Tumors of the Ovary and Fallopian Tube
THE OVARY
HISTOLOGIC RECALL
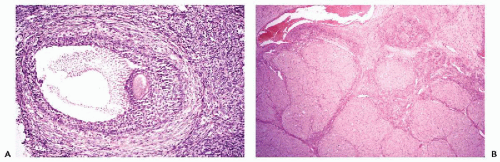
The anatomy of the ovaries was discussed in Chapter 8. Because components of the normal ovary may be observed in cytologic preparations, a brief summary of the histology is provided (Fig. 15-1).
The central portion of the ovaries is formed by the hilum, the site of entry of the vascular supply and lymphatic drainage. The hilum also contains clusters of large endocrine cells with eosinophilic, granular cytoplasm, similar to Leydig cells of the testis, that may contain rod-like Reinke’s crystalloids. The ovary is surrounded by a surface or germinative epithelium. The bulk of the ovary is formed by ovarian stroma.
The surface epithelium, which is closely related to the mesothelium, is composed of a single layer of cuboidal cells with scanty basophilic cytoplasm and spherical nuclei. The surface epithelium often forms invaginations into the cortex of the ovary or small cysts. It should be noted that cortical cysts may be mistaken for ovarian follicles on ultrasound examination and may be incidentally aspirated during the harvest of ova for in vitro fertilization.
The ovarian stroma is composed of small spindly cells, some of which are capable of endocrine function. The superficial part of the ovarian stroma, the cortex, contains ova in various stages of maturation. The ova, numerous at birth, are reduced in number in the mature ovary and reside in the cortical stroma, where each ovum is surrounded by a single layer of epithelial cells, forming a primitive follicle. The maturation of the ova begins at puberty. Under the impact of pituitary follicle-stimulating hormone (FSH), a few select follicles begin to enlarge. It is not known how and why the selection is taking place. The epithelial cells surrounding the ovum begin to multiply, become larger and multilayered, and are named granulosa cells. The ovum is separated from the granulosa cells by a homogeneous membrane, known as the zona pellucida. As the maturation of the ovum progresses, the number of cell layers of the granulosa increase. At the same time stromal cells surrounding the ovum become larger and, named theca cells, form a multilayered envelope around the follicle. The granulosa and theca cells secrete estrogens that induce the proliferative phase in the endometrium (see Chap. 13). As the follicle matures and enlarges, the granulosa cells form a cavity filled with a hormone-rich fluid. The ovum, still surrounded by granulosa cells, now protrudes into the follicular cavity;
the protrusion is named cumulus oophorus (Fig. 15-2A). At this point, the follicle is named after the Dutch anatomist who first described it in the 17th century, a follicle of De Graaf or Graafian follicle. The graafian follicles are now visible on the surface of the ovary as small protrusions, but normally only one of them will spontaneously rupture and discharge the mature ovum together with the follicular fluid into the peritoneal cavity, followed by bleeding into the cavity of the follicle. Again, it is not known how and why the single follicle is selected. The ovulation takes place under the impact of pituitary luteinizing hormone (LH), that also causes enlargement of the granulosa cells that converts the collapsed follicle into a large, grossly visible yellow structure, the corpus luteum, that secretes progesterone, thus inducing the secretory phase of the endometrium (Fig. 15-2B). The yellow color of the corpus luteum is due to a high lipid content of the hormone-producing component cells. The discharged ovum is captured by the fimbria of the fallopian tube, pending fertilization by a spermatozoon in the lumen of the tube. Unless pregnancy intervenes, the corpus luteum undergoes atrophy and fibrosis, resulting in a small white scar [corpus albicans or (plural) corpora albicantia] within the cortex of the ovary. If pregnancy occurs, the corpus luteum persists, becomes larger, and is known as corpus luteum of pregnancy.
the protrusion is named cumulus oophorus (Fig. 15-2A). At this point, the follicle is named after the Dutch anatomist who first described it in the 17th century, a follicle of De Graaf or Graafian follicle. The graafian follicles are now visible on the surface of the ovary as small protrusions, but normally only one of them will spontaneously rupture and discharge the mature ovum together with the follicular fluid into the peritoneal cavity, followed by bleeding into the cavity of the follicle. Again, it is not known how and why the single follicle is selected. The ovulation takes place under the impact of pituitary luteinizing hormone (LH), that also causes enlargement of the granulosa cells that converts the collapsed follicle into a large, grossly visible yellow structure, the corpus luteum, that secretes progesterone, thus inducing the secretory phase of the endometrium (Fig. 15-2B). The yellow color of the corpus luteum is due to a high lipid content of the hormone-producing component cells. The discharged ovum is captured by the fimbria of the fallopian tube, pending fertilization by a spermatozoon in the lumen of the tube. Unless pregnancy intervenes, the corpus luteum undergoes atrophy and fibrosis, resulting in a small white scar [corpus albicans or (plural) corpora albicantia] within the cortex of the ovary. If pregnancy occurs, the corpus luteum persists, becomes larger, and is known as corpus luteum of pregnancy.
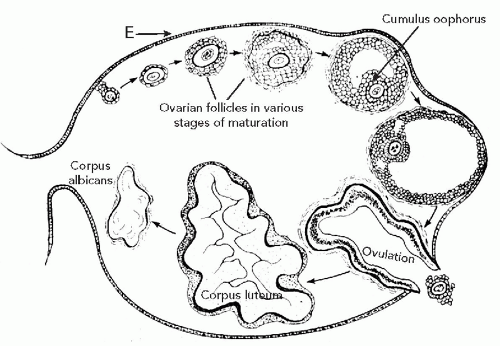 Figure 15-1 Schematic representation of events in ovulation (indicated in arrows) from a primitive follicle to formation of corpus albicans (see text). E, epithelium. |
METHODS OF INVESTIGATION
Cervicovaginal Preparations
In the study of the ovary, cervicovaginal preparations may serve two purposes:
They may contribute to the diagnosis of ovarian tumors that shed recognizable cancer cells.
They allow an assessment of the hormonal status of women, bearers of estrogen-producing tumors. This method occasionally contributes to the recognition of primary or recurrent tumors, particularly of granulosa cell tumors.
Endometrial Aspirations
Occasionally ovarian tumors may be recognized in material aspirated from the endometrium. The techniques were described in Chapter 13.
Transvaginal Aspiration for In Vitro Fertilization
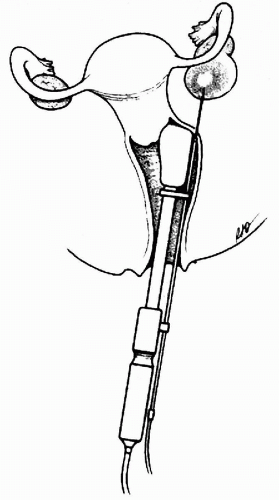
In vitro fertilization requires harvesting ova that are exposed to spermatozoa in vitro and then re-implanted into the suitably primed uterus. The ovary is stimulated by hormonal treatment to achieve maturation of several ova at the same time. The viable ova are harvested by ultrasound-guided transvaginal needle aspirates of maturing Graafian follicles (Fig. 15-3). Cytologic examination of the aspirated material is not warranted unless the aspirated fluid is discolored or the amount is larger than the normal 2 to 3 ml (Greenebaum et al, 1992; Yee et al, 1994). When this occurs, it is assumed that either the aspirated follicle contained a blighted ovum or that a small cortical ovarian cyst has
been aspirated. The main purpose of the cytologic examination is to identify benign or malignant cells in cysts, masquerading as follicles. It should be stressed that malignant tumors diagnosed during harvesting of ova are vanishingly rare (Greenebaum et al, 1992; Rubenchik et al, 1996).
been aspirated. The main purpose of the cytologic examination is to identify benign or malignant cells in cysts, masquerading as follicles. It should be stressed that malignant tumors diagnosed during harvesting of ova are vanishingly rare (Greenebaum et al, 1992; Rubenchik et al, 1996).
Aspiration Biopsy (FNA)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree