PART 17: Neurologic Disorders
SECTION 1 | DIAGNOSIS OF NEUROLOGIC DISORDERS |
437 | Approach to the Patient with Neurologic Disease |
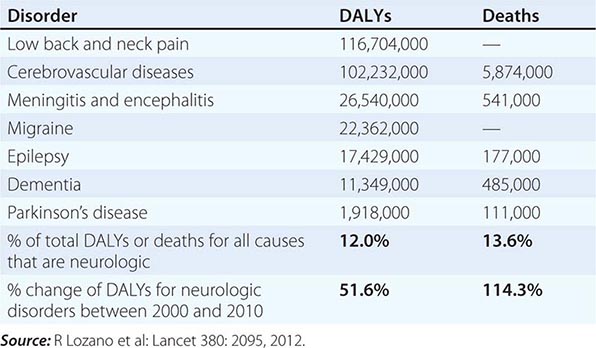
Neurologic diseases are common and costly. According to estimates by the World Health Organization, neurologic disorders affect over 1 billion people worldwide, constitute 12% of the global burden of disease, and cause 14% of global deaths (Table 437-1). These numbers are only expected to increase as the world’s population ages. Most patients with neurologic symptoms seek care from internists and other generalists rather than from neurologists. Because therapies now exist for many neurologic disorders, a skillful approach to diagnosis is essential. Errors commonly result from an overreliance on costly neuroimaging procedures and laboratory tests, which, while useful, do not substitute for an adequate history and examination. The proper approach to the patient with a neurologic illness begins with the patient and focuses the clinical problem first in anatomic and then in pathophysiologic terms; only then should a specific diagnosis be entertained. This method ensures that technology is judiciously applied, a correct diagnosis is established in an efficient manner, and treatment is promptly initiated.
GLOBAL DISABILITY-ADJUSTED LIFE-YEARS (DALYs) AND NUMBER OF ANNUAL DEATHS FOR SELECTED NEUROLOGIC DISORDERS IN 2010 |
THE NEUROLOGIC METHOD
DEFINE THE ANATOMY
The first priority is to identify the region of the nervous system that is likely to be responsible for the symptoms. Can the disorder be mapped to one specific location, is it multifocal, or is a diffuse process present? Are the symptoms restricted to the nervous system, or do they arise in the context of a systemic illness? Is the problem in the central nervous system (CNS), the peripheral nervous system (PNS), or both? If in the CNS, is the cerebral cortex, basal ganglia, brainstem, cerebellum, or spinal cord responsible? Are the pain-sensitive meninges involved? If in the PNS, could the disorder be located in peripheral nerves and, if so, are motor or sensory nerves primarily affected, or is a lesion in the neuromuscular junction or muscle more likely?
The first clues to defining the anatomic area of involvement appear in the history, and the examination is then directed to confirm or rule out these impressions and to clarify uncertainties. A more detailed examination of a particular region of the CNS or PNS is often indicated. For example, the examination of a patient who presents with a history of ascending paresthesias and weakness should be directed toward deciding, among other things, if the location of the lesion is in the spinal cord or peripheral nerves. Focal back pain, a spinal cord sensory level, and incontinence suggest a spinal cord origin, whereas a stocking-glove pattern of sensory loss suggests peripheral nerve disease; areflexia usually indicates peripheral neuropathy but may also be present with spinal shock in acute spinal cord disorders.
Deciding “where the lesion is” accomplishes the task of limiting the possible etiologies to a manageable, finite number. In addition, this strategy safeguards against making serious errors. Symptoms of recurrent vertigo, diplopia, and nystagmus should not trigger “multiple sclerosis” as an answer (etiology) but “brainstem” or “pons” (location); then a diagnosis of brainstem arteriovenous malformation will not be missed for lack of consideration. Similarly, the combination of optic neuritis and spastic ataxic paraparesis suggests optic nerve and spinal cord disease; multiple sclerosis (MS), CNS syphilis, and vitamin B12 deficiency are treatable disorders that can produce this syndrome. Once the question, “Where is the lesion?” is answered, then the question, “What is the lesion?” can be addressed.
IDENTIFY THE PATHOPHYSIOLOGY
Clues to the pathophysiology of the disease process may also be present in the history. Primary neuronal (gray matter) disorders may present as early cognitive disturbances, movement disorders, or seizures, whereas white matter involvement produces predominantly “long tract” disorders of motor, sensory, visual, and cerebellar pathways. Progressive and symmetric symptoms often have a metabolic or degenerative origin; in such cases lesions are usually not sharply circumscribed. Thus, a patient with paraparesis and a clear spinal cord sensory level is unlikely to have vitamin B12 deficiency as the explanation. A Lhermitte symptom (electric shock–like sensations evoked by neck flexion) is due to ectopic impulse generation in white matter pathways and occurs with demyelination in the cervical spinal cord; among many possible causes, this symptom may indicate MS in a young adult or compressive cervical spondylosis in an older person. Symptoms that worsen after exposure to heat or exercise may indicate conduction block in demyelinated axons, as occurs in MS. A patient with recurrent episodes of diplopia and dysarthria associated with exercise or fatigue may have a disorder of neuromuscular transmission such as myasthenia gravis. Slowly advancing visual scotoma with luminous edges, termed fortification spectra, indicates spreading cortical depression, typically with migraine.
THE NEUROLOGIC HISTORY
Attention to the description of the symptoms experienced by the patient and substantiated by family members and others often permits an accurate localization and determination of the probable cause of the complaints, even before the neurologic examination is performed. The history also helps to bring a focus to the neurologic examination that follows. Each complaint should be pursued as far as possible to elucidate the location of the lesion, the likely underlying pathophysiology, and potential etiologies. For example, a patient complains of weakness of the right arm. What are the associated features? Does the patient have difficulty with brushing hair or reaching upward (proximal) or buttoning buttons or opening a twist-top bottle (distal)? Negative associations may also be crucial. A patient with a right hemiparesis without a language deficit likely has a lesion (internal capsule, brainstem, or spinal cord) different from that of a patient with a right hemiparesis and aphasia (left hemisphere). Other pertinent features of the history include the following:
1. Temporal course of the illness. It is important to determine the precise time of appearance and rate of progression of the symptoms experienced by the patient. The rapid onset of a neurologic complaint, occurring within seconds or minutes, usually indicates a vascular event, a seizure, or migraine. The onset of sensory symptoms located in one extremity that spread over a few seconds to adjacent portions of that extremity and then to the other regions of the body suggests a seizure. A more gradual onset and less well-localized symptoms point to the possibility of a transient ischemic attack (TIA). A similar but slower temporal march of symptoms accompanied by headache, nausea, or visual disturbance suggests migraine. The presence of “positive” sensory symptoms (e.g., tingling or sensations that are difficult to describe) or involuntary motor movements suggests a seizure; in contrast, transient loss of function (negative symptoms) suggests a TIA. A stuttering onset where symptoms appear, stabilize, and then progress over hours or days also suggests cerebrovascular disease; an additional history of transient remission or regression indicates that the process is more likely due to ischemia rather than hemorrhage. A gradual evolution of symptoms over hours or days suggests a toxic, metabolic, infectious, or inflammatory process. Progressing symptoms associated with the systemic manifestations of fever, stiff neck, and altered level of consciousness imply an infectious process. Relapsing and remitting symptoms involving different levels of the nervous system suggest MS or other inflammatory processes. Slowly progressive symptoms without remissions are characteristic of neurodegenerative disorders, chronic infections, gradual intoxications, and neoplasms.
2. Patients’ descriptions of the complaint. The same words often mean different things to different patients. “Dizziness” may imply impending syncope, a sense of disequilibrium, or true spinning vertigo. “Numbness” may mean a complete loss of feeling, a positive sensation such as tingling, or even weakness. “Blurred vision” may be used to describe unilateral visual loss, as in transient monocular blindness, or diplopia. The interpretation of the true meaning of the words used by patients to describe symptoms obviously becomes even more complex when there are differences in primary languages and cultures.
3. Corroboration of the history by others. It is almost always helpful to obtain additional information from family, friends, or other observers to corroborate or expand the patient’s description. Memory loss, aphasia, loss of insight, intoxication, and other factors may impair the patient’s capacity to communicate normally with the examiner or prevent openness about factors that have contributed to the illness. Episodes of loss of consciousness necessitate that details be sought from observers to ascertain precisely what has happened during the event.
4. Family history. Many neurologic disorders have an underlying genetic component. The presence of a Mendelian disorder, such as Huntington’s disease or Charcot-Marie-Tooth neuropathy, is often obvious if family data are available. More detailed questions about family history are often necessary in polygenic disorders such as MS, migraine, and many types of epilepsy. It is important to elicit family history about all illnesses, in addition to neurologic and psychiatric disorders. A familial propensity to hypertension or heart disease is relevant in a patient who presents with a stroke. There are numerous inherited neurologic diseases that are associated with multisystem manifestations that may provide clues to the correct diagnosis (e.g., neurofibromatosis, Wilson’s disease, mitochondrial disorders).
5. Medical illnesses. Many neurologic diseases occur in the context of systemic disorders. Diabetes mellitus, hypertension, and abnormalities of blood lipids predispose to cerebrovascular disease. A solitary mass lesion in the brain may be an abscess in a patient with valvular heart disease, a primary hemorrhage in a patient with a coagulopathy, a lymphoma or toxoplasmosis in a patient with AIDS, or a metastasis in a patient with underlying cancer. Patients with malignancy may also present with a neurologic paraneoplastic syndrome (Chap. 122) or complications from chemotherapy or radiotherapy. Marfan’s syndrome and related collagen disorders predispose to dissection of the cranial arteries and aneurysmal subarachnoid hemorrhage; the latter may also occur with polycystic kidney disease. Various neurologic disorders occur with dysthyroid states or other endocrinopathies. It is especially important to look for the presence of systemic diseases in patients with peripheral neuropathy. Most patients with coma in a hospital setting have a metabolic, toxic, or infectious cause.
6. Drug use and abuse and toxin exposure. It is essential to inquire about the history of drug use, both prescribed and illicit. Sedatives, antidepressants, and other psychoactive medications are frequently associated with acute confusional states, especially in the elderly. Aminoglycoside antibiotics may exacerbate symptoms of weakness in patients with disorders of neuromuscular transmission, such as myasthenia gravis, and may cause dizziness secondary to ototoxicity. Vincristine and other antineoplastic drugs can cause peripheral neuropathy, and immunosuppressive agents such as cyclosporine can produce encephalopathy. Excessive vitamin ingestion can lead to disease; examples include vitamin A and pseudotumor cerebri or pyridoxine and peripheral neuropathy. Many patients are unaware that over-the-counter sleeping pills, cold preparations, and diet pills are actually drugs. Alcohol, the most prevalent neurotoxin, is often not recognized as such by patients, and other drugs of abuse such as cocaine and heroin can cause a wide range of neurologic abnormalities. A history of environmental or industrial exposure to neurotoxins may provide an essential clue; consultation with the patient’s coworkers or employer may be required.
7. Formulating an impression of the patient. Use the opportunity while taking the history to form an impression of the patient. Is the information forthcoming, or does it take a circuitous course? Is there evidence of anxiety, depression, or hypochondriasis? Are there any clues to problems with language, memory, insight, comportment, or behavior? The neurologic assessment begins as soon as the patient comes into the room and the first introduction is made.
THE NEUROLOGIC EXAMINATION
The neurologic examination is challenging and complex; it has many components and includes a number of skills that can be mastered only through repeated use of the same techniques on a large number of individuals with and without neurologic disease. Mastery of the complete neurologic examination is usually important only for physicians in neurology and associated specialties. However, knowledge of the basics of the examination, especially those components that are effective in screening for neurologic dysfunction, is essential for all clinicians, especially generalists.
There is no single, universally accepted sequence of the examination that must be followed, but most clinicians begin with assessment of mental status followed by the cranial nerves, motor system, reflexes, sensory system, coordination, and gait. Whether the examination is basic or comprehensive, it is essential that it be performed in an orderly and systematic fashion to avoid errors and serious omissions. Thus, the best way to learn and gain expertise in the examination is to choose one’s own approach and practice it frequently and do it in the same exact sequence each time.
The detailed description that follows describes the more commonly used parts of the neurologic examination, with a particular emphasis on the components that are considered most helpful for the assessment of common neurologic problems. Each section also includes a brief description of the minimal examination necessary to adequately screen for abnormalities in a patient who has no symptoms suggesting neurologic dysfunction. A screening examination done in this way can be completed in 3–5 min.
Several additional points about the examination are worth noting. First, in recording observations, it is important to describe what is found rather than to apply a poorly defined medical term (e.g., “patient groans to sternal rub” rather than “obtunded”). Second, subtle CNS abnormalities are best detected by carefully comparing a patient’s performance on tasks that require simultaneous activation of both cerebral hemispheres (e.g., eliciting a pronator drift of an outstretched arm with the eyes closed; extinction on one side of bilaterally applied light touch, also with eyes closed; or decreased arm swing or a slight asymmetry when walking). Third, if the patient’s complaint is brought on by some activity, reproduce the activity in the office. If the complaint is of dizziness when the head is turned in one direction, have the patient do this and also look for associated signs on examination (e.g., nystagmus or dysmetria). If pain occurs after walking two blocks, have the patient leave the office and walk this distance and immediately return, and repeat the relevant parts of the examination. Finally, the use of tests that are individually tailored to the patient’s problem can be of value in assessing changes over time. Tests of walking a 7.5-m (25-ft) distance (normal, 5–6 s; note assistance, if any), repetitive finger or toe tapping (normal, 20–25 taps in 5 s), or handwriting are examples.
MENTAL STATUS EXAMINATION
• The bare minimum: During the interview, look for difficulties with communication and determine whether the patient has recall and insight into recent and past events.
The mental status examination is under way as soon as the physician begins observing and speaking with the patient. If the history raises any concern for abnormalities of higher cortical function or if cognitive problems are observed during the interview, then detailed testing of the mental status is indicated. The patient’s ability to understand the language used for the examination, cultural background, educational experience, sensory or motor problems, or comorbid conditions need to be factored into the applicability of the tests and interpretation of results.
The Folstein mini-mental status examination (MMSE) is a standardized screening examination of cognitive function that is extremely easy to administer and takes <10 min to complete. Using age-adjusted values for defining normal performance, the test is ~85% sensitive and 85% specific for making the diagnosis of dementia that is moderate or severe, especially in educated patients. When there is sufficient time available, the MMSE is one of the best methods for documenting the current mental status of the patient, and this is especially useful as a baseline assessment to which future scores of the MMSE can be compared.
Individual elements of the mental status examination can be subdivided into level of consciousness, orientation, speech and language, memory, fund of information, insight and judgment, abstract thought, and calculations.
Level of consciousness is the patient’s relative state of awareness of the self and the environment, and ranges from fully awake to comatose. When the patient is not fully awake, the examiner should describe the responses to the minimum stimulus necessary to elicit a reaction, ranging from verbal commands to a brief, painful stimulus such as a squeeze of the trapezius muscle. Responses that are directed toward the stimulus and signify some degree of intact cerebral function (e.g., opening the eyes and looking at the examiner or reaching to push away a painful stimulus) must be distinguished from reflex responses of a spinal origin (e.g., triple flexion response—flexion at the ankle, knee, and hip in response to a painful stimulus to the foot).
Orientation is tested by asking the person to state his or her name, location, and time (day of the week and date); time is usually the first to be affected in a variety of conditions.
Speech is assessed by observing articulation, rate, rhythm, and prosody (i.e., the changes in pitch and accentuation of syllables and words).
Language is assessed by observing the content of the patient’s verbal and written output, response to spoken commands, and ability to read. A typical testing sequence is to ask the patient to name successively more detailed components of clothing, a watch, or a pen; repeat the phrase “No ifs, ands, or buts”; follow a three-step, verbal command; write a sentence; and read and respond to a written command.
Memory should be analyzed according to three main time scales: (1) immediate memory is assessed by saying a list of three items and having the patient repeat the list immediately; (2) short-term memory is tested by asking the patient to recall the same three items 5 and 15 min later; and (3) long-term memory is evaluated by determining how well the patient is able to provide a coherent chronologic history of his or her illness or personal events.
Fund of information is assessed by asking questions about major historic or current events, with special attention to educational level and life experiences.
Abnormalities of insight and judgment are usually detected during the patient interview; a more detailed assessment can be elicited by asking the patient to describe how he or she would respond to situations having a variety of potential outcomes (e.g., “What would you do if you found a wallet on the sidewalk?”).
Abstract thought can be tested by asking the patient to describe similarities between various objects or concepts (e.g., apple and orange, desk and chair, poetry and sculpture) or to list items having the same attributes (e.g., a list of four-legged animals).
Calculation ability is assessed by having the patient carry out a computation that is appropriate to the patient’s age and education (e.g., serial subtraction of 7 from 100 or 3 from 20; or word problems involving simple arithmetic).
CRANIAL NERVE EXAMINATION
• The bare minimum: Check the fundi, visual fields, pupil size and reactivity, extraocular movements, and facial movements.
The cranial nerves (CN) are best examined in numerical order, except for grouping together CN III, IV, and VI because of their similar function.
CN I (Olfactory) Testing is often omitted unless there is suspicion for inferior frontal lobe disease (e.g., meningioma). With eyes closed, ask the patient to sniff a mild stimulus such as toothpaste or coffee and identify the odorant.
CN II (Optic) Check visual acuity (with eyeglasses or contact lens correction) using a Snellen chart or similar tool. Test the visual fields by confrontation, i.e., by comparing the patient’s visual fields to your own. As a screening test, it is usually sufficient to examine the visual fields of both eyes simultaneously; individual eye fields should be tested if there is any reason to suspect a problem of vision by the history or other elements of the examination, or if the screening test reveals an abnormality. Face the patient at a distance of approximately 0.6–1.0 m (2–3 ft) and place your hands at the periphery of your visual fields in the plane that is equidistant between you and the patient. Instruct the patient to look directly at the center of your face and to indicate when and where he or she sees one of your fingers moving. Beginning with the two inferior quadrants and then the two superior quadrants, move your index finger of the right hand, left hand, or both hands simultaneously and observe whether the patient detects the movements. A single small-amplitude movement of the finger is sufficient for a normal response. Focal perimetry and tangent screen examinations should be used to map out visual field defects fully or to search for subtle abnormalities. Optic fundi should be examined with an ophthalmoscope, and the color, size, and degree of swelling or elevation of the optic disc noted, as well as the color and texture of the retina. The retinal vessels should be checked for size, regularity, arteriovenous nicking at crossing points, hemorrhage, exudates, etc.
CN III, IV, VI (Oculomotor, Trochlear, Abducens) Describe the size and shape of pupils and reaction to light and accommodation (i.e., as the eyes converge while following your finger as it moves toward the bridge of the nose). To check extraocular movements, ask the patient to keep his or her head still while tracking the movement of the tip of your finger. Move the target slowly in the horizontal and vertical planes; observe any paresis, nystagmus, or abnormalities of smooth pursuit (saccades, oculomotor ataxia, etc.). If necessary, the relative position of the two eyes, both in primary and multidirectional gaze, can be assessed by comparing the reflections of a bright light off both pupils. However, in practice it is typically more useful to determine whether the patient describes diplopia in any direction of gaze; true diplopia should almost always resolve with one eye closed. Horizontal nystagmus is best assessed at 45° and not at extreme lateral gaze (which is uncomfortable for the patient); the target must often be held at the lateral position for at least a few seconds to detect an abnormality.
CN V (Trigeminal) Examine sensation within the three territories of the branches of the trigeminal nerve (ophthalmic, maxillary, and mandibular) on each side of the face. As with other parts of the sensory examination, testing of two sensory modalities derived from different anatomic pathways (e.g., light touch and temperature) is sufficient for a screening examination. Testing of other modalities, the corneal reflex, and the motor component of CN V (jaw clench—masseter muscle) is indicated when suggested by the history.
CN VII (Facial) Look for facial asymmetry at rest and with spontaneous movements. Test eyebrow elevation, forehead wrinkling, eye closure, smiling, and cheek puff. Look in particular for differences in the lower versus upper facial muscles; weakness of the lower two-thirds of the face with preservation of the upper third suggests an upper motor neuron lesion, whereas weakness of an entire side suggests a lower motor neuron lesion.
CN VIII (Vestibulocochlear) Check the patient’s ability to hear a finger rub or whispered voice with each ear. Further testing for air versus mastoid bone conduction (Rinne) and lateralization of a 512-Hz tuning fork placed at the center of the forehead (Weber) should be done if an abnormality is detected by history or examination. Any suspected problem should be followed up with formal audiometry. For further discussion of assessing vestibular nerve function in the setting of dizziness, hearing loss, or coma, see Chaps. 28, 43, and 328, respectively.
CN IX, × (Glossopharyngeal, Vagus) Observe the position and symmetry of the palate and uvula at rest and with phonation (“aah”). The pharyngeal (“gag”) reflex is evaluated by stimulating the posterior pharyngeal wall on each side with a sterile, blunt object (e.g., tongue blade), but the reflex is often absent in normal individuals.
CN XI (Spinal Accessory) Check shoulder shrug (trapezius muscle) and head rotation to each side (sternocleidomastoid) against resistance.
CN XII (Hypoglossal) Inspect the tongue for atrophy or fasciculations, position with protrusion, and strength when extended against the inner surface of the cheeks on each side.
MOTOR EXAMINATION
• The bare minimum: Look for muscle atrophy and check extremity tone. Assess upper extremity strength by checking for pronator drift and strength of wrist or finger extensors. Assess lower extremity strength by checking strength of the toe extensors and having the patient walk normally and on heels and toes.
The motor examination includes observations of muscle appearance, tone, and strength. Although gait is in part a test of motor function, it is usually evaluated separately at the end of the examination.
Appearance Inspect and palpate muscle groups under good light and with the patient in a comfortable and symmetric position. Check for muscle fasciculations, tenderness, and atrophy or hypertrophy. Involuntary movements may be present at rest (e.g., tics, myoclonus, choreoathetosis), during maintained posture (pill-rolling tremor of Parkinson’s disease), or with voluntary movements (intention tremor of cerebellar disease or familial tremor).
Tone Muscle tone is tested by measuring the resistance to passive movement of a relaxed limb. Patients often have difficulty relaxing during this procedure, so it is useful to distract the patient to minimize active movements. In the upper limbs, tone is assessed by rapid pronation and supination of the forearm and flexion and extension at the wrist. In the lower limbs, while the patient is supine the examiner’s hands are placed behind the knees and rapidly raised; with normal tone, the ankles drag along the table surface for a variable distance before rising, whereas increased tone results in an immediate lift of the heel off the surface. Decreased tone is most commonly due to lower motor neuron or peripheral nerve disorders. Increased tone may be evident as spasticity (resistance determined by the angle and velocity of motion; corticospinal tract disease), rigidity (similar resistance in all angles of motion; extrapyramidal disease), or paratonia (fluctuating changes in resistance; frontal lobe pathways or normal difficulty in relaxing). Cogwheel rigidity, in which passive motion elicits jerky interruptions in resistance, is seen in parkinsonism.
Strength Testing for pronator drift is an extremely useful method for screening upper limb weakness. The patient is asked to hold both arms fully extended and parallel to the ground with eyes closed. This position should be maintained for ~10 s; any flexion at the elbow or fingers or pronation of the forearm, especially if asymmetric, is a sign of potential weakness. Muscle strength is further assessed by having the patient exert maximal effort for the particular muscle or muscle group being tested. It is important to isolate the muscles as much as possible, i.e., hold the limb so that only the muscles of interest are active. It is also helpful to palpate accessible muscles as they contract. Grading muscle strength and evaluating the patient’s effort is an art that takes time and practice. Muscle strength is traditionally graded using the following scale:
0 = no movement
1 = flicker or trace of contraction but no associated movement at a joint
2 = movement with gravity eliminated
3 = movement against gravity but not against resistance
4– = movement against a mild degree of resistance
4 = movement against moderate resistance
4+ = movement against strong resistance
5 = full power
However, in many cases, it is more practical to use the following terms:

Noting the pattern of weakness is as important as assessing the magnitude of weakness. Unilateral or bilateral weakness of the upper limb extensors and lower limb flexors (“pyramidal weakness”) suggests a lesion of the pyramidal tract, bilateral proximal weakness suggests myopathy, and bilateral distal weakness suggests peripheral neuropathy.
REFLEX EXAMINATION
• The bare minimum: Check the biceps, patellar, and Achilles reflexes.
Muscle Stretch Reflexes Those that are typically assessed include the biceps (C5, C6), brachioradialis (C5, C6), and triceps (C7, C8) reflexes in the upper limbs and the patellar or quadriceps (L3, L4) and Achilles (S1, S2) reflexes in the lower limbs. The patient should be relaxed and the muscle positioned midway between full contraction and extension. Reflexes may be enhanced by asking the patient to voluntarily contract other, distant muscle groups (Jendrassik maneuver). For example, upper limb reflexes may be reinforced by voluntary teeth-clenching, and the Achilles reflex by hooking the flexed fingers of the two hands together and attempting to pull them apart. For each reflex tested, the two sides should be tested sequentially, and it is important to determine the smallest stimulus required to elicit a reflex rather than the maximum response. Reflexes are graded according to the following scale:
0 = absent
1 = present but diminished
2 = normoactive
3 = exaggerated
4 = clonus
Cutaneous Reflexes The plantar reflex is elicited by stroking, with a noxious stimulus such as a tongue blade, the lateral surface of the sole of the foot beginning near the heel and moving across the ball of the foot to the great toe. The normal reflex consists of plantar flexion of the toes. With upper motor neuron lesions above the S1 level of the spinal cord, a paradoxical extension of the toe is observed, associated with fanning and extension of the other toes (termed an extensor plantar response, or Babinski sign). However, despite its popularity, the reliability and validity of the Babinski sign for identifying upper motor neuron weakness is limited—it is far more useful to rely on tests of tone, strength, stretch reflexes, and coordination. Superficial abdominal reflexes are elicited by gently stroking the abdominal surface near the umbilicus in a diagonal fashion with a sharp object (e.g., the wooden end of a cotton-tipped swab) and observing the movement of the umbilicus. Normally, the umbilicus will pull toward the stimulated quadrant. With upper motor neuron lesions, these reflexes are absent. They are most helpful when there is preservation of the upper (spinal cord level T9) but not lower (T12) abdominal reflexes, indicating a spinal lesion between T9 and T12, or when the response is asymmetric. Other useful cutaneous reflexes include the cremasteric (ipsilateral elevation of the testicle following stroking of the medial thigh; mediated by L1 and L2) and anal (contraction of the anal sphincter when the perianal skin is scratched; mediated by S2, S3, S4) reflexes. It is particularly important to test for these reflexes in any patient with suspected injury to the spinal cord or lumbosacral roots.
Primitive Reflexes With disease of the frontal lobe pathways, several primitive reflexes not normally present in the adult may appear. The suck response is elicited by lightly touching with a tongue blade the center of the lips, and the root response the corner of the lips; the patient will move the lips to suck or root in the direction of the stimulus. The grasp reflex is elicited by touching the palm between the thumb and index finger with the examiner’s fingers; a positive response is a forced grasp of the examiner’s hand. In many instances, stroking the back of the hand will lead to its release. The palmomental response is contraction of the mentalis muscle (chin) ipsilateral to a scratch stimulus diagonally applied to the palm.
SENSORY EXAMINATION
• The bare minimum: Ask whether the patient can feel light touch and the temperature of a cool object in each distal extremity. Check double simultaneous stimulation using light touch on the hands. Perform the Romberg maneuver.
Evaluating sensation is usually the most unreliable part of the examination because it is subjective and is difficult to quantify. In the compliant and discerning patient, the sensory examination can be extremely helpful for the precise localization of a lesion. With patients who are uncooperative or lack an understanding of the tests, it may be useless. The examination should be focused on the suspected lesion. For example, in spinal cord, spinal root, or peripheral nerve abnormalities, all major sensory modalities should be tested while looking for a pattern consistent with a spinal level and dermatomal or nerve distribution. In patients with lesions at or above the brainstem, screening the primary sensory modalities in the distal extremities along with tests of “cortical” sensation is usually sufficient.
The five primary sensory modalities—light touch, pain, temperature, vibration, and joint position—are tested in each limb. Light touch is assessed by stimulating the skin with single, very gentle touches of the examiner’s finger or a wisp of cotton. Pain is tested using a new pin, and temperature is assessed using a metal object (e.g., tuning fork) that has been immersed in cold and warm water. Vibration is tested using a 128-Hz tuning fork applied to the distal phalanx of the great toe or index finger just below the nail bed. By placing a finger on the opposite side of the joint being tested, the examiner compares the patient’s threshold of vibration perception with his or her own. For joint position testing, the examiner grasps the digit or limb laterally and distal to the joint being assessed; small 1- to 2-mm excursions can usually be sensed. The Romberg maneuver is primarily a test of proprioception. The patient is asked to stand with the feet as close together as necessary to maintain balance while the eyes are open, and the eyes are then closed. A loss of balance with the eyes closed is an abnormal response.
“Cortical” sensation is mediated by the parietal lobes and represents an integration of the primary sensory modalities; testing cortical sensation is only meaningful when primary sensation is intact. Double simultaneous stimulation is especially useful as a screening test for cortical function; with the patient’s eyes closed, the examiner lightly touches one or both hands and asks the patient to identify the stimuli. With a parietal lobe lesion, the patient may be unable to identify the stimulus on the contralateral side when both hands are touched. Other modalities relying on the parietal cortex include the discrimination of two closely placed stimuli as separate (two-point discrimination), identification of an object by touch and manipulation alone (stereognosis), and the identification of numbers or letters written on the skin surface (graphesthesia).
COORDINATION EXAMINATION
• The bare minimum: Observe the patient at rest and during spontaneous movements. Test rapid alternating movements of the hands and feet and finger to nose.
Coordination refers to the orchestration and fluidity of movements. Even simple acts require cooperation of agonist and antagonist muscles, maintenance of posture, and complex servomechanisms to control the rate and range of movements. Part of this integration relies on normal function of the cerebellar and basal ganglia systems. However, coordination also requires intact muscle strength and kinesthetic and proprioceptive information. Thus, if the examination has disclosed abnormalities of the motor or sensory systems, the patient’s coordination should be assessed with these limitations in mind.
Rapid alternating movements in the upper limbs are tested separately on each side by having the patient make a fist, partially extend the index finger, and then tap the index finger on the distal thumb as quickly as possible. In the lower limb, the patient rapidly taps the foot against the floor or the examiner’s hand. Finger-to-nose testing is primarily a test of cerebellar function; the patient is asked to touch his or her index finger repetitively to the nose and then to the examiner’s outstretched finger, which moves with each repetition. A similar test in the lower extremity is to have the patient raise the leg and touch the examiner’s finger with the great toe. Another cerebellar test in the lower limbs is the heel-knee-shin maneuver; in the supine position the patient is asked to slide the heel of each foot from the knee down the shin of the other leg. For all these movements, the accuracy, speed, and rhythm are noted.
GAIT EXAMINATION
• The bare minimum: Observe the patient while walking normally, on the heels and toes, and along a straight line.
Watching the patient walk is the most important part of the neurologic examination. Normal gait requires that multiple systems—including strength, sensation, and coordination—function in a highly integrated fashion. Unexpected abnormalities may be detected that prompt the examiner to return in more detail to other aspects of the examination. The patient should be observed while walking and turning normally, walking on the heels, walking on the toes, and walking heel-to-toe along a straight line. The examination may reveal decreased arm swing on one side (corticospinal tract disease), a stooped posture and short-stepped gait (parkinsonism), a broad-based unstable gait (ataxia), scissoring (spasticity), or a high-stepped, slapping gait (posterior column or peripheral nerve disease), or the patient may appear to be stuck in place (apraxia with frontal lobe disease).
NEUROLOGIC DIAGNOSIS
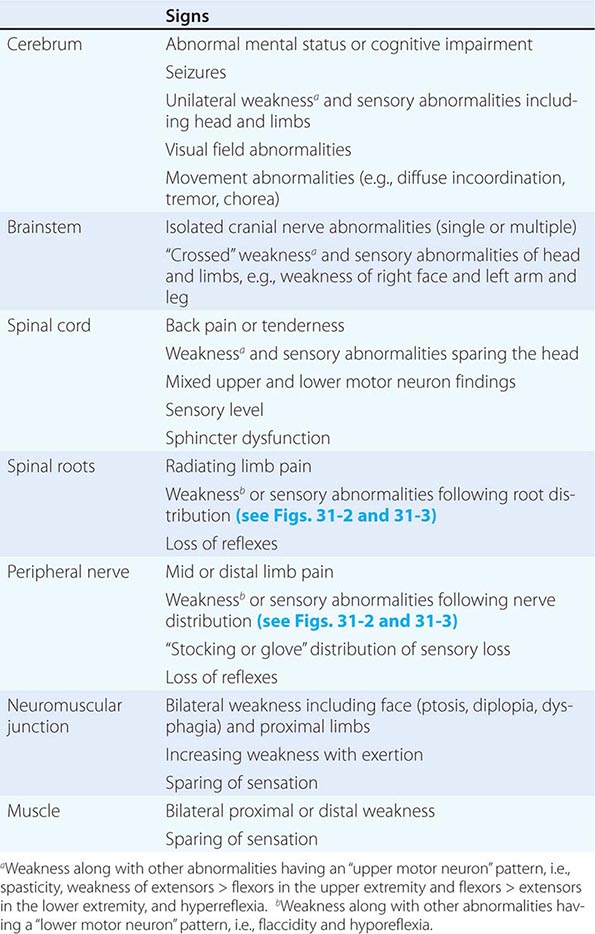
The clinical data obtained from the history and examination are interpreted to arrive at an anatomic localization that best explains the clinical findings (Table 437-2), to narrow the list of diagnostic possibilities, and to select the laboratory tests most likely to be informative. The laboratory assessment may include (1) serum electrolytes; complete blood count; and renal, hepatic, endocrine, and immune studies; (2) cerebrospinal fluid examination; (3) focused neuroimaging studies (Chap. 440e); or (4) electrophysiologic studies (Chap. 442e). The anatomic localization, mode of onset and course of illness, other medical data, and laboratory findings are then integrated to establish an etiologic diagnosis.
FINDINGS HELPFUL FOR LOCALIZATIONS WITHIN THE NERVOUS SYSTEM |
The neurologic examination may be normal even in patients with a serious neurologic disease, such as seizures, chronic meningitis, or a TIA. A comatose patient may arrive with no available history, and in such cases, the approach is as described in Chap. 328. In other patients, an inadequate history may be overcome by a succession of examinations from which the course of the illness can be inferred. In perplexing cases it is useful to remember that uncommon presentations of common diseases are more likely than rare etiologies. Thus, even in tertiary care settings, multiple strokes are usually due to emboli and not vasculitis, and dementia with myoclonus is usually Alzheimer’s disease and not a prion disorder or a paraneoplastic illness. Finally, the most important task of a primary care physician faced with a patient who has a new neurologic complaint is to assess the urgency of referral to a specialist. Here, the imperative is to rapidly identify patients likely to have nervous system infections, acute strokes, and spinal cord compression or other treatable mass lesions and arrange for immediate care.
438e | The Neurologic Screening Exam |
Knowledge of the basic neurologic examination is an essential clinical skill. A simple neurologic screening examination—assessment of mental status, cranial nerves, motor system, sensory system, coordination, and gait—can be reliably performed in 3–5 min. Although the components of the examination may appear daunting at first, skills usually improve rapidly with repetition and practice. In this video, the technique of performing a simple and efficient screening examination is presented.
439e | Video Atlas of the Detailed Neurologic Examination |
The comprehensive neurologic examination is an irreplaceable tool for the efficient diagnosis of neurologic disorders. Mastery of its details requires knowledge of normal nervous system anatomy and physiology combined with personal experience performing orderly and systematic examinations on large numbers of patients and healthy individuals. In the hands of a great clinician, the neurologic examination also becomes a thing of beauty—the pinnacle of the art of medicine. In this video, the most commonly used components of the examination are presented in detail, with a particular emphasis on those elements that are most helpful for assessment of common neurologic problems.
440e | Neuroimaging in Neurologic Disorders |
The clinician caring for patients with neurologic symptoms is faced with myriad imaging options, including computed tomography (CT), CT angiography (CTA), perfusion CT (pCT), magnetic resonance (MR) imaging (MRI), MR angiography (MRA), functional MRI (fMRI), MR spectroscopy (MRS), MR neurography (MRN), diffusion and diffusion tensor imaging, susceptibility-weighted MR imaging (SWI), arterial spin label MRI (ASL) and perfusion MRI (pMRI). In addition, an increasing number of interventional neuroradiologic techniques are available, including angiography catheter embolization, coiling, and stenting of vascular structures, and spine diagnostic and interventional techniques, such as diskography, transforaminal and translaminar epidural and nerve root injections, and blood patches. Multidetector CTA (MDCTA) and gadolinium-enhanced MRA have narrowed the indications for conventional angiography, which is now reserved for patients in whom small-vessel detail is essential for diagnosis or for whom concurrent interventional therapy is planned (Table 440e-1).
GUIDELINES FOR THE USE OF CT, ULTRASOUND, AND MRI |
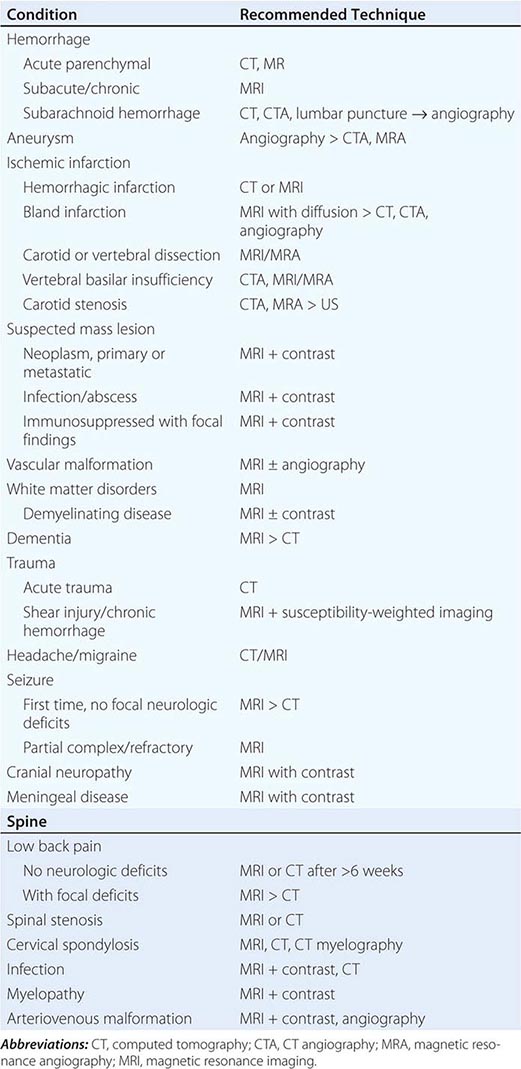
In general, MRI is more sensitive than CT for the detection of lesions affecting the central nervous system (CNS), particularly those of the spinal cord, cranial nerves, and posterior fossa structures. Diffusion MR, a sequence sensitive to the microscopic motion of water, is the most sensitive technique for detecting acute ischemic stroke of the brain or spinal cord, and it is also useful in the detection of encephalitis, abscesses, and prion diseases. CT, however, is quickly acquired and is widely available, making it a pragmatic choice for the initial evaluation of patients with acute changes in mental status, suspected acute stroke, hemorrhage, and intracranial or spinal trauma. CT is also more sensitive than MRI for visualizing fine osseous detail and is indicated in the initial imaging evaluation of conductive hearing loss as well as lesions affecting the skull base and calvarium. MR may, however, add important diagnostic information regarding bone marrow infiltrative processes that are difficult to detect on CT.
COMPUTED TOMOGRAPHY
TECHNIQUE
The CT image is a cross-sectional representation of anatomy created by a computer-generated analysis of the attenuation of x-ray beams passed through a section of the body. As the x-ray beam, collimated to the desired slice width, rotates around the patient, it passes through selected regions in the body. X-rays that are not attenuated by body structures are detected by sensitive x-ray detectors aligned 180° from the x-ray tube. A computer calculates a “back projection” image from the 360° x-ray attenuation profile. Greater x-ray attenuation (e.g., as caused by bone), results in areas of high “density” (whiter) on the scan, whereas soft tissue structures that have poor attenuation of x-rays, such as organs and air-filled cavities, are lower (blacker) in density. The resolution of an image depends on the radiation dose, the detector size, collimation (slice thickness), the field of view, and the matrix size of the display. A modern CT scanner is capable of obtaining sections as thin as 0.5–1 mm with 0.4-mm in-plane resolution at a speed of 0.3 s per rotation; complete studies of the brain can be completed in 1–10 s.
Multidetector CT (MDCT) is now standard in most radiology departments. Single or multiple (from 4 to 320) solid-state detectors positioned opposite to the x-ray source result in multiple slices per revolution of the beam around the patient. The table moves continuously through the rotating x-ray beam, generating a continuous “helix” of information that can be reformatted into various slice thicknesses and planes. Advantages of MDCT include shorter scan times, reduced patient and organ motion, and the ability to acquire images dynamically during the infusion of intravenous contrast, which can be used to construct CT angiograms of vascular structures and perfusion images (Figs. 440e-1B and C). CTA can be displayed in three dimensions to yield angiogram-like images (Figs. 440e-1C, 440e-2E and F, and see Fig. 446-4). CTA has proved useful in assessing the cervical and intracranial arterial and venous anatomy.
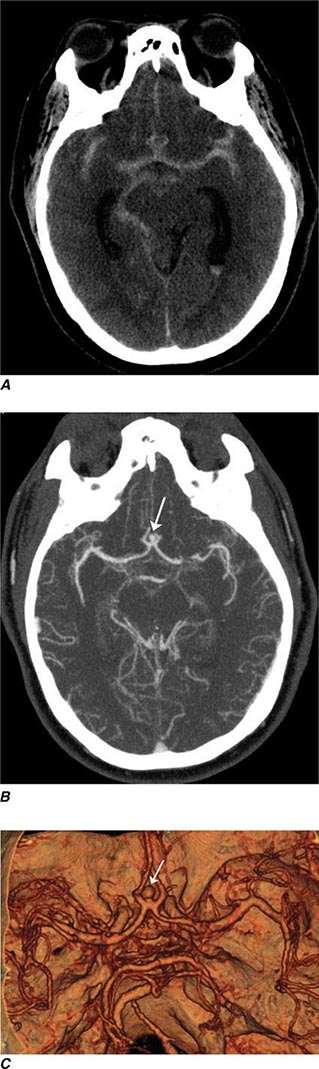
FIGURE 440e-1 Computed tomography (CT) angiography (CTA) of ruptured anterior cerebral artery aneurysm in a patient presenting with acute headache. A. Noncontrast CT demonstrates subarachnoid hemorrhage and mild obstructive hydrocephalus. B. Axial maximum-intensity projection from CTA demonstrates enlargement of the anterior cerebral artery (arrow). C. Three-dimensional surface reconstruction using a workstation confirms the anterior cerebral aneurysm and demonstrates its orientation and relationship to nearby vessels (arrow). CTA image is produced by 0.5- to 1-mm helical CT scans performed during a rapid bolus infusion of intravenous contrast medium.
