http://evolve.elsevier.com/McCuistion/pharmacology
Pain is an unpleasant sensory and emotional experience related to tissue injury. Due to the subjective nature of pain, the nurse must be knowledgeable and skillful in the assessment and measurement of pain to achieve optimal pain management.
Pain management is regarded as such a significant component of nursing care that pain has become known as the “fifth vital sign.” The Joint Commission (TJC) has incorporated the assessment, documentation, and management of pain into its standards, which reflect the importance of this vital sign. The nurse’s role is to assess the patient’s pain level, alleviate the patient’s pain through nonpharmacologic and pharmacologic treatments, thoroughly document the patient’s response to treatment, and teach patients and their significant others to manage pain control when appropriate.
An individual’s pain threshold reflects the level of stimulus needed to create a painful sensation, and individual genetic makeup contributes to the variations in pain threshold from person to person. The mu (μ) opioid receptor gene controls the number of μ-receptors present. When an individual has a large number of μ-receptors, the pain threshold is high, and pain sensitivity is reduced.
The amount of pain a person can endure without having it interfere with normal functioning is called pain tolerance. This psychological aspect of pain varies greatly in individuals because it is subjective and because pain tolerance is influenced by factors such as age, gender, culture, ethnicity, previous experience, anxiety level, and specific circumstances, such as a traumatic event.
Analgesics, both nonopioid and opioid, are prescribed for the relief of pain. The choice of analgesic depends upon the severity of the pain. Mild to moderate pain is frequently relieved with the use of nonopioid, also known as nonnarcotic, analgesics. Moderate to severe pain usually requires an opioid, or narcotic, analgesic.
Drugs used for pain relief are presented in this chapter. Many of the same nonopioid analgesics that are taken for pain, such as the nonsteroidal antiinflammatory drugs (NSAIDs), are also taken for antiinflammatory purposes. This application for these drugs is covered in Chapter 24.
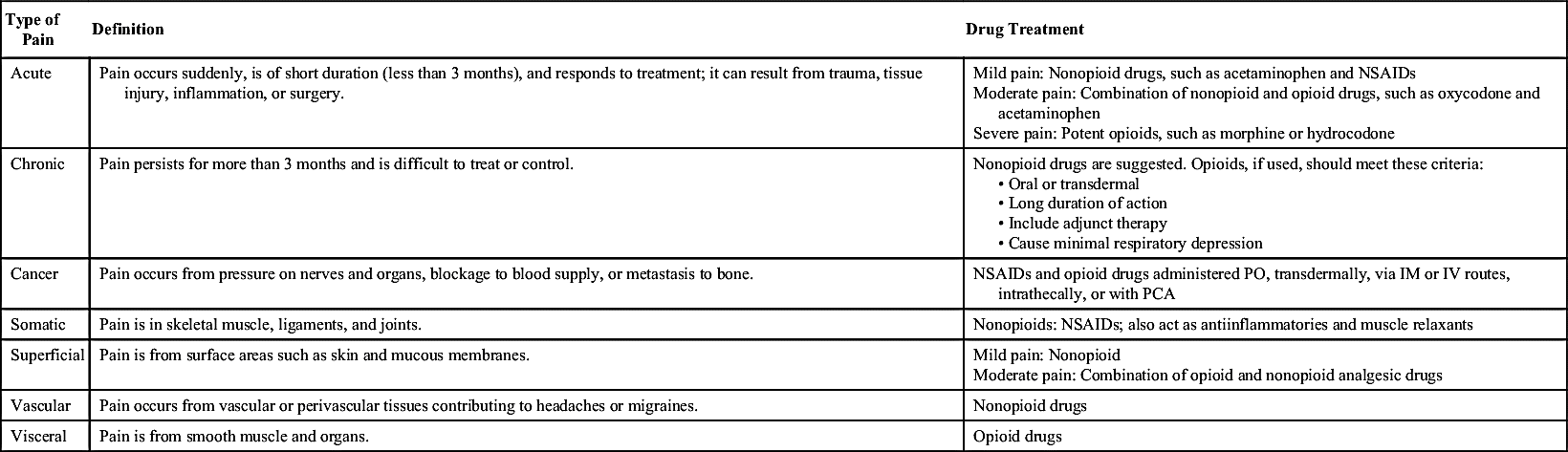
The most common classification of pain is by duration. Acute pain can be mild, moderate, or severe and is usually associated with a specific tissue injury. The onset of acute pain is usually sudden and of short duration lasting less than 3 months. Chronic pain usually has a vague origin and gradual onset with a prolonged duration (more than 3 months) of long-lasting discomfort.
Pain may also be classified by its origin. Nociceptors, sensory receptors for pain, are activated by noxious stimuli—mechanical, thermal, and chemical—in peripheral tissues. When tissue damage occurs, injured cells release chemical mediators that affect the exposed nerve endings of the nociceptors. Pain that originates from tissue injury is nociceptor pain, which includes somatic pain—that is, pain from structural tissues such as bones and muscles—and visceral (organ) pain. Neuropathic pain is an unusual sensory disturbance that often involves neural supersensitivity. This pain is due to injury or disease of the peripheral nervous system (PNS) or central nervous system (CNS). The patient with neuropathic pain usually complains of burning, tingling, or electric shock sensations in the affected area, often triggered by light touch. Diabetic neuropathy associated with diabetes mellitus is an example of peripheral neuropathic pain. Severe, intractable pain from a herniated disk or spinal cord injury is evidence of neuropathic pain from the CNS.
Pathophysiology
The most common pain theory is called the gate theory, proposed by Melzack and Wall in 1965. According to this theory, tissue injury activates nociceptors and causes the release of chemical mediators such as substance P, prostaglandins, bradykinin, histamine, serotonin, acetylcholine, glutamate, adenosine triphosphate, leukotrienes, and potassium. These substances initiate an action potential along a sensory nerve fiber and sensitize pain receptors. Nociceptive action potentials are transmitted via afferent nerve fibers. One type of pain fiber that primarily transmits impulses from the periphery is the A-delta (A-δ) fiber. Because A-δ pain fibers are wrapped in a myelin sheath, they transmit impulses rapidly in acute pain. The C-fiber is a type of pain fiber that is small and unmyelinated, and because C-fibers are unmyelinated, they transmit impulses slowly. C-fibers are more often associated with chronic, dull pain.
A pain signal begins at the nociceptors in the periphery and proceeds throughout the CNS. Knowing how and where pharmacologic agents work is essential to controlling pain. The body produces neurohormones called endorphins (peptides) that naturally suppress pain conduction, although the method is not completely understood. Opioids such as morphine activate the same receptors as endorphins to reduce pain. NSAIDs control pain at the peripheral level by blocking the action of cyclooxygenase, a pain-sensitizing chemical, and interfering with the production of prostaglandins. Cortisone decreases pain by blocking the action of phospholipase, reducing the production of both prostaglandins and leukotrienes. In neuropathic pain, anticonvulsant drugs inhibit the transmission of nerve impulses by stabilizing the neuronal membrane and inactivating peripheral sodium channels.
To ascertain severity of pain, the health care provider should ask the patient to rate the degree of pain on a scale of 0 to 10, with 10 being the worst or most severe pain. For example, a patient who indicates a pain level of 9 may verbalize a decrease in pain to a level of 7 within 30 to 45 minutes after receiving pain medication. Many scales and instruments are available to the nurse for assessment and measurement of the patient’s pain level. Table 25.1 lists the types of pain and the drug groups that may be effective in relieving each type.
Undertreatment of Pain
Undertreatment of pain is a major issue in health care today. The National Pharmaceutical Council and TJC state that up to 75% of patients have unrelieved pain. Some reasons for undertreatment are sociocultural variables that mediate a patient’s willingness to acknowledge being in pain, the patient’s inability to describe pain, the patient’s fear of addiction, the nurse’s inability to measure pain, lack of regular pain-assessment rounds, attitudes of the health care team, an unwillingness to believe the patient’s report of pain, inaccurate knowledge on the part of the health care provider concerning addiction and tolerance, and prescription of an inadequate analgesic dose.
Unrelieved pain leads to a multitude of harmful effects that involve almost all organs of the body. As a result of unrelieved pain, the patient may develop increased respiratory and heart rates, hypertension, increased stress response, urinary retention, fluid overload, electrolyte imbalance, glucose intolerance, hyperglycemia, pneumonia, atelectasis, anorexia, paralytic ileus, constipation, weakness, confusion, and infection.
In addition to psychological and physical suffering, inadequate pain management leads to high health care costs. It is estimated that the cost of extended hospital stays, readmissions to the hospital, and outpatient visits due to inadequate pain management exceeds $200 billion per year.
Nonopioid Analgesics
Nonopioid analgesics such as aspirin, acetaminophen, ibuprofen, and naproxen are less potent than opioid analgesics and are used to treat mild to moderate pain. Nonopioids are usually purchased over the counter, but cyclooxygenase 2 (COX-2) inhibitors require a prescription. Nonopioids are effective for the dull, throbbing pain of headaches, dysmenorrhea (menstrual pain), inflammation, minor abrasions, muscular aches and pain, and mild to moderate arthritis. Most analgesics also have an antipyretic effect and will lower an elevated body temperature. Some, such as aspirin, have antiinflammatory and antiplatelet effects as well.
Nonsteroidal Antiinflammatory Drugs
All NSAIDs have an analgesic effect as well as an antipyretic and antiinflammatory action. NSAIDs such as aspirin, ibuprofen, and naproxen can be purchased as over-the-counter (OTC) drugs. Aspirin, a salicylate NSAID, is the oldest nonopioid analgesic drug still in use. Adolf Bayer marketed the original formulation in 1899, and currently aspirin can be purchased under many names and with added ingredients.
The American Academy of Pediatrics, Centers for Disease Control and Prevention (CDC), U.S. Food and Drug Administration (FDA), National Reye’s Syndrome Foundation, U.S. Surgeon General, and World Health Organization (WHO) recommend aspirin products not be given to children and adolescents younger than 19 years of age during episodes of fever or viral illnesses because of the danger of Reye syndrome. Reye syndrome is a rare but serious condition associated with viral infections treated with salicylates that causes swelling of the brain and liver. In these circumstances, acetaminophen is recommended instead of aspirin.
In addition to its analgesic, antipyretic, and antiinflammatory properties, aspirin decreases platelet aggregation (clotting). Some health care providers may therefore prescribe one 81-mg, 162-mg, or 325-mg aspirin tablet every day or one 325-mg tablet every other day as a preventive measure against transient ischemic attacks (TIAs, or ministrokes), heart attacks, or any thromboembolic episode. Aspirin is discussed in depth in Chapter 24 along with other NSAIDs.
Aspirin and other NSAIDs relieve pain by inhibiting biosynthesis of prostaglandin by different forms of the COX enzyme. As explained in Chapter 24, NSAIDs inhibit or block both COX-1 and COX-2 enzymes, while COX-2 inhibitors are selective and only inhibit COX-2 enzyme. Inhibition of COX-1 decreases protection of the stomach lining while inhibition of COX-2 decreases inflammation and pain. As a result of an NSAID’s inhibition of COX-1, gastric irritation and bleeding may occur. Aspirin is the drug of choice for alleviating pain and inflammation in arthritic conditions, but when given in high doses, severe gastrointestinal (GI) irritation and possible ulceration develop in approximately 20% of patients. Some pharmaceutical companies have developed antiinflammatory and analgesic drugs that inhibit only COX-2. The COX-2 inhibitors were developed to eliminate the GI side effects associated with aspirin and other NSAIDs. COX-2 inhibitors are discussed in depth in Chapter 24.
Side Effects and Adverse Reactions
A common side effect of NSAIDs is gastric distress, including anorexia, nausea, vomiting, and diarrhea. These drugs should be taken with food, at mealtime, or with a full glass of fluid to help reduce this problem. Excessive bleeding might occur as a side effect if an NSAID is taken for dysmenorrhea during the first 2 days of menstruation. Adverse effects of salicylate toxicity includes tinnitus, vertigo, hyperventilation, and potential metabolic acidosis.
Some patients are hypersensitive to aspirin. Dyspnea, bronchospasm, and urticaria are some of the symptoms that indicate anaphylaxis to salicylate products. Certain foods also contain salicylates: prunes, raisins, paprika, and licorice. Those with a hypersensitivity to aspirin and salicylate products may be sensitive to other NSAIDs. This hypersensitivity may be related to inhibition of the COX enzyme by the salicylate.
Acetaminophen
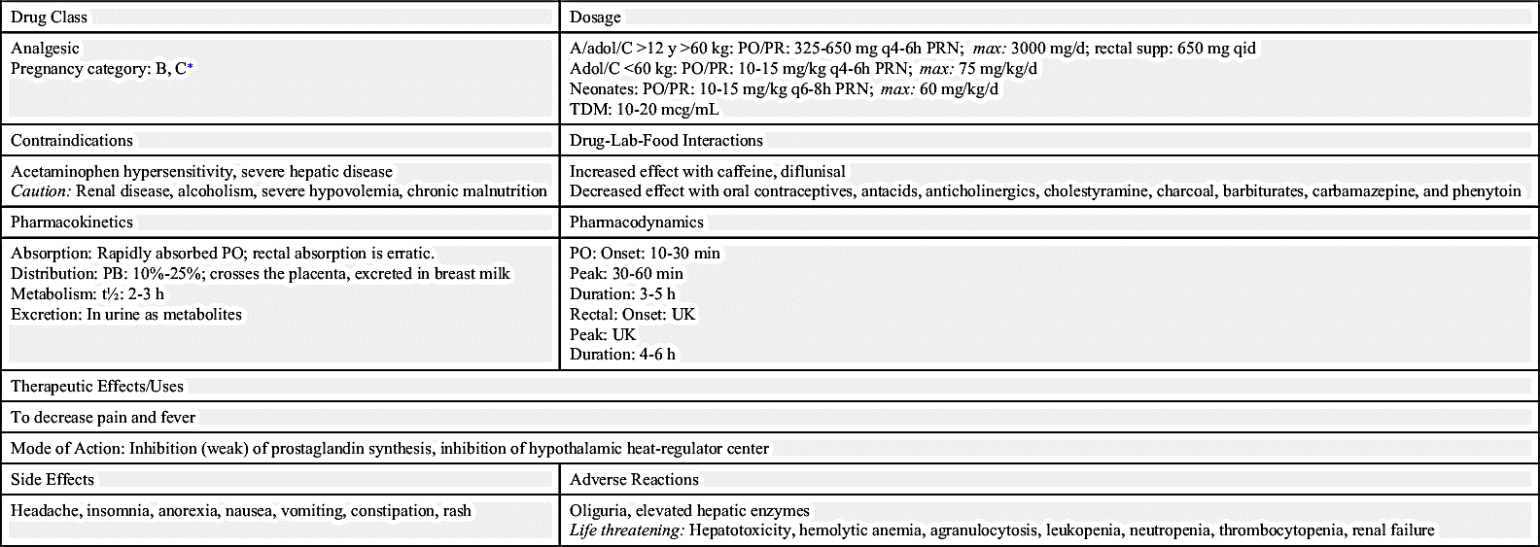
The analgesic acetaminophen, a para-aminophenol derivative, was first marketed in the mid-1950s as an analgesic and antipyretic drug used for muscular aches and pains and for fever caused by viral infections in infants, children, adults, and older adults. It is a popular nonprescription drug: it constitutes 25% of all OTC drugs sold. Acetaminophen is a nonopioid drug, but it is not an NSAID. Because acetaminophen does not have the antiinflammatory properties of aspirin, it is not the drug of choice for any inflammatory process. Acetaminophen is a safe, effective drug when used at therapeutic doses, causes little to no gastric distress, and does not interfere with platelet aggregation. Complementary and Alternative Therapies 25.1 describes the use of these products in pain relief. An intravenous (IV) formulation was approved by the FDA for treating pain and fever. It should be administered undiluted over 15 minutes. There is no link between acetaminophen and Reye syndrome, and unlike aspirin and NSAIDs, it does not increase the potential for excessive bleeding if taken for dysmenorrhea (Prototype Drug Chart 25.1).
Pharmacokinetics
Acetaminophen is well absorbed from the GI tract. Rectal absorption may be erratic because of the presence of fecal material or a decrease in blood flow to the colon. Because of acetaminophen’s short half-life, it can be administered every 4 hours as needed with a maximum dose of 4 g/day for adults. However, it is suggested that a patient who frequently takes acetaminophen limit the dose to 2000 mg/day (2 g/day) to avoid the possibility of hepatic or renal dysfunction. More than 85% of acetaminophen is metabolized to drug metabolites by the liver.
Large doses or overdoses can be toxic to the hepatic cells, so when large doses are administered over a long period, the serum level of acetaminophen should be monitored. The therapeutic serum range is 10 to 20 mcg/mL. Hepatic enzyme levels (aspartate aminotransferase [AST], alanine aminotransferase [ALT], alkaline phosphatase [ALP]) and serum bilirubin should also be monitored. Ingesting alcohol concurrently with acetaminophen may lead to hepatic injury, hepatic failure, and death. When acetaminophen toxicity occurs, acetylcysteine is the antidote, which reduces liver injury by converting toxic metabolites to a nontoxic form.
Pharmacodynamics
Acetaminophen weakly inhibits prostaglandin synthesis, which decreases pain sensation. It is effective in eliminating mild to moderate pain and headaches and is useful for its antipyretic effect. Acetaminophen does not possess antiinflammatory action. When given orally, its onset of action is within 10 to 30 minutes, and the duration of action is 3 to 5 hours. Severe adverse reactions may occur with an overdose, so acetaminophen in liquid or chewable form should be kept out of children’s reach.
Side Effects and Adverse Reactions
An overdose of acetaminophen can be extremely toxic to liver cells; death could occur in 1 to 4 days from hepatic necrosis. If a child or adult ingests excessive amounts of acetaminophen tablets or liquid, a poison control center should be contacted immediately, and the child or adult should be taken to the emergency department. Early symptoms of hepatic damage include nausea, vomiting, diarrhea, and abdominal pain.
Table 25.2 lists the commonly used nonopioid analgesics and their dosages, uses, and considerations.
 Opioid Analgesics
Opioid Analgesics
Opioid analgesics, called opioid agonists, are prescribed for moderate and severe pain. In the United States, the Harrison Opioid Act of 1914 required that all forms of opium be sold with a prescription and that it no longer be used as a nonprescription drug. The Controlled Substances Act of 1970 classified drugs with high abuse potential, opioids among them, in five schedule categories according to their potential for drug abuse (see Chapter 7). Addiction is defined as a psychological and physical dependence upon a substance beyond normal voluntary control, usually after prolonged use of a substance.
Morphine, a prototype opioid, is obtained from the sap of seed pods of the opium poppy plant. Codeine is another drug obtained from opium. In the past decades, many synthetic and semisynthetic opioids have been developed, for example, meperidine.
Although nonopioid analgesics act on the PNS at the pain receptor sites, opioid analgesics act mostly on the CNS. Opioids act primarily by activating the μ-receptors, but they also exert a weak activation of the kappa (κ) receptors. Analgesia, respiratory depression, euphoria, and sedation are effects of μ-receptor activation. Activation of κ-receptors leads to analgesia and sedation but has no effect on respiratory depression and euphoria.
Opioids not only suppress pain impulses but also suppress respiration and coughing by acting on the respiratory and cough centers in the medulla of the brainstem. One example of such an opioid is morphine, a potent analgesic that can readily depress respirations. Codeine is not as potent as morphine (1/15 to 1/20 as potent), but it also relieves mild to moderate pain and suppresses cough, which allows it also to be classified as an antitussive. Most opioids, with the exception of meperidine, have an antitussive (cough suppression) effect. The opioids have two isomers, levo and dextro. The levo-isomers of opioids produce an analgesic effect only; however, both levo- and dextro-isomers possess an antitussive response. The dextro-isomers do not cause physical dependence, but the levo-isomers do. Synthetic cough suppressants are discussed in Chapter 35.
In addition to pain relief and antitussive effects, many opioids possess antidiarrheal effects. Common side effects with high doses of most opioids include nausea and vomiting, particularly in ambulatory patients; constipation; a moderate decrease in blood pressure; and orthostatic hypotension. High doses of opioids may also cause respiratory depression; urinary retention, usually in older adults; and antitussive effects. See Complementary and Alternative Therapies 25.2 for interactions with opioids.
Morphine
Morphine, an extraction from opium, is a potent opioid analgesic (Prototype Drug Chart 25.2). Morphine is effective against acute pain resulting from acute myocardial infarction (AMI) and cancer, relieves dyspnea resulting from pulmonary edema, and may be used as a preoperative medication to relieve anxiety. Although it is effective in relieving severe pain, it can cause respiratory depression, orthostatic hypotension, miosis, urinary retention, constipation resulting from reduced bowel motility, and cough suppression. An antidote for morphine excess or overdose is the opioid antagonist naloxone.