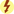http://evolve.elsevier.com/McCuistion/pharmacology/
This chapter discusses the antibacterials and their effects and includes mechanisms of antibacterial action, body defenses, resistance to antibacterials, use of antibacterial combinations, general adverse reactions to antibacterials, and narrow- and broad-spectrum antibiotics.
The groups of antibacterials discussed in this chapter include penicillins, cephalosporins, macrolides (erythromycin, clarithromycin, and azithromycin), lincosamides, glycopeptides, ketolides, tetracyclines, glycylcyclines, aminoglycosides, fluoroquinolones (quinolones), lipopeptides, sulfonamides, and nitroimidazoles.
The penicillins, macrolides, lincosamides, tetracyclines, and sulfonamides are primarily bacteriostatic drugs, those that inhibit bacterial growth, but they may also be bactericidal (bacteria killing), depending on the drug dose, serum level, and the pathogen (the disease-producing microorganism). Cephalosporins, glycopeptides, aminoglycosides, and fluoroquinolones are bactericidal drugs.
Macrolides, lincosamides, glycopeptides, and ketolides are discussed together because they have spectrums of antibiotic effectiveness similar to that of penicillin, although they differ in structure. Drugs from these groups are used as penicillin substitutes, especially in individuals who are allergic to penicillin. Erythromycin is the drug frequently prescribed if the patient has a hypersensitivity to penicillin.
Sulfonamides are one of the oldest antibacterial agents used to combat infection. When penicillin was initially marketed, the sulfonamide drugs were not widely prescribed because penicillin was considered a “miracle drug.” However, use of sulfonamides has increased as a result of newer sulfonamides and drugs that combine a sulfonamide with an antibacterial agent in preparations such as trimethoprim-sulfamethoxazole.
Pathophysiology
Bacteria, known as prokaryotes, are single-celled organisms that lack a true nucleus and nuclear membrane. Most bacteria have a rigid cell wall, and the structure of the cell wall determines the shape of the bacteria. One classification of bacteria involves the appearance or shape under a microscope. A bacillus is a rod-shaped organism, and cocci are spherical. When cocci appear in clusters, they are called staphylococci; when they are arranged in chains, they are called streptococci. Bacteria reproduce by cell division ranging from 12 minutes to 24 hours.
Another classification of bacteria involves staining properties of the cell. The Gram-staining method was devised in 1882 by Hans Christian Gram, a Danish bacteriologist. Gram staining determines the ability of the bacterial cell wall to retain a purple stain by a basic dye. Crystal violet is normally used in the staining process but may be substituted with methylene blue. If bacteria retain a purple stain, they are classified as gram-positive microorganisms. Those bacteria not stained are known as gram-negative microorganisms. Examples of gram-positive bacteria include Staphylococcus aureus, Streptococcus pneumoniae, group B Streptococcus (GBS), and Clostridium perfringens. Examples of gram-negative bacteria include Neisseria meningitides, Escherichia coli, and Haemophilus influenzae.
Bacteria produce toxins that cause cell lysis (cell breakdown). Many bacteria produce the enzyme beta-lactamase, which destroys beta-lactam antibiotics such as penicillins and cephalosporins.
Antibacterial Drugs
Antibacterials/Antibiotics
Although the terms antibacterial, antimicrobial, and antibiotic are frequently used interchangeably, there are some subtle differences in meaning. Antibacterials and antimicrobials are substances that inhibit bacterial growth or kill bacteria and other microorganisms—microscopic organisms that include viruses, fungi, protozoa, and rickettsiae. Technically, the term antibiotic refers to chemicals produced by one kind of microorganism that inhibit the growth of or kill another. For practical purposes, however, these terms may be used interchangeably. Several drugs, including antiinfective and chemotherapeutic agents, have actions similar to those of antibacterial and antimicrobial agents. Antibacterial drugs do not act alone in destroying bacteria. Natural body defenses, surgical procedures to excise infected tissues, and dressing changes may be needed along with antibacterial drugs to eliminate the infecting bacteria.
Antibacterial drugs are either obtained from natural sources or are manufactured. The use of moldy bread on wounds to fight infection dates back 3500 years. In 1928, British bacteriologist Alexander Fleming noted that a mold that had contaminated his bacterial cultures was inhibiting bacterial growth. The mold was Penicillium notatum, thus Fleming called the substance penicillin. Sulfonamide, a synthetic antibacterial, was introduced in 1935. In 1939, Howard Florey expanded on Fleming’s findings and purified penicillin so it could be used commercially. Penicillin was used during World War II and was marketed in 1945.
TABLE 26.1
Mechanisms of Action of Antibacterial Drugs
| Action | Effect | Drugs |
| Inhibition of cell-wall synthesis | Bactericidal effect Enzyme breakdown of cell wall Inhibition of enzyme in synthesis of cell wall | Penicillin Cephalosporins Bacitracin Vancomycin |
| Alteration of membrane permeability | Bacteriostatic or bactericidal effect Increases membrane permeability Cell lysis caused by loss of cellular substances | Amphotericin B Nystatin Polymyxin Colistin |
| Inhibition of protein synthesis | Bacteriostatic or bactericidal effect Interferes with protein synthesis without affecting normal cell Inhibits steps of protein synthesis | Aminoglycosides Tetracyclines Erythromycin Lincomycin |
| Inhibition of synthesis of bacterial RNA and DNA | Inhibits synthesis of RNA and DNA in bacteria Binds to nucleic acid and enzymes needed for nucleic acid synthesis | Fluoroquinolones |
| Interference with cellular metabolism | Bacteriostatic effect Interferes with steps of metabolism within cells | Sulfonamides Trimethoprim Isoniazid Nalidixic acid Rifampin |
For drugs with a narrow therapeutic index, such as the aminoglycosides, peaks and troughs of serum antibiotic levels are monitored to determine whether the drug is within the therapeutic range for its desired effect. If the serum peak level is too high, drug toxicity could occur. If the serum trough level (drawn minutes before administration of the next drug dose) is below the therapeutic range, the patient is not receiving an adequate antibiotic dose to kill the targeted microorganism.
Mechanisms of Antibacterial Action
Five mechanisms of antibacterial action are responsible for the inhibition of growth or destruction of microorganisms: (1) inhibition of bacterial cell-wall synthesis, (2) alteration of membrane permeability, (3) inhibition of protein synthesis, (4) inhibition of the synthesis of bacterial ribonucleic acid (RNA) and deoxyribonucleic acid (DNA), and (5) interference with metabolism within the cell (Table 26.1).
Pharmacokinetics
Antibacterial drugs must not only penetrate the bacterial cell wall in sufficient concentrations but also have an affinity for (attraction to) the binding sites on the bacterial cell. The length of time the drug remains at the binding sites increases the effect of the antibacterial action. This time factor is controlled by the pharmacokinetics—the distribution, half-life, and elimination—of the drug.
Antibacterials that have a longer half-life usually maintain a greater concentration at the binding site, therefore frequent dosing is not required. Most antibacterials are not highly protein bound, with a few exceptions (e.g., oxacillin, ceftriaxone, cefprozil, cloxacillin, nafcillin, clindamycin). Protein binding does not have a major influence on the effectiveness of most antibacterial drugs. The steady state of the antibacterial drug occurs after the fourth to fifth half-lives, and after the seventh half-life, the drug is eliminated from the body, mainly through urine.
Pharmacodynamics
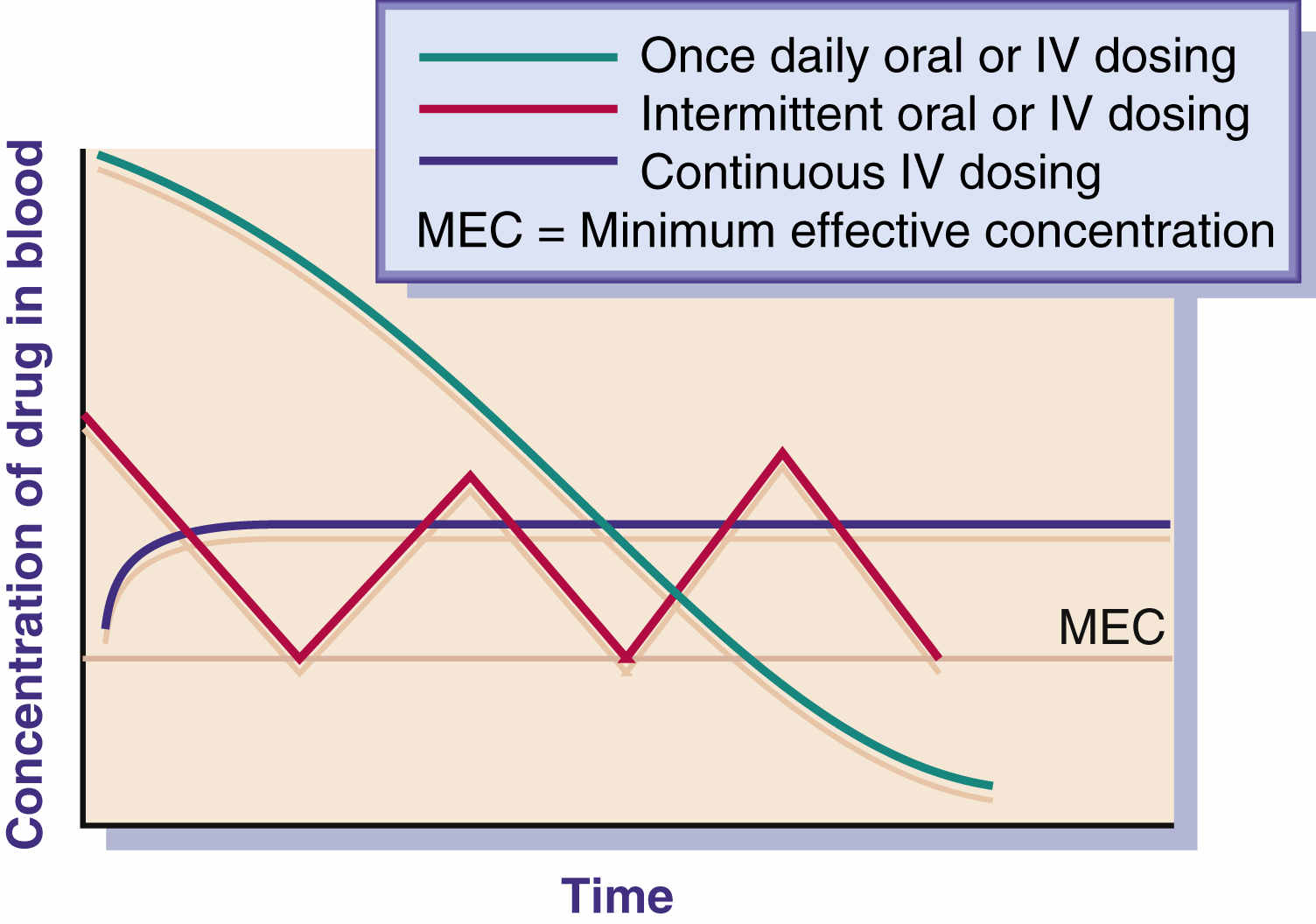
The drug concentration at the site and the exposure time for the drug play important roles in bacterial eradication. Antibacterial drugs are used to achieve the minimum effective concentration (MEC) necessary to halt the growth of a microorganism. Many antibacterials have a bactericidal effect against the pathogen when the drug concentration remains constantly above the MEC during the dosing interval. Duration of use of the antibacterial varies according to the type of pathogen, site of infection, and immunocompetence of the host. With some severe infections, a continuous infusion regimen is more effective than intermittent dosing because of constant drug concentration and time exposure. Once-daily antibacterial dosing—such as with aminoglycosides, macrolides, and fluoroquinolones—has been effective in eradicating pathogens and has not caused severe adverse reactions (ototoxicity, nephrotoxicity) in most cases. The ease of compliance with once- or twice-daily drug dosing also increases the patient’s adherence to the drug regimen.
Fig. 26.1 illustrates the effect of three methods of drug dosing. The drug dose is effective when it remains above the MEC.
Body Defenses
Body defenses and antibacterial drugs work together to stop the infectious process. The effect that antibacterial drugs have on an infection depends not only on the drug but also on the host’s defense mechanisms. Factors such as age, nutrition, immunoglobulins, white blood cells (WBCs), organ function, and circulation influence the body’s ability to fight infection. Older adults and undernourished individuals have less resistance to infection than younger, well-nourished populations. If the host’s natural body defense mechanisms are inadequate, drug therapy might not be as effective. As a result, drug therapy may need to be closely monitored or revised. When circulation is impeded, an antibacterial drug may not be distributed properly to the infected area. In addition, immunoglobulins—antibody proteins such as IgG and IgM—and other elements of the immune response system, such as WBCs needed to combat infections, may be depleted in individuals with poor nutritional status.
Resistance to Antibacterials
Bacteria can be either sensitive or resistant to certain antibacterials. When bacteria are sensitive to a drug, the pathogen is inhibited or destroyed; however, if bacteria are resistant, the pathogen continues to grow despite administration of that antibacterial drug.
Bacterial resistance can result naturally, called inherent resistance, or it may be acquired. A natural, or inherent, resistance occurs without previous exposure to the antibacterial drug. For example, the gram-negative bacterium Pseudomonas aeruginosa is inherently resistant to penicillin G. An acquired resistance is caused by prior exposure to the antibacterial. Although S. aureus was once sensitive to penicillin G, repeated exposures have caused this organism to evolve and become resistant to that drug. Penicillinase, an enzyme produced by the microorganism, is responsible for causing its penicillin resistance. Penicillinase metabolizes penicillin G, causing the drug to be ineffective; however, penicillinase-resistant penicillins that are effective against S. aureus are now available.
Antibiotic resistance is a major problem. In the early 1980s, pharmaceutical companies thought that enough antibiotics were on the market, so they concentrated on developing antiviral and antifungal drugs. As a result, fewer new antibiotics were developed during the 1980s. Now pharmaceutical companies have developed many new antibiotics, but antibiotic resistance continues to develop, especially when antibiotics are used frequently. As bacteria reproduce, some mutation occurs, and eventually the mutant bacteria survive the effects of the drug. One explanation is that the mutant bacterial strain may have grown a thicker cell wall.
In large health care institutions, there is a tendency toward drug resistance in bacteria. Mutant strains of organisms have developed, thus increasing their resistance to antibiotics that were once effective against them. Infections acquired while patients are hospitalized are called health care acquired infections (previously known as nosocomial infections). Many of these infections are caused by drug-resistant bacteria, and this can prolong hospitalization, which is costly to both the patient and third-party health care insurers.
Another problem related to antibiotic resistance is that bacteria can transfer their genetic instructions to another bacterial species, and the other bacterial species then becomes resistant to that antibiotic as well. Bacteria can also pass along high resistance to a more virulent and aggressive bacterium (e.g., S. aureus, enterococci).
Methicillin was the first penicillinase-resistant penicillin; it was developed in 1959 in response to the resistance of S. aureus. In 1968, strains of S. aureus were beginning to become resistant to methicillin. Highly resistant bacteria, so-called methicillin-resistant S. aureus (MRSA), became resistant not only to methicillin but to all penicillins and cephalosporins as well. Resistance that was once found only in hospitals began to emerge in 1981 in the community. Methicillin is now off the market, and the treatment of choice for MRSA is vancomycin. Other effective drugs used to treat MRSA include linezolid, tedizolid, daptomycin, trimethoprim-sulfamethoxazole, doxycycline, clindamycin, and televancin, a glycopeptide antiinfective.
Many enterococcal strains are resistant to penicillin, ampicillin, gentamicin, streptomycin, and vancomycin. Another big resistance problem is vancomycin-resistant Enterococcus faecium (VREF), which can cause death in persons with weakened immune systems. The incidence of VREF in hospitals has increased, and a strain of MRSA has also been reported to be resistant to vancomycin (vancomycin-resistant S. aureus, or VRSA). One antibiotic after another is ineffective against new resistant strains of bacteria, and major medical problems result. As new drugs are developed, drug resistance will probably also develop.
Pharmaceutical companies and biotech firms are working on new classes of drugs to overcome the problem of bacterial resistance to antibiotics. A class of antibiotics known as oxazolidinones was discovered by a pharmaceutical company in 1988, but the company could not overcome the toxicity problems with this class of drug; however, another pharmaceutical company has taken the compound and made it less toxic. This antibiotic, linezolid, is effective against MRSA, VREF, and penicillin-resistant streptococci. Quinupristin-dalfopristin, which consists of two streptogramin antibacterials, is marketed in a 30:70 combination for intravenous (IV) use against life-threatening infection caused by VREF and for treatment of bacteremia, S. aureus, and Streptococcus pyogenes.
Another way to attack antimicrobial resistance is to develop drugs that disable the antibiotic-resistant mechanism in the bacteria. Patients would take the antibiotic-resistance disabler along with the antibiotic already on the market, making the drug effective again. Developing a bacterial vaccine is another way to combat bacteria and lessen the need for antibiotics. The bacterial vaccine against pneumococcus has been effective in decreasing the occurrence of pneumonia and meningitis among various age groups.
Antibiotic misuse, a major problem today, increases antibiotic resistance. Studies reveal that 23% to 37.8% of patients in hospitals receive antibiotics, and 50% of this population is receiving antibiotics inappropriately. When antibiotics are taken unnecessarily—such as for viral infections, when no bacterial infection is present—or incorrectly (e.g., skipping doses, not taking the full antibiotic regimen), resistance to antibacterials may develop. Consumer education is important because many patients demand antibiotics for viral conditions, even though antibiotics are ineffective against viruses; however, viral infections that persist could compromise the body’s immune system and thus promote a secondary bacterial infection. The nurse should teach patients about the proper use of antibiotics to prevent situations that promote drug resistance to bacteria.
Cross-resistance can also occur among antibacterial drugs that have similar actions, such as the penicillins and cephalosporins. To ascertain the effect antibacterial drugs have on a specific microorganism, culture and sensitivity (C&S) or antibiotic susceptibility laboratory testing is performed. C&S can detect the infective microorganism present in a sample (e.g., blood, sputum, swab) and the best drug to kill it. The organism causing the infection is determined by culture, and antibiotics the organism is sensitive to are determined by sensitivity. The susceptibility or resistance of one microorganism to several antibacterials can be determined by the C&S test. Multiantibiotic therapy, or daily use of several antibacterials, delays the development of microorganism resistance.
Use of Antibiotic Combinations
Combination antibiotics should not be routinely prescribed or administered except for specific uncontrollable infections. Usually a single antibiotic will successfully treat a bacterial infection; however, when severe infection persists and is of an unknown origin or has been unsuccessfully treated with several single antibiotics, a combination of two or three antibiotics may be suggested. Before beginning antibiotic therapy, a culture or cultures should be taken to identify the bacteria.
When two antibiotics are combined, the result may be additive, potentiative, or antagonistic. The additive effect is equal to the sum of the effects of two antibiotics. The potentiative effect occurs when one antibiotic potentiates the effect of the second antibiotic, increasing its effectiveness. The antagonistic result is achieved with combination of a drug that is bactericidal, such as penicillin, and a drug that is bacteriostatic, such as tetracycline; when these two drugs are used together, the desired effect may be greatly reduced.
General Adverse Reactions to Antibacterials
Three major adverse reactions associated with the administration of antibacterial drugs are (1) allergic (hypersensitivity, anaphylaxis) reactions, (2) superinfection, and (3) organ toxicity. Table 26.2 describes these adverse reactions, all of which require close monitoring of the patient.
Narrow-Spectrum and Broad-Spectrum Antibiotics
Antibacterial drugs can be classified as either narrow spectrum or broad spectrum. The narrow-spectrum antibiotics are primarily effective against one type of organism. For example, penicillin and erythromycin are used to treat infections caused by gram-positive bacteria. Certain broad-spectrum antibiotics, such as tetracycline and cephalosporins, can be effective against both gram-positive and gram-negative organisms. Because narrow-spectrum antibiotics are selective, they are more active against those single organisms than the broad-spectrum antibiotics. Broad-spectrum antibiotics are frequently used to treat infections when the offending microorganism has not been identified by the C&S test.
TABLE 26.2
General Adverse Reactions to Antibacterial Drugs
| Type | Considerations |
| Allergy or hypersensitivity | Allergic reactions to drugs may be mild or severe. Examples of mild reactions are rash, pruritus, and hives. An example of a severe response is anaphylactic shock, which results in vascular collapse, laryngeal edema, bronchospasm, and cardiac arrest. Severe allergic reaction generally occurs within 20 minutes, and shortness of breath is often the first symptom of anaphylaxis. Mild allergic reaction is treated with an antihistamine, whereas anaphylaxis requires treatment with epinephrine, bronchodilators, and antihistamines. |
| Superinfection | Superinfection is a secondary infection that occurs when the normal microbial flora of the body are disturbed during antibiotic therapy. Superinfections can occur in the mouth, respiratory tract, intestine, genitourinary tract, and skin. Fungal infections frequently result in superinfections, although bacterial organisms (e.g., Proteus, Pseudomonas, Staphylococcus) may be the offending microorganisms. Superinfections rarely develop when drug is administered for less than 1 week, and they occur more commonly with the use of broad-spectrum antibiotics. For fungal infection of the mouth, nystatin is frequently used. |
| Organ toxicity | The liver and kidneys are involved in drug metabolism and excretion, and antibacterials may result in damage to these organs. For example, aminoglycosides can be nephrotoxic and ototoxic. |
Section 26A
Penicillins and Cephalosporins
Penicillins
Penicillin, a natural antibacterial agent obtained from the mold genus Penicillium, was introduced to the military during World War II and is considered to have saved many soldiers’ lives. It became widely used in 1945 and was considered a “miracle drug.” With the advent of penicillin, many patients survived who would have normally died from wound and severe respiratory infections.
Penicillin’s beta-lactam ring structure interferes with bacterial cell-wall synthesis by inhibiting the bacterial enzyme that is necessary for cell division and cellular synthesis. The bacteria die of cell lysis (cell breakdown). The penicillins can be both bacteriostatic and bactericidal, depending on the drug and dosage. Penicillin G is primarily bactericidal.
Penicillins are mainly referred to as beta-lactam antibiotics. Bacteria can produce a variety of enzymes, such as beta-lactamases, that can inactivate penicillin and other beta-lactam antibiotics such as the cephalosporins. The beta-lactamases, which attack penicillins, are called penicillinases.
Penicillin G was the first penicillin administered orally and by injection. With oral administration, only about one third of the dose is absorbed. Because of its poor absorption, penicillin G given by injection (IV and intramuscular [IM]) is more effective in achieving a therapeutic serum penicillin level. Because it is an aqueous drug solution, aqueous penicillin G has a short duration of action, and the IM injection is very painful. As a result, a longer-acting form of penicillin, procaine penicillin (milky color), was produced to extend the activity of the drug. Procaine (an anesthetic) in the penicillin decreases the pain related to injection.
Penicillin V was the next type of penicillin produced. Although two thirds of the oral dose is absorbed by the gastrointestinal (GI) tract, it is a less potent antibacterial drug than penicillin G. Penicillin V is effective against mild to moderate infections, including anthrax as a weapon of bioterrorism.
Initially, penicillin was overused. It was first introduced for the treatment of staphylococcal infections, but after a few years, mutant strains of Staphylococcus developed that were resistant to penicillins G and V because of the bacterial enzyme penicillinase, which destroys penicillin. This led to the development of new broad-spectrum antibiotics with structures similar to penicillin to combat infections resistant to penicillins G and V.
Food in the stomach does not significantly alter absorption of penicillin V, so it should be taken after meals. Amoxicillins are penicillins that are unaffected by food.
Broad-Spectrum Penicillins (Aminopenicillins)
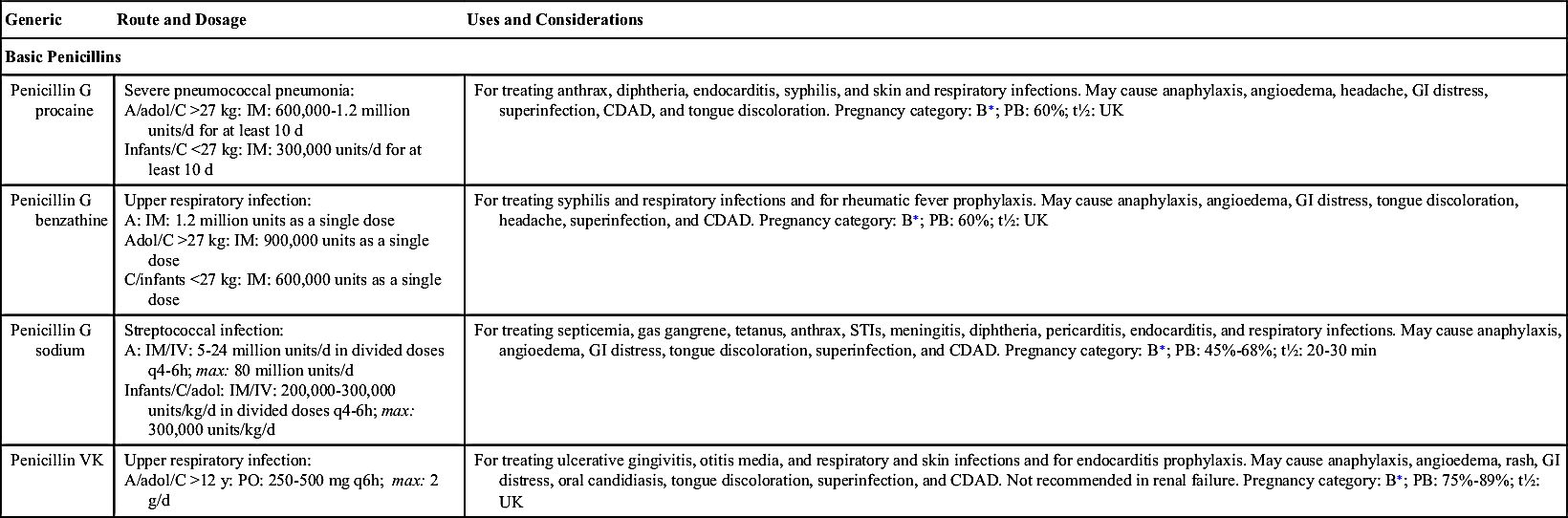
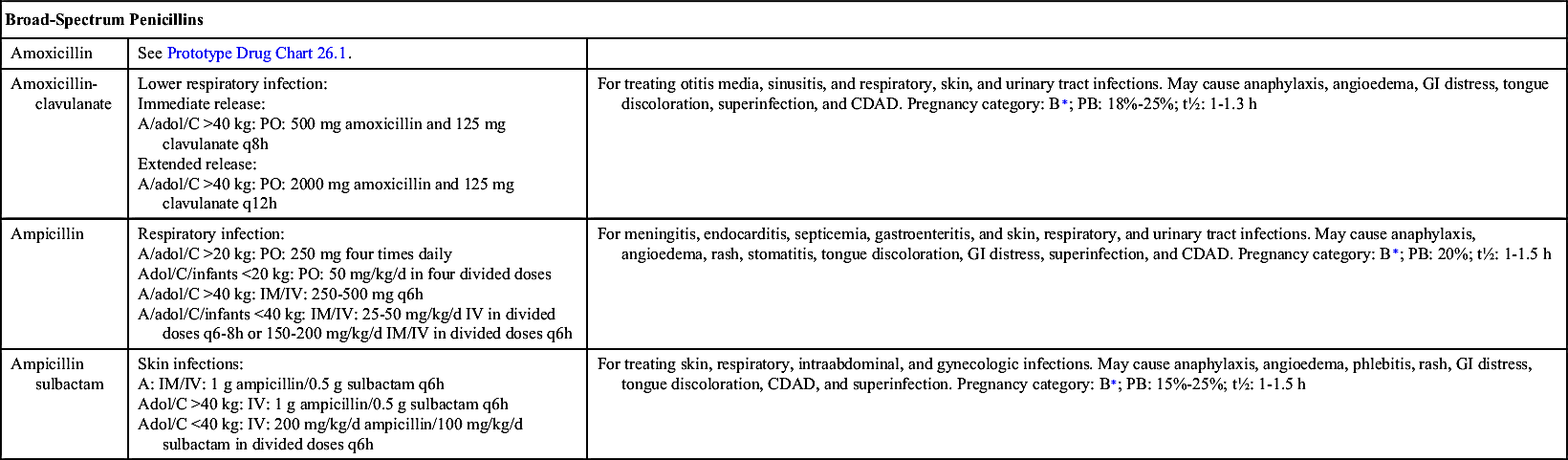
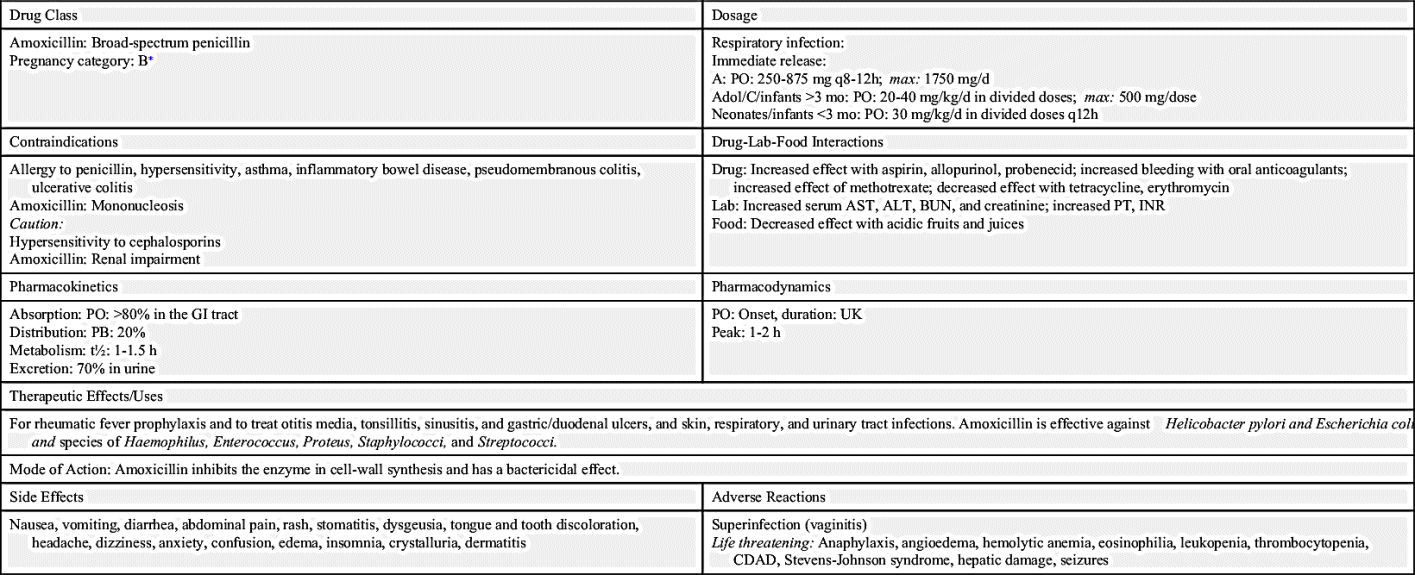
Broad-spectrum penicillins are used to treat both gram-positive and gram-negative bacteria. They are not, however, as broadly effective against all microorganisms as they were once considered to be. This group of drugs is costlier than penicillin and therefore should not be used when ordinary penicillins, such as penicillin G, are effective. The broad-spectrum penicillins are effective against some gram-negative organisms such as E. coli, H. influenzae, Shigella dysenteriae, Proteus mirabilis, and Salmonella species. However, these drugs are not penicillinase resistant; because they are readily inactivated by beta-lactamases, they are ineffective against S. aureus. Examples of this group are ampicillin and amoxicillin (Table 26.3). Amoxicillin is the most prescribed penicillin derivative for adults and children.
Penicillinase-Resistant Penicillins (Antistaphylococcal Penicillins)
Penicillinase-resistant penicillins (antistaphylococcal penicillins) are used to treat penicillinase-producing S. aureus. Dicloxacillin is an oral preparation of these antibiotics, whereas nafcillin and oxacillin are IM and IV preparations. This group of drugs is not effective against gram-negative organisms and is less effective than penicillin G against gram-positive organisms. See Prototype Drug Chart 26.1 and Table 26.3 to compare the similarities and differences in the broad-spectrum penicillin amoxicillin and the penicillinase-resistant penicillin dicloxacillin.
Extended-Spectrum Penicillins (Antipseudomonal Penicillins)
The antipseudomonal penicillins are a group of broad-spectrum antibiotics effective against P. aeruginosa, a gram-negative bacillus that is difficult to eradicate. These drugs are also useful against many gram-negative organisms such as Proteus, Serratia, Enterobacter, and Acinetobacter species and also Klebsiella pneumoniae. The antipseudomonal penicillins are not penicillinase resistant. Their pharmacologic action is similar to that of aminoglycosides, but they are less toxic.
Table 26.3 lists the drugs in the four categories of penicillin-type antibacterials. The administration routes (oral, IM, or IV) of various types of penicillins, along with the cephalosporins, are available on the Evolve website.
Beta-Lactamase Inhibitors
When a broad-spectrum antibiotic such as amoxicillin is combined with a beta-lactamase enzyme inhibitor such as clavulanic acid, the resulting antibiotic combination inhibits the bacterial beta-lactamases, making the antibiotic effective and extending its antimicrobial effect. There are three beta-lactamase inhibitors: (1) clavulanic acid, (2) sulbactam, and (3) tazobactam. These inhibitors are not given alone but are combined with a penicillinase-sensitive penicillin such as amoxicillin, ampicillin, or piperacillin. The combination drugs currently marketed include amoxicillin-clavulanic acid (for oral use) and ampicillin-sulbactam and piperacillin-tazobactam (for use parenterally).
Pharmacokinetics
Amoxicillin is well absorbed from the GI tract, whereas dicloxacillin is only partially absorbed. Protein-binding power differs between the two drugs—amoxicillin is 20% protein bound, but dicloxacillin is highly protein bound (95%). Drug toxicity may result when other highly protein-bound drugs are used with dicloxacillin. Both drugs have short half-lives. Sixty percent of amoxicillin is excreted in the urine; dicloxacillin is excreted in bile and urine.
Pharmacodynamics
Both amoxicillin and dicloxacillin are penicillin derivatives and are bactericidal. These drugs interfere with bacterial cell-wall synthesis, causing cell lysis. Amoxicillin may be produced with or without clavulanic acid, an agent that prevents the breakdown of amoxicillin by decreasing resistance to the antibacterial drug. The addition of clavulanic acid intensifies the effect of amoxicillin. The amoxicillin–clavulanic acid preparation and amoxicillin trihydrate have similar pharmacokinetics and pharmacodynamics as well as similar side effects and adverse reactions. When probenecid is taken with amoxicillin or dicloxacillin, the serum antibacterial levels may be increased. The effects of amoxicillin are decreased when taken with erythromycin and tetracycline. The onset of action, serum peak concentration time, and duration of action for amoxicillin and dicloxacillin are very similar.
Geriatrics
Most beta-lactam antibiotics are excreted via the kidneys. With older adults, assessment of renal function is most important. Serum blood urea nitrogen (BUN) and serum creatinine should be monitored. With a decrease in renal function, the antibiotic dose should be decreased.
Side Effects and Adverse Reactions
Common adverse reactions to penicillin administration are hypersensitivity and superinfection, the occurrence of a secondary infection when the flora of the body are disturbed (see Table 26.2). Anorexia, nausea, vomiting, and diarrhea are common GI disturbances, often referred to as GI distress. This may be alleviated some by taking penicillin with food. Rash is an indicator of a mild to moderate allergic reaction; severe allergic reaction leads to anaphylactic shock. Clinical manifestations of a severe allergic reaction include laryngeal edema, severe bronchoconstriction with stridor, and hypotension. Allergic effects occur in 5% to 10% of persons receiving penicillin compounds, therefore close monitoring during the first and subsequent doses of penicillin is essential.
Drug Interactions
The broad-spectrum penicillins, amoxicillin and ampicillin, may decrease the effectiveness of oral contraceptives. Potassium supplements can increase serum potassium levels when taken with potassium penicillin G or V. When penicillin is mixed with an aminoglycoside in IV solution, the actions of both drugs are inactivated.
Other Beta-Lactam Antibacterials
Like penicillins, these beta-lactam antibacterials preferentially bind to specific penicillin-binding proteins located inside the bacterial cell wall and are bactericidal. This group includes aztrenam, imipenem/cilastatin, and meropenem. Aztrenam’s effectiveness is limited to aerobic gram-positive infections. Imipenem/cilastatin and meropenem are effective against a broader spectrum of activity than many other beta-lactam antibacterials. These three antibacterials are less nephrotoxic than many other antibacterials. Table 26.4 describes these other beta-lactam antibacterials and their dosages, uses, and considerations.
Side Effects and Adverse Reactions
Side effects and adverse reactions to aztrenam, imipenem/cilastatin, and meropenem include headache, nausea, vomiting, diarrhea, anemia, eosinophilia, and neutropenia. Rash may also occur. Severe adverse reactions include anaphylaxis, angioedema, seizures, and Clostridium difficile–associated diarrhea.
TABLE 26.5
Activity of the Five Generations of Cephalosporins
| Generation | Activity |
| First | Effective mostly against gram-positive bacteria (streptococci and most staphylococci) and some gram-negative bacteria (Escherichia coli and species of Klebsiella, Proteus, Salmonella, and Shigella) |
| Second | Same effectiveness as first generation but with a broader spectrum against other gram-negative bacteria such as Haemophilus influenzae, Neisseria gonorrhoeae and N. meningitidis, Enterobacter species, and several anaerobic organisms |
| Third | Same effectiveness as first and second generations and also effective against gram-negative bacteria (Pseudomonas aeruginosa and Serratia and Acinetobacter species) but with increased resistance to destruction by beta-lactamases |
| Fourth | Similar to third-generation drugs and highly resistant to most beta-lactamase bacteria with broad-spectrum antibacterial activity and good penetration to cerebrospinal fluid; effective against E. coli, P. aeruginosa, and Klebsiella, Proteus, and Streptococcus species and certain staphylococci |
| Fifth | Similar characteristics of third and fourth generations, also broad spectrum, and the only cephalosporins effective against methicillin-resistant Staphylococcus aureus (MRSA) |
Cephalosporins
In 1948, a fungus called Emericellopsis minimum (Cephalosporium acremonium) was discovered in seawater at a sewer outlet off the coast of Sardinia. This fungus was found to be active against gram-positive and gram-negative bacteria and resistant to beta-lactamase, an enzyme that acts against the beta-lactam structure of penicillin. In the early 1960s, cephalosporins were used with clinical effectiveness. For cephalosporins to be effective against numerous organisms, their molecules were chemically altered, and semisynthetic cephalosporins were produced. Like penicillin, the cephalosporins have a beta-lactam structure and act by inhibiting the bacterial enzyme necessary for cell-wall synthesis. Lysis to the cell occurs, and the bacterial cell dies.
First-, Second-, Third-, Fourth-, and Fifth-Generation Cephalosporins
Cephalosporins are a major antibiotic group used in hospitals and in health care offices. These drugs are bactericidal with actions similar to penicillin. For antibacterial activity, the beta-lactam ring of cephalosporins is necessary.
Five groups of cephalosporins have been developed, identified as generations. Each generation is effective against a broader spectrum of bacteria, an increased resistance to destruction by beta-lactamases, and an increased ability to reach cerebrospinal fluid (Table 26.5).
Not all cephalosporins are affected by the beta-lactamases. First-generation cephalosporins are effective against most gram-positive bacteria and are destroyed by beta-lactamases, but not all second-generation cephalosporins are affected by beta-lactamases. Second-generation cephalosporins are effective against gram-positive and some gram-negative bacteria. Third-generation cephalosporins are resistant to beta-lactamases. They have broad-spectrum antibacterial activity and are effective against P. aeruginosa. The fourth-generation cephalosporin, cefepime, has broad-spectrum activity, is highly resistant to beta-lactamases, and has good penetration to cerebrospinal fluid. The fifth-generation cephalosporins are broad-spectrum drugs effective against MRSA.
Approximately 10% of persons allergic to penicillin are also allergic to cephalosporins because both groups of antibacterials have similar molecular structures. If a patient is allergic to penicillin and taking a cephalosporin, the nurse should watch for a possible allergic reaction to the cephalosporin, although the likelihood of a reaction is small.
Only a few cephalosporins are administered orally. These include cephalexin, cefadroxil, cefaclor, cefuroxime, cefdinir, cefprozil, cefixime, cefpodoxime, cefditoren, and ceftibuten. The rest of the cephalosporins are administered IM and IV. Prototype Drug Chart 26.2 describes the drug data related to ceftriaxone.
Pharmacokinetics
Cefazolin is administered IM and IV, and cefaclor is given orally. The protein-binding power of cefazolin is greater than that of cefaclor. The half-life of each drug is short, and the drugs are excreted 60% to 80% unchanged in the urine.
Pharmacodynamics
Cefazolin and cefaclor inhibit bacterial cell-wall synthesis and produce a bactericidal action. For IM and IV use of cefazolin, the onset of action is almost immediate, and peak concentration time is 5 to 15 minutes with IV use. The peak concentration time for an oral dose of cefaclor is 30 to 60 minutes.
When probenecid is administered with either of these drugs, urine excretion of cefazolin and cefaclor is decreased, which increases the action of the drug. The effects of cefazolin and cefaclor can be decreased if the drug is given with tetracyclines or erythromycin. These drugs can cause false-positive laboratory results for proteinuria and glucosuria, especially when they are taken in large doses.
Table 26.6 lists the cephalosporins by their designated generation and also gives dosages, uses, and considerations.
Side Effects and Adverse Reactions
The side effects and adverse reactions to cephalosporins include GI disturbances (nausea, vomiting, diarrhea), alteration in blood clotting time (increased bleeding) with administration of large doses, and nephrotoxicity (toxicity to the kidney) in individuals with a preexisting renal disorder.
Drug Interactions
Drug interactions can occur with certain cephalosporins and alcohol. For example, alcohol consumption may cause a disulfiram-like reaction (flushing, dizziness, headache, nausea, vomiting, and muscular cramps) while taking cefotetan. Uricosuric drugs increase the excretion rate of uric acid by inhibiting its reabsorption. Taking uricosurics concurrently can decrease the excretion of cephalosporins, thereby greatly increasing serum levels.