A second fascial layer in the superficial abdominal wall is the deep fascia of Scarpa. Although most commonly considered a distinct anatomic layer, Scarpa fascia actually consists of compressed fibrous components of the superficial fascia.3 The deeper fibrous tissue of the superficial fascia forms the fundiform ligament of the penis (suspensory ligament of the female clitoris), continues onto the penis and scrotum, and ultimately fuses with the superficial fascia of the perineum.

Figure 72-1. The myopectineal orifice. Superior to the inguinal ligament, this area includes the inguinal (Hesselbach) triangle. Inferior to the ligament, the orifice transmits the iliopsoas muscle, the femoral nerve and vessels, and the femoral canal and sheath. (Reproduced with permission from Wantz GE. Atlas of Hernia Surgery. New York, NY: Raven Press; 1991:4.)
The superficial fascia also fuses with the layer of fascia (fascia innominata) investing the external abdominal oblique muscle. This fascia is bound inferiorly to the inguinal ligament and pubis before continuing onto the thigh, where it blends with the fascia lata to seal the space beneath and inferior to the inguinal ligament, which is the inferior portion of the myopectineal orifice (Fig. 72-1). This portion of the inguinal region includes the Hesselbach (inguinal) triangle superiorly and therefore constitutes the weakest aspect of the groin.
The skin of the anterior abdominal wall is segmentally innervated in the familiar dermatome pattern. The nerve branches to this area are derived from the anterior and lateral cutaneous branches of the ventral rami of the 7th to 12th intercostal nerves and from the ventral rami of the first and second lumbar nerves. Disruption of one of these nerves is rarely noted by the postoperative patient because the dermatome fields overlap significantly. The anterior and lateral cutaneous branches reach the subcutaneous layer by coursing between the flat lateral muscles and by piercing the sheath of the rectus abdominis.
Anterior Musculature and Ligaments
The division of the wall into anterior and posterior segments is somewhat artificial because the anterior muscles, with the exception of the rectus abdominis, arise posteriorly and also form part of the posterior wall.
The three muscles of the lateral aspect of the anterior abdominal wall (Fig. 72-2) are composed of a variable amount of muscle with a large aponeurosis. The aponeurosis is the tendon of insertion for the lateral muscles, and it also forms the sheath of the rectus abdominis. The midline decussation of the three aponeuroses forms the linea alba. Fibrous tissue layers are of great importance to the hernia surgeon because of their ability to support sutures. Fascia and aponeurosis are terms commonly used to describe these fibrous structures, but are often confused and used interchangeably. In this chapter, an aponeurosis is defined as the non–muscle-fiber-containing portion of a muscle usually present at insertion points. Muscle fibers are said to “give way” to the corresponding aponeurosis. Fascia, on the other hand, is the fibrous tissue that lines or envelops muscles.

Figure 72-2. Pattern of crossing of the aponeurotic fascicles of the abdominal wall musculature. A: Fascicles from the right external oblique and anterior lamina of the left internal oblique. B: Fascicles from the right transversus abdominis and posterior lamina of the left internal oblique. C: Fascicles between the right and left transversus abdominis muscles.
External Abdominal Oblique Muscle and Associated Ligaments
The external abdominal oblique muscle (Figs. 72-2 and 72-3A,B) is the most superficial of the three lateral abdominal muscles. The external abdominal oblique arises from the posterior aspects of the lower eight ribs and interdigitates with both the serratus anterior and the latissimus dorsi at its origin. The direction of the muscle fibers varies from nearly horizontal in its upper portion to oblique in the middle and lower portions. The mostly horizontal fibers, which originate posteriorly, insert onto the anterior portion of the iliac crest. The obliquely arranged anteroinferior fibers of insertion fold on themselves to form the inguinal ligament. The remaining portion of the aponeurosis inserts into the linea alba after contributing to the anterior portion of the rectus abdominis sheath. Some fibers cross the linea alba to reinforce further the anterior rectus sheath of the opposite side.
The more medial fibers of the aponeurosis of the external oblique divide into a medial and a lateral crus to form the external or superficial inguinal ring. The spermatic cord (or round ligament) and branches of the ilioinguinal and genitofemoral nerves pass through this opening. The inguinal ligament (Fig. 72-4) is worthy of special consideration because of its important role as both a landmark and an integral component of many groin hernia repairs. The inguinal ligament is formed by obliquely oriented anteroinferior aponeurotic fibers of the external abdominal oblique. The ligament is formed when the aponeurosis folds beneath itself. Its lateral attachment is to the anterior superior iliac crest; its medial insertion is primarily on the pubic tubercle.
The medial insertion of the inguinal ligament in most persons is dual. One portion runs along the superior surface of the pubic tubercle and symphysis to form (or at least reinforce) the superior pubic ligament. The other portion is fan shaped and spans the distance between the inguinal ligament proper and the pectineal line of the pubis. This fan-shaped portion of the ligament is called the lacunar ligament (Fig. 72-4). It blends laterally with the pectineal (Cooper) ligament.
Internal Abdominal Oblique Muscle and Aponeurosis
The middle layer of the lateral abdominal group is the internal abdominal oblique muscle (Figs. 72-2 and 72-3B,C). This muscle primarily arises from the iliac fascia along the iliac crest and forms a band of iliac fascia fused with the inguinal ligament. The uppermost fibers course obliquely toward the distal ends of the lower three or four (“floating”) ribs. The muscle fibers of the internal oblique fan out following the shape of the iliac crest so that the lowermost fibers are directed inferiorly. These fibers arch over the round ligament, or spermatic cord. Some of the lower muscle bundles in the male join fibers of the transversus abdominis to form the cremaster muscle. The aponeurosis of the internal oblique (Fig. 72-5A) above the level of the umbilicus splits to envelop the rectus abdominis, re-forming in the midline to join and interweave with the fibers of the linea alba. Below the level of the umbilicus (Fig. 72-5B), the aponeurosis does not split but rather runs anterior to the rectus muscle, continues medially as a single sheet, joins the anterior rectus sheath, and finally contributes to the linea alba. The aponeurotic portion of the internal oblique is widest at the level of the umbilicus.
Transversus Abdominis Muscle and Aponeurosis
The transversus abdominis muscle (Figs. 72-2 and 72-3C) arises from the fascia along the iliac crest and inguinal ligament and from the lower six costal cartilages and ribs, where it interdigitates with the lateral diaphragmatic fibers. The muscle bundles of the transversus abdominis for the most part run horizontally. The lower medial fibers, however, may continue in a more inferomedial course toward the site of insertion on the crest and pecten of the pubis.
The aponeurosis of the transversus abdominis joins the posterior lamina of the internal abdominal oblique, forming above the umbilicus a portion of the posterior rectus sheath. Below the umbilicus, the transversus abdominis aponeurosis is a component of the anterior rectus sheath. The gradual termination of aponeurotic tissue on the posterior aspect of the rectus abdominis forms the arcuate line (of Douglas) (Fig. 72-6). The medial aponeurotic fibers of the transversus abdominis insert on the pecten pubis and the crest of the pubis to form the falx inguinalis. These fibers infrequently are joined by a portion of the internal oblique aponeurosis; only then is a true conjoined tendon formed.4
The arch formed by the termination of the aponeurotic fibers of the transversus abdominis is called the aponeurotic arch (Fig. 72-6). The area beneath the arch varies. A high arch may be a predisposing factor in direct inguinal hernia. Contraction of the transversus abdominis causes the arch to move down toward the inguinal ligament in a kind of shutter mechanism, which reinforces the weakest area of the groin when intra-abdominal pressure is raised.

Figure 72-3. A: External oblique muscle and aponeurosis. B: Internal oblique muscle and aponeurosis. C: Transversus abdominis muscle and aponeurosis. D: Lower rectus abdominis and pyramidalis muscle. The linea alba is formed by the intermeshed fibers of the aponeuroses of the lateral muscle layers; it is tensed by the pyramidalis, which inserts into it.
Rectus Abdominis
The rectus abdominis (Figs. 72-3D and 72-5) forms the central and anchoring muscle mass of the anterior abdomen. The rectus muscle arises from the fifth to the seventh costal cartilages and inserts on the pubic symphysis and pubic crest. Each rectus muscle is segmented by tendinous intersections at the levels of the xiphoid process and the umbilicus and at a point midway between these two. The principal blood supply reaches the muscle from the superior and inferior epigastric arteries (Fig. 72-5), which anastomose just superior to the umbilicus. Other vessels are anterior branches of the intercostal arteries; these reach the muscle by entering the lateral aspect of the rectus sheath. The innervation of the muscle is from the 7th to the 12th intercostal nerves, which laterally pierce the aponeurotic sheath of the muscle. The lateral edge of the muscle is demarcated by a slight depression in the aponeurotic fibers coursing toward the muscle; this depression is the semilunar line.
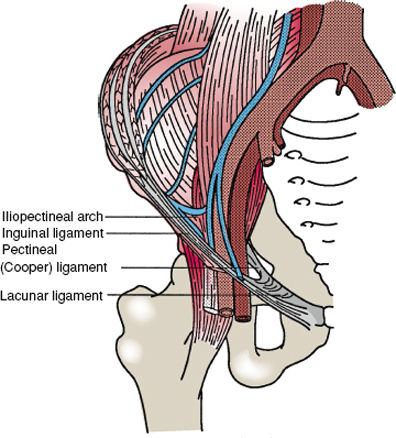
Figure 72-4. Ligamentous structures of the inguinal region. The iliopubic tract is not seen in this view because it is obscured by the inguinal ligament. The lacunar ligament is the expanded medial end of the inguinal ligament; on the pecten pubis, it blends with the inguinal (Cooper) ligament.
The small pyramidalis muscle (Fig. 72-3D) accompanies the rectus abdominis at its origin in a minority of people. The pyramidalis arises from the pubic symphysis. It lies within the rectus sheath and tapers to attach to the linea alba, the conjunction of the two rectus sheaths and the major site of insertion of three aponeuroses from all three lateral muscle layers.
Rectus Sheath
Although the components of the rectus sheath individually have been discussed in relation to the three lateral abdominal muscles, it should also be considered as a distinct entity. Three features of the rectus muscle and its sheath can be observed even topographically in well-muscled or very thin subjects:
1. The semilunar line is a slight depression in the aponeurotic fibers corresponding to the lateral edge of the rectus muscle. It marks the site of initial lateral insertion of the aponeurotic tendons of the lateral abdominal muscles.
2. The tendinous inscriptions divide each muscle into three parts.5 These are the basis of the expression “six pack,” popularized by bodybuilders.
3. The linea alba is the midline confluence of the aponeuroses of the rectus muscles and also the internal and external oblique muscles.
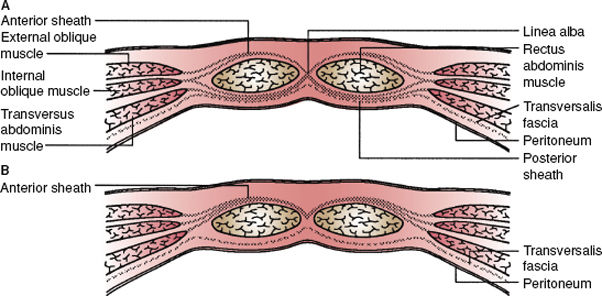
Figure 72-5. A: Immediately superior to the umbilicus, the rectus sheath consists of anterior and posterior components. The anterior sheath is composed of the aponeuroses of the external and internal abdominal oblique muscles, and the posterior sheath consists of the posterior aponeurotic lamina of the internal oblique and the aponeurosis of the transversus abdominis muscle. B: The rectus sheath inferior to the arcuate line (of Douglas) consists of an anterior portion made up of fibers from all aponeurotic layers; the posterior portion at this point comprises only transversalis fascia covered internally by peritoneum.
The composition of the rectus sheath varies depending on the level sampled. The anterior sheath superior to the umbilicus is composed of the aponeurosis of the external abdominal oblique and the anterior lamina of the internal abdominal oblique. The transversalis aponeurosis does not participate in the formation of the anterior sheath at this level. At a variable level inferior to the umbilicus, the anterior sheath is a composite of all the aponeurotic layers.
The posterior sheath of the rectus muscle superior to the umbilicus is a lamination of the posterior lamina of the aponeurosis of the internal abdominal oblique and the transversus abdominis aponeurosis. The external abdominal oblique does not participate in the formation of the posterior portion of the rectus sheath. At a highly variable site inferior to the umbilicus, all the aponeurotic tendons pass anteriorly to form the anterior rectus sheath. The fibers of the posterior sheath are seen to attenuate gradually. The aponeurotic fibers do not end abruptly at the arcuate line. This transfer of connective tissue away from the posterior rectus sheath causes the arcuate line (of Douglas) to form on the posterior surface of the muscle (Fig. 72-6). The tissue covering the deep surface of the rectus muscle inferior to the arcuate line is primarily the transversalis fascia.
Some have questioned this traditional scheme of rectus sheath composition, contending that each of the aponeurotic layers superior to the umbilicus is actually bifid, with both contributing to the anterior and posterior sheaths.6 The fibers of the posterior sheath are seen to attenuate gradually. The concept of rectus sheath composition favored by most is shown in Figure 72-7.7
Innervation and Blood Supply of the Anterior Abdominal Wall
The innervation of the anterior wall muscles is multiple. The lower intercostal and upper lumbar nerves (T7 to T12, L1, L2) contribute most of the innervation to the lateral muscles, the rectus abdominis, and the overlying skin. The nerves pass anteriorly in a plane between the internal abdominal oblique and the transversus abdominis, eventually piercing the lateral aspect of the rectus sheath to innervate the muscle therein. The external oblique muscle receives branches of the intercostal nerves, which penetrate the internal oblique. The anterior ends of the nerves form part of the cutaneous innervation of the abdominal wall. The first lumbar nerve divides into the ilioinguinal and iliohypogastric nerves. These may divide within the psoas major muscle or between the internal oblique and transversus abdominis muscles. The ilioinguinal nerve may communicate with the iliohypogastric nerve before innervating the internal oblique. The ilioinguinal nerve then passes through the external inguinal ring to run with the spermatic cord, whereas the iliohypogastric nerve pierces the external oblique to innervate the skin above the pubis. The cremaster muscle fibers, which are derived from the internal oblique muscle, are innervated by the genitofemoral nerve (L1, L2).
Figure 72-6. The deep inguinal region, pelvis, and anterior abdominal wall from the viewpoint of a surgeon using a laparoscopic technique. The anterior wall folds upward approximately at the iliopubic tract in this illustration.

Figure 72-7. Patterns of midline decussation of the aponeuroses. A: Single anterior and posterior lines of decussation. B: Single anterior and triple posterior lines of decussation. C: Triple anterior and posterior lines of decussation.
The blood supply of the lateral muscles of the anterior wall is primarily from the lower three or four intercostal arteries, the deep circumflex iliac artery, and the lumbar arteries. The rectus abdominis has a complicated blood supply derived from the superior epigastric artery (a terminal branch of the internal thoracic, or internal mammary, artery), the inferior epigastric artery (a branch of the external iliac artery), and the lower intercostal arteries. The latter arteries enter the sides of the muscle after traveling between the oblique muscles. The superior and inferior epigastric arteries enter the rectus sheath and anastomose near the umbilicus.
Posterolateral (Lumbar) Abdominal Wall
The posterolateral or lumbar portion of the abdominal wall (Fig. 72-8) is often overlooked in discussions of abdominal hernia, perhaps because of the much more common occurrence of groin and femoral hernias. The configuration of the muscle layers in the lumbar area also predisposes to hernia formation. For the purposes of this discussion, the lumbar portion of the abdominal wall is defined as the area bounded superiorly by the 12th rib, inferiorly by the iliac crest, and medially by the erector spinae group. Eight muscles arrayed in three layers constitute the posterolateral or lumbar portion of the abdominal wall.
The most superficial layer is composed of the external abdo-minal oblique muscle, which arises from the posteroinferior portion of the lower ribs and inserts in part along the posterior iliac crest. Closely associated with the external oblique in this area is the latissimus dorsi, which arises from the posterior iliac crest, the spinous processes of the sacrum and lumbar vertebrae, and the lumbodorsal fascia. The muscle courses obliquely toward its insertion on the medial aspect of the intertubercular groove of the humerus. The triangular space formed by the two muscles just described and the iliac crest is called the inferior lumbar (Petit) triangle (Fig. 72-8A).

Figure 72-8. The lumbar abdominal wall with the inferior lumbar triangle (A) and the superior lumbar triangle (B).
The middle layer of lumbar abdominal muscles consists of the erector spinae, the internal abdominal oblique, and the extremely thin insignificant serratus posterior inferior. The erector spinae forms a significant portion of the abdominal wall in the lumbar region, with fibers extending nearly the length of the spinal column. The internal abdominal oblique muscle forms the remainder of the layer. The serratus posterior inferior arises from the lumbodorsal fascia and inserts on the lower four ribs. The middle layer of lumbar muscle is associated with the superior lumbar triangle, a more common site of hernia than the inferior lumbar triangle described previously. The superior triangle (Fig. 72-8B) is formed superiorly by the 12th rib, the serratus posterior inferior, and the superior lumbocostal ligament; inferiorly by the upper border of the internal abdominal oblique; and medially by the erector spinae.
The deep layer of the lumbar abdominal wall includes three muscles: the quadratus lumborum, the psoas major, and the transversus abdominis. The quadratus lumborum primarily arises from the posterior iliac crest and inserts on the 12th rib. The psoas major arises from vertebrae T12 through L5 and passes beneath the inguinal ligament to insert on the lesser trochanter of the femur.
Deep Inguinal Region
Laparoscopic View
Deep Aspect of the Anterior Abdominal Wall, Peritoneal Folds, and Associated Structures. If one creates a space in the abdominal cavity by distending it with gas, an excellent view of the anterior wall can be obtained. The umbilical peritoneal folds (Fig. 72-9) in most subjects are very prominent and provide easily identified landmarks. The folds (ligaments) primarily exist because the peritoneum covers underlying structures.

Figure 72-9. The deep inguinal region and the anterior abdominal wall seen from within the abdomen. The urachus, the obliterated portion of the umbilical artery, and the inferior epigastric vessels are covered by peritoneal folds, respectively called the median, medial, and lateral umbilical folds.
The single median umbilical fold extends from the umbilicus to the urinary bladder and covers the urachus, the fibrous remnant of the fetal allantois. The urachus may be patent for a short distance in adults or may open into the umbilical scar in newborns. The medial umbilical fold is formed by the underlying obliterated portion of the fetal umbilical artery. This normally cordlike structure, like the urachus, may be patent for a portion of its length. Indeed, the proximal, patent portion of the artery normally supplies the superior vesicular arteries to the bladder. The lateral fold covers the inferior epigastric arteries as they course toward the posterior rectus sheath, which they enter approximately at the level of the arcuate line.
Between the median and the medial ligaments, a depression is usually found that is called the supravesical fossa. This is the site of hernias of the same name. The fossa formed between the medial and lateral ligaments is the medial fossa; this is the site of direct inguinal hernias. The lateral fossa is less well delineated than the others. The medial border of the fossa is formed by the lateral umbilical ligament and the rectus abdominis. This fossa does not have a lateral border; rather, the concavity slowly attenuates. The deep inguinal ring is located in the lateral fossa and therefore is the site of the congenital or indirect inguinal hernia.
Transversalis Fascia
The transversalis fascia (endoabdominal fascia) is perhaps the most commonly misunderstood structure in the literature devoted to groin hernia. Confusion results because surgeons may actually be referring to very different anatomic structures when discussing various hernia repairs; however, each may use the same anatomic term or eponym. Indeed, perhaps the biggest reservation among surgeons intent on performing a Shouldice repair is a precise definition of what is being sewn to what.
The transversalis fascia proper is a continuous sheet that extends throughout the extraperitoneal space. The term transversalis fascia is generally defined as the deep or endoabdominal fascia covering the internal surface of the transversus abdominis, the iliacus, the psoas muscles, and the obturator internus and portions of the periosteum. One variant of this convention is the use of terms specific to the muscle covered by the fascia (e.g., iliac fascia).
Most authors feel that only one layer of transversalis fascia exists, whereas others maintain that the transversalis fascia comprises two layers, or laminae.8 The posterior lamina is a layer of fibrous connective tissue that widely varies in density and continuity and is interspersed with adipose tissue, as seen in Figure 72-10. This layer is often referred to simply as the preperitoneal fascia. The anterior lamina is more uniform and is adherent to the deep surface of the transversus abdominis and the rectus abdominis. The posterior lamina is contained within the preperitoneal space, which is defined as the space between the peritoneum and the anterior lamina of the transversalis fascia. The inferior epigastric vessels are enclosed by, or interspersed with, the adipose tissue and the fibrous tissue of the posterior lamina of the transversalis fascia. The vessels are in contact anteriorly with the anterior lamina of the transversalis fascia as they course upward to enter the rectus abdominis sheath.
Transversalis Fascia Derivatives
The transversalis fascia analogs or derivatives are the iliopectineal arch, iliopubic tract, and crura of the deep inguinal ring. The superior and inferior crura form a transversalis fascia sling, a structure shaped like a “monk’s hood,” around the deep inguinal ring (Fig. 72-9). The transversalis fascia also contributes the internal spermatic fascia to the spermatic cord at this point. This “sling” has functional significance; when the transversus abdominis contracts, the crura of the ring are pulled upward and laterally, which results in a valvular action that helps to prevent the indirect formation of a hernia.

Figure 72-10. A parasagittal section through the layers of the anterior abdominal wall and groin. Observe that the transversalis fascia is depicted as a bilaminar structure.
The iliopubic tract (Figs. 72-9 and 72-11) has become an increasingly important landmark for surgeons as the use of laparoscopic technology has increased.9,10 The iliopubic tract is the thickened band of transversalis fascia formed at the zone of transition between the deep surfaces of the iliac and transversus abdominis muscles. The structure courses parallel to the more superficially located inguinal ligament, is attached to the iliac crest laterally, and inserts on the pubic tubercle medially. The tract forms along its course a portion of the inferior crus of the deep inguinal ring and then contributes to the anterior and medial walls of the femoral sheath. The tract fuses with the inguinal ligament to form a component of the inferior wall of the inguinal canal. At its insertion on the pubic tubercle, it curves backward slightly to blend with the Cooper pectineal ligament. The pectineal ligament is actually a condensation of periosteum and is not a true analog of the transversalis fascia, but it is reinforced by fibers from the iliopubic tract and inguinal ligament.

Figure 72-11. A schematic representation of the deep inguinal region. The iliopubic tract is shown as a thickening of the transversalis fascia, inferior to which many of the branches of the lumbar plexus exit the pelvis.
The iliopubic tract contains not only fibrous connective tissue but also some elastic fibers.11 In one series, the iliopubic tract was a substantial structure, suitable for use in hernia repairs, in 42% of the specimens examined. The tract, whether substantial or not, can be used as a readily identified landmark.
The iliopubic tract has particular significance because of its importance as a landmark to the laparoscopic surgeon. Many of the branches of the lumbar plexus run inferior to the tract, and damage to these nerves may be the result of aggressive dissection or the placement of tacks or staples to affix a prosthesis below this structure. The tract is not obviously visible in every patient from a laparoscopic view, but its location should always be immediately known to the surgeon because of its constant relationship to the other landmarks in this area.
The iliopectineal arch (Fig. 72-9) is also a condensation of the transversalis fascia. The iliopectineal arch commences at the medial border of the iliacus muscle, where it is continuous with the iliac fascia, itself a portion of the transversalis (endoabdominal) fascia. The arch separates the vascular compartment containing the femoral vessels from the neuromuscular compartment containing the iliopsoas muscle, femoral nerve, and lateral femoral cutaneous nerve. The iliopectineal arch also contributes to the proximal portion of the femoral sheath, thereby joining the iliopubic tract in the formation of the femoral sheath.
Femoral Sheath, Canal, and Ring
The femoral sheath (Fig. 72-12) is primarily composed of extensions of the transversalis fascia. The sheath is best understood in terms of the structures contained within. As the external iliac artery and vein pass beneath the inguinal ligament to become the femoral vessels, they are covered anteriorly by the transversalis fascia proper. This fascial layer is posteriorly and laterally joined by portions of the iliopsoas fascia, which themselves are continuations of the transversalis fascia. At the inguinal ligament, the iliopsoas fascia forms the iliopectineal arch. This arch divides the vascular compartment (lacuna vasorum), containing the femoral vessels, from the muscular portion (lacuna musculorum), which contains the iliopsoas muscle, femoral nerve, and lateral femoral cutaneous nerve. The vascular lacuna is further divided by septa into compartments for the vessels and the femoral branch of the genitofemoral nerve.

Figure 72-12. Schematic view of the femoral sheath, ring, and canal. The transversalis fascia forms the anterior portion of the sheath, and the iliopsoas fascia forms the posterior portion. Septae separate the vessels from each other and the vein from the femoral canal. The femoral ring contains a lymph node. The ring is formed medially by the aponeurosis of the transversus abdominis aponeurosis, anteriorly by the inguinal ligament, posteriorly by the pubic bone, and laterally by the femoral sheath.
The medial border of the femoral sheath follows the transversus abdominis aponeurosis to its insertion just lateral to that of the lacunar ligament and extends inferiorly to fuse eventually with the medial septum and adventitia of the femoral vein. The resultant cone-shaped cul-de-sac is the femoral canal. The canal normally contains only wisps of connective tissue and small lymphatic nodes. The wider proximal part of the canal, the femoral ring, contains a large node, which is often referred to as the Cloquet node.
The femoral ring is the extraperitoneal opening of the canal. The boundaries of the ring are formed medially by the curved edge of the transversus abdominis aponeurosis, not the lacunar ligament, which inserts more medially.12 Laterally, the ring is bounded by the connective tissue septum and the adventitia that is interposed between it and the femoral vein. The anterior boundary is the inguinal ligament; posteriorly, the ring is reinforced by the iliopubic tract and iliopectineal ligament. The canal is not in direct communication with the pelvic cavity. The transversalis fascia is not a component of the roof of the canal because it is diverted at this point to form the femoral sheath. This weakened area is therefore quite prone to hernia formation, especially in female subjects.
Inguinal (Hesselbach) Triangle
The inguinal triangle is the site of direct inguinal hernias. This triangle is most often described from the anterior aspect (Fig. 72-13), in which case the inguinal ligament forms the base of the triangle, the rectus abdominis forms the medial border, and the inferior epigastric vessels form the superolateral border. The triangle as originally described by Hesselbach had the pectineal ligament as its base. The latter description is quite useful to the surgeon viewing the abdomen from within because the inguinal ligament cannot be seen from this viewpoint. When the inguinal triangle is transilluminated, the thinness and translucency of the area of abdominal wall within the triangle underscores its importance in hernia development and repair. In the most translucent area, little or no muscle is present. Only the peritoneum and the transversalis fascia cover the triangle here. The aponeurotic arch of the transversus abdominis crosses the triangle just below the apex in most people. A high aponeurotic arch affords less reinforcement to the triangle and may therefore predispose a person to the formation of a direct inguinal hernia.
Components of the Spermatic Cord
The spermatic cord (Figs. 72-11 and 72-14) is closely associated with the deep inguinal ring. The spermatic cord is most appropriately described at this point because the deep ring itself is formed by derivatives of transversalis fascia, as is the innermost covering layer of the spermatic cord, the internal spermatic fascia. The middle covering layer is called the cremasteric fascia and contains the cremasteric muscle bundles; both are derived from the internal abdominal oblique muscle and fascia. The outermost covering of the spermatic cord is the external spermatic fascia, which is continuous with the investing fascia of the external abdominal oblique muscle.
The tunica vaginalis is initially a component of the cord, but normally it atrophies and closes early in neonatal life. This structure is an evagination of peritoneum. The testicle descends retroperitoneally in fetal life and is merely in contact with the posterior aspect of the tunica. An indirect congenital hernia enters the patent tunica vaginalis.

Figure 72-13. The inguinal (Hesselbach) triangle.

Figure 72-14. The component layers covering the contents of the spermatic cord.
The cord structures enclosed by the coverings described earlier are the ductus (vas) deferens, the pampiniform venous plexus, the testicular artery, and the genital branch of the genitofemoral nerve, a branch of the lumbar plexus (Figs. 72-9, 72-11, and 72-15).
Branches of the Lumbar Plexus
The nerves crossing the iliac fossa are some of the most variable in the body. This variability may be the cause of frequent intraoperative injury to the fragile nerves. The lumbar plexus is formed by roots from the 12th thoracic nerve and the first through fourth lumbar nerves. Cutaneous territories innervated by branches of the lumbar plexus are seen in Figure 72-15A. The five terminal branches commonly encountered in laparoscopic herniorrhaphy can be discerned in many people as they course across the iliacus muscle covered by peritoneum and the iliac fascia (a portion of the transversalis–endopelvic fascia). The nerves form within or deep to the psoas major muscle (Fig. 72-15B), often ramifying with other nerves within or close to the muscle. The nerve branches initially lie within the so-called triangle of pain,13 bordered medially by the psoas muscle, anteriorly and inferiorly by the iliopubic tract, and laterally by the iliac crest. With the exception of the genital branch of the genitofemoral nerve, the branches of the lumbar plexus destined for the thigh run beneath the iliopubic tract.

Figure 72-15. A: The cutaneous territories innervated by several branches of the lumbar plexus. B: Some of the branches of the lumbar plexus seen from within the abdomen.
The most anterior of the nerves encountered, the genitofemoral nerve, is also the most variable. This nerve may occur as a single trunk lying deep to the peritoneum and fascia on the anterior surface of the psoas muscle. The nerve may also divide into its component genital and femoral branches within the muscle. The genital branch travels with the spermatic cord, entering at the deep inguinal ring; it ultimately innervates the cremaster muscle and the lateral scrotum. The femoral branch of the nerve innervates the skin of the proximal mid-thigh.
The lumbar plexus branch encountered immediately deep to the lateral aspect of the psoas muscle is the large femoral nerve. Although not routinely encountered during laparoscopy, the femoral nerve has been injured in some cases.14 The lateral femoral cutaneous nerve crosses the iliac fossa under the iliac fascia to run deep to the iliopubic tract and the inguinal ligament, which it pierces to enter the thigh.
The iliohypogastric nerve typically arises with the ilioinguinal by a common trunk from the first lumbar nerve. They may exchange fibers within the muscle, but they usually diverge immediately to form individual nerves. The iliohypogastric nerve crosses the iliac fossa just inferior to the kidney and pierces the transversus abdominis. The subsequent course of the nerve carries it between the transversus and the internal abdominal oblique until it pierces the aponeuroses of both obliques just above the external inguinal ring.
The ilioinguinal nerve normally crosses the iliac fossa just inferior to the iliohypogastric nerve. In its typical further course, the nerve pierces the transversus abdominis and internal abdominal oblique above the iliac crest and eventually enters the inguinal canal. The nerve may run more diagonally through the iliac fossa and then pierce the iliopubic tract to reach the inguinal canal.15 This path “can obviously render the nerve more vulnerable to iatrogenic injury.”
Vasculature of the Abdominal Wall and Deep Inguinal Region
The vasculature of the deep inguinal region and anterior abdominal wall has been analyzed by surgeons for well over 100 years. The importance and variability of these vessels have been underscored by the ominous mnemonics used to refer to them – “crown of death” (corona mortis) and “triangle of doom.” The primary blood supply to the deep anterior wall is from the inferior epigastric artery. This artery is a branch of the external iliac artery. In many cases, an artery called the “aberrant” obturator artery arises from the inferior epigastric, which joins the “normal” obturator artery and thereby forms a circle – the corona mortis – before entering the obturator foramen. Injury to the circle, usually sustained while the surgeon is working in the area of the Cooper ligament, causes copious bleeding. Recent studies have indicated that aberrant obturator vessels are present in between 60% and 90% of the whole pelves studied.16,17
The veins in this area are also prone to injury because many, especially the iliopubic veins and obturator veins and their tributaries, may be much larger than their accompanying arteries. One network of veins in the area is situated on the inferior deep surface of the rectus muscles. The veins of this network, which anastomose with the pubic branches discussed earlier, have been called the rectusial veins.18
The vessels in the vascular compartment of the deep inguinal region are the external iliac artery and vein. They arise within a triangular area bordered laterally by the gonadal vessels and medially by the ductus deferens. The primary continuations of the external iliac vessels are the femoral artery and vein. The inferior epigastric artery is a branch of the external iliac. The obturator artery may arise from either of these arteries as a replacement or accessory to the obturator branch of the internal iliac artery.
A final vessel to consider in this review is the deep circumflex iliac artery (Fig. 72-10). The origin of this artery is extremely variable, but its course is predictable along the iliopubic tract. It pierces the transversalis fascia and runs along the iliac fossa to anastomose eventually with a deep lumbar artery. Because the deep circumflex artery runs along the iliopubic tract, it can inadvertently be stapled or otherwise injured during laparoscopic herniorrhaphy.
Pelvic Floor and Obturator Muscles
The pelvic musculature normally affords remarkable support to the structures within the true pelvis. Although a myoaponeurotic hammock-like sheet forms the pelvic diaphragm, obturator muscles and membrane, and urogenital diaphragm, herniation of fat or viscera through or around any of these layers occurs. The potential for hernia formation is increased because of the openings through which many structures exit or enter the pelvis.
The Latin term obturator is translated as “stopper for a bottle.” The aptly named obturator internus, along with its membrane and the obturator externus, closes off nearly all the large obturator foramen. The small superolateral aperture through which the obturator vessels and nerve pass is the site where obturator hernias form. The obturator internus arises from the deep surface of parts of all three pelvic bones. The muscle fibers converge on a tendon, which leaves the pelvis through the lesser sciatic foramen to insert on the greater trochanter. The dense internal obturator fascia covers the muscle and is thickened to form the arcuate ligament, from which the levator ani muscles (the pelvic diaphragm) are in part suspended. The obturator internus fascia splits to enclose the pudendal vessels in the pudendal canal. The external obturator muscle arises from the pelvic bones surrounding the obturator foramen and from the anterior portion of the obturator membrane. The external obturator muscle is supplied by the obturator nerve and vessels.
The component muscles of the bowl-shaped pelvic diaphragm, the pubococcygeus, iliococcygeus, and puborectalis, along with the coccygeus form the floor of the pelvis. The pubococcygeus arises from the posterior aspect of the pubis and the thickened portion of the internal obturator fascia, called the tendinous arch (Fig. 72-16), that spans the distance between the pubis and ischial spine. The puborectalis, the midportion of the diaphragm, arises from the pubis and loops around the rectum as the puborectal sling. The iliococcygeus is suspended at its origin from the tendinous arch and inserts on the coccyx. The coccygeus muscle completes the diaphragm posteriorly, arising from the ischial spine and inserting on the sides of the coccyx.
The area remaining between the sacrum and the greater sciatic foramen is filled for the most part by the piriformis muscle. The piriformis arises from the anterior surface of the second through fourth sacral vertebrae and the sacrotuberous ligament. This muscle exits the pelvis through the greater sciatic foramen, which is thereby divided into suprapiriform and infrapiriform portions (Figs. 72-16 and 72-17). The superior gluteal nerves and vessels pass through the suprapiriform foramen, whereas the inferior gluteal nerves and vessels in company with the sciatic nerve pass through the infrapiriform foramen.
The most pronounced deficit in the pelvic diaphragm is situated anteriorly, where an aperture must allow the urogenital structures to pass out of the pelvis. This area is reinforced by the urogenital diaphragm, a structure primarily consisting of the superficial and deep transverse perineal muscles. The deep transverse perineal muscle is enclosed by a weak superior fascia and a sturdier inferior perineal fascia. The urogenital diaphragm recently has been shown to be more funnel shaped than sandwichlike, as previously depicted in many atlases. The urogenital diaphragm exists only in humans because the human pelvic outlet faces inferiorly, unlike that of quadrupeds.

Figure 72-16. A: The pelvic diaphragm (levator ani and the piriformis) and the urogenital diaphragm viewed from within the pelvis. B: Hemisection of the pelvis revealing the levator ani, piriformis, obturator internus, and psoas muscles.
WHY DO HUMAN BEINGS DEVELOP ABDOMINAL WALL HERNIAS?
2 The most common hernias develop at sites where the abdominal wall has natural openings such as the internal inguinal ring, the umbilicus, and the esophageal hiatus. Previous surgical entry sites (incisional hernia) are also common areas where hernias develop. Factors that increase the pressure in the abdominal cavity, such as obesity, heavy lifting, coughing with chronic lung disease, straining during a bowel movement or urination (prostatism), chronic lung disease, and ascites, have traditionally been considered important in the etiology, especially at these natural openings. Developmental phenomena also play a role. For example, in the evolution from a quadruped to a biped, the unprotected groin is more vulnerable to changes in intra-abdominal pressure, predisposing to inguinal herniation. Major risk factors for the development of an abdominal wall hernias include chronic obstructive pulmonary disease, smoking, high intra-abdominal pressure, collagen vascular disease, thoracic or abdominal aortic aneurysm, peritoneal dialysis, matrix metalloproteinase (MMP) abnormalities, and an increased type I: type III collagen ratio.
The role of heavy lifting, especially a single strenuous event, is an unsettled question and has considerable medical–legal ramifications. There is minimal evidence that vigorous abdominal wall activity is an independent risk factor for abdominal wall hernia development despite the overwhelming opinion to the contrary in the lay literature.19–21 In a systematic review of existing evidence performed by Svendsen and colleagues, regarding the effects of occupational and mechanical exposures in relation to inguinal hernias, a causal relationship could not be established between specific mechanical insults and occurrence of inguinal hernia, hernia recurrence, and persistent pain after inguinal hernia repair.22,23 According to the results of another register-based male cohort study in Denmark, increasing cumulative exposure to daily lifting activities and prolonged standing or walking at work was associated with an increased risk of having an indirect inguinal hernia repair.22,23 The authors hypothesized that patent processus vaginalis may be more susceptible to changes in intra-abdominal pressures, whereas in a direct hernia, other mechanisms may be involved in connective tissue degradation and weakening of the transversalis fascia. Still, the role of mechanical activity in the development of abdominal wall hernia is not completely clear, and further research is needed to determine if inguinal hernias can be prevented and if the postoperative prognosis can be improved by reducing occupational mechanical exposures.

Figure 72-17. The gluteal muscles and lateral rotators of the hip. External relations of the sciatic foramen are also evident.
Familial predisposition and the role of connective tissue diseases in hernia development have received considerable attention in recent years. Particularly important for inguinal hernia is a family history as it has been reported to increase the risk up to 8 times.24,25 Various connective tissue disorders, such as osteogenesis imperfecta, Marfan syndrome, Ehlers–Danlos syndrome, and congenital hip dislocation, are associated with hernias. Imbalances in collagen, the basic building block of the abdominal wall, are believed to contribute to hernia disease. While type I collagen confers predominantly tensile strength, type III collagen consists of thinner fibers and is regarded as a temporary matrix during tissue remodeling. A decreased ratio of type I to type III collagens can be detected in fascial and skin specimens obtained from patients with hernias. A similar phenomenon was discovered by Cannon and Read in smokers when they performed biopsies of the rectus sheaths from adult smokers with inguinal hernias and coined the term “metastatic emphysema.”26 The investigators also demonstrated significantly greater levels of circulating serum elastolytic activity in patients who smoke.
Aside from primary defects in collagen synthesis, the imbalances in collagen in patients with hernia can be attributed to altered extracellular matrix (ECM) which is maintained in a dynamic balance of synthesis and degradation by a complex set of enzymes. MMPs,27 which are a group of 23 structurally related zinc-dependent enzymes, protease with collagenolytic activity and have a pivotal role in the integrity and composition of the ECM.
The association between MMP overexpression and abdominal wall hernia was initially demonstrated by Bellon and colleagues, who studied the expression of MMP-1 and MMP-2 in the transversalis fascia of patients with direct and indirect inguinal hernia. Since then, several studies have been conducted, aiming at elucidating the role of MMPs in the development of primary and recurrent abdominal wall hernias. For this purpose, MMP levels in fascia, skin, hernia sac, and blood specimens of patients with inguinal, incisional, and recurrent abdominal wall hernias have been measured. The expression of MMP mRNA in cultured fibroblasts has also been investigated to evaluate cell–cell and cell–matrix interactive mechanisms as causative factors for abnormal MMP production. In addition to this, in-vitro studies have been performed to study the effect of different mesh materials on MMP expression on cultured fibroblasts; in one such study, polypropylene has been shown to induce less MMP2 expression than polygalactin. The function of MMPs is further regulated by a variety of endogenous regulators, the most important ones are known as tissue inhibitors of MMPs (TIMPs).28–30 Studies have shown that an imbalance between MMP and TIMP activity may have a role pathogenesis of abdominal wall hernias.31
Despite the increasing knowledge regarding MMPs in incisional and inguinal hernia, the exact role of MMPs in the pathogenesis of hernia formation remains unclear. Most studies are small and not completely controlled; and there is a remarkable heterogeneity among studies concerning the characteristics of the study populations, the examined tissue specimens, and the biochemical assays used. In addition to MMP and TIMPs, factors that have been implicated in hernia development include deficiency of the elastic fiber system of the transversalis fascia, decreased tropoelastin and lysyl oxidase-like 1 synthesis, elastase overexpression and TGF-beta1 overexpression. It remains unclear whether these alterations in hernia patients are a part of the “cause or effect” phenomenon. Another interesting area of research are drug classes that can suppress MMP expression (e.g., tetracyclines, especially doxycycline, aspirin, statins, and thiazolidinediones) and understanding their role in hernia prevention and treatment.32

Figure 72-18. Reduction of a hernia by taxis. A: Applying pressure on the hernia directly occludes the neck. B: Elongating the neck of the hernia while applying pressure allows reduction.
COMPLICATIONS OF HERNIAS
Hernia Accident
For years, surgeons have been taught that all hernias should be repaired at diagnosis to prevent a hernia accident, which is defined as a bowel obstruction or incarceration with strangulation, because of the perception that patients presenting with these complications have an unacceptable increase in mortality. This thinking, however, has not stood up when tested in randomized controlled trials. For example, for men with asymptomatic inguinal hernias, randomized controlled trials have shown that a strategy of watchful waiting is safe.33
Incarceration
Incarcerated means “trapped” or “imprisoned.” Clinically, an incarcerated hernia is an irreducible hernia. An incarceration is not in and of itself a surgical emergency. Many hernias are chronically incarcerated due to adhesions of contents (e.g., omentum, bowel, ovary, etc.) to the hernia sac. The hernia itself is not necessarily tense to palpation, and the overlying skin appears normal. Normal bowel sounds may be heard within the hernia. It is important to differentiate an incarcerated hernia from a hydrocele of the cord. One can get above the hydrocele with the examining fingers. One cannot get above a hernia, however, as it communicates with the abdominal cavity. Hydroceles will transilluminate clearly, but a hernia will not.
An acutely incarcerated, painful hernia must be managed carefully. An attempt at reduction is reasonable unless there are signs of strangulation, which is not always obvious by clinical examination. Immediate surgical exploration is the safest approach when the diagnosis is not clear. The advantage of reduction followed by elective repair is that edematous tissue associated with an acute incarceration can return to normal, which presumably will translate into a better repair with less chance of infection.34 If an attempt at reduction seems reasonable, the patient is sedated and placed in bed. The Trendelenburg position should facilitate reduction of a groin hernia. An attempt should be made at the initial examination to reduce the hernia. The maneuver of taxis entails grasping the neck of the hernia with the fingers of one hand and then applying intermittent pressure on the most distal part of the hernia with the other hand. Taxis has the effect of elongating the neck of the hernia so that the contents of the hernia may be guided through this area back into the abdominal cavity with a rocking movement. Mere pressure on the most distal part of the hernia causes bulging of the hernial sac around the neck, which can occlude the neck and prevent it from being reduced (Fig. 72-18). The maneuver of taxis should not be performed with excessive pressure or too vigorously. If the hernia is strangulated, gangrenous bowel might be reduced into the abdomen or perforated in the process. One or two gentle attempts should be made at taxis. If they are unsuccessful, this procedure should be abandoned. Rarely, the hernia together with its peritoneal sac and constricting neck may be reduced into the abdomen (reduction en masse). The patient would then have persistent obstruction after reduction of the hernia.
Intestinal Obstruction
One hundred years ago, the most common cause of intestinal obstruction was a hernia. At the present time, hernia is third, after adhesive obstructions and cancer. Hernia is an important cause of obstruction that is not infrequently missed on clinical examination. When a patient with an intestinal obstruction is examined, great emphasis should be placed on adequate exposure of the entire abdominal wall and groin area (from nipples to knees). Proper lighting is essential because previous scars can fade with time and become barely perceptible. The patient with intestinal obstruction as a result of a hernia will have a tense hernia that is irreducible. The abdomen itself will be distended, and high-pitched bowel sounds with frequent rushes will be heard. If the process continues to the complication of strangulation, these signs will disappear. Unlike adhesive small-bowel obstructions, partial small-bowel obstructions secondary to hernia are rare. Most patients will have had vomiting and obstipation.
A plain roentgenogram of the abdomen will reveal the signs of an intestinal obstruction – dilated loops of bowel with air–fluid levels and no bowel gas distal to the obstruction. Frequently on a plain roentgenogram, one can see bowel shadows in the region of the hernia. A lateral view is often useful to demonstrate this feature more clearly. Contrast studies are not usually necessary in this instance. Computed tomography (CT) reliably demonstrates the hernia with characteristic features of obstruction and should be considered if the clinical diagnosis is not certain (Fig. 72-19) because a distal intestinal obstruction secondary to another cause (e.g., adhesions) may result in significant distention of a coincidental nonobstructing hernia of the abdominal wall. Should the examiner focus attention exclusively on the hernia, the real cause of the obstruction may be missed when the hernia is repaired.

Figure 72-19. Computed tomogram showing a left-sided inguinal hernia.
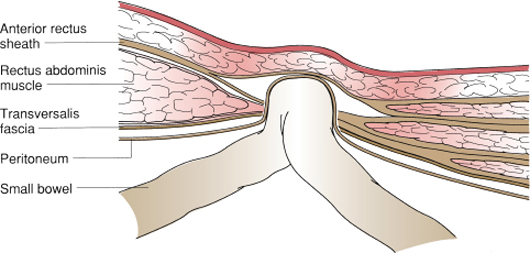
Figure 72-20. Richter hernia. Part of the bowel wall herniates through the defect in the abdominal wall.
The next steps in management include resuscitation followed by urgent surgery. At surgery, an approach directly over the hernia is used. In all patients, the entire gastrointestinal tract must be assessed to eliminate causes of obstruction other than the hernia itself. This is done before the hernia is repaired. The bowel, if viable, is reduced into the abdomen. If difficulty is encountered in reducing the hernia, the neck of the hernia can be widened. In the case of an inguinal hernia, division of the neck with or without ligation and division of the inferior epigastric vessels is safe, and the hernia contents can be reduced into the abdomen. In the case of a femoral hernia, the inguinal ligament can be split anteriorly and the hernia contents reduced into the abdomen. If the bowel is nonviable, then a bowel resection can be performed with anastomosis. The hernia is then repaired.
Strangulation
Strangulation of a hernia is a serious and life-threatening condition in which the hernia contents become ischemic and nonviable. The pathogenesis of this condition involves intra-abdominal contents within the hernia sac. Straining may push more contents into the sac, and the tense sac then causes pressure at the neck. This pressure initially produces venous congestion, resulting in edema. Eventually, the pressure is so great that the arterial supply is obstructed and the contents become gangrenous.
When intestine is involved, in addition to having an irreducible hernia and intestinal obstruction, the patient is toxic, dehydrated, and febrile. Examination of the abdomen reveals the signs of an intestinal obstruction, with distention and increased bowel sounds. Absolute constipation and vomiting are other manifestations. The hernia itself is tense, irreducible, and very tender, and the overlying skin may be discolored with a reddish or bluish tinge. No bowel sounds are heard within the hernia itself. The patient commonly manifests a leukocytosis with a predominance of polymorphonuclear leukocytes. Blood gases may reveal metabolic acidosis.
Management of these patients requires urgent attention to detail. No attempt should be made to reduce the hernia. Rapid resuscitation should commence immediately, with nasogastric suction and replacement of fluids and electrolytes. The patient should be given antibiotics. Once the patient is resuscitated, urgent surgery commences to expose the hernia, open the sac, and assess the viability of the bowel. More bowel can be pulled into the hernia so that viable bowel can be transected and the gangrenous portion removed. An end-to-end anastomosis should be performed and the bowel then reduced into the abdominal cavity. The hernia is then repaired.
Richter Hernia
August Gottlieb Richter in 1785 described a hernia type that bears his name in which the antimesenteric border of the intestine protrudes into the hernia sac without involving the entire circumference of the intestine so that intestinal obstruction does not occur (Fig. 72-20). The most common site is the femoral ring (36% to 88%), followed by the inguinal canal (12% to 36%), and an abdominal wall incision (4% to 25%). Miscellaneous locations include umbilical, obturator, supravesical, Spigelian, triangle of Petit, sacral foramen, Morgagni, internal, and diaphragmatic.35 The routine use of laparoscopy by general surgeons has resulted in an increased incidence at trocar sites such that most surgeons will repair the fascia for trocar sleeves greater than 5 mm. The surgical treatment is to expose the herniated bowel by opening the sac. The neck of the sac is enlarged to allow delivery of the bowel into the wound. Any areas of gangrene are excised and the bowel wall reconstituted. The hernia is then repaired.
Prosthetic Materials
The development of a wide variety of materials for abdominal wall reconstruction makes it possible to individualize the selection of prostheses so that they can be used in almost all clinical situations. Currently available prosthetic materials can be divided into synthetic or biologic depending on their source. The synthetic prostheses can be further classified as noncomposite heavyweight plastic, noncomposite heavyweight membrane, noncomposite lightweight plastic, composite, and coated prosthesis. Although surgeons use the term mesh to refer to all varieties of hernia prostheses, this is inaccurate as only prostheses that have large grossly visible interstices should be termed meshes. Microporous material such as expanded polytetrafluoroethylene (ePTFE) have no visible interstices, do not promote ingrowth as with the mesh prosthesis, and act more like a membrane. In the absence of infection, synthetic material is preferred to biologic prostheses. The materials that have the longest track record for routine use in hernia surgery include polyester, polypropylene, either monofilament (Marlex, Prolene) or polyfilament (Surgipro); Dacron (Mersilene); and ePTFE (Gore-Tex). Synthetic materials such as polypropylene or polyester work by inciting an intense fibroplastic response to form a strong scar plate interface. Because of this response, they should not be used adjacent to the viscera because of the propensity of these materials to erode into intra-abdominal organs, most commonly intestine, resulting in fistulization. The weight of the polypropylene or polyester meshes, as well as the size of the pores, is a controversial issue currently. As an example, a 7.5- × 15-cm polypropylene mesh (Prolene, Ethicon, Inc.) contains about 80 g/m2 of polypropylene, while a polypropylene–poliglecaprone-25 (Monocryl) lightweight mesh of the same size (UltraPro, Ethicon, Inc.) contains less than 30 g/m2. One of the ways of reducing the amount of nonabsorbable material in a mesh is to increase the size of the pores. Many authorities believe that the inflammatory response incited by the small-pore, heavyweight plastic meshes can lead to chronic pain; a sensation of being able to feel the mesh; increased stiffness of the abdominal wall with loss of compliance; and shrinkage, which can lead to recurrence. There is increasing evidence that decreasing the density of polypropylene and increasing the size of the pores reduces this foreign body response, resulting in less long-term pain than normal mesh with a similar recurrence rate.36–39 Recently, there have been reports of “burst mesh” or central mesh failures with the use of lightweight mesh in the repair of large ventral hernias and thus, one should exercise caution while employing them in repair of massive hernias.40
Table 72-2 Classification and Examples of Prosthetic Materials Currently Available for Hernia Repair

When contact of mesh with viscera cannot be avoided, either a nonmesh material such as ePTFE alone or a dual-layered prosthesis with a standard plastic mesh on the side facing the abdominal wall and ePTFE facing the viscera should be used. Because ePTFE heals by encapsulation rather than incorporation as with the mesh prostheses, it does not have the propensity to erode or fistulize which makes it suitable for use adjacent to abdominal viscera. This lack of incorporation has a downside in terms of increased susceptibility of ePTFE to infection, and when these do get infected, they almost always have to be removed. On the other hand, the incorporated mesh materials such as polypropylene can commonly be salvaged when infected. To address this issue, manufacturers have developed temporary or biodegradable adhesion barrier materials to coat the mesh on the side facing the viscera to prevent adhesions without the need for ePTFE (Table 72-2). The injured peritoneum forms a mesothelial layer as quickly as 5 to 7 days with the adhesion barrier products being degraded over the course of an average of 30 (range 28 to 240) days.41 Neither the adhesion barriers nor ePTFE can completely eliminate adhesions. Patients who undergo laparotomy after either type of prosthesis has been placed intra-abdominally are routinely found to have significant adhesions to the device. However, these adhesions tend to be filmy and easily taken down. It remains to be seen if the long-term performance of coated prostheses compares to ePTFE with its long track record.
The role of biologic tissue matrix in ventral hernia repair has evolved over the last few years. These materials are derived from human, porcine, or fetal bovine skin; porcine small intestine submucosa; and bovine pericardium and are processed to remove hair, cells, and cell components as well as other antigens present in the matrix, leaving only the highly organized collagen architecture with the surrounding extracellular ground tissue.32 The matrix acts as a scaffold to allow native tissue and neovascularization to infiltrate the healing wound and promote strong tissue in-growth that limits contraction. These properties, theoretically, lead to increased resistance to infection. Some biologics undergo further processing to increase the crosslinking between collagen fibers and to decrease susceptibility to degradation by collagenase. In general, these materials possess the physical and mechanical characteristics of a clinically acceptable surgical mesh in that have sufficient mechanical strength to withstand the physiologic and anatomic stresses of the abdominal wall.42–44
Because of the properties mentioned above, it is felt by some surgeons that the best indication for biologic prostheses is in contaminated wounds, where a synthetic prosthesis may be contraindicated. They are not useful in grossly infected wounds presumably because of the high collagenase content present, which destroys them. It should be noted that none of the biologic prostheses are regulated by the U.S. Food and Drug Administration (FDA) as a medical device and are typically approved under a 510 (K) approval process which is primarily based on safety. These materials were introduced into clinical practice and used in contaminated fields without prior FDA approval or clearance for this indication.45 Another major argument against them is their expense, as they generally cost 20 to 30 times that of the plastic prostheses. The majority of evidence for the use of biologic prostheses for hernia repair is based on retrospective studies and mostly in clean procedures. Pooled results from 635 patients treated with three different biologic materials have revealed a recurrence rate of up to 18% to 30% at 2 years with biologic prostheses.43,45 Retrospective study with long-term follow-up (>5 years) with the use of porcine acellular dermal matrix for repair of incisional hernias at risk for infection showed a recurrence rate of 20% and 53% when used as an onlay or intraperitoneal sublay technique compared to 80% when used as a bridge in either inlay or sublay position, with particularly high recurrence rates seen in contaminated (71%) and grossly infected (100%) wounds.46 Although these materials allow a single stage repair of hernias in a contaminated field, with no or little need for mesh removal in case of subsequent wound infection, the high rate of recurrence with the use of biologics, especially in contaminated fields, remains a concern.
A major factor in the popularity of biologic materials for hernia repair is reluctance among many surgeons regarding the use of synthetic mesh in clean-contaminated and contaminated fields, although currently available evidence does not support this view. A retrospective review evaluating lightweight polypropylene mesh placed in a sublay position, in clean-contaminated and contaminated cases revealed a 14% surgical site infection rate (7.1% for clean-contaminated and 34% for contaminated cases) and a 7% recurrence rate at a mean follow-up of 10 months.47 Results from this and other similar experiences challenge the surgical dictum contraindicated in a contaminated surgical field. Even when infectious complications occur, synthetic macroporous mesh materials can often be salvaged without the need for removal.
Another interesting area of research is development of novel materials which integrate the desirable qualities of both biologic and synthetic mesh materials. Examples of such materials include Phasix® mesh (Davol, Warwick RI) which is a long-term resorbable mesh composed of monofilament poly-4-hydroxybutyrate, TIGR Matrix Surgical Mesh® (Novus Scientific) which is composed of fast and slow degrading synthetic reabsorbable fibers (Glycolide, lactide and trimethylene carbonate). These materials are gradually reabsorbed by the body in a predictable fashion. Preclinical in vivo and in vitro studies have shown favorable results with these materials, and further clinical evaluation with these materials is currently underway.48 Fast absorbable prostheses such as polyglactin are not durable and almost never result in long-term correction of an abdominal wall defect but are sometimes useful as a temporary substitute when a nonabsorbable prosthesis is contraindicated (i.e., a grossly infected wound).
GROIN HERNIAS
Groin hernias are described as inguinal and femoral, the inguinal hernias being further subdivided into direct and indirect hernias (some authorities refer to these as medial and lateral hernias, respectively). Groin hernias may be primary or recurrent. An indirect hernia occurs as a protrusion of abdominal contents through the internal ring, lateral to the inferior epigastric vessels, into the inguinal canal. Indirect inguinal hernias (lateral hernias) are situated within the spermatic cord and therefore may extend into the scrotum. In female patients, the hernia follows the round ligament and may present as a swelling in the labium. A direct hernia (medial hernia) is a protrusion through the triangle of Hesselbach medial to the inferior epigastric vessels. These hernias develop through an area where the endoabdominal fascia is not protected by overlying muscle. Direct hernias do not usually involve the cord, as they tend to protrude forward. However, they occasionally track alongside the cord down the entire length of the inguinal canal and even enter the scrotum. For this reason, the only absolute distinction between a direct and an indirect hernia is the relationship to the inferior epigastric vessels. Inguinal hernias are more common in men (with a 15% lifetime risk among men aged >25 years) with a male to female age-adjusted ratio of 7.5:1.49 A femoral hernia protrudes through the femoral canal, which is bordered by the inguinal ligament superiorly, the pubic ramus medially and inferiorly, and the femoral vein laterally. This hernia presents below the inguinal ligament (Fig. 72-21). In a sliding hernia, part of the sac is formed by the viscera, on the left side the sigmoid colon or bladder, and on the right side the cecum or bladder (Fig. 72-22).

Figure 72-21. A: Inguinal hernia. This presents above the inguinal ligament and extends below it. B: Femoral hernia. This presents below the inguinal ligament.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree



