PART 14: Disorders of the Gastrointestinal System
SECTION 1 | DISORDERS OF THE ALIMENTARY TRACT |
344 | Approach to the Patient with Gastrointestinal Disease |
ANATOMIC CONSIDERATIONS
The gastrointestinal (GI) tract extends from the mouth to the anus and is composed of several organs with distinct functions. Specialized independently controlled thickened sphincters that assist in gut compartmentalization separate the organs. The gut wall is organized into well-defined layers that contribute to functional activities in each region. The mucosa is a barrier to luminal contents or a site for transfer of fluids or nutrients. Gut smooth muscle in association with the enteric nervous system mediates propulsion from one region to the next. Many GI organs possess a serosal layer that provides a supportive foundation but that also permits external input.
Interactions with other organ systems serve the needs both of the gut and the body. Pancreaticobiliary conduits deliver bile and enzymes into the duodenum. A rich vascular supply is modulated by GI tract activity. Lymphatic channels assist in gut immune activities. Intrinsic gut wall nerves provide the basic controls for propulsion and fluid regulation. Extrinsic neural input provides volitional or involuntary control to degrees that are specific for each gut region.
FUNCTIONS OF THE GASTROINTESTINAL TRACT
The GI tract serves two main functions—assimilating nutrients and eliminating waste. The gut anatomy is organized to serve these functions. In the mouth, food is processed, mixed with salivary amylase, and delivered to the gut lumen. The esophagus propels the bolus into the stomach; the lower esophageal sphincter prevents oral reflux of gastric contents. The esophageal mucosa has a protective squamous histology, which does not permit significant diffusion or absorption. Propulsive esophageal activities are exclusively aboral and coordinate with relaxation of the upper and lower esophageal sphincters on swallowing.
The stomach furthers food preparation by triturating and mixing the bolus with pepsin and acid. Gastric acid also sterilizes the upper gut. The proximal stomach serves a storage function by relaxing to accommodate the meal. The distal stomach exhibits phasic contractions that propel solid food residue against the pylorus, where it is repeatedly propelled proximally for further mixing before it is emptied into the duodenum. Finally, the stomach secretes intrinsic factor for vitamin B12 absorption.
The small intestine serves most of the nutrient absorptive function of the gut. The intestinal mucosa exhibits villus architecture to provide maximal surface area for absorption and is endowed with specialized enzymes and transporters. Triturated food from the stomach mixes with pancreatic juice and bile in the duodenum to facilitate digestion. Pancreatic juice contains the main enzymes for carbohydrate, protein, and fat digestion as well as bicarbonate to optimize the pH for activation of these enzymes. Bile secreted by the liver and stored in the gallbladder is essential for intestinal lipid digestion. The proximal intestine is optimized for rapid absorption of nutrient breakdown products and most minerals, whereas the ileum is better suited for absorption of vitamin B12 and bile acids. The small intestine also aids in waste elimination. Bile contains by-products of erythrocyte degradation, toxins, metabolized and unmetabolized medications, and cholesterol. Motor function of the small intestine delivers indigestible food residue and sloughed enterocytes into the colon for further processing. The small intestine terminates in the ileocecal junction, a sphincteric structure that prevents coloileal reflux and maintains small-intestinal sterility.
The colon prepares the waste material for controlled evacuation. The colonic mucosa dehydrates the stool, decreasing daily fecal volumes from 1000–1500 mL delivered from the ileum to 100–200 mL expelled from the rectum. The colonic lumen possesses a dense bacterial colonization that ferments undigested carbohydrates and short-chain fatty acids. Whereas transit times in the esophagus are on the order of seconds and times in the stomach and small intestine range from minutes to a few hours, propagation through the colon takes more than 1 day in most individuals. Colonic motor patterns exhibit a to-and-fro character that facilitates slow fecal desiccation. The proximal colon serves to mix and absorb fluid, while the distal colon exhibits peristaltic contractions and mass actions that function to expel the stool. The colon terminates in the anus, a structure with volitional and involuntary controls to permit retention of the fecal bolus until it can be released in a socially convenient setting.
EXTRINSIC MODULATION OF GUT FUNCTION
GI function is modified by influences outside of the gut. Unlike other organ systems, the gut is in continuity with the outside environment. Thus, protective mechanisms are vigilant against deleterious effects of foods, medications, toxins, and infectious organisms. Mucosal immune mechanisms include chronic lymphocyte and plasma cell populations in the epithelial layer and lamina propria backed up by lymph node chains to prevent noxious agents from entering the circulation. Antimicrobial peptides secreted by Paneth cells in the intestine further contribute to the defense mechanisms against pathogens in the lumen. All substances absorbed into the bloodstream are filtered through the liver via the portal venous circulation. In the liver, many drugs and toxins are detoxified by a variety of mechanisms. Although intrinsic nerves control most basic gut activities, extrinsic neural input modulates many functions. Two activities under voluntary control are swallowing and defecation. Many normal GI reflexes involve extrinsic vagus or splanchnic nerve pathways. The brain-gut axis further alters function in regions not under volitional regulation. As an example, stress has potent effects on gut motor, secretory, and sensory functions.
OVERVIEW OF GASTROINTESTINAL DISEASES
GI diseases develop as a result of abnormalities within or outside of the gut and range in severity from those that produce mild symptoms and no long-term morbidity to those with intractable symptoms or adverse outcomes. Diseases may be localized to one organ or exhibit diffuse involvement at many sites.
CLASSIFICATION OF GI DISEASES
GI diseases are manifestations of alterations in nutrient assimilation or waste evacuation or in the activities supporting these main functions.
Impaired Digestion and Absorption Diseases of the stomach, intestine, biliary tree, and pancreas can disrupt digestion and absorption. The most common intestinal maldigestion syndrome, lactase deficiency, produces gas and diarrhea after ingestion of dairy products and has no adverse outcomes. Other intestinal enzyme deficiencies produce similar symptoms after ingestion of other simple sugars. Conversely, celiac disease, bacterial overgrowth, infectious enteritis, Crohn’s ileitis, and radiation damage, which affect digestion and/or absorption more diffusely, produce anemia, dehydration, electrolyte disorders, or malnutrition. Gastric hypersecretory conditions such as Zollinger-Ellison syndrome damage the intestinal mucosa, impair pancreatic enzyme activation, and accelerate transit due to excess gastric acid. Biliary obstruction from stricture or neoplasm impairs fat digestion. Impaired pancreatic enzyme release in chronic pancreatitis or pancreatic cancer decreases intraluminal digestion and can lead to malnutrition.
Altered Secretion Selected GI diseases result from dysregulation of gut secretion. Gastric acid hypersecretion occurs in Zollinger-Ellison syndrome, G cell hyperplasia, retained antrum syndrome, and some individuals with duodenal ulcers. Conversely, patients with atrophic gastritis or pernicious anemia release little or no gastric acid. Inflammatory and infectious small-intestinal and colonic diseases produce fluid loss through impaired absorption or enhanced secretion. Common intestinal and colonic hypersecretory conditions cause diarrhea and include acute bacterial or viral infection, chronic Giardia or cryptosporidia infections, small-intestinal bacterial overgrowth, bile salt diarrhea, microscopic colitis, diabetic diarrhea, and abuse of certain laxatives. Less common causes include large colonic villus adenomas and endocrine neoplasias with tumor overproduction of secretagogue transmitters like vasoactive intestinal polypeptide.
Altered Gut Transit Impaired gut transit may be secondary to mechanical obstruction. Esophageal occlusion often results from acid-induced stricture or neoplasm. Gastric outlet obstruction develops from peptic ulcer disease or gastric cancer. Small-intestinal obstruction most commonly results from adhesions but may also occur with Crohn’s disease, radiation- or drug-induced strictures, and less likely malignancy. The most common cause of colonic obstruction is colon cancer, although inflammatory strictures develop in patients with inflammatory bowel disease, after certain infections such as diverticulitis, or with some drugs.
Retardation of propulsion also develops from disordered motor function. Achalasia is characterized by impaired esophageal body peristalsis and incomplete lower esophageal sphincter relaxation. Gastroparesis is the symptomatic delay in gastric emptying of meals due to impaired gastric motility. Intestinal pseudoobstruction causes marked delays in small-bowel transit due to enteric nerve or intestinal smooth-muscle injury. Slow-transit constipation is produced by diffusely impaired colonic propulsion. Constipation also is produced by outlet abnormalities such as rectal prolapse, intussusception, or dyssynergia—a failure of anal or puborectalis relaxation upon attempted defecation.
Disorders of rapid propulsion are less common than those with delayed transit. Rapid gastric emptying occurs in postvagotomy dumping syndrome, with gastric hypersecretion, and in some cases of functional dyspepsia and cyclic vomiting syndrome. Exaggerated intestinal or colonic motor patterns may be responsible for diarrhea in irritable bowel syndrome. Accelerated transit with hyperdefecation is noted in hyperthyroidism.
Immune Dysregulation Many inflammatory GI conditions are consequences of altered gut immune function. The mucosal inflammation of celiac disease results from dietary ingestion of gluten-containing grains. Some patients with food allergy also exhibit altered immune populations. Eosinophilic esophagitis and eosinophilic gastroenteritis are inflammatory disorders with prominent mucosal eosinophils. Ulcerative colitis and Crohn’s disease are disorders of uncertain etiology that produce mucosal injury primarily in the lower gut. The microscopic colitides, lymphocytic and collagenous colitis, exhibit colonic subepithelial infiltrates without visible mucosal damage. Bacterial, viral, and protozoal organisms may produce ileitis or colitis in selected patient populations.
Impaired Gut Blood Flow Different GI regions are at variable risk for ischemic damage from impaired blood flow. Rare cases of gastroparesis result from blockage of the celiac and superior mesenteric arteries. More commonly encountered are intestinal and colonic ischemia that are consequences of arterial embolus, arterial thrombosis, venous thrombosis, or hypoperfusion from dehydration, sepsis, hemorrhage, or reduced cardiac output. These may produce mucosal injury, hemorrhage, or even perforation. Chronic ischemia may result in intestinal stricture. Some cases of radiation enterocolitis exhibit reduced mucosal blood flow.
Neoplastic Degeneration All GI regions are susceptible to malignant degeneration to varying degrees. In the United States, colorectal cancer is most common and usually presents after age 50 years. Worldwide, gastric cancer is prevalent especially in certain Asian regions. Esophageal cancer develops with chronic acid reflux or after an extensive alcohol or tobacco use history. Small-intestinal neoplasms are rare and occur with underlying inflammatory disease. Anal cancers arise after prior anal infection or inflammation. Pancreatic and biliary cancers elicit severe pain, weight loss, and jaundice and have poor prognoses. Hepatocellular carcinoma usually arises in the setting of chronic viral hepatitis or cirrhosis secondary to other causes. Most GI cancers exhibit carcinomatous histology; however, lymphomas and other cell types also are observed.
Disorders Without Obvious Organic Abnormalities The most common GI disorders show no abnormalities on biochemical or structural testing and include irritable bowel syndrome, functional dyspepsia, functional chest pain, and functional heartburn. These disorders exhibit altered gut motor function; however, the pathogenic relevance of these abnormalities is uncertain. Exaggerated visceral sensory responses to noxious stimulation may cause discomfort in these disorders. Symptoms in other patients result from altered processing of visceral pain sensations in the central nervous system. Functional bowel patients with severe symptoms may exhibit significant emotional disturbances on psychometric testing. Subtle immunologic defects may contribute to functional symptoms as well.
Genetic Influences Although many GI diseases result from environmental factors, others exhibit hereditary components. Family members of inflammatory bowel disease patients show a genetic predisposition to disease development themselves. Colonic and esophageal malignancies arise in certain inherited disorders. Rare genetic dysmotility syndromes are described. Familial clustering is even observed in the functional bowel disorders, although this may be secondary learned familial illness behavior rather than a true hereditary factor.
SYMPTOMS OF GASTROINTESTINAL DISEASE
The most common GI symptoms are abdominal pain, heartburn, nausea and vomiting, altered bowel habits, GI bleeding, and jaundice (Table 344-1). Others are dysphagia, anorexia, weight loss, fatigue, and extraintestinal symptoms.
COMMON CAUSES OF COMMON GASTROINTESTINAL (GI) SYMPTOMS |
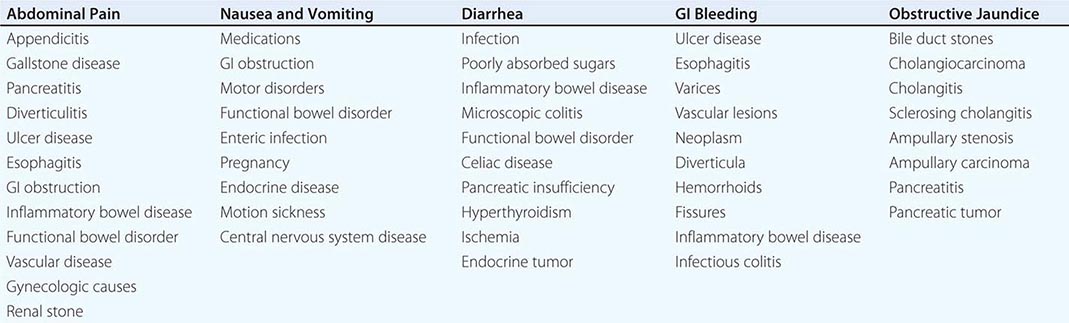
Abdominal Pain Abdominal pain results from GI disease and extra-intestinal conditions involving the genitourinary tract, abdominal wall, thorax, or spine. Visceral pain generally is midline in location and vague in character, whereas parietal pain is localized and precisely described. Common inflammatory diseases with pain include peptic ulcer, appendicitis, diverticulitis, inflammatory bowel disease, and infectious enterocolitis. Other intraabdominal causes of pain include gallstone disease and pancreatitis. Noninflammatory visceral sources include mesenteric ischemia and neoplasia. The most common causes of abdominal pain are irritable bowel syndrome and functional dyspepsia.
Heartburn Heartburn, a burning substernal sensation, is reported intermittently by at least 40% of the population. Classically, heartburn is felt to result from excess gastroesophageal reflux of acid. However, some cases exhibit normal esophageal acid exposure and may result from reflux of nonacidic material or heightened sensitivity of esophageal mucosal nerves.
Nausea and Vomiting Nausea and vomiting are caused by GI diseases, medications, toxins, acute and chronic infection, endocrine disorders, labyrinthine conditions, and central nervous system disease. The best-characterized GI etiologies relate to mechanical obstruction of the upper gut; however, disorders of propulsion including gastroparesis and intestinal pseudoobstruction also elicit prominent symptoms. Nausea and vomiting also are commonly reported by patients with irritable bowel syndrome and functional disorders of the upper gut (including chronic idiopathic nausea and functional vomiting).
Altered Bowel Habits Altered bowel habits are common complaints of patients with GI disease. Constipation is reported as infrequent defecation, straining with defecation, passage of hard stools, or a sense of incomplete fecal evacuation. Causes of constipation include obstruction, motor disorders of the colon, medications, and endocrine diseases such as hypothyroidism and hyperparathyroidism. Diarrhea is reported as frequent defecation, passage of loose or watery stools, fecal urgency, or a similar sense of incomplete evacuation. The differential diagnosis of diarrhea is broad and includes infections, inflammatory causes, malabsorption, and medications. Irritable bowel syndrome produces constipation, diarrhea, or an alternating bowel pattern. Fecal mucus is common in irritable bowel syndrome, whereas pus characterizes inflammatory disease. Steatorrhea develops with malabsorption.
GI Bleeding Hemorrhage may develop from any gut organ. Most commonly, upper GI bleeding presents with melena or hematemesis, whereas lower GI bleeding produces passage of bright red or maroon stools. However, briskly bleeding upper sites can elicit voluminous red rectal bleeding, whereas slowly bleeding ascending colon sites may produce melena. Chronic slow GI bleeding may present with iron deficiency anemia. The most common upper GI causes of bleeding are ulcer disease, gastroduodenitis, and esophagitis. Other etiologies include portal hypertensive causes, malignancy, tears across the gastroesophageal junction, and vascular lesions. The most prevalent lower GI sources of hemorrhage include hemorrhoids, anal fissures, diverticula, ischemic colitis, and arteriovenous malformations. Other causes include neoplasm, inflammatory bowel disease, infectious colitis, drug-induced colitis, and other vascular lesions.
Jaundice Jaundice results from prehepatic, intrahepatic, or posthepatic disease. Posthepatic causes of jaundice include biliary diseases, such as choledocholithiasis, acute cholangitis, primary sclerosing cholangitis, other strictures, and neoplasm, and pancreatic disorders, such as acute and chronic pancreatitis, stricture, and malignancy.
Other Symptoms Other symptoms are manifestations of GI disease. Dysphagia, odynophagia, and unexplained chest pain suggest esophageal disease. A globus sensation is reported with esophagopharyngeal conditions, but also occurs with functional GI disorders. Weight loss, anorexia, and fatigue are nonspecific symptoms of neoplastic, inflammatory, gut motility, pancreatic, small-bowel mucosal, and psychiatric conditions. Fever is reported with inflammatory illness, but malignancies also evoke febrile responses. GI disorders also produce extraintestinal symptoms. Inflammatory bowel disease is associated with hepatobiliary dysfunction, skin and eye lesions, and arthritis. Celiac disease may present with dermatitis herpetiformis. Jaundice can produce pruritus. Conversely, systemic diseases can have GI consequences. Systemic lupus may cause gut ischemia, presenting with pain or bleeding. Overwhelming stress or severe burns may lead to gastric ulcer formation.
EVALUATION OF THE PATIENT WITH GASTROINTESTINAL DISEASE
Evaluation of the patient with GI disease begins with a careful history and examination. Subsequent investigation with a variety of tools designed to test gut structure or function are indicated in selected cases. Some patients exhibit normal findings on diagnostic testing. In these individuals, validated symptom profiles are used to confidently diagnose a functional bowel disorder.
HISTORY
The history of the patient with suspected GI disease has several components. Symptom timing suggests specific etiologies. Symptoms of short duration commonly result from acute infection, toxin exposure, or abrupt inflammation or ischemia. Long-standing symptoms point to underlying chronic inflammatory or neoplastic conditions or functional bowel disorders. Symptoms from mechanical obstruction, ischemia, inflammatory bowel disease, and functional bowel disorders are worsened by meals. Conversely, ulcer symptoms may be relieved by eating or antacids. Symptom patterns and duration may suggest underlying etiologies. Ulcer pain occurs at intermittent intervals lasting weeks to months, whereas biliary colic has a sudden onset and lasts up to several hours. Pain from acute inflammation as with acute pancreatitis is severe and persists for days to weeks. Meals elicit diarrhea in some cases of inflammatory bowel disease and irritable bowel syndrome. Defecation relieves discomfort in inflammatory bowel disease and irritable bowel syndrome. Functional bowel disorders are exacerbated by stress. Sudden awakening from sound sleep suggests organic rather than functional disease. Diarrhea from malabsorption usually improves with fasting, whereas secretory diarrhea persists without oral intake.
Symptom relation to other factors narrows the list of diagnostic possibilities. Obstructive symptoms with prior abdominal surgery raise concern for adhesions, whereas loose stools after gastrectomy or gallbladder excision suggest dumping syndrome or postcholecystectomy diarrhea. Symptom onset after travel prompts a search for enteric infection. Medications may produce pain, altered bowel habits, or GI bleeding. Lower GI bleeding likely results from neoplasms, diverticula, or vascular lesions in an older person and from anorectal abnormalities or inflammatory bowel disease in a younger individual. Celiac disease is prevalent in people of northern European descent, whereas inflammatory bowel disease is more common in certain Jewish populations. A sexual history may raise concern for sexually transmitted diseases or immunodeficiency.
For more than two decades, working groups have been convened to devise symptom criteria to improve the confident diagnosis of functional bowel disorders and to minimize the numbers of unnecessary diagnostic tests performed. The most widely accepted symptom-based criteria are the Rome criteria. When tested against findings of structural investigations, the Rome criteria exhibit diagnostic specificities exceeding 90% for many of the functional bowel disorders.
PHYSICAL EXAMINATION
The physical exam complements information from the history. Abnormal vital signs provide diagnostic clues and determine the need for acute intervention. Fever suggests inflammation or neoplasm. Orthostasis is found with significant blood loss, dehydration, sepsis, or autonomic neuropathy. Skin, eye, or joint findings may point to specific diagnoses. Neck exam with swallowing assessment evaluates dysphagia. Cardiopulmonary disease may present with abdominal pain or nausea; thus lung and cardiac exams are important. Pelvic examination tests for a gynecologic source of abdominal pain. Rectal exam may detect blood, indicating gut mucosal injury or neoplasm or a palpable inflammatory mass in appendicitis. Metabolic conditions and gut motor disorders have associated peripheral neuropathy.
Inspection of the abdomen may reveal distention from obstruction, tumor, or ascites or vascular abnormalities with liver disease. Ecchymoses develop with severe pancreatitis. Auscultation can detect bruits or friction rubs from vascular disease or hepatic tumors. Loss of bowel sounds signifies ileus, whereas high-pitched, hyperactive sounds characterize intestinal obstruction. Percussion assesses liver size and can detect shifting dullness from ascites. Palpation assesses for hepatosplenomegaly as well as neoplastic or inflammatory masses. Abdominal exam is helpful in evaluating unexplained pain. Intestinal ischemia elicits severe pain but little tenderness. Patients with visceral pain may exhibit generalized discomfort, whereas those with parietal pain or peritonitis have directed pain, often with involuntary guarding, rigidity, or rebound. Patients with musculoskeletal abdominal wall pain may note tenderness exacerbated by Valsalva or straight-leg lift maneuvers.
TOOLS FOR PATIENT EVALUATION
Laboratory, radiographic, and functional tests can assist in diagnosis of suspected GI disease. The GI tract also is amenable to internal evaluation with upper and lower endoscopy and to examination of luminal contents. Histopathologic exams of GI tissues complement these tests.
Laboratory Selected laboratory tests facilitate the diagnosis of GI disease. Iron-deficiency anemia suggests mucosal blood loss, whereas vitamin B12 deficiency results from small-intestinal, gastric, or pancreatic disease. Either also can result from inadequate oral intake. Leukocytosis and increased sedimentation rates and C-reactive protein levels are found in inflammatory conditions, whereas leukopenia is seen in viremic illness. Severe vomiting or diarrhea elicits electrolyte disturbances, acid-base abnormalities, and elevated blood urea nitrogen. Pancreaticobiliary or liver disease is suggested by elevated pancreatic or liver chemistries. Thyroid chemistries, cortisol, and calcium levels are obtained to exclude endocrinologic causes of GI symptoms. Pregnancy testing is considered for women with unexplained nausea. Serologic tests can screen for celiac disease, inflammatory bowel disease, rheumatologic diseases like lupus or scleroderma, and paraneoplastic dysmotility syndromes. Hormone levels are obtained for suspected endocrine neoplasia. Intraabdominal malignancies produce other tumor markers including the carcinoembryonic antigen CA 19-9 and α-fetoprotein. Blood testing also monitors medication therapy in some diseases, as with thiopurine metabolite levels in inflammatory bowel disease. Other body fluids are sampled under certain circumstances. Ascitic fluid is analyzed for infection, malignancy, or findings of portal hypertension. Cerebrospinal fluid is obtained for suspected central nervous system causes of vomiting. Urine samples screen for carcinoid, porphyria, and heavy metal intoxication.
Luminal Contents Luminal contents can be examined for diagnostic clues. Stool samples are cultured for bacterial pathogens, examined for leukocytes and parasites, or tested for Giardia antigen. Duodenal aspirates can be examined for parasites or cultured for bacterial overgrowth. Fecal fat is quantified in possible malabsorption. Stool electrolytes can be measured in diarrheal conditions. Laxative screens are done when laxative abuse is suspected. Gastric acid is quantified to rule out Zollinger-Ellison syndrome. Esophageal pH testing is done for refractory symptoms of acid reflux, whereas impedance techniques assess for nonacidic reflux. Pancreatic juice is analyzed for enzyme or bicarbonate content to exclude pancreatic exocrine insufficiency.
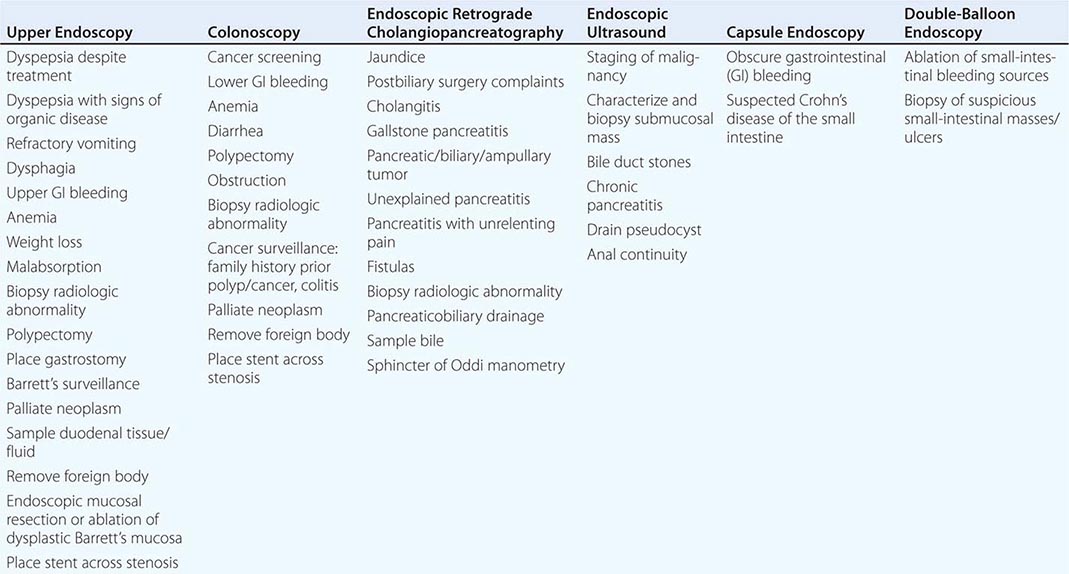
Endoscopy The gut is accessible with endoscopy, which can provide the diagnosis of the causes of bleeding, pain, nausea and vomiting, weight loss, altered bowel function, and fever. Table 344-2 lists the most common indications for the major endoscopic procedures. Upper endoscopy evaluates the esophagus, stomach, and duodenum, whereas colonoscopy assesses the colon and distal ileum. Upper endoscopy is advocated as the initial structural test performed in patients with suspected ulcer disease, esophagitis, neoplasm, malabsorption, and Barrett’s metaplasia because of its ability to directly visualize as well as biopsy the abnormality. Colonoscopy is the procedure of choice for colon cancer screening and surveillance as well as diagnosis of colitis secondary to infection, ischemia, radiation, and inflammatory bowel disease. Sigmoidoscopy examines the colon up to the splenic flexure and is currently used to exclude distal colonic inflammation or obstruction in young patients not at significant risk for colon cancer. For elusive GI bleeding secondary to arteriovenous malformations or superficial ulcers, small-intestinal examination is performed with push enteroscopy, capsule endoscopy, or double-balloon enteroscopy. Capsule endoscopy also can visualize small-intestinal Crohn’s disease in individuals with negative barium radiography. Endoscopic retrograde cholangiopancreaticography (ERCP) provides diagnoses of pancreatic and biliary disease. Endoscopic ultrasound is useful for evaluating extent of disease in GI malignancy as well as exclusion of choledocholithiasis, evaluation of pancreatitis, drainage of pancreatic pseudocysts, and assessment of anal continuity.
COMMON INDICATIONS FOR ENDOSCOPY |
Radiography/Nuclear Medicine Radiographic tests evaluate diseases of the gut and extraluminal structures. Oral or rectal contrast agents like barium provide mucosal definition from the esophagus to the rectum. Contrast radiography also assesses gut transit and pelvic floor dysfunction. Barium swallow is the initial procedure for evaluation of dysphagia to exclude subtle rings or strictures and assess for achalasia, whereas small-bowel contrast radiology reliably diagnoses intestinal tumors and Crohn’s ileitis. Contrast enemas are performed when colonoscopy is unsuccessful or contraindicated. Ultrasound and computed tomography (CT) evaluate regions not accessible by endoscopy or contrast studies, including the liver, pancreas, gallbladder, kidneys, and retroperitoneum. These tests are useful for diagnosis of mass lesions, fluid collections, organ enlargement, and, in the case of ultrasound, gallstones. CT and magnetic resonance (MR) colonography are being evaluated as alternatives to colonoscopy for colon cancer screening. MR imaging assesses the pancreaticobiliary ducts to exclude neoplasm, stones, and sclerosing cholangitis, and the liver to characterize benign and malignant tumors. Specialized CT or MR enterography can assess intensity of inflammatory bowel disease. Angiography excludes mesenteric ischemia and determines spread of malignancy. Angiographic techniques also access the biliary tree in obstructive jaundice. CT and MR techniques can be used to screen for mesenteric occlusion, thereby limiting exposure to angiographic dyes. Positron emission tomography can facilitate distinguishing malignant from benign disease in several organ systems.
Scintigraphy both evaluates structural abnormalities and quantifies luminal transit. Radionuclide bleeding scans localize bleeding sites in patients with brisk hemorrhage so that therapy with endoscopy, angiography, or surgery may be directed. Radiolabeled leukocyte scans can search for intraabdominal abscesses not visualized on CT. Biliary scintigraphy is complementary to ultrasound in the assessment of cholecystitis. Scintigraphy to quantify esophageal and gastric emptying is well established, whereas techniques to measure small-intestinal or colonic transit are less widely used.
Histopathology Gut mucosal biopsies obtained at endoscopy evaluate for inflammatory, infectious, and neoplastic disease. Deep rectal biopsies assist with diagnosis of Hirschsprung’s disease or amyloid. Liver biopsy is indicated in cases with abnormal liver chemistries, in unexplained jaundice, following liver transplant to exclude rejection, and to characterize the degree of inflammation in patients with chronic viral hepatitis prior to initiating antiviral therapy. Biopsies obtained during CT or ultrasound can evaluate for other intraabdominal conditions not accessible by endoscopy.
Functional Testing Tests of gut function provide important data when structural testing is nondiagnostic. In addition to gastric acid and pancreatic function testing, functional testing of motor activity is provided by manometric techniques. Esophageal manometry is useful for suspected achalasia, whereas small-intestinal manometry tests for pseudoobstruction. A wireless motility capsule is now available to measure transit and contractile activity in the stomach, small intestine, and colon in a single test. Anorectal manometry with balloon expulsion testing is used for unexplained incontinence or constipation from outlet dysfunction. Anorectal manometry and electromyography also assess anal function in fecal incontinence. Biliary manometry tests for sphincter of Oddi dysfunction with unexplained biliary pain. Measurement of breath hydrogen while fasting and after oral mono- or oligosaccharide challenge can screen for carbohydrate intolerance and small-intestinal bacterial overgrowth.
345 | Gastrointestinal Endoscopy |
Gastrointestinal endoscopy has been attempted for over 200 years, but the introduction of semirigid gastroscopes in the middle of the twentieth century marked the dawn of the modern endoscopic era. Since then, rapid advances in endoscopic technology have led to dramatic changes in the diagnosis and treatment of many digestive diseases. Innovative endoscopic devices and new endoscopic treatment modalities continue to expand the use of endoscopy in patient care.
Current flexible endoscopes provide an electronic video image generated by a charge-coupled device in the tip of the endoscope. Operator controls permit deflection of the endoscope tip; fiberoptic bundles or light-emitting diodes bring light to the tip of the endoscope; and working channels allow washing, suctioning, and the passage of instruments. Progressive changes in the diameter and stiffness of endoscopes have improved the ease and patient tolerance of endoscopy.
ENDOSCOPIC PROCEDURES
UPPER ENDOSCOPY
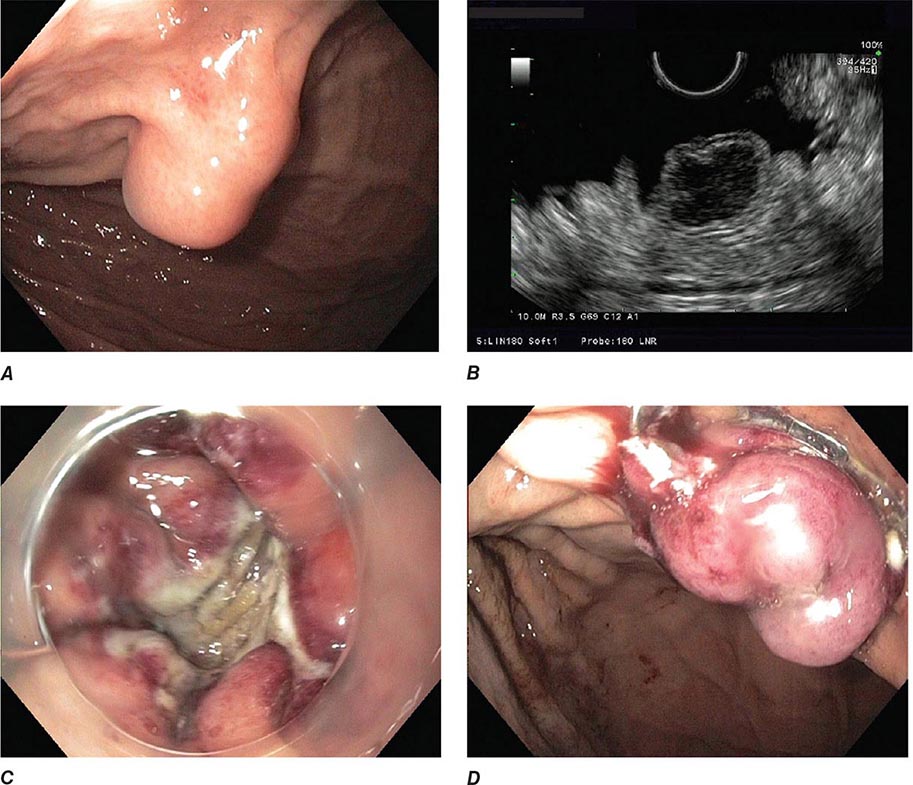
Upper endoscopy, also referred to as esophagogastroduodenoscopy (EGD), is performed by passing a flexible endoscope through the mouth into the esophagus, stomach, and duodenum. The procedure is the best method for examining the upper gastrointestinal mucosa. While the upper gastrointestinal radiographic series has similar accuracy for diagnosis of duodenal ulcer (Fig. 345-1), EGD is superior for detection of gastric ulcers (Fig. 345-2) and flat mucosal lesions such as Barrett’s esophagus (Fig. 345-3), and it permits directed biopsy and endoscopic therapy. Intravenous conscious sedation is given to most patients in the United States to ease the anxiety and discomfort of the procedure, although in many countries EGD is routinely performed with topical pharyngeal anesthesia only. Patient tolerance of unsedated EGD is improved by the use of an ultrathin, 5-mm diameter endoscope that can be passed transorally or transnasally.
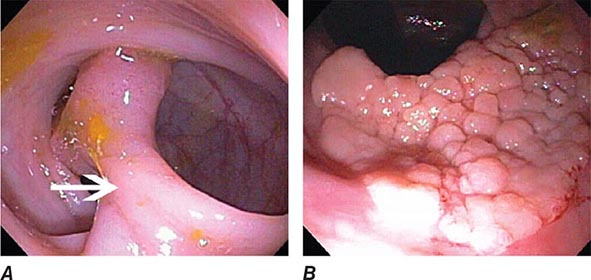
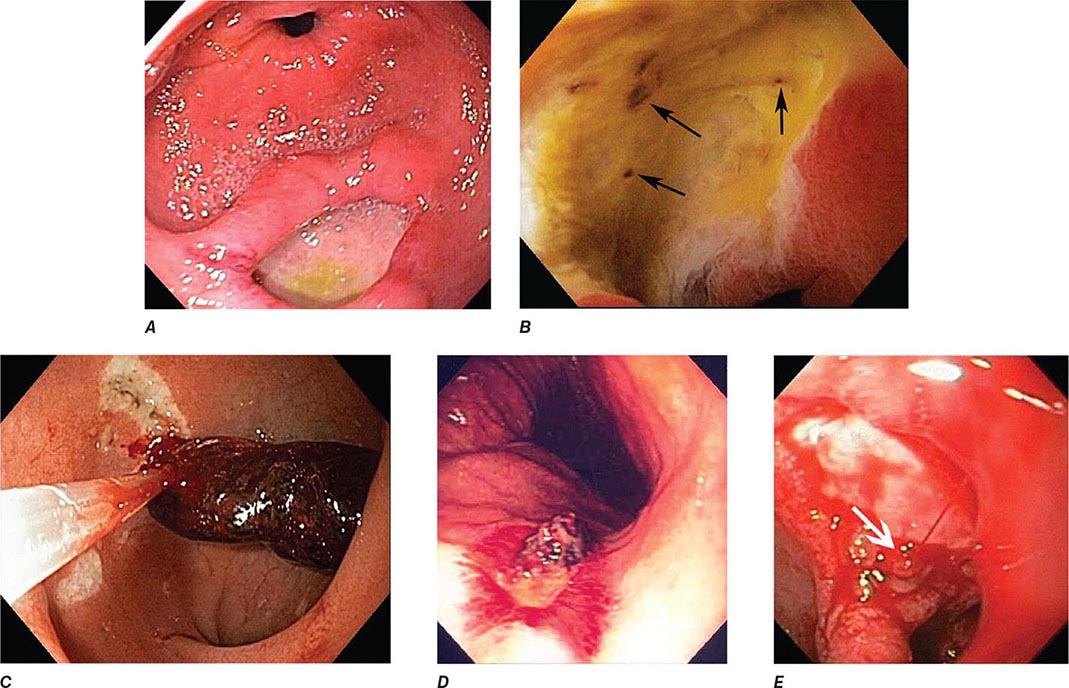
FIGURE 345-1 Duodenal ulcers. A. Ulcer with a clean base. B. Ulcer with a visible vessel (arrow) in a patient with recent hemorrhage.
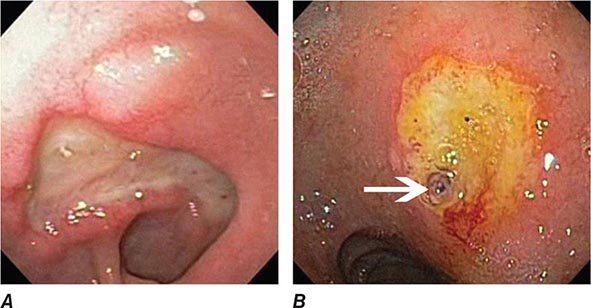
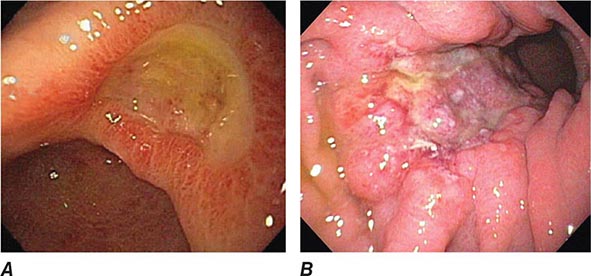
FIGURE 345-2 Gastric ulcers. A. Benign gastric ulcer. B. Malignant gastric ulcer involving greater curvature of stomach.
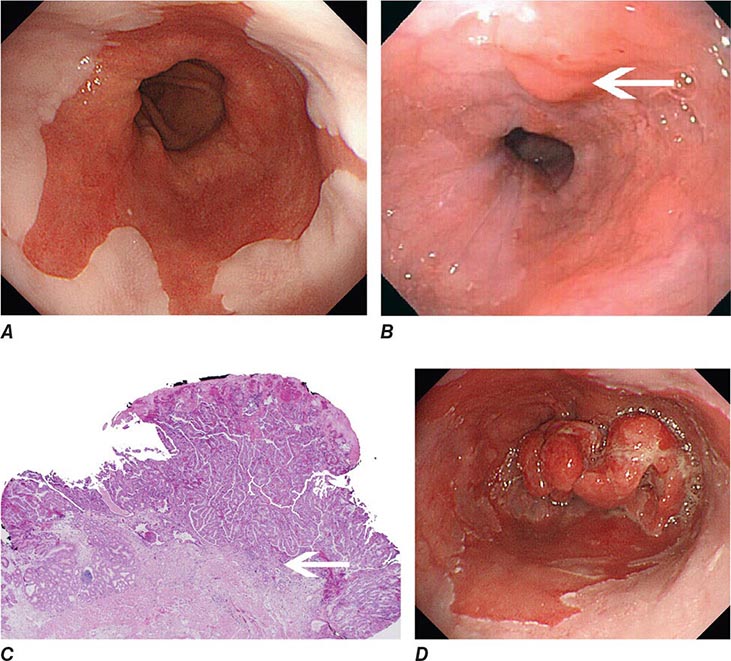
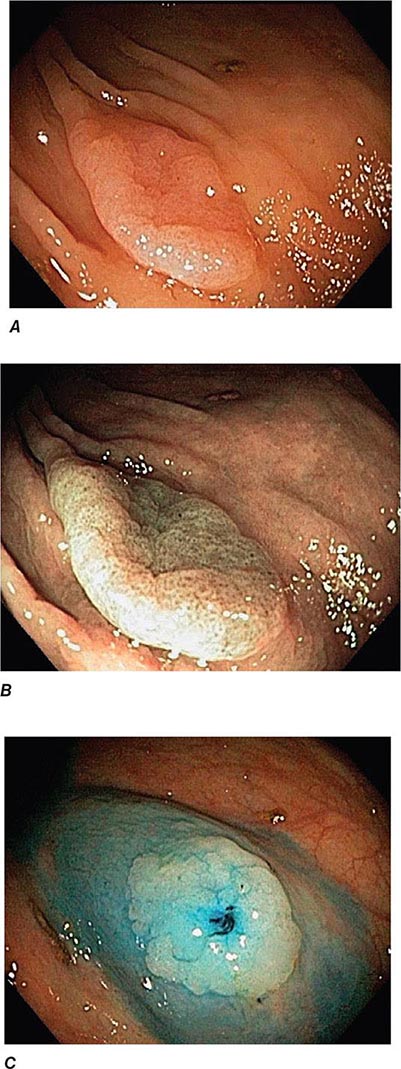
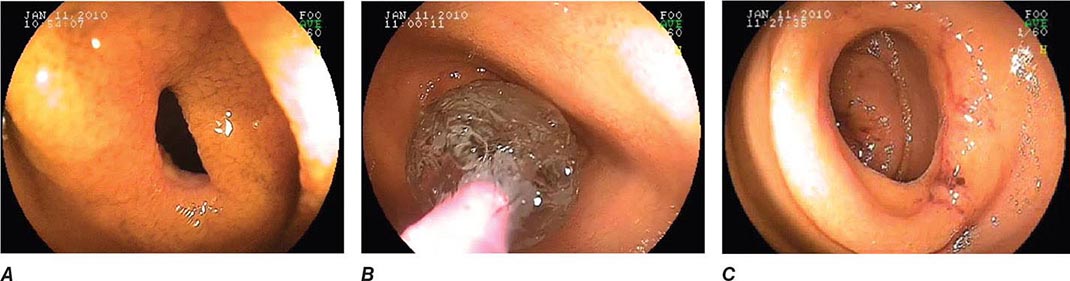
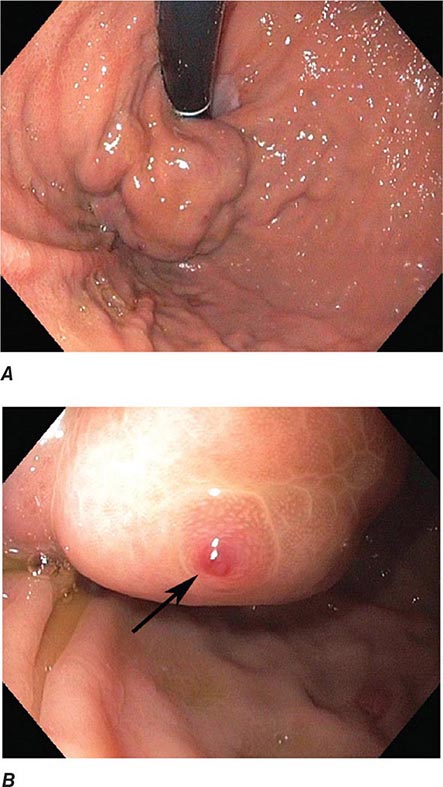
FIGURE 345-3 Barrett’s esophagus. A. Pink tongues of Barrett’s mucosa extending proximally from the gastroesophageal junction. B. Barrett’s esophagus with a suspicious nodule (arrow) identified during endoscopic surveillance. C. Histologic finding of intramucosal adenocarcinoma in the endoscopically resected nodule. Tumor extends into the esophageal submucosa (arrow). D. Barrett’s esophagus with locally advanced adenocarcinoma.
COLONOSCOPY
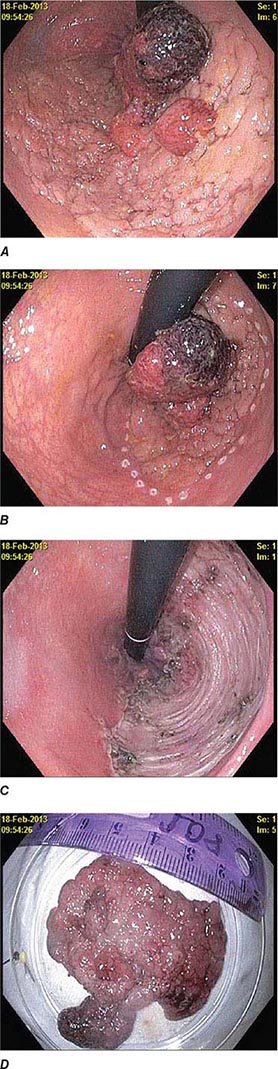
Colonoscopy is performed by passing a flexible colonoscope through the anal canal into the rectum and colon. The cecum is reached in >95% of cases, and the terminal ileum can often be examined. Colonoscopy is the gold standard for imaging the colonic mucosa. Colonoscopy has greater sensitivity than barium enema for colitis (Fig. 345-4), polyps (Fig. 345-51), and cancer (Fig. 345-6). Computed tomography (CT) colonography is an emerging technology that rivals the accuracy of colonoscopy for detection of some polyps and cancer, although it may not be sensitive for the detection of flat lesions, such as serrated polyps (Fig. 345-7). Conscious sedation is usually given before colonoscopy in the United States, although a willing patient and a skilled examiner can complete the procedure without sedation in many cases.
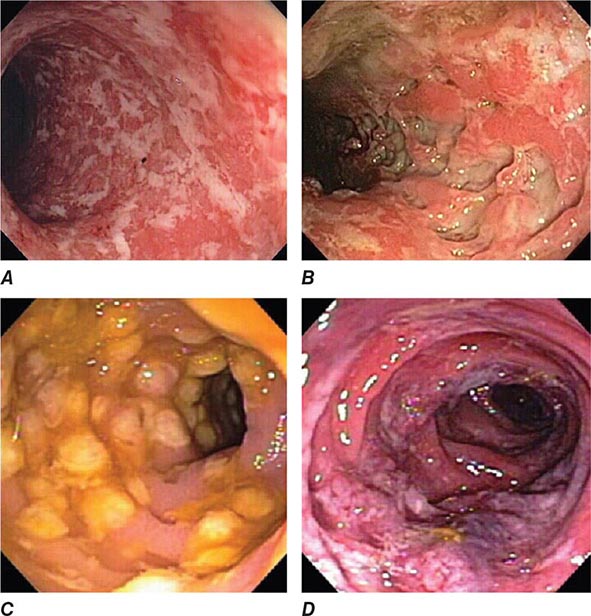
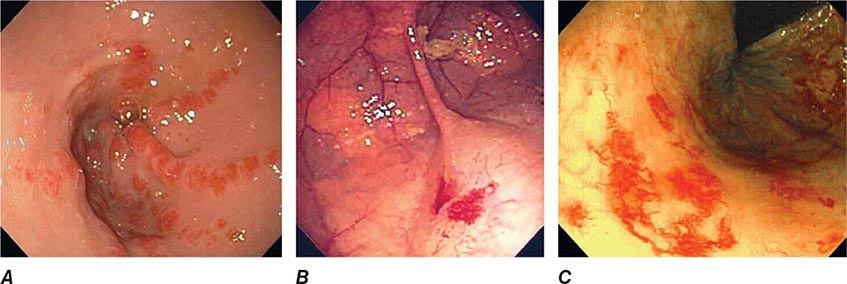
FIGURE 345-4 Causes of colitis. A. Chronic ulcerative colitis with diffuse ulcerations and exudates. B. Severe Crohn’s colitis with deep ulcers. C. Pseudomembranous colitis with yellow, adherent pseudomembranes. D. Ischemic colitis with patchy mucosal edema, subepithelial hemorrhage, and cyanosis.
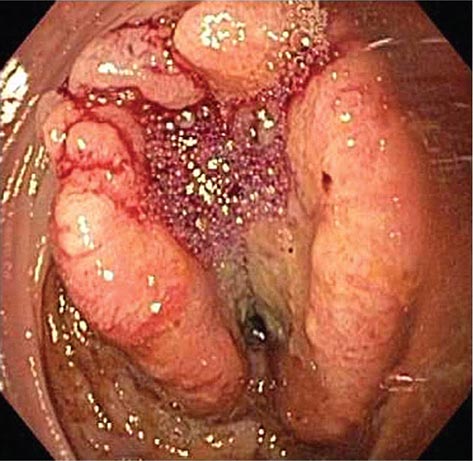
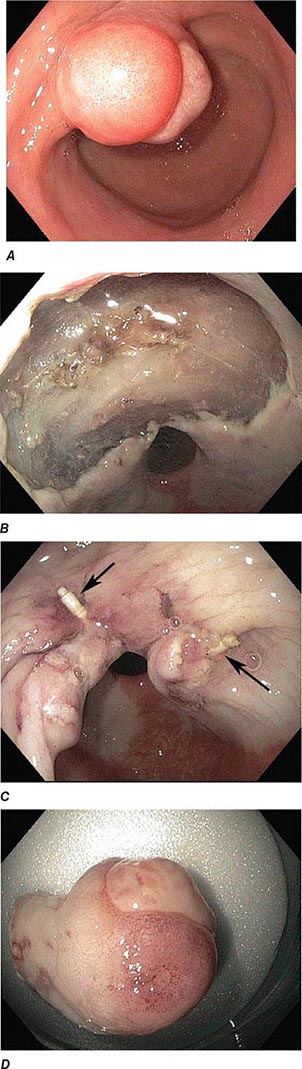
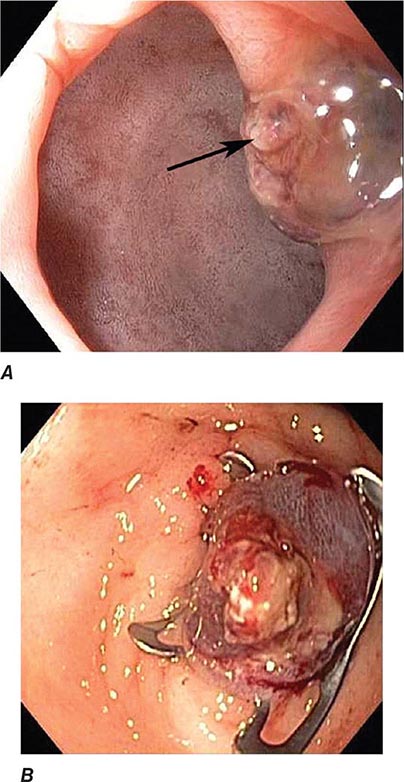
FIGURE 345-5 Colonic polyps. A. Pedunculated colon polyp on a thick stalk covered with normal mucosa (arrow). B. Sessile rectal polyp.
FIGURE 345-6 Colon adenocarcinoma growing into the lumen.
FIGURE 345-7 Flat serrated polyp in the cecum. A. Appearance of the lesion under conventional white-light imaging. B. Mucosal patterns and boundary of the lesion enhanced with narrow band imaging. C. Submucosal lifting of the lesion with dye (methylene blue) injection prior to resection.
FLEXIBLE SIGMOIDOSCOPY
Flexible sigmoidoscopy is similar to colonoscopy, but visualizes only the rectum and a variable portion of the left colon, typically to 60 cm from the anal verge. This procedure causes abdominal cramping, but it is brief and is usually performed without sedation. Flexible sigmoidoscopy is primarily used for evaluation of diarrhea and rectal outlet bleeding.
SMALL-BOWEL ENDOSCOPY
Three endoscopic techniques are currently used to evaluate the small intestine, most often in patients presenting with presumed small-bowel bleeding. For capsule endoscopy, the patient swallows a disposable capsule that contains a complementary metal oxide silicon (CMOS) chip camera. Color still images (Fig. 345-8) are transmitted wirelessly to an external receiver at several frames per second until the capsule’s battery is exhausted or it is passed into the toilet. Capsule endoscopy enables visualization of the small-bowel mucosa beyond the reach of a conventional endoscope and, at present, is solely a diagnostic procedure.
FIGURE 345-8 Capsule endoscopy image of jejunal vascular ectasia.
Push enteroscopy is performed with a long endoscope similar in design to an upper endoscope. The enteroscope is pushed down the small bowel, sometimes with the help of a stiffening overtube that extends from the mouth to the small intestine. The proximal to mid-jejunum is usually reached, and the instrument channel of the endoscope allows for biopsy or endoscopic therapy.
Deeper insertion into the small bowel can be accomplished by single– or double-balloon enteroscopy or spiral enteroscopy (Fig. 345-9). These instruments enable pleating of the small intestine onto an overtube (see Video 346e-1). With balloon-assisted enteroscopy, the entire intestinal tract can be visualized in some patients when both the oral and anal routes of insertion are used. Biopsies and endoscopic therapy can be performed throughout the visualized small bowel (Fig. 345-10).
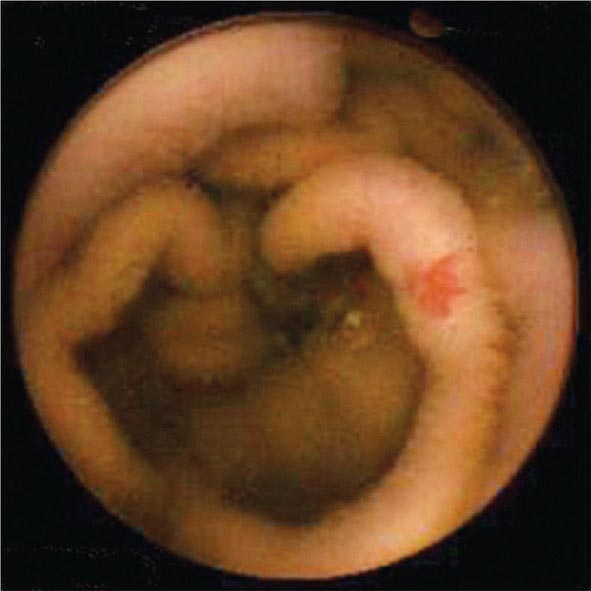
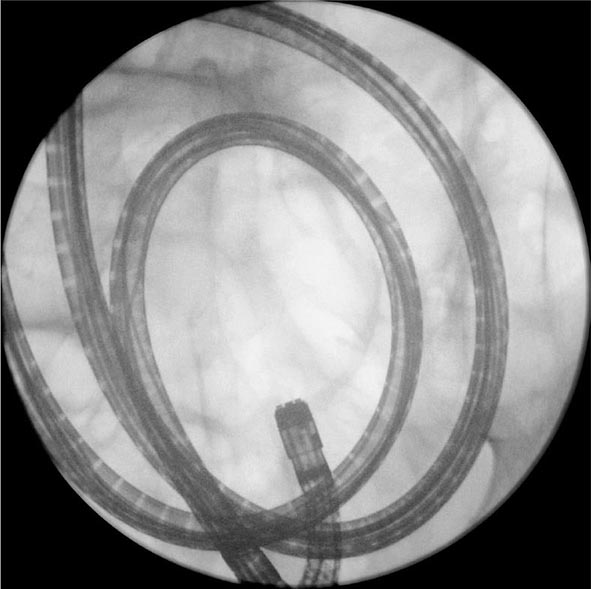
FIGURE 345-9 Radiograph of a double-balloon enteroscope in the small intestine.
FIGURE 345-10 Nonsteroidal anti-inflammatory drug (NSAID)–induced proximal ileal stricture diagnosed by double-balloon endoscopy. A. Ileal stricture causing obstructive symptoms. B. Balloon dilatation of the ileal stricture. C. Appearance of stricture after dilatation.
ENDOSCOPIC RETROGRADE CHOLANGIOPANCREATOGRAPHY (ERCP)
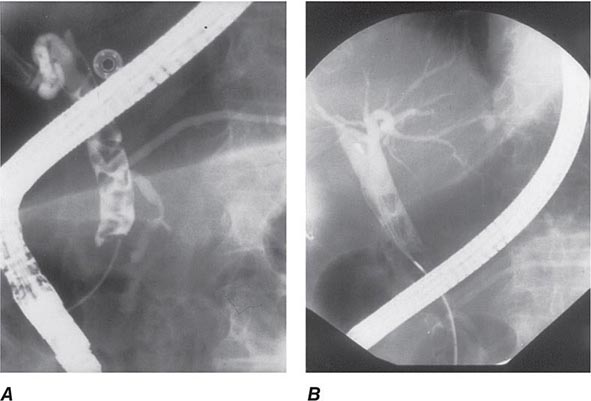
During ERCP a side-viewing endoscope is passed through the mouth to the duodenum, the ampulla of Vater is identified and cannulated with a thin plastic catheter, and radiographic contrast material is injected into the bile duct and pancreatic duct under fluoroscopic guidance (Fig. 345-11). When indicated, the sphincter of Oddi can be opened using the technique of endoscopic sphincterotomy (Fig. 345-12). Stones can be retrieved from the ducts (see Video 346e-15), biopsies can be performed, strictures can be dilated and/or stented (Fig. 345-13), and ductal leaks can be stented (Fig. 345-14). ERCP is often performed for therapy but remains important in diagnosis, especially for sphincter of Oddi dysfunction and for tissue sampling of ductal strictures.
FIGURE 345-11 Endoscopic retrograde cholangiopancreatography (ERCP) for bile duct stones with cholangitis. A. Faceted bile duct stones are demonstrated in the common bile duct. B. After endoscopic sphincterotomy, the stones are extracted with a Dormia basket. A small abscess communicates with the left hepatic duct.
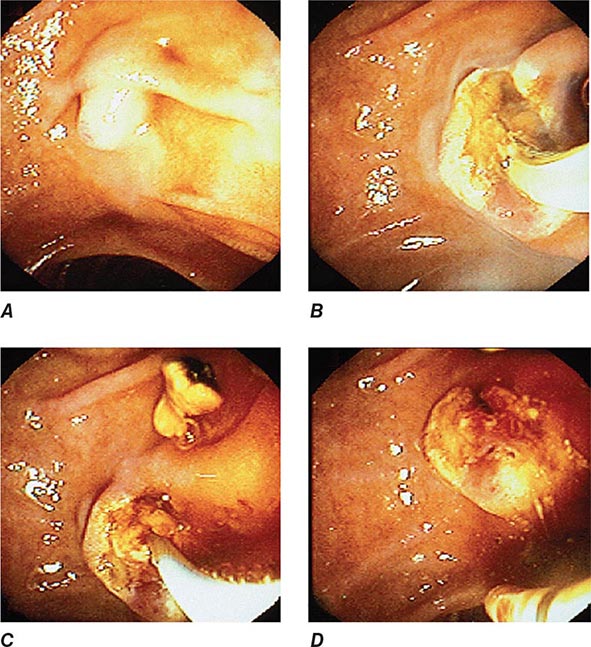
FIGURE 345-12 Endoscopic sphincterotomy. A. A normal-appearing ampulla of Vater. B. Sphincterotomy is performed with electrocautery. C. Bile duct stones are extracted with a balloon catheter. D. Final appearance of the sphincterotomy.
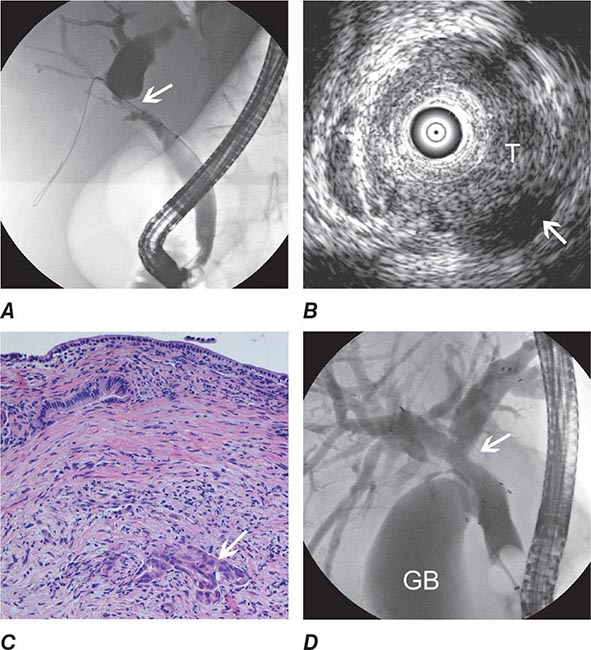
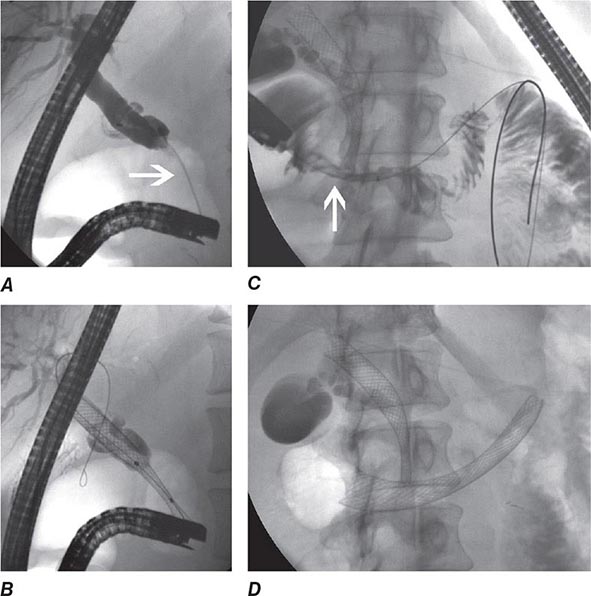
FIGURE 345-13 Endoscopic diagnosis, staging, and palliation of hilar cholangiocarcinoma. A. Endoscopic retrograde cholangiopancreatography (ERCP) in a patient with obstructive jaundice demonstrates a malignant-appearing stricture of the biliary confluence extending into the left and right intrahepatic ducts. B. Intraductal ultrasound of the biliary stricture demonstrates marked bile duct wall thickening due to tumor (T) with partial encasement of the hepatic artery (arrow). C. Intraductal biopsy obtained during ERCP demonstrates malignant cells infiltrating the submucosa of the bile duct wall (arrow). D. Endoscopic placement of bilateral self-expanding metal stents (arrow) relieves the biliary obstruction. GB, gallbladder. (Image C courtesy of Dr. Thomas Smyrk; with permission.)
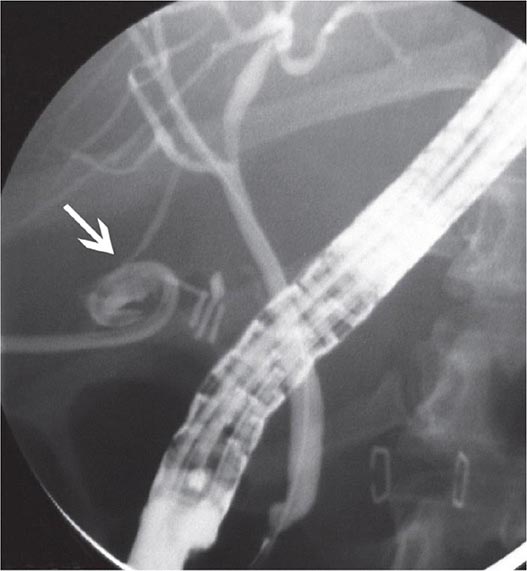
FIGURE 345-14 Bile leak (arrow) from a duct of Luschka after laparoscopic cholecystectomy. Contrast leaks from a small right intrahepatic duct into the gallbladder fossa and then flows into the pigtail of a percutaneous drainage catheter.
ENDOSCOPIC ULTRASOUND (EUS)
EUS utilizes high-frequency ultrasound transducers incorporated into the tip of a flexible endoscope. Ultrasound images are obtained of the gut wall and adjacent organs, vessels, and lymph nodes. By sacrificing depth of ultrasound penetration and bringing the ultrasound transducer close to the area of interest via endoscopy, high-resolution images are obtained. EUS provides the most accurate preoperative local staging of esophageal, pancreatic, and rectal malignancies (Fig. 345-15), although it does not detect most distant metastases. EUS is also useful for diagnosis of bile duct stones, gallbladder disease, submucosal gastrointestinal lesions, and chronic pancreatitis. Fine-needle aspirates and core biopsies of masses and lymph nodes in the posterior mediastinum, abdomen, pancreas, retroperitoneum, and pelvis can be obtained under EUS guidance (Fig. 345-16). EUS-guided therapeutic procedures are increasingly performed, including drainage of abscesses, pseudocysts, and pancreatic necrosis into the gut lumen (see Video 346e-2), celiac plexus neurolysis for treatment of pancreatic pain, ethanol ablation of pancreatic neuroendocrine tumors, treatment of gastrointestinal hemorrhage, and drainage of obstructed biliary and pancreatic ducts.
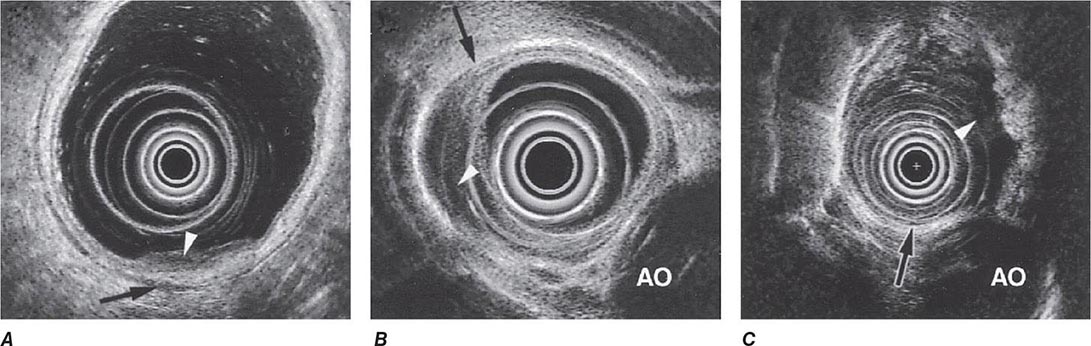
FIGURE 345-15 Local staging of gastrointestinal cancers with endoscopic ultrasound. In each example, the white arrowhead marks the primary tumor and the black arrow indicates the muscularis propria of the intestinal wall. A. T1 gastric cancer. The tumor does not invade the mp. B. T2 esophageal cancer. The tumor invades the muscularis propria. C. T3 esophageal cancer. The tumor extends through the muscularis propria into the surrounding tissue and focally abuts the aorta. AO, aorta.
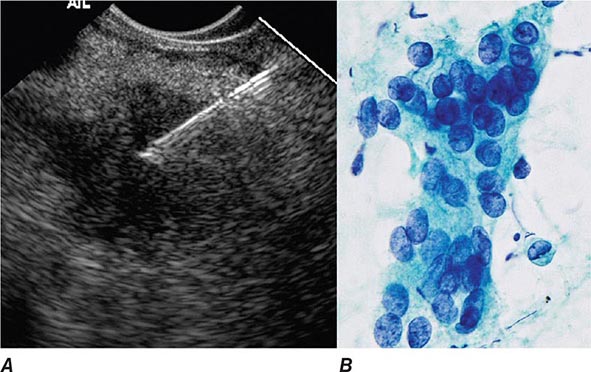
FIGURE 345-16 Endoscopic ultrasound (EUS)–guided fine-needle aspiration (FNA). A. Ultrasound image of a 22-gauge needle passed through the duodenal wall and positioned in a hypoechoic pancreatic head mass. B. Micrograph of aspirated malignant cells. (Image B courtesy of Dr. Michael R. Henry; with permission.)
NATURAL ORIFICE TRANSLUMINAL ENDOSCOPIC SURGERY (NOTES)
NOTES is an evolving collection of endoscopic methods that entail passage of an endoscope or its accessories into or through the wall of the gastrointestinal tract to perform diagnostic or therapeutic interventions. Some NOTES procedures, such as percutaneous endoscopic gastrostomy (PEG) or endoscopic necrosectomy of pancreatic necrosis, are well-established clinical procedures (see Video 346e-2); others, such as per-oral endoscopic myotomy (POEM) and endoscopic full-thickness resection of gastrointestinal mural lesions (Fig. 345-17, see Video 346e-3), are emerging as viable clinical therapeutic options; and still others, such as endoscopic appendectomy, cholecystectomy, and tubal ligation, are in development, and their ultimate clinical application is presently unclear. NOTES is currently an area of intense innovation and endoscopic research.
FIGURE 345-17 Endoscopic full-thickness resection of a gastrointestinal stromal tumor. A. Subepithelial lesion in the proximal stomach. B. Hypoechoic lesion arising from the fourth layer (muscularis propria) at endoscopic ultrasound. C. Full-thickness resection defect. D. Closure of defect using an over-the-scope clip.
ENDOSCOPIC RESECTION AND CLOSURE TECHNIQUES
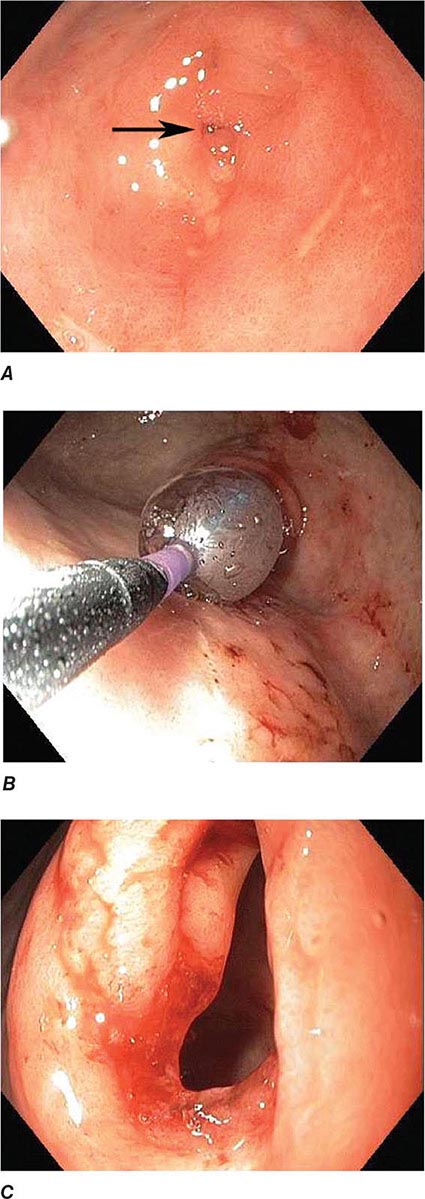
Endoscopic mucosal resection (EMR) (see Video 346e-4) and endoscopic submucosal dissection (ESD) (Fig. 345-18, see Video 346e-5) are two commonly used techniques for the resection of benign and early-stage malignant gastrointestinal neoplasms. In addition to providing larger specimens for more accurate histopathologic assessment and diagnosis, these techniques can be potentially curative for certain dysplastic lesions and focal intramucosal carcinomas involving the esophagus, stomach, and colon. Several devices are also available for closure of EMR and ESD defects, as well as gastrointestinal fistulas and perforations. Endoscopic clips deployed through the working channel of an endoscope have been used for many years to treat bleeding lesions, but the development of more robust over-the-scope clips has facilitated endoscopic closure of gastrointestinal fistulas and perforations not previously amenable to endoscopic therapy (see Video 346e-6). Endoscopic suturing is also feasible, and the technique can be used to close perforations and large defects (Fig. 345-19, see Video 346e-7), anastomotic leaks, and fistulas. Other potential indications for endoscopic suturing include stent fixation to prevent its migration (Fig. 345-20), and endoscopic bariatric procedures. These technologies are likely to have an expanding role in patient care.
FIGURE 345-18 Endoscopic submucosal dissection. A. Large flat distal rectal adenoma with central lobulation. B. Marking the periphery of the lesion with coagulation dots. C. Rectal defect following endoscopic submucosal dissection. D. Specimen resected en bloc.
FIGURE 345-19 Closure of large defect using an endoscopic suturing device. A. Ulcerated inflammatory fibroid polyp in the antrum. B. Large defect following endoscopic submucosal dissection of the lesion. C. Closure of the defect using endoscopic sutures (arrows). D. Resected specimen.
FIGURE 345-20 Prevention of stent migration using endoscopic sutures. A. Esophagogastric anastomotic stricture refractory to balloon dilation. B. Temporary placement of covered esophageal stent. C. Endoscopic suturing device to anchor stent to esophageal wall. D. Stent fixation with endoscopic sutures (arrows).
RISKS OF ENDOSCOPY
Medications used during conscious sedation may cause respiratory depression or allergic reactions. All endoscopic procedures carry some risk of bleeding and gastrointestinal perforation. The risk is small with diagnostic upper endoscopy and colonoscopy (<1:1000 procedures), but ranges from 0.5 to 5% when therapeutic procedures, such as EMR and ESD, control of hemorrhage, or stricture dilatation, are performed. Bleeding and perforation are rare adverse events with flexible sigmoidoscopy. The risk of adverse events for diagnostic EUS (without needle aspiration) is similar to that for diagnostic upper endoscopy.
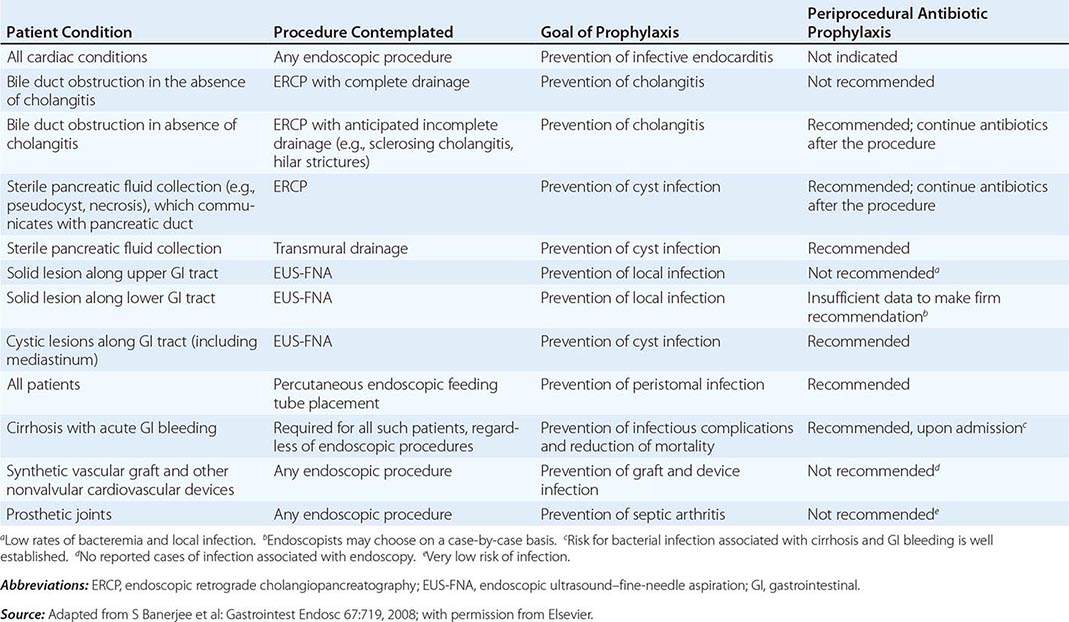
Infectious complications are uncommon with most endoscopic procedures. Some procedures carry a higher incidence of postprocedure bacteremia, and prophylactic antibiotics may be indicated (Table 345-1). Management of antithrombotic agents prior to endoscopic procedures should take into account the procedural risk of hemorrhage, the agent, and the patient condition, as summarized in Table 345-2.
ANTIBIOTIC PROPHYLAXIS FOR ENDOSCOPIC PROCEDURES |
MANAGEMENT OF ANTITHROMBOTIC DRUGS BEFORE ENDOSCOPIC PROCEDURES |
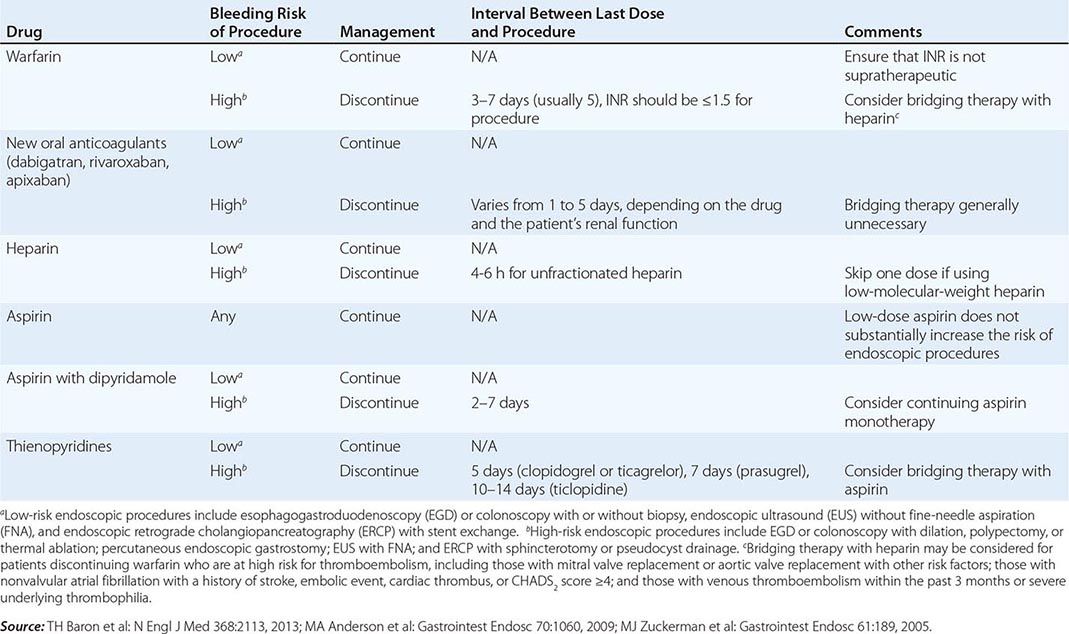
ERCP carries additional risks. Pancreatitis occurs in about 5% of patients undergoing the procedure and in up to 30% of patients with sphincter of Oddi dysfunction. Young anicteric patients with normal ducts are at increased risk. Post-ERCP pancreatitis is usually mild and self-limited, but may result in prolonged hospitalization, surgery, diabetes, or death when severe. Bleeding occurs in 1% of endoscopic sphincterotomies. Ascending cholangitis, pseudocyst infection, retroperitoneal perforation, and abscess formation may occur as a result of ERCP.
Percutaneous gastrostomy tube placement during EGD is associated with a 10–15% incidence of adverse events, most often wound infections. Fasciitis, pneumonia, bleeding, buried bumper syndrome, and colonic injury may result from gastrostomy tube placement.
URGENT ENDOSCOPY
ACUTE GASTROINTESTINAL HEMORRHAGE
Endoscopy is an important diagnostic and therapeutic technique for patients with acute gastrointestinal hemorrhage. Although gastrointestinal bleeding stops spontaneously in most cases, some patients will have persistent or recurrent hemorrhage that may be life-threatening. Clinical predictors of rebleeding help identify patients most likely to benefit from urgent endoscopy and endoscopic, angiographic, or surgical hemostasis.
Initial Evaluation The initial evaluation of the bleeding patient focuses on the severity of hemorrhage as reflected by the postural vital signs, the frequency of hematemesis or melena, and (in some cases) findings on nasogastric lavage. Decreases in hematocrit and hemoglobin lag behind the clinical course and are not reliable gauges of the magnitude of acute bleeding. This initial evaluation, completed well before the bleeding source is confidently identified, guides immediate supportive care of the patient, triage to the ward or intensive care unit, and timing of endoscopy. The severity of the initial hemorrhage is the most important indication for urgent endoscopy, since a large initial bleed increases the likelihood of ongoing or recurrent bleeding. Patients with resting hypotension or orthostatic change in vital signs, repeated hematemesis, or bloody nasogastric aspirate that does not clear with large-volume lavage, or those requiring blood transfusions, should be considered for urgent endoscopy. In addition, patients with cirrhosis, coagulopathy, or respiratory or renal failure and those over 70 years of age are more likely to have significant rebleeding.
Bedside evaluation also suggests an upper or lower gastrointestinal source of bleeding in most patients. Over 90% of patients with melena are bleeding proximal to the ligament of Treitz, and about 85% of patients with hematochezia are bleeding from the colon. Melena can result from bleeding in the small bowel or right colon, especially in older patients with slow colonic transit. Conversely, some patients with massive hematochezia may be bleeding from an upper gastrointestinal source, such as a gastric Dieulafoy lesion or duodenal ulcer, with rapid intestinal transit. Early upper endoscopy should be considered in such patients.
Endoscopy should be performed after the patient has been resuscitated with intravenous fluids and transfusions, as necessary. Marked coagulopathy or thrombocytopenia is usually treated before endoscopy, since correction of these abnormalities may lead to resolution of bleeding, and techniques for endoscopic hemostasis are limited in such patients. Metabolic derangements should also be addressed. Tracheal intubation for airway protection should be considered before upper endoscopy in patients with repeated recent hematemesis, encephalopathy, and suspected variceal hemorrhage.
Most patients with significant hematochezia can undergo colonoscopy after a rapid colonic purge with a polyethylene glycol solution; the preparation fluid may be administered via a nasogastric tube. Colonoscopy has a higher diagnostic yield than radionuclide bleeding scans or angiography in lower gastrointestinal bleeding, and endoscopic therapy can be applied in some cases. In a minority of cases, endoscopic assessment is hindered by poor visualization due to persistent vigorous bleeding with recurrent hemodynamic instability, and other techniques (such as angiography or emergent subtotal colectomy) must be employed. In such patients, massive bleeding originating from an upper gastrointestinal source should also be considered and excluded by upper endoscopy. The anal and rectal mucosa should be visualized endoscopically early in the course of massive rectal bleeding, because bleeding lesions in or close to the anal canal may be identified that are amenable to endoscopic or surgical transanal hemostatic techniques.
Peptic Ulcer The endoscopic appearance of peptic ulcers provides useful prognostic information and guides the need for endoscopic therapy in patients with acute hemorrhage (Fig. 345-21). A clean-based ulcer is associated with a low risk (3–5%) of rebleeding; patients with melena and a clean-based ulcer are often discharged home from the emergency room or endoscopy suite if they are young, reliable, and otherwise healthy. Flat pigmented spots and adherent clots covering the ulcer base have a 10% and 20% risk of rebleeding, respectively. Endoscopic therapy is often considered for an ulcer with an adherent clot. When a fibrin plug is seen protruding from a vessel wall in the base of an ulcer (so-called sentinel clot or visible vessel), the risk of rebleeding from the ulcer is 40%. This finding generally leads to endoscopic therapy to decrease the rebleeding rate. Occasionally, active spurting from an ulcer is seen, with >90% risk of ongoing bleeding without therapy.
FIGURE 345-21 Stigmata of hemorrhage in peptic ulcers. A. Gastric antral ulcer with a clean base. B. Duodenal ulcer with flat pigmented spots (arrows). C. Duodenal ulcer with a dense adherent clot. D. Gastric ulcer with a pigmented protuberance/visible vessel. E. Duodenal ulcer with active spurting (arrow).
Endoscopic therapy of ulcers with high-risk stigmata typically lowers the rebleeding rate to 5–10%. Several hemostatic techniques are available, including injection of epinephrine or a sclerosant into and around the vessel, “coaptive coagulation” of the vessel in the base of the ulcer using a thermal probe that is pressed against the site of bleeding, placement of hemoclips (Fig. 345-22), or a combination of these modalities (see Video 346e-8). In conjunction with endoscopic therapy, the administration of a proton pump inhibitor decreases the risk of rebleeding and improves patient outcome.
FIGURE 345-22 Endoscopic hemostasis of ulcer bleeding. A. Pyloric channel ulcer with visible vessel (arrow). B. Ulcer hemostasis with placement of an over-the-scope clip.
Varices Two complementary strategies guide therapy of bleeding varices: local treatment of the bleeding varices and treatment of the underlying portal hypertension. Local therapies, including endoscopic variceal band ligation, endoscopic variceal sclerotherapy, and balloon tamponade with a Sengstaken-Blakemore tube, effectively control acute hemorrhage in most patients, although therapies that decrease portal pressure (pharmacologic treatment, surgical shunts, or radiologically placed intrahepatic portosystemic shunts) also play an important role.
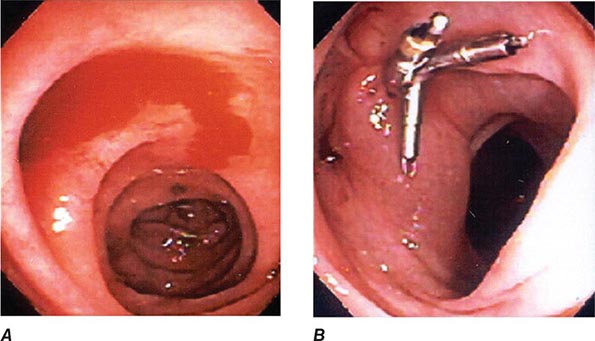
Endoscopic variceal ligation (EVL) is indicated for the prevention of a first bleed (primary prophylaxis) from large esophageal varices (Figs. 345-23 and 24), particularly in patients in whom beta blockers are contraindicated or not tolerated. EVL is also the preferred endoscopic therapy for control of active esophageal variceal bleeding and for subsequent eradication of esophageal varices (secondary prophylaxis). During EVL, a varix is suctioned into a cap fitted on the end of the endoscope, and a rubber band is released from the cap, ligating the varix (Fig. 345-24, see Video 346e-9). EVL controls acute hemorrhage in up to 90% of patients. Complications of EVL, such as postbanding ulcer bleeding and esophageal stenosis, are uncommon. Endoscopic variceal sclerotherapy (EVS) involves the injection of a sclerosing, thrombogenic solution into or next to esophageal varices. EVS also controls acute hemorrhage in most patients, but it is generally used as salvage therapy when band ligation fails because of its higher complication rate compared to EVL. These techniques are used when varices are actively bleeding during endoscopy or (more commonly) when varices are the only identifiable cause of acute hemorrhage. Bleeding from large gastric fundic varices (Fig. 345-25) is best treated with endoscopic cyanoacrylate (“glue”) injection (see Video 346e-10), because EVL or EVS of these varices is associated with a high rebleeding rate. Complications of cyanoacrylate injection include infection and glue embolization to other organs, such as the lungs, brain, and spleen.
FIGURE 345-23 Esophageal varices.
FIGURE 345-24 Endoscopic band ligation of esophageal varices. A. Large esophageal varices with stigmata of recent bleeding. B. Band ligation of varices.
FIGURE 345-25 Gastric varices. A. Large gastric fundal varices. B. Stigmata of recent bleeding from the same gastric varices (arrow).
After treatment of the acute hemorrhage, an elective course of endoscopic therapy can be undertaken with the goal of eradicating esophageal varices and preventing rebleeding months to years later. However, this chronic therapy is less successful, preventing long-term rebleeding in ~50% of patients. Pharmacologic therapies that decrease portal pressure have similar efficacy, and the two modalities may be combined.
Dieulafoy’s Lesion This lesion, also called persistent caliber artery, is a large-caliber arteriole that runs immediately beneath the gastrointestinal mucosa and bleeds through a pinpoint mucosal erosion (Fig. 345-26). Dieulafoy’s lesion is seen most commonly on the lesser curvature of the proximal stomach, causes impressive arterial hemorrhage, and may be difficult to diagnose; it is often recognized only after repeated endoscopy for recurrent bleeding. Endoscopic therapy, such as thermal coagulation or band ligation, is typically effective for control of bleeding and ablation of the underlying vessel once the lesion has been identified (see Video 346e-11). Rescue therapies, such as angiographic embolization or surgical oversewing, are considered in situations where endoscopic therapy has failed.
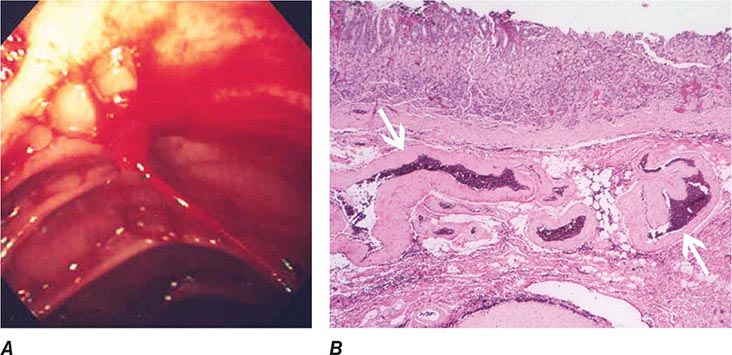
FIGURE 345-26 Dieulafoy’s lesion. A. Actively spurting jejunal Dieulafoy’s lesion. There is no underlying mucosal lesion. B. Histology of a gastric Dieulafoy’s lesion. A persistent caliber artery (arrows) is present in the gastric submucosa, immediately beneath the mucosa.
Mallory-Weiss Tear A Mallory-Weiss tear is a linear mucosal rent near or across the gastroesophageal junction that is often associated with retching or vomiting (Fig. 345-27). When the tear disrupts a submucosal arteriole, brisk hemorrhage may result. Endoscopy is the best method of diagnosis, and an actively bleeding tear can be treated endoscopically with epinephrine injection, coaptive coagulation, band ligation, or hemoclips (see Video 346e-12). Unlike peptic ulcer, a Mallory-Weiss tear with a nonbleeding sentinel clot in its base rarely rebleeds and thus does not necessitate endoscopic therapy.
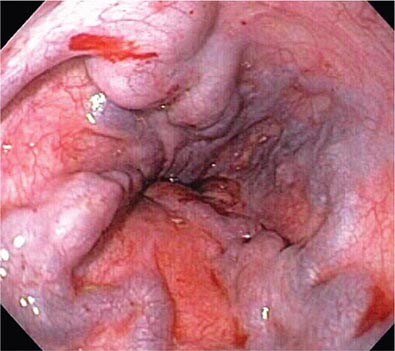
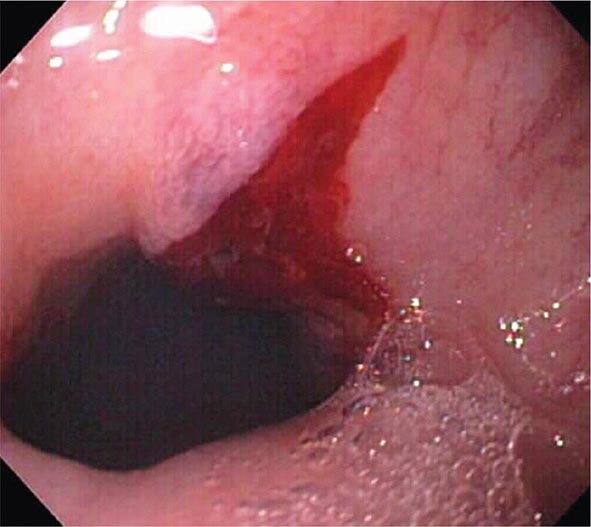
FIGURE 345-27 Mallory-Weiss tear at the gastroesophageal junction.
Vascular Ectasias Vascular ectasias are flat mucosal vascular anomalies that are best diagnosed by endoscopy. They usually cause slow intestinal blood loss and occur either in a sporadic fashion or in a well-defined pattern of distribution (e.g., gastric antral vascular ectasia [GAVE] or “watermelon stomach”) (Fig. 345-28). Cecal vascular ectasias, GAVE, and radiation-induced rectal ectasias are often responsive to local endoscopic ablative therapy, such as argon plasma coagulation (see Video 346e-13). Patients with diffuse small-bowel vascular ectasias (associated with chronic renal failure and with hereditary hemorrhagic telangiectasia) may continue to bleed despite endoscopic treatment of easily accessible lesions by conventional endoscopy. These patients may benefit from deep enteroscopy with endoscopic therapy, pharmacologic treatment with octreotide or estrogen/progesterone therapy, or intraoperative enteroscopy.
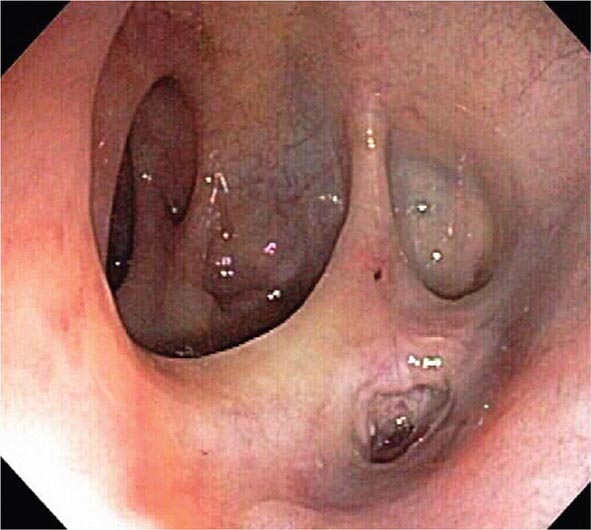
FIGURE 345-28 Gastrointestinal vascular ectasias. A. Gastric antral vascular ectasia (“watermelon stomach”) characterized by stripes of prominent flat or raised vascular ectasias. B. Cecal vascular ectasias. C. Radiation-induced vascular ectasias of the rectum in a patient previously treated for prostate cancer.
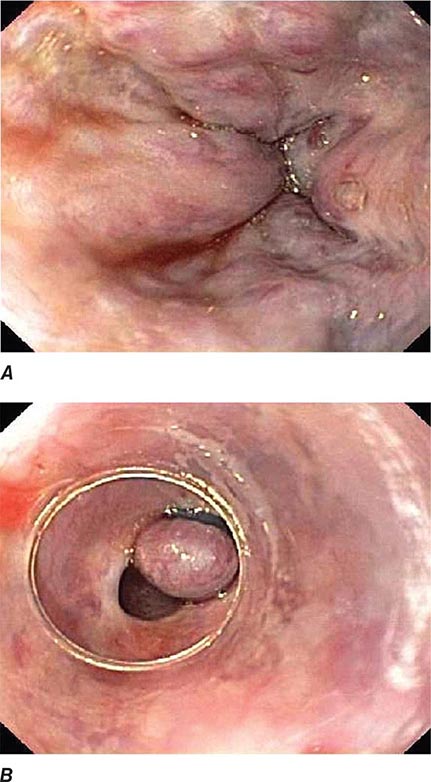
Colonic Diverticula Diverticula form where nutrient arteries penetrate the muscular wall of the colon en route to the colonic mucosa (Fig. 345-29). The artery found in the base of a diverticulum may bleed, causing painless and impressive hematochezia. Colonoscopy is indicated in patients with hematochezia and suspected diverticular hemorrhage, because other causes of bleeding (such as vascular ectasias, colitis, and colon cancer) must be excluded. In addition, an actively bleeding diverticulum may be seen and treated during colonoscopy (Fig. 345-30, see Video 346e-14).
FIGURE 345-29 Colonic diverticula.
FIGURE 345-30 Diverticular hemorrhage. A. Actively bleeding sigmoid diverticulum. B. Hemostasis achieved using endoscopic clips.
GASTROINTESTINAL OBSTRUCTION AND PSEUDOOBSTRUCTION
Endoscopy is useful for evaluation and treatment of some forms of gastrointestinal obstruction. An important exception is small-bowel obstruction due to surgical adhesions, which is generally not diagnosed or treated endoscopically. Esophageal, gastroduodenal, and colonic obstruction or pseudoobstruction can all be diagnosed and often managed endoscopically.
Acute Esophageal Obstruction
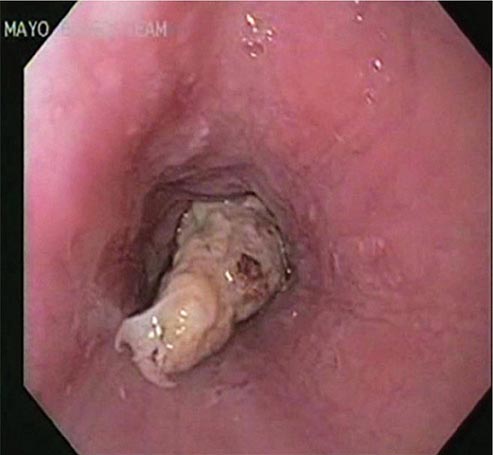
Esophageal obstruction by impacted food (Fig. 345-31) or an ingested foreign body is a potentially life-threatening event and represents an endoscopic emergency. Left untreated, the patient may develop esophageal ulceration, ischemia, and perforation. Patients with persistent esophageal obstruction often have hypersalivation and are usually unable to swallow water; endoscopy is generally the best initial test in such patients, because endoscopic removal of the obstructing material is usually possible, and the presence of an underlying esophageal pathology can often be determined. Radiographs of the chest and neck should be considered before endoscopy in patients with fever, obstruction for ≥24 h, or ingestion of a sharp object, such as a fishbone. Radiographic contrast studies interfere with subsequent endoscopy and are not advisable in most patients with a clinical picture of esophageal obstruction. Sips of a carbonated beverage, sublingual nifedipine or nitrates, or intravenous glucagon may resolve an esophageal food impaction, but in most patients, an underlying web, ring, or stricture is present and endoscopic removal of the obstructing food bolus is necessary.
FIGURE 345-31 Esophageal food (meat) impaction.
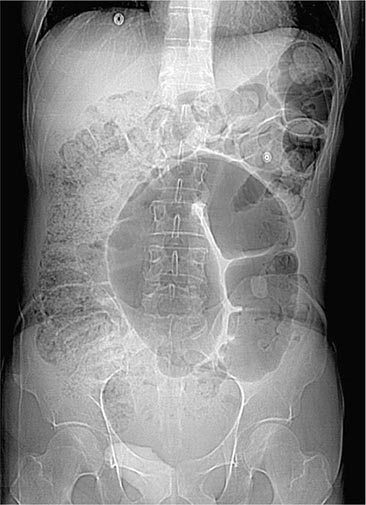
Gastric Outlet Obstruction Obstruction of the gastric outlet is commonly caused by gastric, duodenal, or pancreatic malignancy or chronic peptic ulceration with stenosis of the pylorus (Fig. 345-32). Patients vomit partially digested food many hours after eating. Gastric decompression with a nasogastric tube and subsequent lavage for removal of retained material is the first step in treatment. The diagnosis can then be confirmed with a saline load test, if desired. Endoscopy is useful for diagnosis and treatment. Patients with benign pyloric stenosis may be treated with endoscopic balloon dilatation of the pylorus, and a course of endoscopic dilatation results in long-term relief of symptoms in about 50% of patients. Malignant gastric outlet obstruction can be relieved with endoscopically placed expandable stents in patients with inoperable malignancy (Fig. 345-33).
FIGURE 345-32 Gastric outlet obstruction due to pyloric stenosis. A. Sequela of nonsteroidal anti-inflammatory drug (NSAID)–induced ulcer disease with severe stenosis of the pylorus (arrow). B. Balloon dilation of the stenosis. C. Appearance of pyloric ring after dilation.
FIGURE 345-33 Biliary and duodenal self-expanding metal stents (SEMS) for obstruction caused by pancreatic cancer. A. Endoscopic retrograde cholangiopancreatography (ERCP) demonstrates a distal bile duct stricture (arrow). B. A biliary SEMS is placed. C. Contrast injection demonstrates a duodenal stricture (arrow). D. Biliary and duodenal SEMS in place.
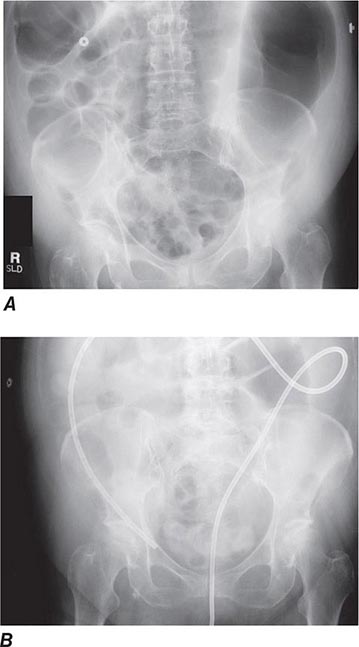
Colonic Obstruction and Pseudoobstruction These both present with abdominal distention and discomfort; tympany; and a dilated, air-filled colon on plain abdominal radiography. The radiographic appearance can be characteristic of a particular condition, such as sigmoid volvulus (Fig. 345-34). Both structural obstruction and pseudoobstruction may lead to colonic perforation if left untreated. Acute colonic pseudoobstruction is a form of colonic ileus that is usually attributable to electrolyte disorders, narcotic and anticholinergic medications, immobility (as after surgery), and retroperitoneal hemorrhage or mass. Multiple causative factors are often present. Colonoscopy, water-soluble contrast enema, or CT may be used to assess for an obstructing lesion and differentiate obstruction from pseudoobstruction. One of these diagnostic studies should be strongly considered if the patient does not have clear risk factors for pseudoobstruction, if radiographs do not show air in the rectum, or if the patient fails to improve when underlying causes of pseudoobstruction have been addressed. The risk of cecal perforation in pseudoobstruction rises when the cecal diameter exceeds 12 cm, and decompression of the colon may be achieved using intravenous neostigmine or via colonoscopic decompression (Fig. 345-35). Most patients should receive a trial of conservative therapy (with correction of electrolyte disorders, removal of offending medications, and increased mobilization) before undergoing an invasive decompressive procedure for colonic pseudoobstruction.
FIGURE 345-34 Sigmoid volvulus with the characteristic radiologic appearance of a “bent inner tube.”
FIGURE 345-35 Acute colonic pseudoobstruction. A. Acute colonic dilatation occurring in a patient soon after knee surgery. B. Colonoscopic placement of decompression tube with marked improvement in colonic dilatation.
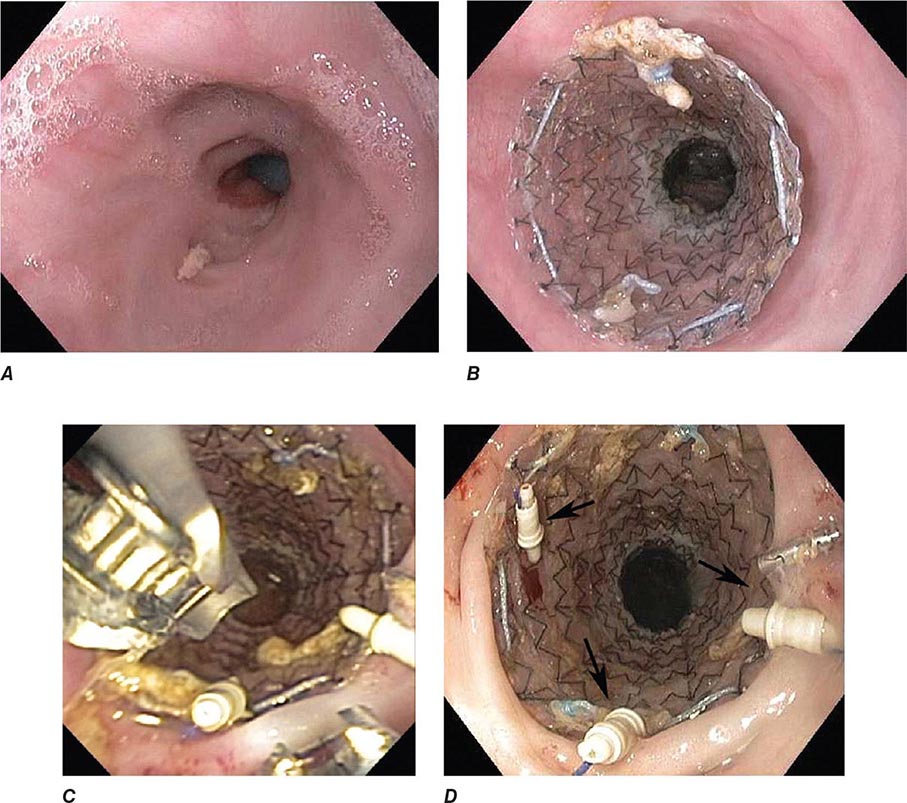
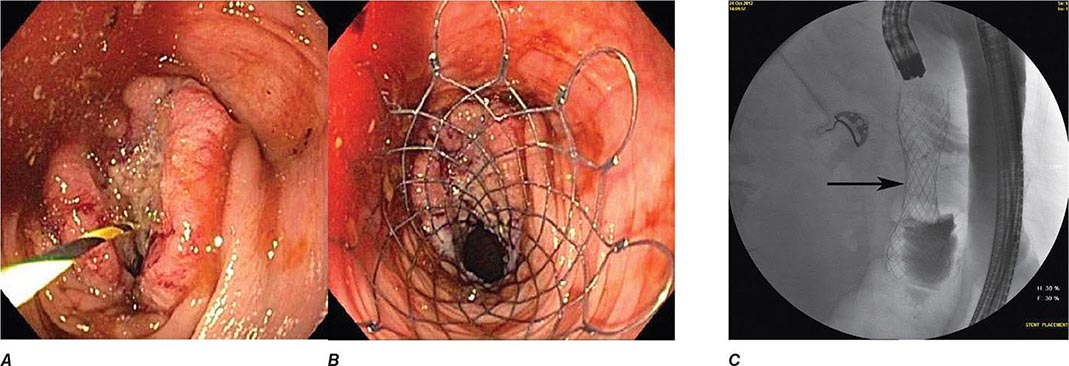
Colonic obstruction is an indication for urgent intervention. In the past, emergent diverting colostomy was usually performed with a subsequent second operation after bowel preparation to treat the underlying cause of obstruction. Colonoscopic placement of an expandable stent is now a widely used alternative that can relieve malignant colonic obstruction without emergency surgery and permit bowel preparation for an elective one-stage operation (Fig. 345-36, see Video 346e-15).
FIGURE 345-36 Obstructing colonic carcinoma. A. Colonic adenocarcinoma causing marked luminal narrowing of the distal transverse colon. B. Endoscopic placement of a self-expandable metal stent. C. Radiograph of expanded stent across the obstructing tumor with a residual waist (arrow).
ACUTE BILIARY OBSTRUCTION
The steady, severe pain that occurs when a gallstone acutely obstructs the common bile duct often brings patients to a hospital. The diagnosis of a ductal stone is suspected when the patient is jaundiced or when serum liver tests or pancreatic enzyme levels are elevated; it is confirmed by EUS, magnetic resonance cholangiography (MRCP), or direct cholangiography (performed endoscopically, percutaneously, or during surgery). ERCP is currently the primary means of diagnosing and treating common bile duct stones in most hospitals in the United States (Figs. 345-11 and 345-12).
Bile Duct Imaging Whereas transabdominal ultrasound diagnoses only a minority of bile duct stones, MRCP and EUS are >90% accurate and have an important role in diagnosis. Examples of these modalities are shown in Fig. 345-37.
FIGURE 345-37 Methods of bile duct imaging. Arrows mark bile duct stones. Arrowheads indicate the common bile duct, and the asterisk marks the portal vein. A. Endoscopic ultrasound (EUS). B. Magnetic resonance cholangiopancreatography (MRCP). C. Helical computed tomography (CT).
If the suspicion for a bile duct stone is high and urgent treatment is required (as in a patient with obstructive jaundice and biliary sepsis), ERCP is the procedure of choice, because it remains the gold standard for diagnosis and allows for immediate treatment (see Video 346e-16). If a persistent bile duct stone is relatively unlikely (as in a patient with gallstone pancreatitis), ERCP may be supplanted by less invasive imaging techniques, such as EUS, MRCP, or intraoperative cholangiography performed during cholecystectomy, sparing patients the risk and discomfort of ERCP.
Ascending Cholangitis Charcot’s triad of jaundice, abdominal pain, and fever is present in about 70% of patients with ascending cholangitis and biliary sepsis. These patients are managed initially with fluid resuscitation and intravenous antibiotics. Abdominal ultrasound is often performed to assess for gallbladder stones and bile duct dilation. However, the bile duct may not be dilated early in the course of acute biliary obstruction. Medical management usually improves the patient’s clinical status, providing a window of approximately 24 h during which biliary drainage should be established, typically by ERCP. Undue delay can result in recrudescence of overt sepsis and increased morbidity and mortality rates. In addition to Charcot’s triad, the additional presence of shock and confusion (Reynolds’s pentad) is associated with high mortality rate and should prompt urgent intervention to restore biliary drainage.
Gallstone Pancreatitis Gallstones may cause acute pancreatitis as they pass through the ampulla of Vater. The occurrence of gallstone pancreatitis usually implies passage of a stone into the duodenum, and only about 20% of patients harbor a persistent stone in the ampulla or the common bile duct. Retained stones are more common in patients with jaundice, rising serum liver tests following hospitalization, severe pancreatitis, or superimposed ascending cholangitis.
Urgent ERCP decreases the morbidity rate of gallstone pancreatitis in a subset of patients with retained bile duct stones. It is unclear whether the benefit of ERCP is mainly attributable to treatment and prevention of ascending cholangitis or to relief of pancreatic ductal obstruction. ERCP is warranted early in the course of gallstone pancreatitis if ascending cholangitis is suspected, especially in a jaundiced patient. Urgent ERCP may also benefit patients predicted to have severe pancreatitis using a clinical index of severity, such as the Glasgow or Ranson score. Because the benefit of ERCP is limited to patients with a retained bile duct stone, a strategy of initial MRCP or EUS for diagnosis decreases the utilization of ERCP in gallstone pancreatitis and improves clinical outcomes by limiting the occurrence of ERCP-related adverse events.
ELECTIVE ENDOSCOPY
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree