DRUG CLASSES
Selective serotonin reuptake inhibitors (SSRIs)
Serotonin/norepinephrine or dopamine/norepinephrine reuptake inhibitors (SNRIs or DNRIs)
Tricyclic antidepressants (TCAs)
Monoamine oxidase inhibitors (MAOIs)
PHARMACOLOGY IN PRACTICE

Mr. Phillip has been severely depressed for about 2 months, following the death of his wife in a car accident. One week ago, the primary care provider prescribed Zoloft 100 mg orally daily. His family is concerned because he is still depressed. Mr. Phillip’s daughter is concerned because he feels drowsy and can’t seem to get up until 11 a.m. or noon on most days. They are requesting that the dosage be increased. Read about the antidepressants and determine what should happen next.
Depression may be described as feeling sad, unhappy, or “down in the dumps.” Most of us feel this way at one time or another for short periods. Major depressive disorder is the medical diagnosis for one of the mood disorders (a spectrum of conditions that range from severe debilitation to an exhaustive elation). Also called clinical depression by many people, symptoms are not the result of normal bereavement, such as the loss of a loved one, or another disease, such as hypothyroidism. When the major depressive episode is a depressed or dysphoric (extreme or exaggerated sadness, anxiety, or unhappiness) mood that interferes with daily functioning, it is termed unipolar depression. For a diagnosis of major depressive disorder, five or more of the symptoms listed in Display 22.1 need to occur daily or nearly every day for a period of 2 weeks or more.
The depressive symptoms of mood disorders are treated with antidepressant drugs and psychotherapy. The four classes of antidepressants are:
• Selective serotonin reuptake inhibitors (SSRIs)
• Serotonin/norepinephrine or dopamine/norepinephrine reuptake inhibitors (SNRIs or DNRIs)
• Tricyclic antidepressants (TCAs)
• Monoamine oxidase inhibitors (MAOIs)
For several years it was thought that antidepressants blocked the reuptake of the endogenous (produced by the body) neurotransmitters norepinephrine and serotonin. This action resulted in stimulation of the central nervous system (CNS) and alleviation of the depressed mood. This theory is now being questioned. Research indicates that the effects of antidepressants are related to slow adaptive changes in norepinephrine and serotonin receptor systems (Fuller, 1985). Treatment with antidepressants is thought to produce complex changes in the sensitivities of both presynaptic and postsynaptic receptor sites. Antidepressants increase the sensitivity of postsynaptic alpha (α)-adrenergic and serotonin receptors and decrease the sensitivity of the presynaptic receptor sites. This enhances recovery from the depressive episode by making neurotransmission activity more effective, as visualized in Figure 22.1.
Display 22.1 Symptoms of Depression
• Feelings of hopelessness or helplessness
• Diminished interest in activities of life
• Significant weight loss or gain (without dieting)
• Insomnia (inability to sleep) or hypersomnia (excessive sleeping)
• Agitation, restlessness, or irritability
• Fatigue or loss of energy
• Feelings of worthlessness
• Excessive or inappropriate guilt
• Diminished ability to think or concentrate, or indecisiveness
• Recurrent thoughts of death or suicide (or suicide attempt)
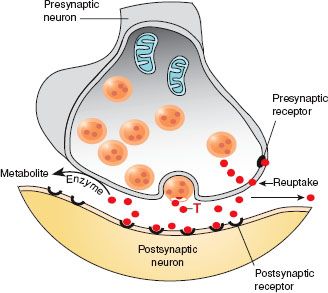
Figure 22.1 Antidepressant drugs inhibit the reuptake of the neurotransmitter (T), thus making it available to the postsynaptic neuron.
Selective Serotonin Reuptake Inhibitors
Actions
The SSRIs inhibit CNS uptake of serotonin (a CNS neurotransmitter). The increase in serotonin levels is thought to act as a stimulant to reverse depression.
Uses
The SSRIs are used in the treatment of the following:
• Depressive episodes
• Obsessive-compulsive disorder (OCD)
• Bulimia nervosa
Unlabeled uses include panic disorders, premenstrual syndrome and posttraumatic stress disorders, generalized anxiety and social phobias, Raynaud’s disease, migraine headaches, diabetic neuropathy, and hot flashes. These drugs may be used with psychotherapy in severe cases.
Adverse Reactions
Neuromuscular System Reactions
• Somnolence, dizziness
• Headache, insomnia
• Tremor, weakness
Gastrointestinal and Genitourinary System Reactions
• Constipation, dry mouth, nausea
• Pharyngitis and runny nose
• Urinary retention and abnormal ejaculation
Contraindications
The SSRIs are contraindicated in patients with a hypersensitivity to the drugs and during pregnancy (pregnancy category C). Patients taking cisapride (Propulsid), pimozide (Orap), or carbamazepine (Tegretol) should not take fluoxetine (Prozac). Due to a specific enzyme reaction, grapefruit or its juice should not be consumed if the patient is on sertraline (Zoloft).
Precautions
The SSRI antidepressants should be used cautiously in patients with type 2 diabetes, cardiac disease, or impaired liver or kidney function and in those at risk for suicidal ideation or behavior. Patients should not be switched to an SSRI antidepressant drug within 2 weeks of stopping an MAOI antidepressant.
Interactions
The following interactions may occur when an SSRI is administered with another agent:
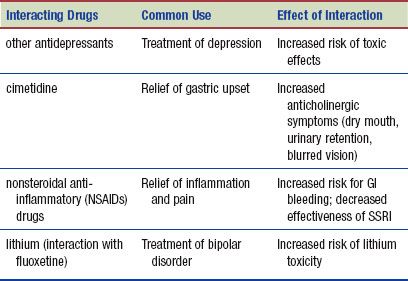
Note that the effectiveness of fluoxetine is decreased in patients who smoke cigarettes during administration of the drug.
Serotonin/Norepinephrine or dopamine/Norepinephrine Reuptake Inhibitors
Actions
It is thought that the mechanism of action of most of the SNRIs/DNRIs is to affect neurotransmission of serotonin, norepinephrine, and dopamine. Examples in this group of drugs include venlafaxine (Effexor XR) and bupropion (Wellbutrin).
Uses
SNRIs/DNRIs may be used in conjunction with psychotherapy in severe cases or alone in the treatment of the following:
• Depressive episodes
• Depression accompanied by anxiety disorders
• Diabetic peripheral neuropathic pain
Unlabeled uses include enhancing weight loss and treating aggressive behaviors, neuropathic pain, menstrual disorders, cocaine withdrawal and alcohol cravings, fibromyalgia, and stress incontinence.
Adverse Reactions
Neuromuscular System Reactions
• Somnolence, migraine headache
• Hypotension, dizziness, lightheadedness, and vertigo
• Blurred vision, photosensitivity, insomnia, nervousness or agitation, and tremor
Gastrointestinal System Reactions
• Nausea, dry mouth, anorexia, thirst
• Diarrhea, constipation, bitter taste
Other System Reactions
• Fatigue, tachycardia, and palpitations
• Change in libido, impotence
• Skin rash, itching, vasodilation resulting in flushing and excessive sweating
Additional adverse reactions and adverse reactions associated with the use of all the antidepressant drugs are listed in the Summary Drug Table: Antidepressants.
Contraindications
SNRIs/DNRIs are contraindicated in patients with known hypersensitivity to the drugs. Among the SNRIs/DNRIs antidepressants, bupropion and maprotiline are pregnancy category B drugs. Other antidepressants discussed in this section are in pregnancy category C. Safe use of the antidepressants during pregnancy has not been established. They should be used during pregnancy only when the potential benefits outweigh the potential hazards to the developing fetus. Maprotiline should not be used with patients who have a seizure disorder or during the acute phase of a myocardial infarction. Patients taking cisapride, pimozide, or carbamazepine should not take nefazodone because of risk for hepatic failure. Due to a specific enzyme reaction, grapefruit or its juice should not be taken if the patient is on vilazodone or trazodone.
 NURSING ALERT
NURSING ALERT
The smoking cessation product Zyban is a form of the antidepressant drug bupropion. Smokers should not use Zyban if they are currently taking bupropion for management of depression because of the possibility of bupropion overdose.
Precautions
SNRIs/DNRIs should be used cautiously in patients with cardiac disease, renal or hepatic impairment, or hyperthyroid disease or those who are at risk of suicidal ideation or behavior.
Interactions
The following interactions may occur when an SNRI/DNRI is administered with another agent:
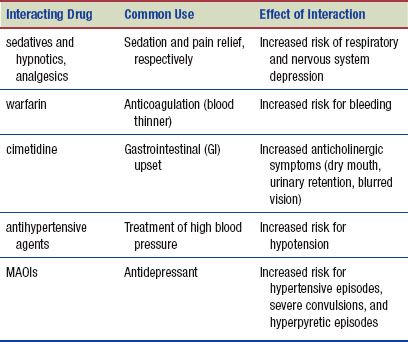
Discovery of the role of dopamine in depression and the ability to be more selective in reuptake make the following classes of drugs less frequently prescribed than those already discussed.
TRICYCLIC ANTIDEPRESSANTS
Actions
The TCAs, such as amitriptyline and doxepin, inhibit reuptake of norepinephrine or serotonin in the brain.
Uses
Tricyclic antidepressant drugs are used in the treatment of the following:
• Depressive episodes
• Bipolar disorder
• OCD
• Chronic neuropathic pain
• Depression accompanied by anxiety disorders
• Enuresis
Unlabeled uses include peptic ulcer disease, sleep apnea, panic disorder, bulimia nervosa, premenstrual symptoms, and some dermatologic problems. These drugs may be used with psychotherapy in severe cases.
Adverse Reactions
Generalized reactions include:
• Anticholinergic effects (e.g., sedation, dry mouth, visual disturbances, urinary retention)
• Constipation and photosensitivity
 NURSING ALERT
NURSING ALERT
Although the TCAs are not considered antipsychotic agents, the drug amoxapine has been associated with tardive dyskinesia and neuroleptic malignant syndrome (NMS). Tardive dyskinesia is a syndrome of involuntary movement that may be irreversible. Symptoms of NMS are similar and include muscle rigidity, altered mental status, and autonomic system problems, such as tachycardia or sweating. These syndromes tend to occur more readily in elderly women; the drug should be discontinued, the primary health care provider notified immediately, and treatment of adverse effects begun quickly.
Contraindications
The TCAs are contraindicated in patients with known hypersensitivity to the drugs. The TCAs are not given within 14 days of the MAOI antidepressants, to patients with a recent myocardial infarction, or to children or lactating mothers. These drugs are in pregnancy categories C and D, and the safety of their use during pregnancy has not been established. Doxepin is contraindicated in patients with glaucoma or in those with a tendency for urinary retention.
Precautions
The TCAs should be used cautiously in patients with cardiac disease, hepatic or renal impairment, hyperthyroid disease, history of seizure activity, narrow-angle glaucoma or increased intraocular pressure, or urinary retention and in those at risk of suicidal ideation or behavior.
 NURSING ALERT
NURSING ALERT
The TCAs can cause cardiac-related adverse reactions, such as tachycardia and heart block. Give these drugs with caution to older adults or the person with pre-existing cardiac disease.
Interactions
The following interactions may occur when a TCA is administered with another agent:
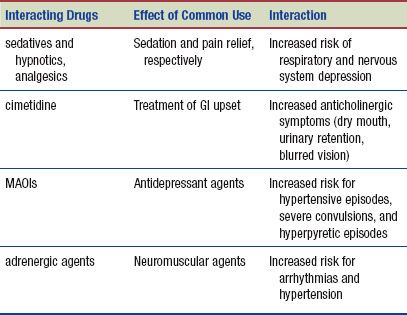
MONOAMINE OXIDASE INHIBITORS
Actions
Drugs classified as MAOIs inhibit the activity of monoamine oxidase, a complex enzyme system responsible for inactivating certain neurotransmitters. Blocking monoamine oxidase results in an increase in endogenous epinephrine, norepinephrine, dopamine, and serotonin in the nervous system. An increase in these neurohormones (secreted rather than transmitted neurosubstances) stimulates the CNS.
Uses
The MAOI antidepressants are used in the treatment of depressive episodes and may be used in conjunction with psychotherapy in severe cases. Unlabeled uses include bulimia, night terrors, migraine headaches, seasonal affective disorder, and multiple sclerosis.
Adverse Reactions
• Neuromuscular reactions include orthostatic hypotension, dizziness, vertigo, headache, and blurred vision.
• GI and genitourinary system reactions include constipation, dry mouth, nausea, diarrhea, and impotence.
• A serious adverse reaction associated with MAOIs is hypertensive crisis (extremely high blood pressure), which may occur when foods containing tyramine (an amino acid) are eaten (see Display 22.2 for a list of foods containing tyramine).
Display 22.2 Foods Containing Tyramine to Be Avoided if Taking MAOI Antidepressant
• Aged cheese (e.g., blue, Camembert, cheddar, Emmenthaler, mozzarella, parmesan, Romano, Stilton, Swiss)
• Sour cream
• Yogurt
• Beef or chicken livers
• Pickled herring
• Fermented meats (e.g., bologna, pepperoni, salami, dried fish)
• Undistilled alcoholic beverages (e.g., imported beer; ale; red wine, especially Chianti and sherry)
• Caffeinated beverages (e.g., coffee, tea, colas)
• Chocolate
• Certain fruits and vegetables (e.g., avocado, bananas, fava beans, figs, raisins, sauerkraut)
• Yeast extracts
• Soy sauce
 NURSING ALERT
NURSING ALERT
One of the earliest symptoms of hypertensive crisis is headache (usually occipital), followed by a stiff or sore neck, nausea, vomiting, sweating, fever, chest pain, dilated pupils, and bradycardia or tachycardia. If a hypertensive crisis occurs, immediate medical intervention is necessary to reduce the blood pressure. Strokes (cerebrovascular accidents) and death have been reported.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