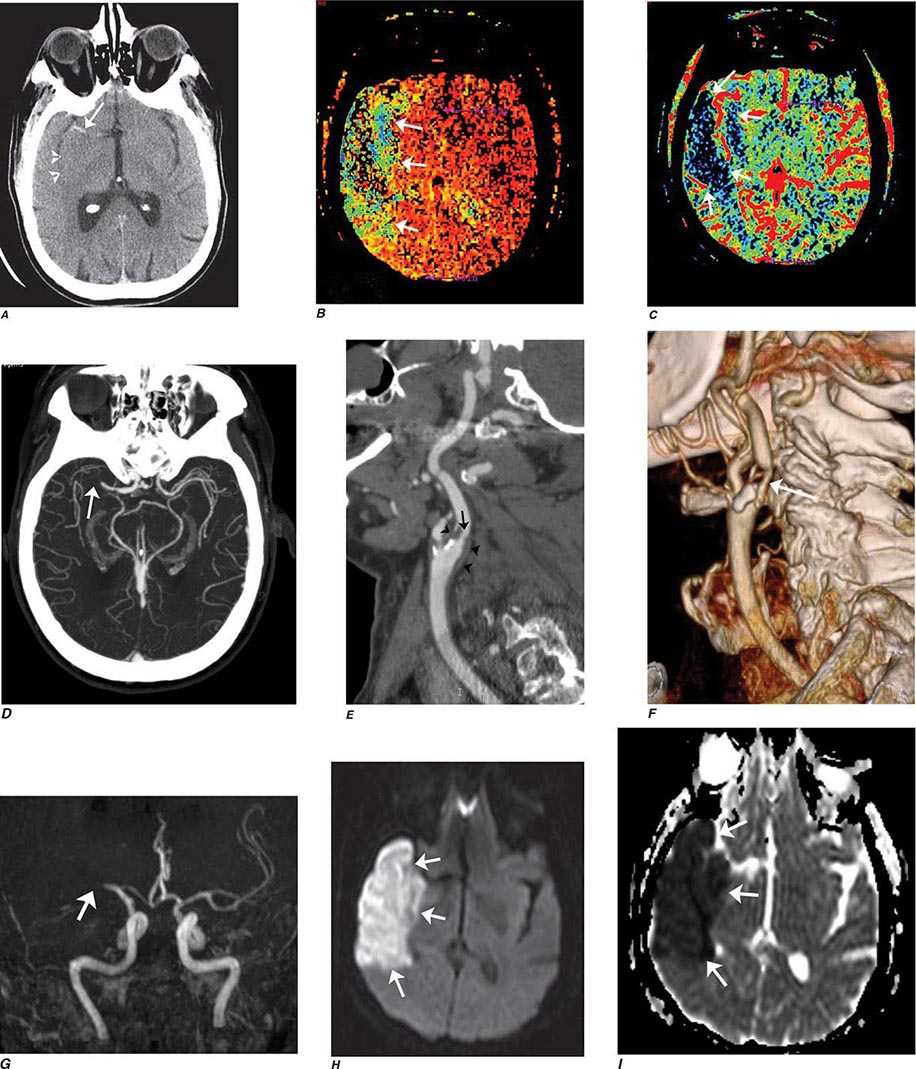
FIGURE 440e-2 Acute left hemiparesis due to middle cerebral artery occlusion. A. Axial noncontrast computed tomography (CT) scan demonstrates high density within the right middle cerebral artery (arrowheads). B. Mean transit time CT perfusion parametric map indicating prolonged mean transit time involving the right middle cerebral territory (arrows). C. Cerebral blood volume (CBV) map shows reduced CBV involving an area within the defect shown in B, indicating a high likelihood of infarction (arrows). D. Axial maximum-intensity projection from a CT angiography (CTA) study through the circle of Willis demonstrates an abrupt occlusion of the proximal right middle cerebral artery (arrow). E. Sagittal reformation through the right internal carotid artery demonstrates a low-density lipid-laden plaque (arrowheads) narrowing the lumen (black arrow). F. Three-dimensional surface-rendered CTA image demonstrates calcification and narrowing of the right internal carotid artery (arrow), consistent with atherosclerotic disease. G. Coronal maximum-intensity projection from magnetic resonance angiography shows right middle cerebral artery (MCA) occlusion (arrow). H. and I. Axial diffusion-weighted image (H) and apparent diffusion coefficient image (I) documents the presence of a right middle cerebral artery infarction.
Intravenous iodinated contrast is often administered to identify both vascular structures and to detect defects in the blood-brain barrier (BBB) that are caused by tumors, infarcts, and infections. In the normal CNS, only vessels and structures lacking a BBB (e.g., the pituitary gland, choroid plexus, and dura) enhance after contrast administration. The use of iodinated contrast agents carries a small risk of allergic reaction and adds additional expense. While helpful in characterizing mass lesions as well as essential for the acquisition of CTA studies, the decision to use contrast material should always be considered carefully.
INDICATIONS
CT is the primary study of choice in the evaluation of an acute change in mental status, focal neurologic findings, acute trauma to the brain and spine, suspected subarachnoid hemorrhage, and conductive hearing loss (Table 440e-1). CT is complementary to MR in the evaluation of the skull base, orbit, and osseous structures of the spine. In the spine, CT is useful in evaluating patients with osseous spinal stenosis and spondylosis, but MRI is often preferred in those with neurologic deficits. CT can also be obtained following intrathecal contrast injection to evaluate the intracranial cisterns (CT cisternography) for cerebrospinal fluid (CSF) fistula, as well as the spinal subarachnoid space (CT myelography), although intrathecal administration of gadolinium combined with MR may also be complementary.
COMPLICATIONS
CT is safe, fast, and reliable. Radiation exposure depends on the dose used but is normally between 2 and 5 mSv (millisievert) for a routine brain CT study. Care must be taken to reduce exposure when imaging children. With the advent of MDCT, CTA, and CT perfusion, the benefit must be weighed against the increased radiation doses associated with these techniques. Advanced noise reduction software now permits acceptable diagnostic CT scans at 30–40% lower radiation doses.
The most frequent complications are those associated with use of intravenous contrast agents. While two broad categories of contrast media, ionic and nonionic, are in use, ionic agents have been largely replaced by safer nonionic compounds.
Contrast nephropathy may result from hemodynamic changes, renal tubular obstruction and cell damage, or immunologic reactions to contrast agents. A rise in serum creatinine of at least 85 μmol/L (1 mg/dL) within 48 h of contrast administration is often used as a definition of contrast nephropathy, although other causes of acute renal failure must be excluded. The prognosis is usually favorable, with serum creatinine levels returning to baseline within 1–2 weeks. Risk factors for contrast nephropathy include advanced age (>80 years), preexisting renal disease (serum creatinine exceeding 2 mg/dL), solitary kidney, diabetes mellitus, dehydration, paraproteinemia, concurrent use of nephrotoxic medication or chemotherapeutic agents, and high contrast dose. Patients with diabetes and those with mild renal failure should be well hydrated prior to the administration of contrast agents, although careful consideration should be given to alternative imaging techniques such as MRI, noncontrast CT, or ultrasound (US). Nonionic, low-osmolar media produce fewer abnormalities in renal blood flow and less endothelial cell damage but should still be used carefully in patients at risk for allergic reaction. Estimated glomerular filtration rate (eGFR) is a more reliable indicator of renal function compared to creatinine alone because it takes into account age, race, and sex. In one study, 15% of outpatients with a normal serum creatinine had an estimated creatinine clearance of 50 mL/min/1.73 m2 or less (normal is ≥90 mL/min/1.73 m2). The exact eGFR threshold, below which withholding intravenous contrast should be considered, is controversial. The risk of contrast nephropathy increases in patients with an eGFR <60 mL/min/1.73 m2; however, the majority of these patients will only have a temporary rise in creatinine. The risk of dialysis after receiving contrast significantly increases in patients with eGFR <30 mL/min/1.73 m2. Thus, an eGFR threshold between 60 and 30 mL/min/1.73 m2 is appropriate; however, the exact number is somewhat arbitrary. A creatinine of 1.6 in a 70-year-old, non-African-American male corresponds to an eGFR of approximately 45 mL/min/1.73 m2. The American College of Radiology suggests using an eGFR of 45 mL/min/1.73 m2 as a threshold below which iodinated contrast should not be given without serious consideration of the potential for contrast nephropathy. If contrast must be administered to a patient with an eGFR below 45 mL/min/1.73 m2, the patient should be well hydrated, and a reduction in the dose of contrast should be considered. Use of other agents such as bicarbonate and acetylcysteine may reduce the incidence of contrast nephropathy.
Allergy Immediate reactions following intravenous contrast media can occur through several mechanisms. The most severe reactions are related to allergic hypersensitivity (anaphylaxis) and range from mild hives to bronchospasm and death. The pathogenesis of allergic hypersensitivity reactions is thought to include the release of mediators such as histamine, antibody-antigen reactions, and complement activation. Severe allergic reactions occur in ~0.04% of patients receiving nonionic media, sixfold lower than with ionic media. Risk factors include a history of prior contrast reaction (fivefold increased likelihood), food and or drug allergies, and atopy (asthma and hay fever). The predictive value of specific allergies, such as those to shellfish, once thought important, actually is now recognized to be unreliable. Nonetheless, in patients with a history worrisome for potential allergic reaction, a noncontrast CT or MRI procedure should be considered as an alternative to contrast administration. If iodinated contrast is absolutely required, a nonionic agent should be used in conjunction with pretreatment with glucocorticoids and antihistamines (Table 440e-2); however, pretreatment does not guarantee safety. Patients with allergic reactions to iodinated contrast material do not usually react to gadolinium-based MR contrast material, although such reactions can occur. It would be wise to pretreat patients with a prior allergic history to MR contrast administration in a similar fashion. Nonimmediate (>1 h after injection) reactions are frequent and probably related to T cell–mediated immune reactions. These are typically urticarial but can occasionally be more severe. Drug provocation and skin testing may be required to determine the culprit agent involved as well as determine a safe alternative.
GUIDELINES FOR PREMEDICATION OF PATIENTS WITH PRIOR CONTRAST ALLERGY |
Other side effects of CT scanning are rare but include a sensation of warmth throughout the body and a metallic taste during intravenous administration of iodinated contrast media. Extravasation of contrast media, although rare, can be painful and lead to compartment syndrome. When this occurs, consultation with plastic surgery is indicated. Patients with significant cardiac disease may be at increased risk for contrast reactions, and in these patients, limits to the volume and osmolality of the contrast media should be considered. Patients who may undergo systemic radioactive iodine therapy for thyroid disease or cancer should not receive iodinated contrast media if possible, because this will decrease the uptake of the radioisotope into the tumor or thyroid (see the American College of Radiology Manual on Contrast Media, Version 9, 2013; http://www.acr.org/~/media/ACR/Documents/PDF/QualitySafety/Resources/Contrast%20Manual/2013_Contrast_Media.pdf).
MAGNETIC RESONANCE IMAGING
TECHNIQUE
MRI is a complex interaction between hydrogen protons in biologic tissues, a static magnetic field (the magnet), and energy in the form of radiofrequency (Rf) waves of a specific frequency introduced by coils placed next to the body part of interest. Images are made by computerized processing of resonance information received from protons in the body. Field strength of the magnet is directly related to signal-to-noise ratio. While 1.5-T magnets have become the standard high-field MRI units, 3-T magnets are now widely available and have distinct advantages in the brain and musculoskeletal systems. Even higher field magnets (7-T) and positron emission tomography (PET) MR machines promise increased resolution and anatomic-functional information on a variety of disorders. Spatial localization is achieved by magnetic gradients surrounding the main magnet, which impart slight changes in magnetic field throughout the imaging volume. Rf pulses transiently excite the energy state of the hydrogen protons in the body. Rf is administered at a frequency specific for the field strength of the magnet. The subsequent return to equilibrium energy state (relaxation) of the hydrogen protons results in a release of Rf energy (the echo), which is detected by the coils that delivered the Rf pulses. Fourier analysis is used to transform the echo into the information used to form an MR image. The MR image thus consists of a map of the distribution of hydrogen protons, with signal intensity imparted by both density of hydrogen protons and differences in the relaxation times (see below) of hydrogen protons on different molecules. Although clinical MRI currently makes use of the ubiquitous hydrogen proton, research into sodium and carbon imaging and spectroscopy appears promising.
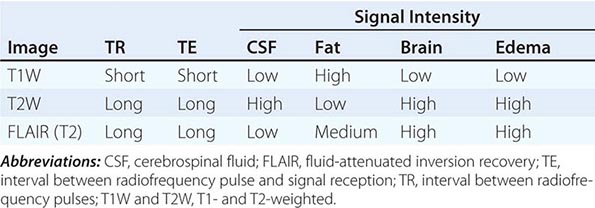
T1 and T2 Relaxation Times The rate of return to equilibrium of perturbed protons is called the relaxation rate. The relaxation rate varies among normal and pathologic tissues. The relaxation rate of a hydrogen proton in a tissue is influenced by local interactions with surrounding molecules and atomic neighbors. Two relaxation rates, T1 and T2, influence the signal intensity of the image. The T1 relaxation time is the time, measured in milliseconds, for 63% of the hydrogen protons to return to their normal equilibrium state, whereas the T2 relaxation is the time for 63% of the protons to become dephased owing to interactions among nearby protons. The intensity and image contrast of the signal within various tissues can be modulated by altering acquisition parameters such as the interval between Rf pulses (TR) and the time between the Rf pulse and the signal reception (TE). T1-weighted (T1W) images are produced by keeping the TR and TE relatively short, whereas using longer TR and TE times produces T2-weighted (T2W) images. Fat and subacute hemorrhage have relatively shorter T1 relaxation rates and thus higher signal intensity than brain on T1W images. Structures containing more water, such as CSF and edema, have long T1 and T2 relaxation rates, resulting in relatively lower signal intensity on T1W images and higher signal intensity on T2W images (Table 440e-3). Gray matter contains 10–15% more water than white matter, which accounts for much of the intrinsic contrast between the two on MRI (Fig. 440e-6B). T2W images are more sensitive than T1W images to edema, demyelination, infarction, and chronic hemorrhage, whereas T1W imaging is more sensitive to subacute hemorrhage and fat-containing structures.
SOME COMMON INTENSITIES ON T1- AND T2-WEIGHTED MRI SEQUENCES |
Many different MR pulse sequences exist, and each can be obtained in various planes (Figs. 440e-2, 440e-3, and 440e-4). The selection of a proper protocol that will best answer a clinical question depends on an accurate clinical history and indication for the examination. Fluid-attenuated inversion recovery (FLAIR) is a useful pulse sequence that produces T2W images in which the normally high signal intensity of CSF is suppressed (Fig. 440e-6B). FLAIR images are more sensitive than standard spin echo images for any water-containing lesions or edema. Susceptibility-weighted imaging, such as gradient echo imaging, is very sensitive to magnetic susceptibility generated by blood, calcium, and air and routinely obtained in patients suspected of pathology that might result in microhemorrhages, such as amyloid, hemorrhagic metastases, and thrombotic states (Fig. 440e-5C). MR images can be generated in any plane without changing the patient’s position. Each sequence, however, must be obtained separately and takes 1–10 min on average to complete. Three-dimensional volumetric imaging is also possible with MRI, resulting in a three-dimensional volume of data that can be reformatted in any orientation to highlight certain disease processes.
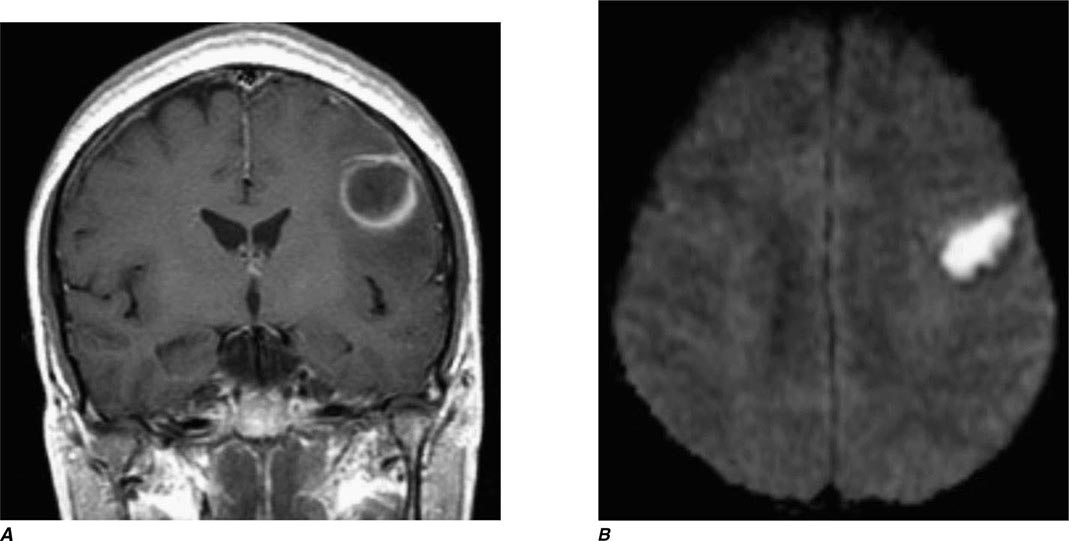
FIGURE 440e-3 Cerebral abscess in a patient with fever and a right hemiparesis. A. Coronal postcontrast T1-weighted image demonstrates a ring-enhancing mass in the left frontal lobe. B. Axial diffusion-weighted image demonstrates restricted diffusion (high signal intensity) within the lesion, which in this setting is highly suggestive of cerebral abscess.
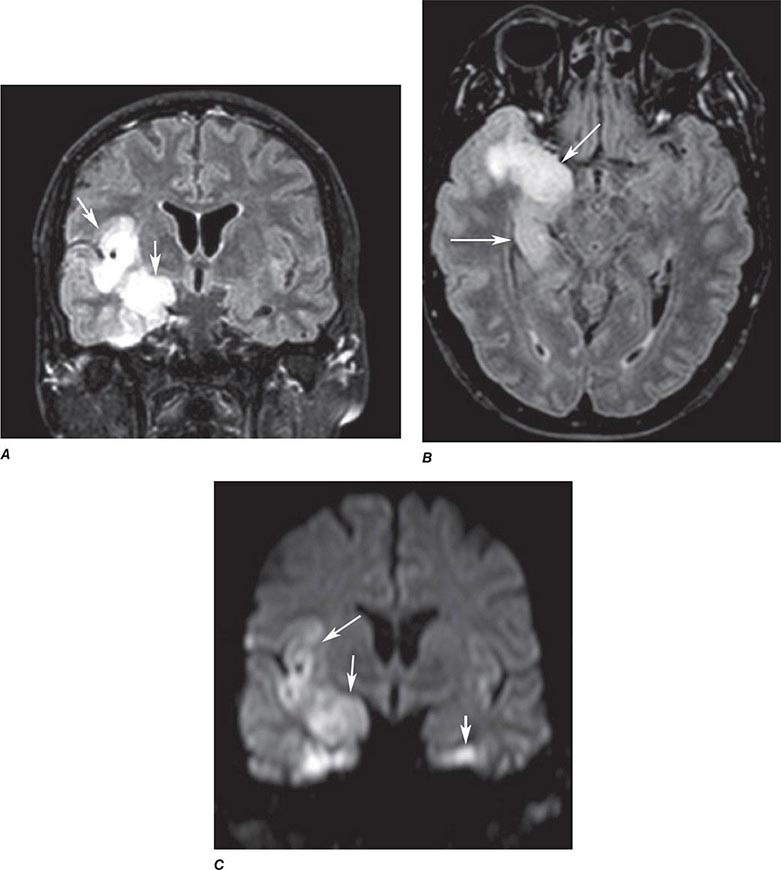
FIGURE 440e-4 Herpes simplex encephalitis in a patient presenting with altered mental status and fever. A. and B. Coronal (A) and axial (B) T2-weighted fluid-attenuated inversion recovery images demonstrate expansion and high signal intensity involving the right medial temporal lobe and insular cortex (arrows). C. Coronal diffusion-weighted image demonstrates high signal intensity indicating restricted diffusion involving the right medial temporal lobe and hippocampus (arrows) as well as subtle involvement of the left inferior temporal lobe (arrowhead). This is most consistent with neuronal death and can be seen in acute infarction as well as encephalitis and other inflammatory conditions. The suspected diagnosis of herpes simplex encephalitis was confirmed by cerebrospinal fluid polymerase chain reaction analysis.
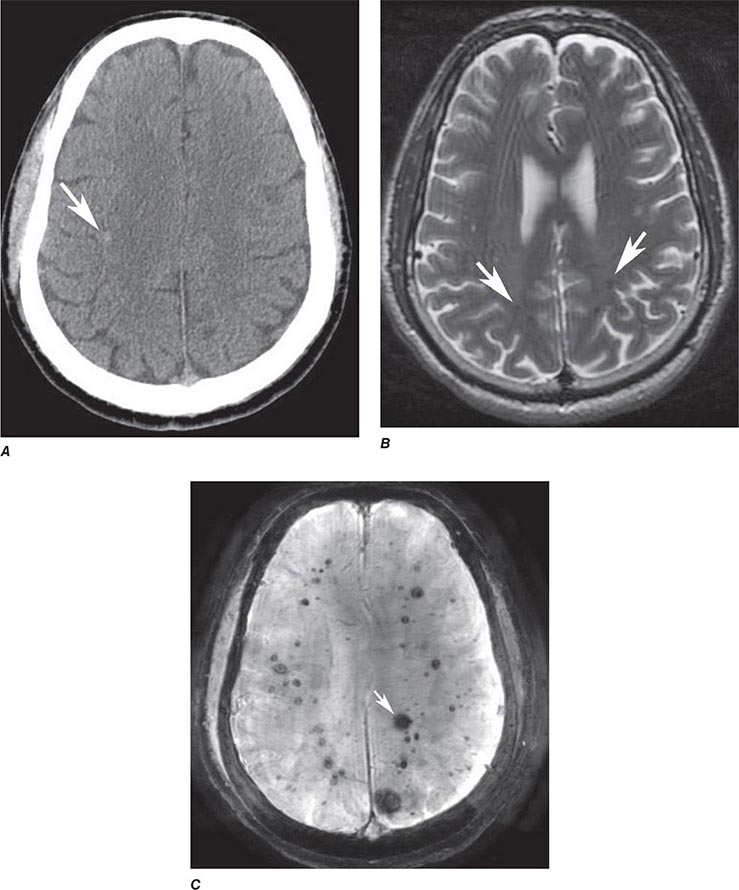
FIGURE 440e-5 Susceptibility-weighted imaging in a patient with familial cavernous malformations. A. Noncontrast computed tomography scan shows one hyperdense lesion in the right hemisphere (arrow). B. T2-weighted fast spin echo image shows subtle low-intensity lesions (arrows). C. Susceptibility-weighted image shows numerous low-intensity lesions consistent with hemosiderin-laden cavernous malformations (arrow).
MR Contrast Material The heavy-metal element gadolinium forms the basis of all currently approved intravenous MR contrast agents. Gadolinium is a paramagnetic substance, which means that it reduces the T1 and T2 relaxation times of nearby water protons, resulting in a high signal on T1W images and a low signal on T2W images (the latter requires a sufficient local concentration, usually in the form of an intravenous bolus). Unlike iodinated contrast agents, the effect of MR contrast agents depends on the presence of local hydrogen protons on which it must act to achieve the desired effect. There are nine different gadolinium agents approved in the United States for use with MRI. These differ according the attached chelated moiety, which also affects the strength of chelation of the otherwise toxic gadolinium element. The chelating carrier molecule for gadolinium can be classified by whether it is macrocyclic or has linear geometry and whether it is ionic or nonionic. Most of these are excreted by the renal system. Cyclical agents are less likely to release the gadolinium element, and thus are considered the safest category.
ALLERGIC HYPERSENSITIVITY Gadolinium-DTPA (diethylenetriaminepentaacetic acid) does not normally cross the intact BBB immediately but will enhance lesions lacking a BBB (Fig. 440e-3A) as well as areas of the brain that normally are devoid of the BBB (pituitary, dura, choroid plexus). However, gadolinium contrast has been noted to slowly cross an intact BBB over time and especially in the setting of reduced renal clearance or inflamed meninges. The agents are generally well tolerated; overall adverse events after injection range from 0.07–2.4%. True allergic reactions are rare (0.004–0.7%) but have been reported. Severe life-threatening reactions are exceedingly rare; in one report, only 55 reactions out of 20 million doses occurred. However, the adverse reaction rate in patients with a prior history of reaction to gadolinium is eight times higher than normal. Other risk factors include atopy or asthma (3.7%); although there is no cross-reactivity to iodinated contrast material, those with a prior allergic response to iodine should be considered at higher risk. Gadolinium contrast material can be administered safely to children as well as adults, although these agents are generally avoided in those under 6 months of age.
NEPHROTOXICITY Contrast-induced renal failure does not occur with gadolinium agents. A rare complication, nephrogenic systemic fibrosis (NSF), has occurred in patients with severe renal insufficiency who have been exposed to gadolinium contrast agents. The onset of NSF has been reported between 5 and 75 days following exposure; histologic features include thickened collagen bundles with surrounding clefts, mucin deposition, and increased numbers of fibrocytes and elastic fibers in skin. In addition to dermatologic symptoms, other manifestations include widespread fibrosis of the skeletal muscle, bone, lungs, pleura, pericardium, myocardium, kidney, muscle, bone, testes, and dura. The American College of Radiology recommends that a glomerular filtration rate (GFR) assessment be obtained within 6 weeks prior to elective gadolinium-based MR contrast agent administration in patients with:
1. A history of renal disease (including solitary kidney, renal transplant, renal tumor)
2. Age >60 years
3. History of hypertension
4. History of diabetes
5. History of severe hepatic disease, liver transplant, or pending liver transplant; for these patients, it is recommended that the patient’s GFR assessment be nearly contemporaneous with the MR examination
The incidence of NSF in patients with severe renal dysfunction (GFR <30) varies from 0.19 to 4%. Other risk factors for NSF include acute kidney injury, the use of nonmacrocyclic agents, and repeated or high-dose exposure to gadolinium. The American College of Radiology Committee on Drugs and Contrast Media states that patients receiving any gadolinium-containing agent should be considered at risk of NSF if they are on dialysis (of any form); have severe or end-stage chronic renal disease (eGFR <30 mL/min/1.73 m2) without dialysis; eGFR of 30–40 mL/min/1.73 m2 without dialysis (as the GFR may fluctuate); or have acute renal insufficiency.
COMPLICATIONS AND CONTRAINDICATIONS
From the patient’s perspective, an MRI examination can be intimidating, and a higher level of cooperation is required than with CT. The patient lies on a table that is moved into a long, narrow gap within the magnet. Approximately 5% of the population experiences severe claustrophobia in the MR environment. This can be reduced by mild sedation but remains a problem for some. Because it takes between 3 and 10 min per sequence, movement of the patient during an MR exam distorts all of the images; therefore, uncooperative patients should either be sedated for the MR study or scanned with CT. Generally, children under the age of 8 years usually require conscious sedation in order to complete the MR examination without motion degradation.
MRI is considered safe for patients, even at very high field strengths. Serious injuries have been caused, however, by attraction of ferromagnetic objects into the magnet, which act as missiles if brought too close to the magnet. Likewise, ferromagnetic implants, such as aneurysm clips, may torque within the magnet, causing damage to vessels and even death. Metallic foreign bodies in the eye have moved and caused intraocular hemorrhage; screening for ocular metallic fragments is indicated in those with a history of metal work or ocular metallic foreign bodies. Implanted cardiac pacemakers are generally a contraindication to MRI owing to the risk of induced arrhythmias; however, some newer pacemakers have been shown to be safe. All health care personnel and patients must be screened and educated thoroughly to prevent such disasters because the magnet is always “on.” Table 440e-4 lists common contraindications for MRI.
COMMON CONTRAINDICATIONS TO MAGNETIC RESONANCE IMAGING |
Note: See also http://www.mrisafety.com.
MAGNETIC RESONANCE ANGIOGRAPHY
MR angiography is a general term describing several MR techniques that result in vascular-weighted images. These provide a vascular flow map rather than the anatomic map shown by conventional angiography. On routine spin echo MR sequences, moving protons (e.g., flowing blood, CSF) exhibit complex MR signals that range from high- to low-signal intensity relative to background stationary tissue. Fast-flowing blood returns no signal (flow void) on routine T1W or T2W spin echo MR images. Slower-flowing blood, as occurs in veins or distal to arterial stenosis, may appear high in signal. However, using special pulse sequences called gradient echo sequences, it is possible to increase the signal intensity of moving protons in contrast to the low signal background intensity of stationary tissue. This creates angiography-like images, which can be manipulated in three dimensions to highlight vascular anatomy and relationships.
So called time-of-flight (TOF) MRA relies on the suppression of nonmoving tissue to provide a low-intensity background for the high signal intensity of flowing blood entering the section; arterial or venous structures may be highlighted. A typical TOF MRA sequence results in a series of contiguous, thin MR sections (0.6–0.9 mm thick), which can be viewed as a stack and manipulated to create an angiographic image data set that can be reformatted and viewed in various planes and angles, much like that seen with conventional angiography (Fig. 440e-2G).
Phase-contrast MRA has a longer acquisition time than TOF MRA, but in addition to providing anatomic information similar to that of TOF imaging, it can be used to reveal the velocity and direction of blood flow in a given vessel. Through the selection of different imaging parameters, differing blood velocities can be highlighted; selective venous and arterial MRA images can thus be obtained. One advantage of phase-contrast MRA is the excellent suppression of high-signal-intensity background structures.
MRA can also be acquired during infusion of contrast material. Advantages include faster imaging times (1–2 min vs 10 min), fewer flow-related artifacts, and higher resolution images. Recently, contrast-enhanced MRA has become the standard for extracranial vascular MRA. This technique entails rapid imaging using coronal three-dimensional TOF sequences during a bolus infusion of gadolinium contrast agent. Proper technique and timing of acquisition relative to bolus arrival are critical for success.
MRA has lower spatial resolution compared with conventional film-based angiography, and therefore the detection of small-vessel abnormalities, such as vasculitis and distal vasospasm, is problematic. MRA is also less sensitive to slowly flowing blood and thus may not reliably differentiate complete from near-complete occlusions. Motion, either by the patient or by anatomic structures, may distort the MRA images, creating artifacts. These limitations notwithstanding, MRA has proved useful in evaluation of the extracranial carotid and vertebral circulation as well as of larger-caliber intracranial arteries and dural sinuses. It has also proved useful in the noninvasive detection of intracranial aneurysms and vascular malformations.
ECHO-PLANAR MRI
Recent improvements in gradients, software, and high-speed computer processors now permit extremely rapid MRI of the brain. With echo-planar MRI (EPI), fast gradients are switched on and off at high speeds to create the information used to form an image. In routine spin echo imaging, images of the brain can be obtained in 5–10 min. With EPI, all of the information required for processing an image is accumulated in milliseconds, and the information for the entire brain can be obtained in less than 1–2 min, depending on the degree of resolution required or desired. Fast MRI reduces patient and organ motion and is the basis of perfusion imaging during contrast infusion and kinematic motion studies. EPI is also the sequence used to obtain diffusion imaging and tractography, as well as fMRI and arterial spin-labeled studies (Figs. 440e-2H, 440e-3, 440e-4C, and 440e-6; and see Fig. 446-16).
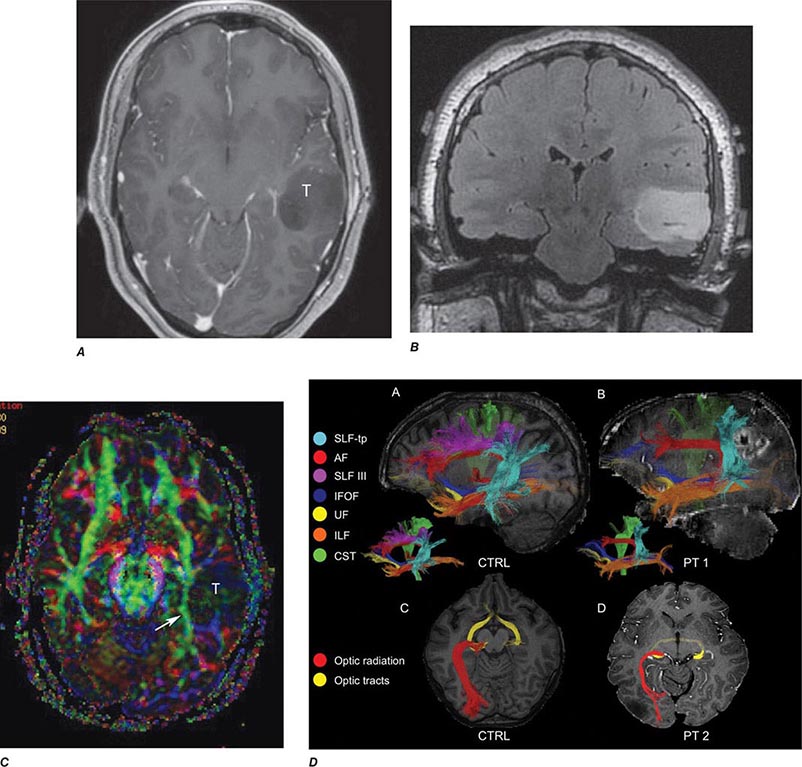
FIGURE 440e-6 Diffusion tractography in cerebral glioma. Associative and descending pathways in a healthy subject (A) and in a patient with parietal lobe glioblastoma (B) presenting with a language deficit: the mass causes a disruption of the arcuate-SLF complex, in particular of its anterior portion (SLF III). Also shown are bilateral optic tract and left optic radiation pathways in a healthy subject (C) and in a patient with left occipital grade II oligoastrocytoma (D): the mass causes a disruption of the left optic radiation. Shown in neurologic orientation, i.e., the left brain appears on the left side of the image. AF, long segment of the arcuate fascicle; CST, corticospinal tract; IFOF: inferior fronto-occipital fascicle; ILF, inferior longitudinal fascicle; SLF III, superior longitudinal fascicle III or anterior segment of the arcuate fascicle; SLF-tp, temporo-parietal portion of the superior longitudinal fascicle or posterior segment of the arcuate fascicle; T, tumor; UF, uncinated fascicle. (Part D courtesy of Eduardo Caverzasi and Roland Henry.)
Perfusion and diffusion imaging are EPI techniques that are useful in early detection of ischemic injury of the brain and may be useful together to demonstrate infarcted tissue as well as ischemic but potentially viable tissue at risk of infarction (e.g., the ischemic penumbra). Diffusion-weighted imaging (DWI) assesses microscopic motion of water; abnormal restriction of motion appears as relative high-signal intensity on diffusion-weighted images. Infarcted tissue reduces the water motion within cells and in the interstitial tissues, resulting in high signal on DWI. DWI is the most sensitive technique for detection of acute cerebral infarction of <7 days in duration (Fig. 440e-2H). It is also quite sensitive for detecting dying or dead brain tissue secondary to encephalitis, as well as abscess formation (Fig. 440e-3B).
Perfusion MRI involves the acquisition of fast echo planar gradient images during a rapid intravenous bolus of gadolinium contrast material. Relative cerebral blood volume, mean transit time, and cerebral blood flow maps are then derived. Delay in mean transit time and reduction in cerebral blood volume and cerebral blood flow are typical of infarction. In the setting of reduced blood flow, a prolonged mean transit time of contrast but normal or elevated cerebral blood volume may indicate tissue supplied by collateral flow that is at risk of infarction. Perfusion MRI imaging can also be used in the assessment of brain tumors to differentiate intraaxial primary tumors, whose BBB is relatively intact, from extraaxial tumors or metastases, which demonstrate a relatively more permeable BBB.
Diffusion tensor imaging is derived from diffusion MRI imaging sequences, which assesses the direction of microscopic motion of water along white matter tracts. This technique has great potential in the assessment of brain maturation as well as disease entities that undermine the integrity of the white matter architecture. It has proven valuable in preoperative assessment of subcortical white matter tract anatomy prior to brain tumor surgery (Fig. 440e-6).
fMRI of the brain is an EPI technique that localizes regions of activity in the brain following task activation. Neuronal activity elicits a slight increase in the delivery of oxygenated blood flow to a specific region of activated brain. This results in an alteration in the balance of oxyhemoglobin and deoxyhemoglobin, which yields a 2–3% increase in signal intensity within veins and local capillaries. Further studies will determine whether these techniques are cost effective or clinically useful, but currently, preoperative somatosensory and auditory cortex localization is possible. This technique has proved useful to neuroscientists interested in interrogating the localization of certain brain functions.
ARTERIAL SPIN LABELING
ASL is a quantitative noninvasive MR technique that measures cerebral blood flow. Blood traversing in the neck is labeled by an MR pulse and then imaged in the brain after a short delay. The signal in the brain is reflective of blood flow. ASL is an especially important technique for patients with kidney failure and for pediatric patients in whom the use of radioactive tracers or exogenous contrast agents is contraindicated. Increased cerebral flow is more easily identified than slow flow, which can be sometimes difficult to quantify. This technique has also been shown useful in detecting arterial venous shunting in arteriovenous malformations and arteriovenous fistulas.
MAGNETIC RESONANCE NEUROGRAPHY
MRN is a T2W MR technique that shows promise in detecting increased signal in irritated, inflamed, or infiltrated peripheral nerves. Images are obtained with fat-suppressed fast spin echo imaging or short inversion recovery sequences. Irritated or infiltrated nerves will demonstrate high signal on T2W imaging. This is indicated in patients with radiculopathy whose conventional MR studies of the spine are normal, or in those suspected of peripheral nerve entrapment or trauma.
POSITRON EMISSION TOMOGRAPHY
PET relies on the detection of positrons emitted during the decay of a radionuclide that has been injected into a patient. The most frequently used moiety is 2-[18F]fluoro-2-deoxy-D-glucose (FDG), which is an analogue of glucose and is taken up by cells competitively with 2-deoxyglucose. Multiple images of glucose uptake activity are formed after 45–60 min. Images reveal differences in regional glucose activity among normal and pathologic brain structures. FDG-PET is used primarily for the detection of extracranial metastatic disease; however, a lower activity of FDG in the parietal lobes is associated with Alzheimer’s disease, a finding that may simply reflect atrophy that occurs in the later stages of the disease. Combination PET-CT scanners, in which both CT and PET are obtained at one sitting, have largely replaced PET scans alone for most clinical indications. MR-PET scanners have also been developed and may prove useful for imaging the brain and other organs without the radiation exposure of CT. More recent PET ligand developments include amyloid tracers, such as Pittsburgh compound B (PIB) and 18-F AV-45 (florbetapir), and tau PET tracers, such as 18F-T807 and T808. Studies have shown an increased percentage of amyloid deposition in patients with Alzheimer’s disease compared with mild cognitive impairment and healthy controls; however, up to 25% of cognitively “normal” patients show abnormalities on amyloid PET imaging. This may either reflect subclinical disease processes or variation of normal. Tau imaging may be more specific for Alzheimer’s disease, and clinical studies are under way.
MYELOGRAPHY
TECHNIQUE
Myelography involves the intrathecal instillation of specially formulated water-soluble iodinated contrast medium into the lumbar or cervical subarachnoid space. CT scanning is typically performed after myelography (CT myelography) to better demonstrate the spinal cord and roots, which appear as filling defects in the opacified subarachnoid space. Low-dose CT myelography, in which CT is performed after the subarachnoid injection of a small amount of relatively dilute contrast material, has replaced conventional myelography for many indications, thereby reducing exposure to radiation and contrast media. Newer multidetector scanners now obtain CT studies quickly so that reformations in sagittal and coronal planes, equivalent to traditional myelography projections, are now routine.
INDICATIONS
Myelography has been largely replaced by CT myelography and MRI for diagnosis of diseases of the spinal canal and cord (Table 440e-1). Remaining indications for conventional plain-film myelography include the evaluation of suspected meningeal or arachnoid cysts and the localization of CSF fistulas. Conventional myelography and CT myelography provide the most precise information in patients with prior spinal fusion and spinal fixation hardware.
CONTRAINDICATIONS
Myelography is relatively safe; however, it should be performed with caution in any patient with elevated intracranial pressure, evidence of a spinal block, or a history of allergic reaction to intrathecal contrast media. In patients with a suspected spinal block, MR is the preferred technique. If myelography is necessary, only a small amount of contrast medium should be instilled below the lesion in order to minimize the risk of neurologic deterioration. Lumbar puncture is to be avoided in patients with bleeding disorders, including patients receiving anticoagulant therapy, as well as in those with infections of the overlying soft tissues (Chap. 443e).
COMPLICATIONS
Headache is the most frequent complication of myelography and is reported to occur in 5–30% of patients. Nausea and vomiting may also occur rarely. Postural headache (post–lumbar puncture headache) is generally due to leakage of CSF from the puncture site, resulting in CSF hypotension. A higher incidence is noted among younger women and with the use of larger gauge cutting-type spinal needles. If significant headache persists for longer than 48 h, placement of an epidural blood patch should be considered. Management of lumbar puncture headache is discussed in Chap. 21. Vasovagal syncope may occur during lumbar puncture; it is accentuated by the upright position used during lumbar myelography. Adequate hydration before and after myelography will reduce the incidence of this complication.
Hearing loss is a rare complication of myelography. It may result from a direct toxic effect of the contrast medium or from an alteration of the pressure equilibrium between CSF and perilymph in the inner ear. Puncture of the spinal cord is a rare but serious complication of cervical (C1–2) or high lumbar puncture. The risk of cord puncture is greatest in patients with spinal stenosis, Chiari malformations, or conditions that reduce CSF volume. In these settings, a low-dose lumbar injection followed by thin-section CT or MRI is a safer alternative to cervical puncture. Intrathecal contrast reactions are rare, but aseptic meningitis and encephalopathy are reported complications. The latter is usually dose related and associated with contrast entering the intracranial subarachnoid space. Seizures occur following myelography in 0.1–0.3% of patients. Risk factors include a preexisting seizure disorder and the use of a total iodine dose of >4500 mg. Other reported complications include hyperthermia, hallucinations, depression, and anxiety states. These side effects have been reduced by the development of nonionic, water-soluble contrast agents as well as by head elevation and generous hydration following myelography.
SPINE INTERVENTIONS
DISKOGRAPHY
The evaluation of back pain and radiculopathy may require diagnostic procedures that attempt either to reproduce the patient’s pain or relieve it, indicating its correct source prior to lumbar fusion. Diskography is performed by fluoroscopic placement of a 22- to 25-gauge needle into the intervertebral disk and subsequent injection of 1–3 mL of contrast media. The intradiskal pressure is recorded, as is an assessment of the patient’s response to the injection of contrast material. Typically little or no pain is felt during injection of a normal disk, which does not accept much more than 1 mL of contrast material, even at pressures as high as 415–690 kPa (60–100 lb/in2). CT and plain films are obtained following the procedure. Concerns have been raised that diskography may contribute to an accelerated rate of disk degeneration.
SELECTIVE NERVE ROOT AND EPIDURAL SPINAL INJECTIONS
Percutaneous selective nerve root and epidural blocks with glucocorticoid and anesthetic mixtures may be both therapeutic and diagnostic, especially if a patient’s pain is relieved. Typically, 1–2 mL of an equal mixture of a long-acting glucocorticoid such as betamethasone and a long-acting anesthetic such as bupivacaine 0.75% is instilled under CT or fluoroscopic guidance in the intraspinal epidural space or adjacent to an existing nerve root.
ANGIOGRAPHY
Catheter angiography is indicated for evaluating intracranial small-vessel pathology (such as vasculitis), for assessing vascular malformations and aneurysms, and in endovascular therapeutic procedures (Table 440e-1). Angiography has been replaced for many indications by CT/CTA or MRI/MRA.
Angiography carries the greatest risk of morbidity of all diagnostic imaging procedures, owing to the necessity of inserting a catheter into a blood vessel, directing the catheter to the required location, injecting contrast material to visualize the vessel, and removing the catheter while maintaining hemostasis. Therapeutic transcatheter procedures (see below) have become important options for the treatment of some cerebrovascular diseases. The decision to undertake a diagnostic or therapeutic angiographic procedure requires careful assessment of the goals of the investigation and its attendant risks.
To improve tolerance to contrast agents, patients undergoing angiography should be well hydrated before and after the procedure. Because the femoral route is used most commonly, the femoral artery must be compressed after the procedure to prevent a hematoma from developing. The puncture site and distal pulses should be evaluated carefully after the procedure; complications can include thigh hematoma or lower extremity emboli.
COMPLICATIONS
A common femoral arterial puncture provides retrograde access via the aorta to the aortic arch and great vessels. The most feared complication of cerebral angiography is stroke. Thrombus can form on or inside the tip of the catheter, and atherosclerotic thrombus or plaque can be dislodged by the catheter or guide wire or by the force of injection and can embolize distally in the cerebral circulation. Risk factors for ischemic complications include limited experience on the part of the angiographer, atherosclerosis, vasospasm, low cardiac output, decreased oxygen-carrying capacity, advanced age, and prior history of migraine. The risk of a neurologic complication varies but is ~4% for transient ischemic attack and stroke, 1% for permanent deficit, and <0.1% for death.
Ionic contrast material injected into the cerebral vasculature can be neurotoxic if the BBB is breached, either by an underlying disease or by the injection of hyperosmolar contrast agent. Ionic contrast media are less well tolerated than nonionic media, probably because they can induce changes in cell membrane electrical potentials. Patients with dolichoectasia of the basilar artery can suffer reversible brainstem dysfunction and acute short-term memory loss during angiography, owing to the slow percolation of the contrast material and the consequent prolonged exposure of the brain. Rarely, an intracranial aneurysm ruptures during an angiographic contrast injection, causing subarachnoid hemorrhage, perhaps as a result of injection under high pressure.
SPINAL ANGIOGRAPHY
Spinal angiography may be indicated to evaluate vascular malformations and tumors and to identify the artery of Adamkiewicz (Chap. 456) prior to aortic aneurysm repair. The procedure is lengthy and requires the use of relatively large volumes of contrast; the incidence of serious complications, including paraparesis, subjective visual blurring, and altered speech, is ~2%. Gadolinium-enhanced MRA has been used successfully in this setting, as has iodinated contrast CTA, which has promise for replacing diagnostic spinal angiography for some indications.
INTERVENTIONAL NEURORADIOLOGY
This rapidly developing field is providing new therapeutic options for patients with challenging neurovascular problems. Available procedures include detachable coil therapy for aneurysms, particulate or liquid adhesive embolization of arteriovenous malformations, stent retrieval systems for embolectomy, balloon angioplasty and stenting of arterial stenosis or vasospasm, transarterial or transvenous embolization of dural arteriovenous fistulas, balloon occlusion of carotid-cavernous and vertebral fistulas, endovascular treatment of vein-of-Galen malformations, preoperative embolization of tumors, and thrombolysis of acute arterial or venous thrombosis. Many of these disorders place the patient at high risk of cerebral hemorrhage, stroke, or death.
The highest complication rates are found with the therapies designed to treat the highest risk diseases. The advent of electrolytically detachable coils has ushered in a new era in the treatment of cerebral aneurysms. Two randomized trials found reductions of morbidity and mortality at 1 year among those treated for aneurysm with detachable coils compared with neurosurgical clipping. It remains to be determined what the role of coils will be relative to surgical options, but in many centers, coiling has become standard therapy for many aneurysms.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree