Figure 70-1. Fascial attachments of the rectum.
Understanding the various types of epithelium within the anal canal is important for the diagnosis of both benign and malignant conditions of the anus. The histology of tumors of the anal canal is critical to understanding the typical behavior and management of the disease. Squamous cell lesions that arise at the anal margin are often treated as skin cancers (wide excision), whereas those arising within the anal canal are treated with chemoradiation. Adenocarcinomas of the anal canal typically arise from the upper anus and are treated similar to adenocarcinomas of the distal rectum. Fistulas of the anal canal typically originate from the crypts at the dentate line, however fistulas associated with Crohn’s disease will often arise from the columnar epithelium of the upper anal canal, and those associated with hidradenitis will originate from the mucosa or skin below the dentate line.

Figure 70-2. Arrangement of the external sphincter muscles.
The rectum and anus receive abundant blood supply from the superior, middle, and inferior rectal arteries (Fig. 70-5). The superior rectal (hemorrhoidal) artery starts as the terminal branch of the inferior mesenteric artery, descending posterior to the rectum where it bifurcates to supply the rectum and upper anal canal. The middle rectal arteries arise from the internal pudendal or inferior gluteal arteries (branches of the internal iliac artery). The inferior rectal arteries also arise from the internal pudendal arteries (in Alcock canal) and traverse the ischioanal fossa to supply the anal sphincters and anal canal. There are multiple communications among these three vessels.
The venous drainage of the anus is via both the portal and system venous systems (Fig. 70-6). The superior rectal vein drains the rectum and upper anal canal into the portal venous system through the inferior mesenteric vein. The middle and inferior rectal veins drain into the systemic venous system via the internal iliac veins. Because of the rich communication between these veins, patients with portal hypertension may develop porto-systemic shunts resulting in bleeding rectal varices.
Lymphatic drainage of the rectum and anus mirrors the arterial supply (Figs. 70-7 and 70-8). Drainage from the upper and middle rectum flows to the inferior mesenteric lymph nodes. The lower rectum and upper anal canal may drain to the inferior mesenteric nodes, or may drain laterally to the internal iliac nodes. Lymph from the anus below the level of the dentate line typically drains to the inguinal lymph nodes, however drainage may also occur to the inferior mesenteric and internal iliac nodes. Clinically this drainage pattern is important in patients with squamous cell cancers of the anus, as palpation of enlarged inguinal lymph nodes may signify metastatic disease.
The rectum and urogenital organs are supplied by a vast network of both sympathetic and parasympathetic nerves (Fig. 70-9). The sympathetic nerves branch from the sympathetic trunk from the first three segments of the lumbar spinal cord and synapse at the preaortic plexus. Some postsynaptic nerves innervate the upper rectum, while others travel distally to form the hypogastric plexus, which extends laterally along the rectum to form the pelvis plexus. The parasympathetic nerves (nervi erigentes) originate from the second, third, and fourth sacral nerve roots. These nerves then join the sympathetic nerves in the pelvic plexus to supply both the lower rectum and urogenital organs. This plexus lies within the lateral attachments of the rectum and along Denonvillers fascia.

Figure 70-4. Muscles of the pelvic floor.
Preservation of both the hypogastric and pelvic plexuses during proctectomy is essential in maintaining normal sexual function.2 In men, parasympathetic innervation is responsible for erection, while a complex interaction between sympathetic, parasympathetic, and somatic pathways produces ejaculation. Injury to the nervi erigentes may result in impotence, while injury to the hypogastric sympathetic trunks may cause retrograde ejaculation. Injury to the pelvic nerves may also lead to bladder dysfunction. In women, dysfunction following nerve injury is less well understood, however some patients do report dyspareunia and decreased lubrication during intercourse following pelvic surgery.
The IAS receives both sympathetic and parasympathetic nerves, and maintains tonic contraction at rest. The external anal sphincter is innervated by the inferior rectal branch of the pudendal nerve and perineal branch of the fourth sacral nerve. The inferior rectal nerve also carries sensory information from the anal canal. The majority of sensory nerve endings within the anal canal are located distal to the dentate line, however patients may sense painful stimuli up to 1.5 cm proximal to this area. Clinically this anatomy is relevant, as external hemorrhoids (below the dentate line) are sensitive to touch, while internal hemorrhoids (above the dentate line) may often be manipulated (banding, etc.) without the need for anesthesia. In addition, sensory receptors within the anal canal allow the discrimination between solid or liquid stool and flatus.

Figure 70-5. Arterial supply of the rectum and anal canal.

Figure 70-6. Venous drainage of the rectum and anal canal.

Figure 70-7. Lymphatic drainage of the rectum.
Physiology
The principal function of the anus is the maintenance of continence and regulation of defecation. The physiology of the defecation and maintenance of continence are complex, and involve both voluntary and involuntary muscular activities. Continence is also affected by the compliance and tone of the rectum, the consistency and volume of the stool, the sensation of the anorectum, and the muscular activity of the pelvic floor muscles. The hemorrhoidal plexus may also contribute to continence by acting as a “cushion” to keep the anus closed. Alterations of any of these mechanisms of continence may result in impaired continence.
The primary function of the rectum is to act as a reservoir for stool until it is a socially acceptable time and place for evacuation. When stool enters the rectum from the colon and the rectum becomes distended, receptors within the puborectal muscle initiate the rectoanal inhibitory reflex. This distension leads to simultaneous relaxation of the IAS and contraction of the external anal sphincter, allowing the stool to descend toward the anal canal. This sphincter response allows for “sampling,” or determination by the sensory epithelium of the anal canal as to whether the distension is from gas or stool. If defecation is deferred, the musculature of the rectum relaxes, thereby decreasing the pressure within the rectum; this is referred to as accommodation.
Continence is maintained by the fact that the pressure within the anal canal is higher than the pressure within the rectum. This high-pressure zone (between 40 and 70 mm Hg) within the anal canal is primarily accounted for by the IAS, however the external sphincter and puborectal muscle contribute as well. Autonomic innervation keeps the internal sphincter under tonic contraction and cannot be altered by voluntary control. Contraction of the external anal sphincter will often more than double the intra-anal pressure, however this muscle can typically be contracted voluntarily for only 40- to 60-second periods. The angle between the anus and rectum also may aid in the maintenance of continence by creating a flap valve and preventing stool from entering the anus. This anteroposterior angle is maintained by the contraction of the sling-shaped pubrectalis muscle.

Figure 70-8. Lymphatic drainage of the anal canal.
Defecation involves both voluntary and involuntary mechanisms. If the call to defecate is answered, either the sitting or squatting position is assumed, thereby helping to straighten the anorectal angle. Straining by increasing the intra-abdominal pressure leads to a reflex relaxation of the puborectal muscle which further opens the anorectal angle and shortens the anal canal. Both the internal and external anal sphincters relax, the pelvic floor descends, and a funneling occurs, allowing the rectal contents to be expelled. After completion of rectal evacuation, a “closing reflex” occurs, where transient contraction of the external anal sphincter and puborectalis help to restore tonic contracture of the IAS (Fig. 70-10).
Diagnostic Evaluation of the Anus
Accurate diagnosis of anorectal disorders requires a detailed history and physical examination, and may include both anatomical and functional testing. Underlying illness or mediation use may present with symptoms in the anal area, and it is important to know about travel history and sexual activity. The most common presenting symptoms of anorectal disease are pain, bleeding, discharge, and change in bowel habits.

Figure 70-9. Sympathetic and parasympathetic nerve supply of the rectum.
Bleeding may be seen as the result of an anal condition, or may represent bleeding from a more proximal source in the gastrointestinal system. Obtaining a history of the type and frequency of bleeding may help in making the diagnosis. Bleeding that is bright red and is not mixed with the stool may indicate an anal source, while blood in the form of clots or melena is more indicative of a colonic source. Even in patients with rectal bleeding in whom an anal disorder is found, it is important to consider colonoscopic evaluation of the colon to exclude other sources, particularly in patients who are at increased risk for cancer.
Anal pain associated with swelling that may or may not be related to defecation may be indicative of a thrombosed external hemorrhoid or an abscess. Pain that occurs during or immediately after defecation is often secondary to an anal fissure. Episodic pain (unrelated to bowel movements) that lasts for a short duration may be due to a condition known as proctalgia fugax or levator ani syndrome.
Physical examination of the anus should include a visual inspection of the perianal area as well as a digital examination (Table 70-1). Patients can be examined in either the left lateral decubitus or prone jack-knife position. Simple explanation and reassurance about the planned examination helps to ensure cooperation of the patient and minimize discomfort. Inspection of the anus may demonstrate skin tags or external hemorrhoids, fissures, scars, or excoriation of the skin. Straining during inspection may help to demonstrate rectal prolapse or perineal descent. Digital examination with a well-lubricated gloved index finger will give information including the tone of the anal sphincters as well identification of any rectal masses.
More detailed evaluation of the anus includes anoscopy, rigid or flexible proctosigmoidoscopy, and ultrasonography, as well as physiologic tests such as anal manometry, electromyography, pudendal nerve assessment, and defecography. Use of these tests for specific conditions will be discussed later in the chapter.
BENIGN ANORECTAL DISEASE
Hemorrhoids
Hemorrhoids are cushions of vascular tissue found in the anal canal found from birth. Histologically, this tissue contains vascular structures whose walls do not contain muscle, and are therefore considered sinusoids instead of veins. The number and location of hemorrhoids may vary, however most commonly there are three pillars located in the left lateral, right anterior, and right posterior quadrants.
The term “hemorrhoids” typically refers to clinical situations where these vascular cushions are abnormal and cause symptoms. External hemorrhoids are defined by their location distal to the dentate line and are covered with squamous epithelium. Although these may swell and make anal hygiene difficult, they may also cause pain secondary to formation of clot within the sinusoid (thrombosed external hemorrhoids). Internal hemorrhoids are located proximal to the dentate line and are covered by transitional epithelium. As there are no somatic nerve endings here, internal hemorrhoids do not cause pain, but may present with bleeding or downward displacement of the cushion during defecation (referred to as prolapse). Hemorrhoids are classified according to the degree of prolapse (Table 70-2).

Figure 70-10. Mechanics of defecation.
Clinical Manifestations
1 Although patients may present with complaints of “hemorrhoids,” many of the symptoms of pain, itching, burning, and swelling may not be related to hemorrhoidal disease, instead being due to anal fissure, prolapsed anal papilla, or pruritus ani. The most common symptoms of internal hemorrhoids are bleeding with bowel movements and prolapse of tissue with defecation. Patients will often describe blood dripping into the bowl and staining the toilet water bright red. After passing a firm stool or a forceful straining, bleeding may continue with bowel movements for several days and then resolve for a variable length of time. It is rare that hemorrhoidal bleeding is severe enough to result in anemia. Patients may also complain of a sensation of incomplete evacuation of the rectum, and in chronic cases of prolapse may complain of mucous drainage or incontinence.
Physical examination should include inspection both at rest and during straining, digital rectal examination, and anoscopy. Evaluation may be performed in either the left lateral or prone jack-knife position. Inspection will help to exclude other pathology, including anal fissure, external opening of an anal fistula, and perianal excoriation, as well as to evaluate for the presence of skin tags, external hemorrhoids, and prolapse. Internal hemorrhoids are soft and cannot be reliable diagnosed with the examining finger, however digital rectal examination may help to rule out a low rectal or anal neoplasm. If prolapse is present, it is possible to reduce the hemorrhoidal tissue during the examination. Anoscopy provides definitive examination of the anal canal, however this should be avoided in patients with anal pain due to fissure or abscess. While anoscopy may provide the diagnosis of hemorrhoids or other intra-anal pathology, patients presenting with rectal bleeding should also undergo assessment of the large intestine. In young patients, a flexible sigmoidoscopy may be sufficient, however in patients over the age of 50, those with significant risk factors for polyps or malignancy, or patients with inflammatory bowel disease, a colonoscopy should be performed.
Table 70-1 The Complete Digital Rectal Examination

CLASSIFICATION
Table 70-2 Hemorrhoids Classification

Treatment
Depending on the severity of the symptoms and the grade of the hemorrhoids, patients may be treated through nonoperative measures, office-based procedures, or operative therapy.
Nonoperative Management
As constipation and excessive straining during defecation are often the underlying causes of hemorrhoids, symptoms can be reduced or eliminated in many patients by altering their dietary intake and lifestyle. This goal is often achieved in patients with grade 1 or 2 hemorrhoids by increasing fluid and fiber in the diet, increasing physical exercise, and adding supplemental fiber. Fiber helps to add moisture to the stool to decrease constipation, and the addition of fiber has been shown in a double-blind, placebo controlled trial to be effective in reducing hemorrhoidal bleeding.3 Warm sitz baths, suppositories, and topical creams or ointments may also help to alleviate symptoms, however they often do not provide a long-term solution.
2 In patients with first-, second-, or third-degree internal hemorrhoids with no symptomatic external disease who fail conservative therapy, office-based therapies such as rubber band ligation, sclerotherapy, or photocoagulation may be an option. Rubber band ligation can be performed through an anoscope in the office without the use of anesthesia. The band is placed around the hemorrhoid above the dentate line, causing localized ischemia of the intervening tissue (Fig. 70-11). More than one hemorrhoid may be banded at one time, however multiple synchronous bands may lead to increased pain. A portion of the hemorrhoid and the rubber band are passed during defecation 48 to 72 hours following application. Fibrosis that occurs at the site of the banding causes fixation of the remaining hemorrhoidal tissue, which helps to prevent further prolapse and bleeding. Relief of symptoms may be achieved in up to 80% of patients who undergo banding, however recurrence rates as high as 30% have been reported.4 Complications of hemorrhoidal banding include severe pain requiring removal of the band, increased bleeding, and thrombosis of the hemorrhoids. Severe perianal sepsis is a rare complication, and should be suspected in any patient who develops worsening pain, fever, or the inability to void.

Figure 70-11. Rubber band ligation of an internal hemorrhoid.
Sclerotherapy involves the injection of a sclerosing agent into the submucosa that leads to fibrosis of the surrounding tissue. Many different injection agents have been described, however phenol is most commonly used. Injection is performed through an anoscope, and may be an option for symptomatic hemorrhoids that are too small to band. It is important not to inject the sclerosant directly into the hemorrhoids or thrombosis may ensue. Infrared photocoagulation causes thrombosis and tissue destruction within the anal canal. The probe is applied just proximal to the internal hemorrhoids through an anoscope. Results with both sclerotherapy and photocoagulation are varied and tend to be temporary only.
Operative Treatment
3 Excisional hemorrhoidectomy should be considered in patients who fail conservative or office-based procedures, who have combined internal and external hemorrhoids, who have hemorrhoids that require manual reduction (grade 3), or who have associated pathology such as ulceration, fissures, or fistulas. Hemorrhoidectomy is typically an outpatient procedure, and in most cases local or regional (spinal) anesthesia may be used, however general anesthesia may also be employed. The patient is placed in the prone jack-knife position with the buttocks taped apart. An elliptical incision is made, beginning on the anoderm to remove any external component of the hemorrhoid, and is continued to the base of the hemorrhoid above the dentate line (Fig. 70-12). It is critical to preserve the underlying IAS muscle, as damage to the IAS may lead to postoperative incontinence. One, two, or three hemorrhoidal bundles may be excised at once, however it is also critical to ensure that normal anoderm is left between the excision sites; failure to preserve this anoderm may lead to postoperative anal stenosis. The dissection may be undertaken with the use of a scalpel, scissors, electrocautery, or controlled electrical energy such as ultrasonic shears or a bipolar vessel sealing device. The wound may be left open or, more commonly, may be closed with absorbable suture.
Excisional hemorrhoidectomy is superior to office-based therapies in achieving complete remission of hemorrhoidal symptoms, however complications including stenosis and hemorrhage are more common.5 Patients who undergo hemorrhoidectomy are also less likely to require multiple treatments. Surgical treatment of hemorrhoids can unfortunately also result in significant postoperative pain, and patients often require narcotic pain medication and may require up to 2 to 4 weeks to recover.
Stapled hemorrhoidopexy, while initially described as a treatment of mucosal prolapse, is also an option for prolapsing and bleeding hemorrhoids. Also known as the Procedure for Prolapse and Hemorrhoids (or PPH), this technique involves the removal of a circumferential sleeve of mucosa and submucosa of the distal rectum and anus. A circular stapler creates an anastomosis which elevates the anal canal and fixes the anal cushions into their normal anatomic positions. Because the resection is performed above the level of the dentate line, this technique is advantageous as it may result in less postoperative pain. It has no effect on external hemorrhoids, and therefore its use is somewhat limited.
Stapled hemorrhoidopexy is begun by placing a purse string suture in the mucosa and submucosa approximately 4 cm above the dentate line, incorporating all the redundant tissue circumferentially (Fig. 70-13). Correct placement of this suture is critical to prevent placement of the stapler too close to the dentate line, which can result in chronic pain. The anvil of a specialized circular stapler is then passed above the purse string, and the suture is used to pull the mucosa and submucosa into the stapler head. The stapler is then fired, which excises the sleeve of tissue and creates the anastomosis.

Figure 70-12. Technique of internal closed hemorrhoidectomy. A: Exposure of hemorrhoid with elliptic excision starting at perianal skin and extending to anorectal ring. B: Submucosal hemorrhoidal plexus dissected from the internal sphincter, anoderm, and mucosa. C: Wound closed with a running suture.
A meta-analysis of prospective randomized studies demonstrated that when compared with excisional hemorrhoidectomy, stapled hemorrhoidopexy offers some short-term benefits including less postoperative pain and earlier return to normal activity.6 In long-term follow-up, however, stapled hemorrhoidopexy has been associated with a higher rate of recurrent symptoms. Although PPH has been associated with several unique complications (rectovaginal fistula, staple line bleeding, and chronic pain), the overall rates of postoperative complications did not differ between the procedures.
Doppler-guided transanal hemorrhoid devascularization involves suture ligation of each hemorrhoidal column with resection of the hemorrhoid. A specific anoscope that includes a Doppler probe is used to identify the signal of the vessel feeding each hemorrhoidal column, typically above or just at the top of the column. Once the vessel is identified, it is then ligated with a suture and the Doppler probe is again used to confirm the disappearance of the signal. Typically the hemorrhoidal column is then also oversewn with this suture. The purported benefit of this procedure is that because the ligation occurs above the dentate line, postoperative pain should be significantly lower than with an excisional hemorrhoidectomy. Long-term results with this procedure are limited, however some authors have reported effectiveness in 90%.7

Figure 70-13. Stapled circular hemorrhoidectomy or procedure for prolapsing hemorrhoids. A: A purse-string suture is placed in the rectal mucosa proximal to hemorrhoids. B: The stapler anvil is placed proximal to the purse string, and the purse string is tied down to draw hemorrhoidal tissue into the staple line. C: The stapler is fired and removed, excising a sleeve of distal rectal tissue and creating a stapled anastomosis.
Special Hemorrhoid Situations
Thrombosed External Hemorrhoids. Thrombosed external hemorrhoids are a relatively common complication of hemorrhoidal disease. The exact etiology is unknown, however their presence is often associated with physical exertion or straining (heavy exercise, moving or lifting furniture) or a bout of severe constipation. Patients typically present with a painful, tender mass in the perianal area. Frequently, the thrombosis will lead to necrosis of the overlying skin and patients will complain of bleeding that is independent of bowel movements.
The management of thrombosed external hemorrhoids depends on when in the course of the disease the patient presents. The pain associated with this condition typically peaks within 48 hours and normally begins to subside after 4 days. If left untreated, the clot in the thrombosed vessels will dissolve within several weeks. Following resolution, large thrombosed hemorrhoids may remain as skin tags.
Because this condition is self-limiting, management is typically conservative and includes pain control with a mild analgesic, warm sitz baths, and a bulk-producing agent (usually psyllium fiber). If the patient presents within the first 48 hours, the procedure of choice is excision of the entire thrombosed hemorrhoid (Fig. 70-14). This procedure may be performed in the office or emergency room with the use of a local anesthetic. Using scissors, the thrombosed hemorrhoid is excised with the underlying vein; it is important to excise the entire thrombus in order to prevent recurrence. The skin edges are then reapproximated with the use of an absorbable suture or may be left open.
Incarcerated Hemorrhoids. Rarely, patients will present with prolapsed fourth-degree hemorrhoids which have become incarcerated. While internal hemorrhoids do not typically cause pain, patients with incarcerated hemorrhoids often have severe pain and may develop urinary retention. Edematous prolapsed hemorrhoids are seen, often in combination with large external components as well. Traditionally this condition has been treated with an urgent or emergent hemorrhoidectomy, however there is concern that leaving inadequate anoderm between the excision sites may lead to postoperative stenosis. Instead, patients may be treated with oral or intravenous pain medications and stool softeners, allowing the edema to resolve and the prolapse to reduce. These patients should then be offered an elective hemorrhoidectomy to prevent recurrence.
Anal Fissure
5 Anal fissure is an ulcer-like tear in the mucosal lining of the anal canal distal to the dentate line. Although more commonly found in younger patients, fissures can occur at any age and equally afflict male and females. Fissures can be classified as acute or chronic, and as primary or secondary. Primary anal fissures are almost always located in the posterior or anterior midline, and are not associated with any underlying disease. Secondary fissures may occur in a lateral position, and should alert the clinician to the possibility of Crohn’s disease, HIV infection, tuberculosis, syphilis, or a hematologic malignancy. Anal fissures that have been present for longer than 6 weeks duration are arbitrarily classified as chronic.
The exact etiology is not known, however several mechanisms are thought to lead to the development of anal fissure. Trauma to the anal canal appears to be the initiating factor, most commonly as the result of passage of hard stool, as a low fiber diet seems to be associated with the development of anal fissure.8 Fissure may also occur following prolonged bouts of diarrhea, or following childbirth, most likely the result of forces from the fetus on the anal canal.
Hypertonicity of the IAS with resultant ischemia has also been implicated in development of chronic anal fissure. Ninety percent of fissures are found in the posterior midline of the anal canal, where Doppler flow studies and cadaver vascular injections have demonstrated relatively low perfusion. Studies have shown that when compared with normal subjects, patients with chronic anal fissure have higher resting pressure of the IAS. If the pressure within the anal sphincter approaches or exceeds the intra-arterial pressure of the inferior rectal artery, this may lead to relative ischemia and the development of an ischemic ulcer. This theory is strengthened by the fact that reduction of anal pressure following sphincterotomy improves blood flow to the anal canal, thereby promoting healing of the fissure.9
Clinical Manifestations
Anal fissure typically presents with pain during defecation and rectal bleeding. Patients often describe the pain as knifelike or as a tearing sensation, which may persist for several hours or longer after bowel movements. Rectal bleeding is typically bright red, and is separate from the stool and often seen only after wiping. Constipation is a common complaint of patients with anal fissure, and is frequently both a precipitating event and a result of patients’ fear of a painful bowel movement.
The diagnosis of anal fissure can be made on physical examination by gently spreading the buttocks apart to visualize the anal verge. Fissures will appear as a longitudinal or oval-shaped tear and may be associated with a sentinel pile, a protruding skin tag at the distal end of the fissure. Fibers of the IAS may be visible in chronic anal fissures. Once a diagnosis of fissure has been made, digital examination or anoscopy adds little other than increased pain for the patient, and should therefore be avoided.
Management
Medical Management. Symptomatic relief of anal pain from fissures may be obtained by warm sitz baths two to three times per day. Topical anesthetics or anti-inflammatory ointments may also be of benefit. The majority of patients with acute anal fissure will respond to conservative measures including sitz baths and the addition of a bulking agent such as psyllium fiber. Acute anal fissures often heal within 6 weeks, although the recurrence rate approaches 20%.10
As anal fissure is associated with hypertonicity of the anal sphincter, medical therapy is directed as decreasing resting anal pressures. Nitric oxide is a potent neurotransmitter that induces relaxation of the IAS. Application of 0.2% nitroglycerin ointment twice daily has been shown to induce healing in as many as 85% of patients,11 however, patient compliance is often low due to nitrate-induced headache. Calcium-channel blockers have also been shown to reduce anal pressures. Topical diltiazem or nifedipine has been shown to have healing rates equivalent or superior to nitroglycerin12 without the associated side effects. Unfortunately, recurrence rates with all of these therapies approach 50% in many series.
Botulinum toxin is an endopeptidase that blocks acetylcholine release at the neuromuscular junction, resulting in temporary paralysis of skeletal muscle. Although its exact mechanism of action in smooth muscle is not understood, injection of the toxin also results in relaxation of the IAS. The technique, dose, and success rates have widely varied in the literature, however some authors have reported success in 60% to 80% of patients. The most common side effect is temporary anal incontinence, typically only to flatus. The use of botulinum toxin may be an option in patients who have failed topical treatment, but who may be at a high risk of complications from surgery.
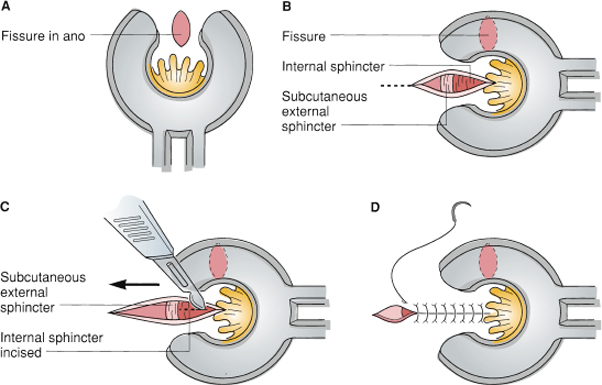
Figure 70-15. Lateral internal sphincterotomy (open method). A: The fissure in the midline is left alone. B: With a speculum used to expose the left lateral quadrant, an incision is made through the subcutaneous tissue to expose both the subcutaneous external sphincter and the internal sphincter. C: The internal sphincter is incised to its full thickness; care is taken not to cut the external sphincter. D: The wound is closed.
Surgical Management. In patients with chronic or recurrent fissures that fail to heal with medical management, surgical intervention is warranted. The most commonly performed procedure is the lateral internal sphincterotomy, which is an outpatient procedure that may be performed under local, spinal, or general anesthesia. When performed in an open fashion, an incision is made overlying the anal sphincter complex in the lateral position away from the fissure (Fig. 70-15). The internal sphincter is identified, and a portion of the muscle is cut typically extending cephalad to the level of the apex of the fissure. The wound is then closed with an absorbable suture. The procedure can also be performed in a closed fashion, where the scalpel is inserted into the intersphincteric groove, turned horizontally, and used to cut the internal sphincter as the anal canal is stretched open with a speculum. Healing rates following sphincterotomy range from 90% to 100%, however, impairment of continence following the procedure has been reported to be as high as 47%.13
In patients who are at high risk of experiencing postoperative incontinence, an alternative approach may be the use of an advancement flap. This procedure involves excision of the fissure, surrounding scar tissue, and any skin tags. A variety of flap configurations including V-Y, Y-V, house, and others have been employed to help close the defect. Healing rates following fissurectomy and advancement flap are comparable to those following sphincterotomy.
Anorectal Abscess
The vast majority of anorectal abscesses result from infection of the glands that empty into the anal crypts within the anal canal at the level of the dentate line. Other causes of anorectal abscess include inflammatory bowel disease, trauma including iatrogenic (usually gynecologic or surgical) trauma, infections such as tuberculosis and actinomycosis, and malignancy. Since these glands lie within the intersphincteric space, blockage of a duct results first in an intersphincteric abscess which can then spread to the surrounding spaces (Fig. 70-16). The most commonly encountered anorectal abscess occurs in the perianal area, followed in frequency by ischioanal, intersphincteric, and supralevator (Fig. 70-17).

Figure 70-16. Pathways of infection start in the intersphincteric space (A) and then spread to perianal spaces, forming perianal abscesses (B).

Figure 70-17. Classification of anorectal abscess.
Clinical Manifestations
Most patients with anal abscess present with pain. In patients with perianal or ischioanal abscesses, pain may be accompanied by swelling and erythema. Conversely, patients with supralevator abscess are less likely to present with swelling and may have accompanying fever. Patients with severe rectal pain may also develop urinary retention.
Management
Treatment of anorectal abscesses requires incision and drainage; antibiotics alone are ineffective and may allow the suppurative process to progress to a more complicated abscess. The use of antibiotics in conjunction with drainage may be considered in the patient with concomitant cellulitis or in immunocompromised patients.
Most patients with perianal abscesses can be treated in an outpatient setting under local anesthesia. An elliptical or cruciate incision is made over the most tender area or area of fluctuance, and the skin edges are trimmed to prevent early closure of the wound, and all loculations are drained. Routine culture of the drainage is not recommended. Packing is also not necessary; rather, patients are instructed to soak the area in warm water and simply keep the wound covered with gauze.
Patients with superficial ischioanal abscesses may be treated in a similar fashion, however patients with deeper or more complex abscesses typically require an examination under anesthesia with drainage in the operating room. Less commonly, patients may present with bilateral ischioanal abscess, a condition referred to as a horseshoe abscess. These infections typically begin in the deep postanal space, which spread to both ischioanal areas. Treatment requires that the deep postanal space be drained, which is accomplished by making a longitudinal incision in the skin between the tip of the coccyx and the anus and exposing the anococcygeal ligament. The ligament is then divided and the deep postanal space drained; counter incisions are then made overlying the ischioanal areas as well.
Patients with intersphincteric abscess often present with severe anorectal pain, however no indurated or fluctuant area is evident. These patients also typically require an examination under anesthesia. In the operating room, an intersphincteric abscess can be diagnosed by palpation of a protrusion into the anal canal or aspiration of purulent fluid from the intersphincteric space. Drainage is performed by dividing a portion of the internal sphincter muscle along the length of the abscess cavity.
Supralevator abscesses are less common and can often be difficult to diagnose. Because the location of the abscess is adjacent to the abdominal cavity, patients may present with abdominal or pelvic pain. Digital examination may demonstrate induration or a tender mass located in the distal rectum above the level of the anorectal ring. The etiology of a supralevator abscess dictates its management. These abscesses may result as an extension of an intersphincteric abscess, extension of an ischioanal abscess, or may result from an intra-abdominal abscess from perforated diverticulitis, appendicitis, or Crohn’s disease. Supralevator abscess secondary to extension from an ischioanal abscess should be drained through the ischioanal area. Supralevator abscess secondary to extension from an intersphincteric abscess should be drained into the rectum, as drainage through the ischioanal area may result in a complex suprasphincteric fistula. Percutaneous drainage may be an option in patients with supralevator abscess secondary to an intra-abdominal source.
Rarely, anorectal abscess can result in necrotizing infection and death, a condition referred to as Fournier gangrene. This situation may result from a delay in diagnosis and management of an anorectal abscess, infection with a highly virulent organism, or may be due to patient factors such as diabetes or compromised immune function. The infection superficially spreads around the perineum, causing necrosis of the skin and underlying muscle and fascia. Treatment includes empiric broad-spectrum intravenous antibiotics and prompt surgical debridement of the necrotic tissue until healthy tissue is encountered.
Anal Fistula
6 A fistula is generally defined as an abnormal communication between two epithelialized surfaces. Anal fistula (or fistula-in-ano) represents a communication between the anorectal canal and the perianal skin as the result from spontaneous or surgical drainage of an anorectal abscess. The incidence of fistulas following abscess drainage ranges from 5% to 83% in the medical literature, but is generally thought to occur in one-quarter to one-third of patients.14 Anal fistulas may also occur in up to 30% of patients with Crohn’s disease.
Classification
Treatment of anal fistulas is in part dictated by the type of fistula. Fistulas are classified by their relation to the anal sphincter complex (Fig. 70-18).15 The most common type is the intersphincteric fistula, followed by transsphincteric, suprasphincteric, and extrasphincteric (Box 70-1).
Clinical Manifestations
The majority of patients with anal fistula have a history of previous anorectal abscess which either spontaneously drained or were surgically incised. Recurrence of anorectal abscess in the same location often indicates the presence of an underlying fistula. Patients complain of drainage, intermittent swelling, pain with defecation, and occasional bleeding. Physical examination often demonstrates an external opening on the perianal skin with granulation tissue. Drainage of fecal, purulent, or serosanguinous drainage may be evident or can be elicited with digital rectal examination or compression of the fistula tract.

Figure 70-18. The four main anatomic types of fistula.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree




