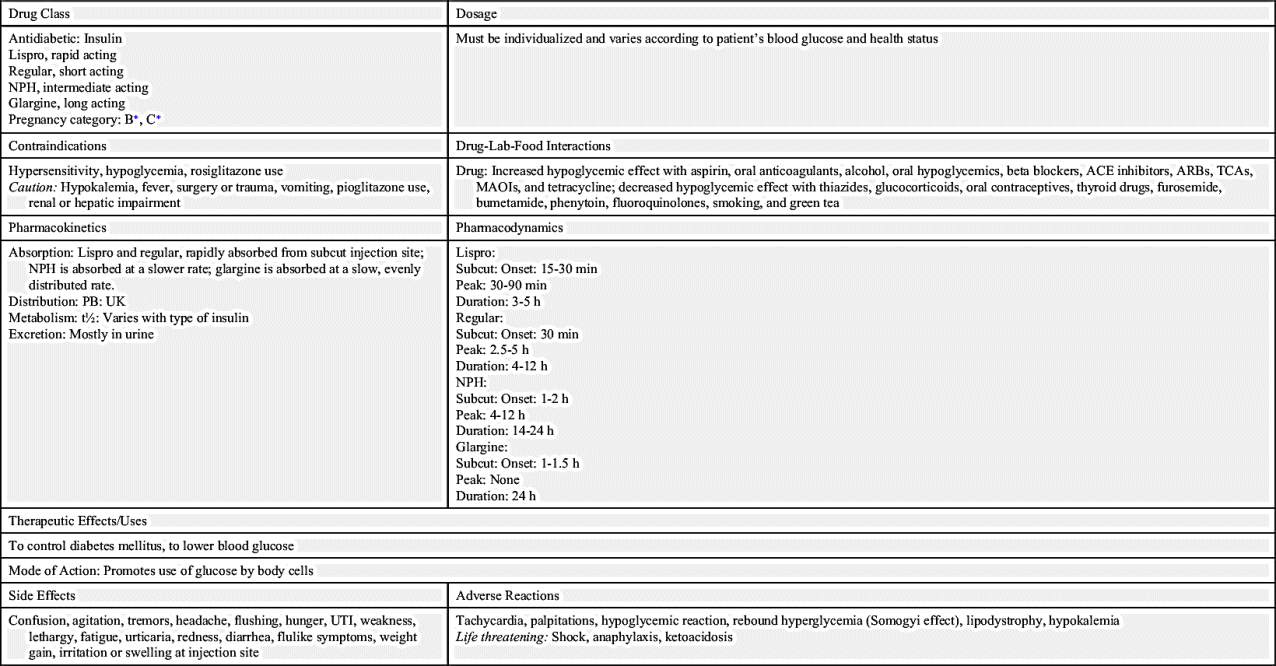
Prototype Drug Chart 47.1 lists the pharmacologic data for the different types of insulin.
Pharmacokinetics
All insulins can be administered subcutaneously, but only regular insulin can be given intravenously. The half-life varies. Insulin is metabolized by the liver and muscle and is excreted in the urine.
Pharmacodynamics
Insulin lowers blood glucose by promoting the use of glucose by the body’s cells. Insulin is also active in the storage of glucose as glycogen in muscles. The onset of action of rapid-acting insulin given subcutaneously is approximately 10 to 30 minutes. The onset of action of regular insulin is 30 minutes given subcutaneously and 15 minutes given intravenously. The onset of action of intermediate-acting insulin is 1.5 hours. The peak action of insulins is important because of the possibility of a hypoglycemic reaction (insulin shock) occurring during that time. The peak action for rapid-acting insulin is approximately 30 to 90 minutes. The peak time for regular insulin is 1.5 to 3.5 hours; for intermediate-acting insulin, the peak time is 4 to 12 hours. The nurse needs to assess for signs and symptoms of hypoglycemic reaction, such as nervousness, tremors, confusion, sweating, and increased pulse rate. Orange juice, sugar-sweetened beverages, or hard candy should be kept available to be given if a reaction occurs. If the patient is unable to ingest fast-acting carbohydrates, glucagon may be given. Glucagon promotes the breakdown of glycogen in the liver and raises blood glucose within 10 minutes.
Regular insulin can be given several times a day, especially during the regulation of insulin dosage. Intermediate- and long-acting insulins are usually administered once a day. Regular insulin can be mixed with intermediate-acting NPH insulin, especially if rapid onset of action is needed. When switching from one type of insulin to another, the patient may require a dose adjustment because human insulin has a shorter duration of action.
Sliding-Scale Insulin Coverage
Insulin may be administered in adjusted doses that depend on individual blood glucose test results. When the diabetic patient has extreme variances in insulin requirements—such as with stress from hospitalization, surgery, illness, or infection—adjusted dosing or sliding-scale insulin coverage provides a more constant blood glucose level. Blood glucose testing is performed several times a day at specified intervals, usually before meals. A preset scale usually involves directions for the administration of rapid- or short-acting insulin.
Drug Interactions
Drugs such as thiazide diuretics, glucocorticoids (cortisone preparations), thyroid agents, and estrogen increase blood glucose, therefore the insulin dosage may need adjustment. Drugs that decrease insulin needs are tricyclic antidepressants (TCAs), monoamine oxidase inhibitors (MAOIs), aspirin products, and oral anticoagulants.
Table 47.2 lists the rapid-, short-, intermediate-, and long-acting insulins with their dosages, uses, and considerations.
Side Effects and Adverse Reactions: Hypoglycemic Reactions and Ketoacidosis
When more insulin is administered than is needed for glucose metabolism, a hypoglycemic reaction, or insulin shock, occurs. The person may exhibit nervousness, trembling, and lack of coordination; have cold and clammy skin; and complain of a headache. Some patients become combative and incoherent. Giving sugar orally or intravenously increases the use of insulin, and the symptoms disappear immediately.
In response to an excessive dose of insulin, diabetic patients may develop what is known as the Somogyi effect, a hypoglycemic condition that usually occurs in the predawn hours of 2:00 am to 4:00 am wherein a rapid decrease in blood glucose during the nighttime hours stimulates a release of hormones (cortisol, glucagon, and epinephrine) to increase blood glucose by lipolysis, gluconeogenesis, and glycogenolysis. Management of the Somogyi effect involves monitoring blood glucose between 2:00 am and 4:00 am and reducing the bedtime insulin dosage.
Hyperglycemia on awakening is known as the dawn phenomenon. The patient usually awakens with a headache and reports night sweats and nightmares. Management of the dawn phenomenon involves increasing the bedtime dose of insulin.
With an inadequate amount of insulin, sugar cannot be metabolized, and fat catabolism occurs. The use of fatty acids (ketones) for energy causes ketoacidosis (diabetic acidosis or diabetic coma). Table 47.3 lists the signs and symptoms of hypoglycemic reaction and ketoacidosis.
Insulin Pen Injectors
An insulin pen resembles a fountain pen but contains a disposable needle and a disposable insulin-filled cartridge. Insulin pens come in two types, prefilled and reusable, and are considered to deliver a more accurate dose than the traditional 100-unit syringe and vial.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree