The Gastrointestinal Tract
The parts of the gastrointestinal tract that are accessible to cytologic investigation are the esophagus, stomach, duodenum, colon, and the biliary and pancreatic ducts (Fig. 24-1). The methods of sampling are similar for all the organs, except colon.
METHODS OF SAMPLING
The introduction of flexible fiberglass optics instruments has not only revolutionized the endoscopy of the entire gastrointestinal tract but also allowed direct sampling of any visible lesion by cytology or tissue biopsy. A variety of instruments specially adapted to the inspection of the esophagus,
stomach, duodenum, and colon are available. Ultrathin endoscopes with a diameter smaller than 6 mm, provided with a video camera, are now available (Van Damm and Brugge, 1999). These procedures can be performed on any patients with radiographic abnormalities or clinical symptoms, or as a part of cancer prevention programs in asymptomatic patients at risk for stomach (in Japan) or colon carcinomas. The instruments may also be used for laser treatment of superficial lesions.
stomach, duodenum, and colon are available. Ultrathin endoscopes with a diameter smaller than 6 mm, provided with a video camera, are now available (Van Damm and Brugge, 1999). These procedures can be performed on any patients with radiographic abnormalities or clinical symptoms, or as a part of cancer prevention programs in asymptomatic patients at risk for stomach (in Japan) or colon carcinomas. The instruments may also be used for laser treatment of superficial lesions.
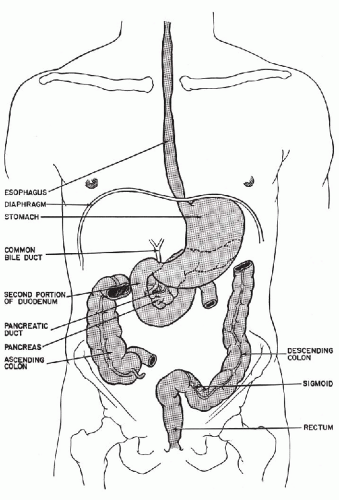 Figure 24-1 Schematic representation of portions of the gastrointestinal tract accessible to cytologic investigation. The transverse colon is omitted, to demonstrate the pancreas and the duodenum. |
The principles of collection of material for diagnostic sampling are the same for all organs. The instruments are provided with an accessory channel through which a brush, a plastic tube, or a small biopsy forceps may be passed. Thus, brushing, lavage, or a biopsy of any area in the gastrointestinal tract may be performed under direct visual control. Cytologic sampling must be performed prior to biopsy.
Washings of the esophagus or stomach without endoscopic control are nowadays very rarely used, although this simple technique led to the monumental contributions by Schade (1956, 1959, and 1960A,B) to the diagnosis of early gastric cancer. An equal volume of 95% alcohol must be added to the fluid immediately after aspiration to preserve the cells.
Endoscopically directed needle aspiration biopsy for the diagnosis of esophageal and gastric lesions was described by Layfield et al (1992) in a small number of patients with moderate success. A transbronchial aspiration needle was used in the procedures. There is no evidence that this technique achieved widespread acceptance.
Endoscopic ultrasound-guided needle aspiration biopsy has been proven effective in sampling small lesions of various organs of the gastrointestinal tract (Kosch et al, 1992; Chang et al, 1994; Mallery and Van Damm, 1999; Jhala et al, 2003). Perhaps the most important application of this technique is in sampling of intramural lesions and in assessing small lesions of the pancreas (Gress et al, 2001; Afify et al, 2003). For comments on the application of this technique to lesions of the biliary tree, see below.
“Salvage cytology” is a technique proposed by Graham et al (1979) consisting of washing the channel of the endoscopic instrument with saline and collecting the fluid for cytologic analysis into a suction trap. Initially, the technique was intended for use after biopsies (Graham and Spjut, 1979) but Caos et al (1986) reported its successful application without biopsies.
The application of esophageal and gastro-esophageal balloons and similar techniques for purposes of cancer detection are described below.
Special methods of cytologic investigation of the colon and rectum and the biliary tract are described further on in this chapter.
PREPARATION OF SMEARS
Preparation of smears from material obtained by endoscopic instruments is an important part of diagnostic evaluation of patients. Special skills are required for handling of brushes and direct needle aspirates. Brushing is usually performed to sample a visible lesion. Upon completion of brushing, the brush must be carefully, yet rapidly, withdrawn from the instrument and the smears prepared without delay, either by the physician or a trained assistant. Usually a brush yields two to four smears. Alternately, the entire brush may be forwarded to the laboratory for further processing (see Chap. 44). Preparation of smears from aspirates is best performed by a trained cytopathologist or technologist by methods described in Chapter 28.
THE ESOPHAGUS
ANATOMY AND HISTOLOGY
The esophagus is a tubular structure with muscular walls which extends from the pharynx across the diaphragm to the cardia of the stomach. The esophageal lumen is slightly narrowed at the level of thyroid cartilage, the bifurcation of the trachea, and the diaphragm. These are the sites wherein esophageal carcinoma tends to occur (see below). The esophagus is in close proximity to many vital structures. In the neck, the larynx and the trachea are immediately anterior; the recurrent nerves run along lateral walls of the esophagus, and the vagus nerves descend along its anterior and posterior walls. In the upper thorax, the esophagus is in contact with the trachea and the arch of the aorta. At the level of the heart, the left auricle is in close proximity. Thus, cancers of esophagus not only may obstruct its lumen, but also may invade and damage several vital organs.
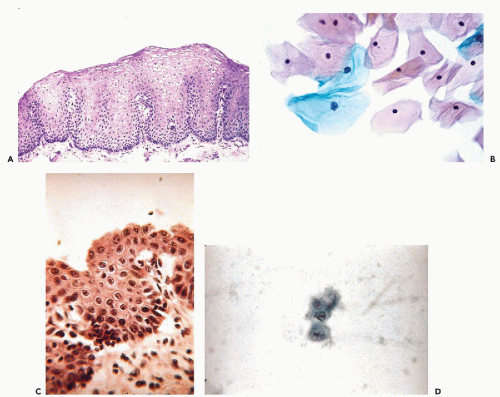
The esophagus is lined by nonhornifying squamous epithelium (Fig. 24-2A). Islands of gastric epithelium may be found in the areas immediately adjacent to the cardia and, rarely, elsewhere within the esophagus. Small, mucus-producing glands are found in the submucosa. The epithelium rests on a connective tissue layer, the submucosa or the lamina propria, which separates the epithelium from the muscle layers or the muscularis propria.
NORMAL CYTOLOGY
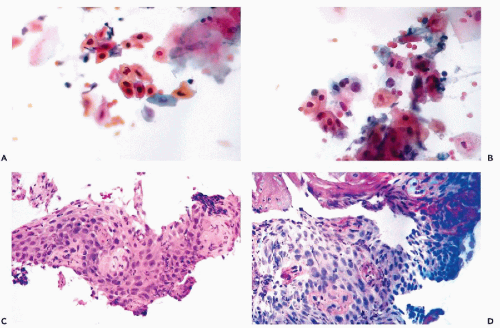
The cytology of the esophageal aspirates, brushings, and washings in the absence of disease is extremely simple. The smears are composed essentially of superficial squamous cells with vesicular nuclei, identical to those observed in sputum samples (Fig. 24-2B). Less frequent are smaller squamous cells derived from the deeper layers of the epithelium and provided with nuclei of similar sizes and configuration to the superficial cells. Occasionally, benign squamous “pearls,” may be noted. It is not unusual to find swallowed cells of respiratory origin, such as dust-containing macrophages and ciliated bronchial cells (see Chap. 19). Also, gastric epithelial cells, singly or in clusters, may occur (see Fig. 24-16). Foreign material, especially plant (vegetable) cells, may be present if there is an obstruction of the esophageal lumen. For description of these contaminants, see Chapter 19.
BENIGN DISORDERS
Acute and Chronic Erosive Esophagitis
This group of diseases of varying etiology is very important in diagnostic cytology, because it produces cells that may be confused with cancer. Esophagitis may have different causes such as trauma, acid reflux from the stomach, a reaction to swallowed corrosive liquids, cardiospasm of long standing, Plummer-Vinson syndrome (also known as sideropenic dysphagia, a syndrome of atrophy of the esophageal epithelium and iron deficiency anemia), some forms of avitaminosis, scleroderma (or systemic sclerosis of connective tissue, affecting primarily the skin), and hiatus hernia. It is of interest that, in older women, systemic sclerosis may be associated with the presence of persisting fetal DNA, suggesting that this disease may be an immune response to remote past pregnancies (Artlett et al, 1998). It is not known whether chronic erosive esophagitis, a rare, sometimes fatal disease, is related to any of these disorders. A drug, alendronate, an inhibitor of bone resorption (osteoporosis) has been shown to cause a chemical esophagitis in some patients with erosions and ulcerations of esophageal mucosa and thickening of the esophageal wall. In some cases, the disease is severe and disabling (De Groen et al, 1996).
Histology
The histologic lesion in esophagitis is mucosal erosions or ulcerations varying in number, depth, and configuration. There is moderate infiltration of the stroma with inflammatory cells. The surface of the lesion may be covered with granulation tissue or fibrin. Chronic erosive esophagitis is characterized by loss of superficial epithelial layers (mucosal erosions) (Fig. 24-2C). Squamous metaplasia of the submucosal glands may occur in this disorder.
Cytology
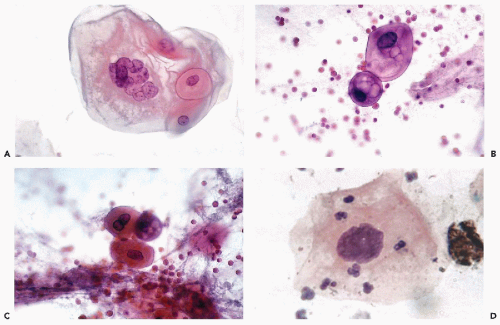
Cells desquamating from the eroded esophageal epithelium are derived from the deep layers and are of the same size as parabasal squamous cells, occurring singly and in small clusters. The cytoplasm is evenly distributed and the cells do not vary much in size. In clusters, there is good adherence of the cells to each other. The most outstanding features of the relatively large, somewhat hyperchromatic nuclei of these cells, sometimes show isolated clumps of chromatin and, occasionally, large nucleoli that may lead to an erroneous diagnosis of cancer (Fig. 24-2D). Careful attention to cellular detail helps in the correct interpretation of the cytologic findings. The cells seen in erosive esophagitis
may be similar to those observed in “repair” of squamous epithelium or in pemphigus (see Fig. 21-10).
may be similar to those observed in “repair” of squamous epithelium or in pemphigus (see Fig. 21-10).
A recently recognized entity, eosinophilic esophagitis, has been observed mainly in children but also in adults (Liacouras, 2003; Dahms, 2004). Clinically, the disease causes painful reflux esophagitis that mimics gastroesophageal reflux disease (GERD) but does not respond to antiacid treatment. Dense eosinophilic infiltrate of the esophageal wall has been observed in biopsies (Straumann et al, 2004). There are no reported cases of this entity diagnosed on cytologic sampling.
Herpetic Esophagitis
Clinical Findings and Histology
Many years ago, Berg (1955) emphasized the occurrence of this disease in cancer patients who, in the course of treatment, sustained a surgical or radiation injury to the esophagus. Immunosuppressive therapy, cytotoxic anticancer drugs, and acquired immunodeficiency syndrome (AIDS) have contributed to an increased frequency of this disease (Lightdale et al, 1977). Most patients have vague complaints referable to the esophagus, such as retrosternal pain or mild dysphagia. Herpetic esophagitis produces extensive, although superficial, ulcerations of the esophageal mucosa. In biopsies, intranuclear eosinophilic inclusions are observed within the epithelial cells. The use of cytology resulted in primary diagnosis in several cases. Surprisingly, in some patients, there was no past history of immune deficiency or immunosuppression. It is, therefore, possible that the disease is more common than hitherto anticipated. We have observed apparent simultaneous involvement of the esophagus and the bronchial tree.
Cytology
The disease may be diagnosed in material obtained from the esophagus by washings, or brushing. The cytologic findings
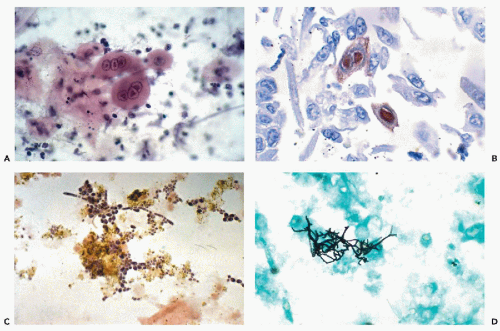
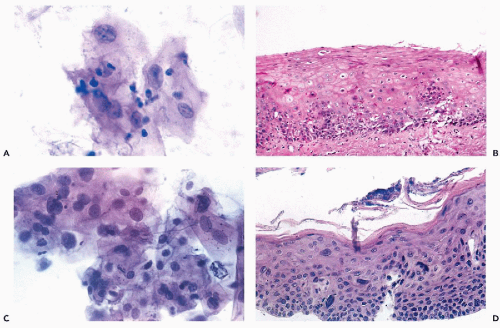
are identical with those observed in the material from the female genital tract (see Fig. 10-29), and the respiratory tract (see Chap. 19 and Fig. 19-36). Multinucleated cells with molded “ground-glass,” opaque nuclei and cells with intranuclear eosinophilic inclusions are observed (Fig. 24-3A,B).
are identical with those observed in the material from the female genital tract (see Fig. 10-29), and the respiratory tract (see Chap. 19 and Fig. 19-36). Multinucleated cells with molded “ground-glass,” opaque nuclei and cells with intranuclear eosinophilic inclusions are observed (Fig. 24-3A,B).
Esophageal Infections in AIDS
Esophagitis is a common early manifestation of AIDS. Candidiasis (moniliasis) (Fig. 24-3C), herpetic esophagitis (see above), cytomegalovirus infection (see Fig. 24-17B), and other infectious agents such as Nocardia may be identified in cytologic samples obtained during esophagoscopy (Fig. 24-3D). Geisinger (1995) stressed that cytologic techniques are much superior to biopsies in the diagnosis of Candida esophagitis. Teot et al (1993) reported a case with simultaneous herpetic and cytomegalovirus esophagitis in AIDS. Borczuk et al (1998) described a case of esophagitis caused by Trichomonas, diagnosed in esophageal brushings, in a male patient with AIDS.
Tuberculous esophagitis has been described in India where this disease is prevalent (Jain et al, 1999). The finding of clusters of elongated epithelioid cells and occasional Langhans’ type giant cells, in a background of inflammatory exudate, is suggestive of this rare event. Because of a marked increase of tuberculosis in AIDS patients, it may be anticipated that such cases will soon occur in the Western world as well.
Esophageal Diverticula
Esophageal diverticulum is an outpouching of the esophageal epithelium through the muscular wall of the organ. A diverticulum distended by accumulated food particles may produce symptoms of esophageal obstruction similar to those of cancer. There are no cytologic abnormalities known to occur in the presence of a diverticulum. However, rare cancers originating within the diverticula may be diagnosed by cytology (see below).
BARRETT’S ESOPHAGUS (COLUMNARLINED ESOPHAGEAL EPITHELIUM)
Clinical and Histologic Data
The syndrome, first described by Barrett in 1950, consists of a replacement of the distal esophageal squamous epithelium by columnar epithelium of gastric or intestinal
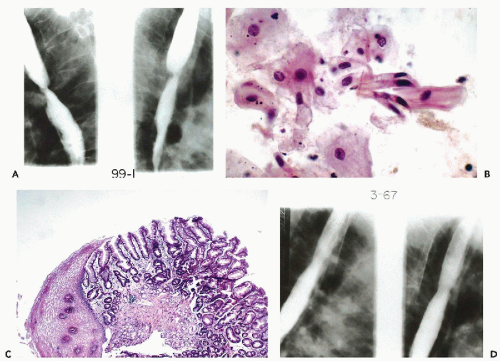
type, associated mainly with chronic gastroesophageal reflux disease (GERD), and sometimes with hiatus hernia and esophageal stricture (Mossberg, 1966; Burgess et al, 1971; Spechler and Goyal, 1986; Chandrasoma et al, 2001; Shaheen and Ransohoff, 2002). The segment of the replaced esophageal mucosa may be short or long (recent summaries in Glickman et al, 2001 and Spechler, 2002). The symptoms associated with Barrett’s syndrome are dysphagia, regurgitation, heartburn, and pain. Episodes of acute obstruction may occur, sometimes caused by peptic ulcers, similar to gastric ulcers (Fig. 24-4). In such cases, the radiologic examination may reveal a stricture that may mimic to perfection the appearance of an esophageal carcinoma (see Fig. 24-4A), although there is usually a preservation of esophageal peristalsis above and below. After treatment, the stricture may regress significantly (Fig. 24-4D). Barrett’s syndrome is, per se, a benign disorder, but has now been recognized as a risk factor for adenocarcinoma of the esophagus and, to a lesser extent, of gastric cardia (Lagergren et al, 1999). Cytologic techniques may be used to monitor patients with Barrett’s esophagus to identify malignant transformation and precancerous states, as described below.
type, associated mainly with chronic gastroesophageal reflux disease (GERD), and sometimes with hiatus hernia and esophageal stricture (Mossberg, 1966; Burgess et al, 1971; Spechler and Goyal, 1986; Chandrasoma et al, 2001; Shaheen and Ransohoff, 2002). The segment of the replaced esophageal mucosa may be short or long (recent summaries in Glickman et al, 2001 and Spechler, 2002). The symptoms associated with Barrett’s syndrome are dysphagia, regurgitation, heartburn, and pain. Episodes of acute obstruction may occur, sometimes caused by peptic ulcers, similar to gastric ulcers (Fig. 24-4). In such cases, the radiologic examination may reveal a stricture that may mimic to perfection the appearance of an esophageal carcinoma (see Fig. 24-4A), although there is usually a preservation of esophageal peristalsis above and below. After treatment, the stricture may regress significantly (Fig. 24-4D). Barrett’s syndrome is, per se, a benign disorder, but has now been recognized as a risk factor for adenocarcinoma of the esophagus and, to a lesser extent, of gastric cardia (Lagergren et al, 1999). Cytologic techniques may be used to monitor patients with Barrett’s esophagus to identify malignant transformation and precancerous states, as described below.
Histologic findings show an abrupt transition from the normal squamous epithelium to mucus-producing columnar epithelium of gastric type or of intestinal type with goblet cells (see Fig. 24-4C). The epithelium may form cysts wherein Rubio et al (1989, 1992) observed occasional presence of ciliated columnar cells, although this finding was much more common in papillary carcinomas (see below).
Cytology
Although the diagnosis of uncomplicated Barrett’s esophagus is usually established by esophagoscopy and biopsies, cytologic examination of esophageal brush specimens, aspirates or washings, may be valuable in establishing baseline data for comparison with future abnormalities occurring during the follow-up of these patients. The smears, containing mucus-producing benign columnar cells and goblet cells, usually in clusters, characteristic of mucus-producing intestinal epithelium, are diagnostic of Barrett’s esophagus (see Fig. 24-4B). The glandular cells have small, spherical, nuclei of even sizes. The peripheral nuclei of the goblet cells may appear a bit darker. We have not
observed the ciliated columnar cells described by Rubio (1989), except for swallowed cells of bronchial origin. Fennerty et al (1995) attempted to diagnose this disorder by esophageal balloon technique (described below) without success. Malignant changes in Barrett’s esophagus are described below.
observed the ciliated columnar cells described by Rubio (1989), except for swallowed cells of bronchial origin. Fennerty et al (1995) attempted to diagnose this disorder by esophageal balloon technique (described below) without success. Malignant changes in Barrett’s esophagus are described below.
BENIGN TUMORS
Focal Hyperplasia and Papillomas (Condylomas) of Squamous Epithelium
In 1982, Syrjänen et al described a rare papilloma-like lesion of the squamous epithelium of the esophagus, akin to a condyloma acuminatum, and postulated that human papillomavirus (HPV) may be a factor in the genesis of this tumor. Winkler et al (1985) confirmed this hypothesis by documenting the presence of HPV antigen in two such lesions and in 11 of 73 “focal hyperplasias,” some of which resembled “flat condylomas” with significant koilocytosis and nuclear abnormalities. Further evidence that HPV is present in papillomas was provided in case reports (Yamada et al, 1995; Politoske, 1992) and by Lavergne and de Villiers (1999) who confirmed the presence of the virus in 4 of 11 papillomas of the esophagus by molecular analysis. Apparently, none of these lesions progressed to cancer. It is of note, though, that Syrjänen (1982) observed “flat condylomas” at the periphery of invasive esophageal cancers. There is no known cytologic presentation of these lesions but it is likely that koilocytes should be observed in smears.
Granular Cell Tumor or Myoblastoma
A case of granular cell myoblastoma of esophagus diagnosed on esophageal brushings was described by Cordoba et al (1998). The lesion is very unusual in this location. The cytologic presentation of this tumor has been described in reference to the lung (see Chap. 20) and the breast (see Chap. 29).
Leiomyoma
We had the opportunity to study esophageal lavage and brush specimens from several cases of leiomyomas of the wall of the esophagus. The tumors could not be recognized in cytologic samples.
SQUAMOUS CARCINOMA AND ITS PRECURSORS
Epidemiology and Clinical Aspects
Squamous carcinoma is, by far, the most common type of esophageal cancer. The disease may affect any part of the esophagus, but occurs preferentially in segments of slight narrowing: at the level of the thyroid cartilage, bifurcation of the trachea, and the diaphragm. As a general rule, fully developed esophageal cancers cause obstruction of the esophagus, resulting in difficulties of swallowing and dysphagia.
Squamous cancer of the esophagus is a quasi-endemic disorder in northeastern Iran, in parts of China, among the Chinese in Singapore, among Africans in southern Africa, and among men in Brittany (France) (Enzinger and Mayer, 2003). In the United States, the disease is relatively uncommon; African American men appear to be more prone to it than other ethnic groups. In general, the disease is more common in males than in females. Epidemiologic data suggest that intake of hot beverages, cigarette smoking, and alcohol consumption are possible risk factors (Tavani et al, 1994; Enzinger and Mayer, 2003). Recent studies in China failed to reveal any consistent risk factors except, perhaps, diet (Li et al, 1989; Yu et al, 1993). Auerback et al (1965) demonstrated a high frequency of squamous carcinoma in situ of the esophagus among smokers.
The prognosis of esophageal squamous carcinoma is stage related. The overall survival is about 20% (Lerut et al, 1992; Goldminc et al, 1993). Izbicki et al (1997) pointed out that the prognosis of patients with clinical stage I disease, confined to the wall of the esophagus, may be modified by finding occult micrometastasis in regional lymph nodes stained with an epithelial antibody. Stockeld et al (2002) described the use of fine needle aspiration (FNA) biopsy technique for prognosis of esophageal squamous cancer. Aspirates of the esophageal wall, obtained at 2 cm intervals, led to the discovery of microscopic tumor spread in onethird of the 52 investigated patients. The results were more accurate than synchronous esophageal brushing or multiple biopsies. The ratio of benign to malignant cells in the aspirated samples appeared to be of prognostic significance.
Kwong et al (2004) studied chromosomal aberrations in esophageal squamous cancer by comparative genomic hybridization. Numerous gains and losses were observed but gain in the short arm of chromosome 12 (+ 12p) predicted poor prognosis after surgery, at least among the Chinese.
Adenocarcinoma occurring in Barrett’s esophagus is discussed below.
Human Papillomavirus in Squamous Cancer of the Esophagus
Invariably, as with all squamous cancers, the question of human papillomavirus (HPV) as a factor in the genesis of this tumor was raised (Syrjänen, 1987). HPV DNA presence in five invasive esophageal cancers was first reported by Kulski et al (1986). Subsequently, Chang et al (1992) reported the presence of HPV in 25 of 51 (49%) biopsies from Chinese patients with invasive esophageal carcinoma. In 16 of these 25 specimens, HPV types 16 and 18 were documented by in situ hybridization. Other types of HPV were observed in the remaining 7 patients. In the same study, 53 of 80 cytologic preparations, also from asymptomatic Chinese patients from a high-risk area, were positive for HPV by filter in situ hybridization. HPV was also detected in cells of 2 of 9 patients without cytologic abnormalities, in 3 of 6 patients with “mild dysplasia,” in 25 of 31
patients with “moderate dysplasia,” in 19 of 28 patients with “severe dysplasia,” and in 4 of 6 patients with invasive carcinoma. In an update of this study, Chang et al (2000) reported that 16.9% of 700 Chinese patients with esophageal carcinoma were HPV positive, with 27% of the positive samples containing the “high risk” HPV types 16 and 18.
patients with “moderate dysplasia,” in 19 of 28 patients with “severe dysplasia,” and in 4 of 6 patients with invasive carcinoma. In an update of this study, Chang et al (2000) reported that 16.9% of 700 Chinese patients with esophageal carcinoma were HPV positive, with 27% of the positive samples containing the “high risk” HPV types 16 and 18.
These data were either confirmed or contested in several papers. Thus, Cooper (1994) and He (1997) observed HPV in about 15% of esophageal cancers. De Villiers et al (1999), known for impeccable laboratory technique, confirmed the presence of HPV of various types in 17% of samples from Chinese patients. Takahashi et al (1998) and Kawaguchi et al (2000) observed that the presence of HPV was associated with mutations of the p53 gene.
On the other hand, Smits et al (1995) from the Netherlands, Benamouzig et al (1995) from France, and Mizobuchi et al (1997) from Japan, were unable to identify HPV in their many samples. In a study of 51 patients with squamous carcinoma from three North American cities, only one patient’s tumor contained HPV type 16 (Turner et al, 1997).
It is quite evident that the issue of the role of HPV in esophageal carcinoma has not been definitely settled but there appears to be little doubt that in tumors from some patients, mainly Chinese and Japanese, the virus is present (Galloway and Daling, 1996). Since a person-to-person transmission of HPV is unlikely in these patients, an activation of the latent viral infection is a more likely explanation of these findings.
It is of note that Wang et al (1999) detected Epstein-Barr virus (EBV) in squamous cancer in Taiwan.
Histology
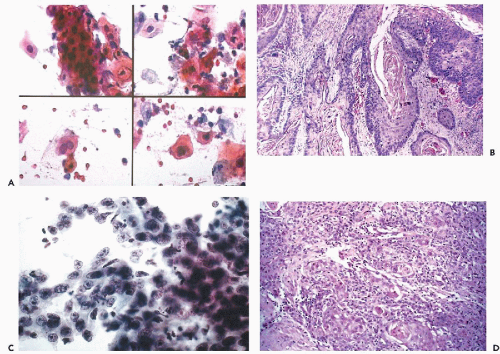
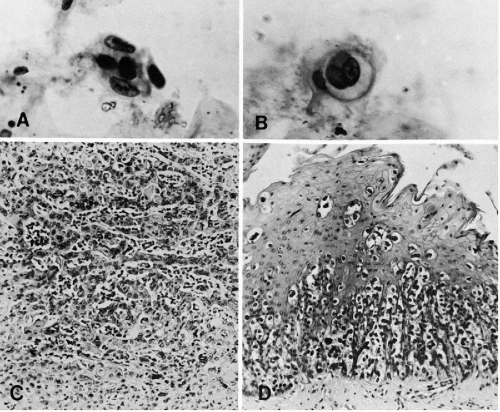
Histologic appearance of squamous cancer may vary in the degree of differention from well differentiated, highly keratinized (verrucous) types (Fig. 24-5B) to poorly differentiated squamous cancer (Fig. 24-5D) and, rarely, small-cell (oat cell) type of carcinoma (Rosen et al, 1975; Bogomoletz et al, 1989). A basaloid variant, resembling a basal cell carcinoma but with a highly malignant behavior, was described (see Fig. 24-6D) (Abe et al, 1996). Focal
glandular features may be observed in a substantial proportion of epidermoid carcinomas (Kuwano et al, 1988). Some of the poorly differentiated carcinomas may show endocrine granules in electron microscopy (Reyes et al, 1980).
glandular features may be observed in a substantial proportion of epidermoid carcinomas (Kuwano et al, 1988). Some of the poorly differentiated carcinomas may show endocrine granules in electron microscopy (Reyes et al, 1980).
Cytology
As discussed above, these tumors may occur in a variety of grades and degrees of differentiation. The cytologic findings in esophageal washings or brushings closely reflect these structural variants and are similar to those described for bronchogenic carcinomas of similar histologic types (see Chap. 20).
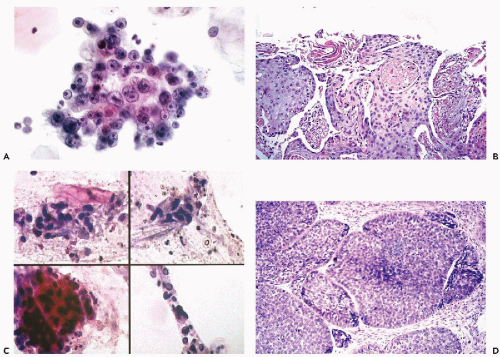
The well-differentiated squamous carcinoma produces heavily keratinized abnormal squamous cells, singly or in clusters, with either completely pyknotic, hyperchromatic nuclei, or with nuclear shadows, much in the manner described for similar cancers of the bronchus (Fig. 24-5A; see Chap. 20). Koilocyte-like cells with large, hyperchromatic nuclei and perinuclear clear zones or halos are sometimes observed in such tumors. There is no good correlation between these cells and the presence of HPV.
Less well-differentiated squamous cancers of large cell type (epidermoid carcinomas) are characterized by smaller cancer cells with very scanty basophilic cytoplasm, often forming clusters, particularly in brush specimens (Figs. 24-5C and 24-6A). The nuclear abnormalities in the form of enlargement, hyperchromasia and large nucleoli are usually quite evident. The diagnosis of tumor type depends largely on the finding of squamous cancer cells with eosinophilic cytoplasm, which may be very scarce.
Small cell carcinomas, the most anaplastic varieties of squamous cancer, produce cancer cells that often are very small, with abnormally large, hyperchromatic nuclei and very scanty cytoplasm (Fig. 24-6C). The corresponding tissue sections may sometimes show the “basaloid” tumor pattern (Fig. 24-6D). Horai et al (1978), Reid et al (1980), and Imai et al (1978) described several examples of such carcinomas. Hoda and Hajdu (1992) pointed out that, contrary to oat cell carcinoma of the bronchus, cell molding
was uncommon in the esophageal tumors and that the evidence of endocrine activity in these tumors was insecure.
was uncommon in the esophageal tumors and that the evidence of endocrine activity in these tumors was insecure.
Squamous or epidermoid carcinomas of the distal end of the esophagus may extend into the gastric cardia and fail to produce radiographic abnormalities of cancer on cursory examination. Cytologic examination, either by esophageal or gastric brushings, may be of critical diagnostic importance (Fig. 24-7).
The use of fine needle aspiration of the esophageal wall, for diagnosis and prognosis of squamous cancer, proposed by Stockeld et al (2002) was described above.
Precursor Lesions of Squamous Carcinoma and Their Detection: Lessons From China
In the 1961 and 1968 editions of this book, it was anticipated that carcinoma of the esophagus must be preceded by precancerous epithelial changes, such as carcinoma in situ and related abnormalities. In the Western world, the knowledge of precancerous squamous lesions of the esophagus is scarce. There are several cases on record in which squamous carcinoma in situ and related lesions had been observed as incidental findings (previous editions of this book; Imbriglia and Lopusniak, 1949; Auerback et al, 1965; Ushigome et al, 1967; Koss et al, 1998) or as a lesion accompanying invasive carcinoma (Suckow et al, 1962; Kuwano et al, 1988). However, the hypothesis could be confirmed only by the extensive cytologic and histologic studies of esophageal cancer conducted by Chinese investigators.
The stimulus for the Chinese studies was the very high prevalence rate of esophageal cancer in certain areas of central and northern China (Yang, 1980; Shu 1984, 1985). It is of incidental interest that, in the same areas of China, chickens are susceptible to cancer-like tumors of the gullet. The relationship of these tumors to human cancer is not understood. Except for its possible association with diet and human papillomavirus, the causes of human esophageal cancer in China and its relationship to the tumors in poultry, remain unknown at the time of this writing (2004).
The Chinese investigators proposed that prevention of esophageal cancer could be based on the same principles as detection of precursor stages of cancer of the uterine cervix. If cytologic samples, obtained in asymptomatic, high-risk populations, could lead to the discovery of precancerous lesions, then early surgical intervention or photodynamic therapy could prevent invasive esophageal cancer with its very high mortality rate (Yang et al, 2002). The instruments used in the cytologic investigations were small,
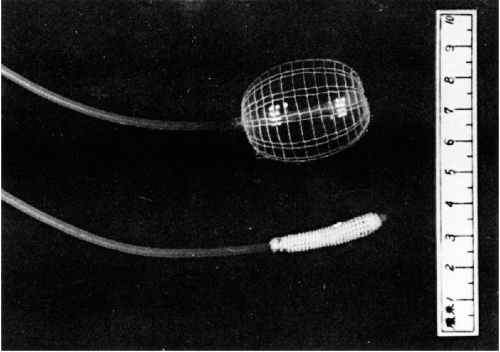
inflatable plastic balloons with abrasive surface (Fig. 24-8), modifications of a gastric balloon that was described in 1950 by Panico et al. The balloon was attached to a narrowcaliber tube with color markers to indicate the position of the balloon in the esophagus. The balloon could be easily swallowed in deflated state, moved by peristalsis to the cardia, inflated, and slowly withdrawn to the level of the cricoid cartilage. At this point, the balloon is deflated and withdrawn. The abrasive surfaces of the balloon contained cells scraped from the surface epithelium that could be examined in smears. The method, which was tested in our institution, caused trivial discomfort to the patients and was well accepted (Greenebaum, 1984).
inflatable plastic balloons with abrasive surface (Fig. 24-8), modifications of a gastric balloon that was described in 1950 by Panico et al. The balloon was attached to a narrowcaliber tube with color markers to indicate the position of the balloon in the esophagus. The balloon could be easily swallowed in deflated state, moved by peristalsis to the cardia, inflated, and slowly withdrawn to the level of the cricoid cartilage. At this point, the balloon is deflated and withdrawn. The abrasive surfaces of the balloon contained cells scraped from the surface epithelium that could be examined in smears. The method, which was tested in our institution, caused trivial discomfort to the patients and was well accepted (Greenebaum, 1984).
The accuracy of balloon sampling was tested in China on 1,861 patients with overt esophageal cancer, documented by biopsy. The accuracy varied from 87.2% to 99.0%, averaging 94.9% (summary in Shu 1984, 1985). As reported by Shu in 1984, the cytologic sampling by balloon proved to be much superior to either endoscopy or radiologic examination for the diagnosis of precursor lesions and early invasive squamous carcinoma. In an elaborate statistical analysis, Dawsey et al (1994A,B) compared the results of cytologic sampling with the incidence rates of esophageal squamous carcinoma in the Linxian province of China and concluded that the esophageal balloon cytology successfully identified persons at increased risk for esophageal cancer. This was confirmed for the Anyang County of China by Yang et al (2002). It is not known how the balloon method would compare with contemporary endoscopy, which because of cost and limited availability could not be used on a very large scale for purposes of esophageal cancer detection.
Several other methods to study esophageal cytology were developed. Jaskiewicz et al (1987) used a small sponge, attached to a string and packaged in an easy-to-swallow gelatin capsule to study patients in South Africa. A similar system was described by Sepehr et al (2000) as better acceptable to patients. Qin and Zhou (1992) described an elastic plastic tube for esophageal sampling and reported an accuracy of 96% in the diagnosis of cancer.
Results of Screening
The first results of the population survey were presented in the Fourth International Cancer Congress in Florence, Italy in 1974 by an anonymous group representing the Chinese Academy of Medical Sciences. A cytologic survey of 17,471 persons over 30 years of age was conducted in the Henan Province in northern China. “Dysplasia” of the esophageal epithelium was observed in 276 patients, mostly below the age of 40, whereas invasive carcinoma in this population usually occurred in patients older than 40. Follow-up study of the patients with “dysplasia,” some over a period of 7 to 10 years, disclosed that 30.3% of them developed esophageal carcinoma, in 27.3% the original lesion persisted unchanged, and in 42.4% the changes either regressed to mild dysplasia or reverted to normal. In histologic studies of 67 patients, the progression of dysplasia of various types to carcinoma in situ could be observed in many specimens. It was the conclusion of this study that “marked dysplasia” must be considered a precancerous lesion. During the intervening years and changing political conditions in China, the names of the investigators became known (summary in Shu 1984, 1985), and the results of several surveys became available.
As related by Shu in 1984, there is no doubt that mass screening for esophageal carcinoma in high-risk areas of-China had a major beneficial effect. Before screening was instituted, the diagnosis of carcinoma in situ or early invasive carcinoma was 2 per 1,000 in low-risk areas and 10 per 1,000 in high-risk areas. Screening of 81,187 asymptomatic people over the age of 30 in the high-risk Henan Province resulted in the discovery of 880 esophageal cancers (a huge prevalence rate of 1%!), of which 649 (73.7%) were early and treatable by surgery (Shu, 1984). Less is known about survival of these patients, but Dr. Shu assured me that most of the treated patients survived 5 years or longer with a good quality of life. This information must be compared with a survival of about 10% to 25% of patients with invasive squamous cancer of the esophagus commonly observed in the Western world (Ide et al, 1994; Lieberman et al, 1995). Kwong et al (2004) reported that, among Chinese patients with esophagus cancer, those showing a gain of the short arm of chromosome 12 (+p12) in the tumor had poor outcome after surgical treatment, regardless of stage of disease.
Screening for Esophageal Squamous Cancer in Countries Other Than China
The accomplishments of the Chinese scholars found several imitators. Thus, Berry et al (1981) attempted a similar project in South Africa (where the rate of esophageal cancer is very high among some black populations), resulting in the discovery of 15 occult invasive carcinomas and carcinomas in situ in 500 patients studied. Dysplasia was illustrated, but the clinical significance of the lesion was not discussed. Similar results were reported by Jaskiewicz et al (1987) from a high-risk rural population in Transkei (South Africa); in
five patients, dysplastic changes progressed to invasive cancer.
five patients, dysplastic changes progressed to invasive cancer.
To our knowledge, the Chinese experience has not been duplicated in the Western countries, except for the work in this laboratory. Greenebaum et al (1984) studied 96 high-risk Montefiore Hospital patients in New York City by the balloon technique. The selected patients had prior cancers of the larynx or pharynx, or were alcoholics and heavy cigarette smokers. Greenebaum unexpectedly found three occult recurrent oropharyngeal cancers and one carcinoma in situ of the esophagus, observed in a man with prior history of squamous carcinoma of the larynx. The biopsy of the esophagus disclosed fragments of squamous cancer in the absence of radiologic abnormalities.
Classification of Precancerous Lesions in China
Based on cytologic and histologic criteria, the Chinese investigators divided the precancerous lesions into two groups: dysplasia and carcinoma in situ. The criteria were derived from the classification of precancerous lesions of the uterine cervix (see Chap. 11). Lesions with more orderly epithelial growth, surface differentiation, and relatively minor nuclear abnormalities were classified as dysplasia and lesions with more significant atypia were classed as carcinoma in situ.
The dysplasias were further subdivided into mild, moderate, and severe, based mainly on cytologic criteria (see below). The true significance of dysplasia is not clear. Although, in some patients, the lesions either failed to progress or regress, there is no doubt that, in a substantial number of untreated patients, invasive cancer of the esophagus was subsequently observed (Shu 1984, 1985). The same conclusion was reached by Sugimachi et al (1995), who considered “dysplasia” as an early carcinoma of the esophagus. In any event, the insecure behavior of the precancerous lesions of the esophagus is remarkably similar to lesions of the uterine cervix (see Chap. 11).
Cytology
Much of the current knowledge of cytology of precursor lesions comes from Chinese sources (summaries in Shu, 1984, 1985; Shen, 1984). There is a remarkable similarity between the cytologic presentation of carcinoma in situ and related lesions of the esophagus and those of the uterine cervix (see Chap. 11). As in the uterine cervix, the lesions may be conveniently divided into high grade and low grade.
High-Grade Lesions
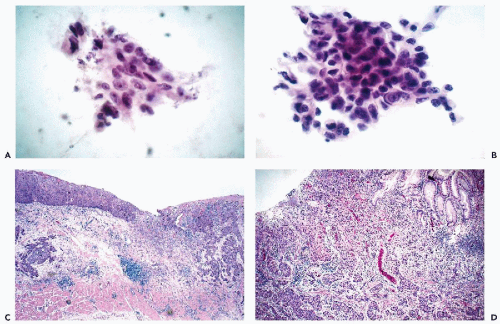
The squamous cancer cells derived from high grade lesions (high grade dysplasia, carcinoma in situ) are of the parabasal variety. The nuclear abnormalities consist of enlargement and hyperchromasia; the cytoplasm is scanty, resulting in a high nucleocytoplasmic ratio (Fig. 24-9). Cell clustering is common. Shu (1984, 1985) illustrated several examples of progression of dysplasias to carcinoma in situ and, in some cases, to invasive carcinomas over a period of 2 to 4 years.
There are very few cases of carcinoma in situ diagnosed by cytology in the Western world. Besides the case reported by Imbriglia and Lopusniak (1949), one case of a lesion approaching carcinoma in situ was personally observed in esophageal washings in a 59-year-old man with an esophageal diverticulum and symptoms of obstruction, leading to the clinical diagnosis of esophageal cancer. The lesion was characterized by typical dyskaryotic (dysplastic) superficial and parabasal squamous cells. The biopsies, which, unfortunately, were obtained after an initial short course of radiotherapy, localized the lesion to the diverticulum, which was subsequently successfully resected. The histologic appearance of the epithelium disclosed nuclear abnormalities and some degree of disarrangement of the component cells. One carcinoma in situ was recognized during balloon screening of a high risk population by Greenebaum et al (1984), and another during analysis of esophageal cytologic abnormalities by a neural net-based scanning system (Koss et al, 1998). In both cases, the smears contained squamous cancer cells, singly and in clusters, that could not be differentiated from an invasive squamous cancer (Fig. 24-10). The last patient who had a history of esophageal stricture and necrotizing esophagitis, was alive without evidence of esophageal cancer 3 years after the diagnosis. These anecdotal cases confirm the insecure prognosis of precursor lesions of esophageal squamous carcinoma.
Low-Grade Lesions
Low-grade lesions of the esophagus (mild or moderate dysplasia; Fig. 24-11) are characterized by well-differentiated superficial and intermediate squamous cells with marked nuclear enlargement and hyperchromasia. In some patients, koilocytes may be observed. The resemblance of these cell abnormalities to dyskaryosis (dysplasia) of cervical squamous cells is remarkable (see Chap. 11).
ADENOCARCINOMA AND ITS PRECURSORS
Clinical Data and Natural History
Although only about 3% of esophageal cancers are adenocarcinomas, this disease has generated an enormous amount of attention because of its association with “columnarlined epithelium” or Barrett’s esophagus (Haggitt et al, 1978; recent reviews in Shaheen and Ransohoff, 2002; Enzinger and Mayer, 2003). It is estimated that the presence of this abnormality increases the chances of adenocarcinoma about 50-fold and that the risk factor is in proportion to the size of the lesion (Menke-Pluymers et al, 1993; Lagergren et al, 1999). The association of Barrett’s esophagus with carcinoma is sometimes referred to as Dawson’s syndrome. However, adenocarcinomas of the esophagus may also occur in the absence of Barrett’s syndrome. The prognosis of esophageal adenocarcinoma is poor with 1-year survival
estimated at 44% but 5-year survival at only 13% (Eloubeidi et al, 2003D).
estimated at 44% but 5-year survival at only 13% (Eloubeidi et al, 2003D).
Histology
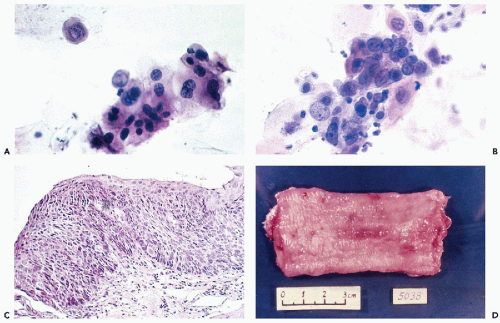
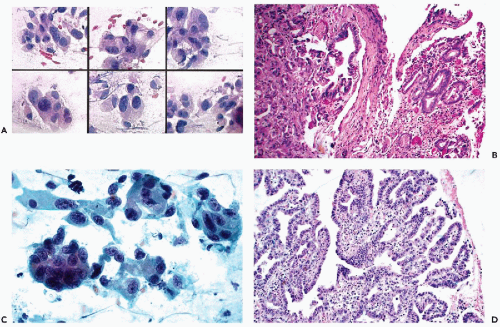
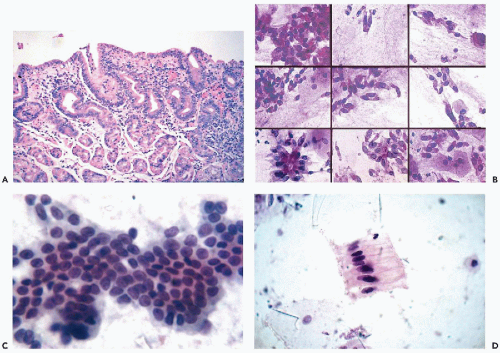
Most adenocarcinomas of the esophagus occur in the area of the cardia and originate in islands of gastrointestinal mucosa, less often in the submucosal glands, and usually are histologically similar to gastric adenocarcinoma and its various histologic patterns, described below. Most tumors are well differentiated and signet ring type of carcinoma is very rare. Occasionally, papillary adenocarcinomas, composed of large columnar cancer cells, may be observed (Fig. 24-12), also possibly related to Barrett’s esophagus wherein precancerous lesions of a similarhistologic and cytologic type have been observed (Belladonna et al, 1974). Rubio et al (1989, 1992) observed ciliated glandular cells in dilated glands of papillary adenocarcinomas in patients with Barrett’s esophagus.
As frequently happens in areas of the body where two different types of mucosa meet, tumors that have the properties of both glandular and squamous epithelium may occur in the lower esophagus. These cancers may be best classified as mucoepidermoid. There is no evidence that their behavior is in any way different from the behavior of pure epidermoid or pure mucus-producing varieties of cancer.
Precursor Lesions
The sequence of morphologic events in the genesis of adenocarcinoma became the subject of numerous scientific communications. Briefly summarized, morphologic precancerous abnormalities in columnar epithelium (named dysplasia, rather than carcinoma in situ) precede invasive carcinoma (Smith et al, 1984; Lee, 1985). These lesions have been subdivided into “low grade” and “high grade.” The criteria of this classification have been published by Reid et al (1980) and tested as reproducible among expert pathologists by Montgomery et al (2001A).
“High-grade dysplasia” (that we would prefer to classify as adenocarcinoma in situ) consist of nuclear enlargement and hyperchromasia in the columnar epithelial cells, occasionally with branching or distortion of the affected glands and a marked increase in abnormal mitoses (Reid et al, 1988; Rubio and Riddell, 1989). The lesions are very similar to precancerous abnormalities and carcinoma in situ of the gastric epithelium (see Fig. 24-26). The problem with high-grade esophageal dysplasia is its separation from frank adenocarcinoma, which is not
easy either in biopsies or in cytologic samples. Prospective studies of patients with severe dysplasia indicate a high level of progression to adenocarcinomas of the gastrointestinal type (Smith et al, 1984; Lee, 1985; Spechler and Goyal, 1986; Rusch et al, 1994; Montgomery et al, 2001B).
easy either in biopsies or in cytologic samples. Prospective studies of patients with severe dysplasia indicate a high level of progression to adenocarcinomas of the gastrointestinal type (Smith et al, 1984; Lee, 1985; Spechler and Goyal, 1986; Rusch et al, 1994; Montgomery et al, 2001B).
Elaborate molecular studies suggested that high grade dysplasia and early adenocarcinoma show similar genetic alterations, regardless whether the Barrett lesion involved short or long segments of the esophagus (Nobukawa et al, 2001). Also, mutation of p53 gene was shown to be frequent in high grade dysplasia and adenocarcinoma than in low-grade dysplasia (Bian et al, 2001). Surgical resection of “severe dysplasia” is curative of the disease but it is not without major complications (Rush et al, 1994). Currently, such lesions may also be treated by endoscopic ablation, using photodynamic therapy or laser surgery (Van Dam and Brugge, 1999).
Much less secure is the identification of low grade or “mild dysplasia” which is described as slight atypia of the columnar epithelial cells (Robey et al, 1988). According to Montgomery et al (2001A, 2001B), the diagnosis of low-grade dysplasia is fairly reproducible among expert pathologists and shows a 15% to 20% progression rate to invasive cancer. Patients with Barrett’s syndrome are monitored by endoscopic biopsies and by cytologic studies of brush specimens (Spechler, 2002).
Cytology
Invasive Adenocarcinomas and Mucoepidermoid Carcinomas
In brush specimens, the cancer cells derived from adenocarcinomas are usually well-differentiated columnar cells with conspicuous nuclear abnormalities in the form of enlarged, sometimes hyperchromatic nuclei with large single or multiple nucleoli (Fig. 24-12A,C). In advanced carcinomas, these cells occur singly and in small clusters, usually accompanied by evidence of necrosis. Smaller, more spherical cancer cells may also occur in less well-differentiated tumors. Shurbaji and Erozan (1991) noted that the findings were not consistent and that nuclear hyperchromasia and large nucleoli were not observed in all the cases.
In mucoepidermoid carcinomas, the cells of glandular derivation are mixed with occasional squamous cancer cells. In many such cases, it is not clear whether the tumor is primarily squamous or glandular, although the malignant nature of the process is evident.
Precursor Lesions
Cytologic definition of “dysplasia” of columnar epithelium is insecure. Wang et al (1992) described small clusters
of mildly pleomorphic cells with enlarged nuclei with occasional multiple nucleoli. In a more recent thoughtful review of this topic by Hughes and Cohen (1998), the definition of cytologic abnormalities in dysplasia was not particularly helpful. These authors reviewed the literature and pointed out that the accumulated experience with comparative cytology-histology of these lesions is very small.
of mildly pleomorphic cells with enlarged nuclei with occasional multiple nucleoli. In a more recent thoughtful review of this topic by Hughes and Cohen (1998), the definition of cytologic abnormalities in dysplasia was not particularly helpful. These authors reviewed the literature and pointed out that the accumulated experience with comparative cytology-histology of these lesions is very small.
Personal experience suggests that the morphologic distinction of “high-grade or severe dysplasia” from adenocarcinoma is highly subjective. In cases with clusters of mildly or moderately atypical columnar cells with somewhat enlarged, somewhat hyperchromatic nuclei and the presence of multiple small or single large nucleoli, the diagnosis of “atypia” of columnar cells is justified (Fig. 12-13A,B). If the atypical cells form clusters, the possibility of an adenocarcinoma cannot be ruled out (Fig. 12-13C,D). In all such cases, the tentative cytologic diagnosis must be confirmed by biopsies. The situation is reminiscent of the problems with recognition of early neoplastic abnormalities in endocervical epithelium, discussed at length in Chapter 12.
There is virtually no cytologic experience with the identification of “mild dysplasia.” Hughes and Cohen (1998) point out that the separation of the possible neoplastic from reactive changes in the columnar cells is virtually impossible.
Monitoring of precursors of adenocarcinoma by balloon cytology was attempted with diverse results. Fennerty et al (1995) failed to obtain informative samples in a small number of patients. Falk et al (1997), in a much larger series of patients, compared the inexpensive balloon cytology with brushings. Brush cytology recognized all of 11 patients with high-grade dysplasia and carcinoma whereas balloon samples failed to recognize two of these patients. For low-grade dysplasia, both brushings and balloon cytology failed in a substantial proportion of cases. In two of the patients without disease, atypical cells were observed. This study documented the problem with cytologic diagnosis of low-grade abnormalities.
The monitoring of epithelial DNA content by flow cytometry disclosed abnormal, aneuploid DNA histograms in dysplasias and in carcinomas (Haggitt et al, 1988). However, securing adequate cell samples for this procedure by esophageal brushings is difficult. Another approach to progression of dysplasia to adenocarcinoma in tissue samples was discussed by Polkowski et al (1995). These authors reported an
increase in the expression of mutated p53 gene and an increase in the proliferative fraction of cells as measured by expression of Ki67 antigen with progression of esophageal dysplasia to adenocarcinoma. To our knowledge, this approach has not been tested in cytologic samples.
increase in the expression of mutated p53 gene and an increase in the proliferative fraction of cells as measured by expression of Ki67 antigen with progression of esophageal dysplasia to adenocarcinoma. To our knowledge, this approach has not been tested in cytologic samples.
CYTOLOGIC FOLLOW-UP OF PATIENTS WITH TREATED ESOPHAGEAL CARCINOMA
The customary treatment of invasive carcinoma of the esophagus is either by surgery, radiotherapy alone or by radiotherapy followed by an attempt at surgical removal of the lesion. Extensive surgical resection for esophageal adenocarcinoma did not improve the survival rate (Hulscher et al, 2002). Combining chemotherapy with cisplatin and radiotherapy (chemoradiotherapy) did not improve overall survival of patients (Bosset et al, 1997). In several cases so treated, the observation has been made that, in spite of the remarkable clinical improvement following radiotherapy, the smears remained positive. In several of the surgically removed esophagi, there was disappearance of much of the invasive tumor, but areas of carcinoma in situ were not affected by therapy. This situation is reminiscent of the results of radiation treatment of carcinoma of the bladder, discussed in Chapter 23. Undoubtedly, the persisting carcinomas in situ are at the origin of treatment failure in many cases. Stockeld et al (2002) used serial needle aspiration biopsies of various parts of the esophagus to rule out the presence of occult foci of cancer. The presence of cancer cells in areas of the esophagus grossly free of tumor correlated with poor prognosis.
Brien et al (2001) described gastric dysplasia-like epithelial atypia associated with chemoradiotherapy of esophageal cancer. For further discussion of gastric dysplasia, see below.
Radiation changes observed in esophageal cytologic material are closely similar to those described for the uterine cervix (see Chapter 18) and the respiratory tract (see Chap. 19) (Fig. 24-14). The changes may affect both benign and malignant cells (Cabré-Fiol, 1970). The diagnosis of persisting or residual carcinoma should be made only on cancer cells showing no effect of radiation.
Radiomimetic changes in esophageal epithelium were described by O’Morchoe et al (1983) in patients receiving cytotoxic drug therapy for tumors other than esophageal cancer. Some of these patients also had herpetic esophagitis and infections with fungi of the Candida species.
EFFECTIVENESS OF CYTOLOGY IN THE DIAGNOSIS OF ESOPHAGEAL CANCER
The effectiveness of balloon cytology in the detection of precancerous lesions of the esophagus has been documented in the studies from the People’s Republic of China, cited above.
The contributions of cytology in symptomatic patients before widespread use of fiberoptic instruments are best assessed by comparison with other diagnostic techniques: a few cases of esophageal carcinoma with negative radiographic findings were observed in this laboratory. The results, of historical interest only, were reported from our laboratory and Papanicolaou’s laboratory by Johnson et al in 1955. The cytologic results in 148 cases of esophageal cancer and 135 controls are summarized in Tables 24-1 and 24-2. In a significant percentage of cases (12%), cytology yielded positive results, whereas the biopsy was either negative or impossible to obtain. We also reported three false-positive cases, based on the presence of atypical squamous cells in cases of erosive esophagitis (see above).
Other older studies (Raskin et al, 1959; Prolla and Kirsner, 1972; Cabré-Fiol, 1970; Drake, 1985) also show a very high rate of accuracy in the diagnosis of esophageal carcinoma, ranging from 90% to 95%. It is of interest that the introduction of fiberoptics and of direct brushing resulted in only slight improvement in the accuracy of cytologic diagnoses of esophageal cancer, when compared with the simpler methods of washings and aspirations. Geisinger (1995) compared the results of cytologic brushings with endoscopic biopsies in specimens from a broad spectrum of esophageal diseases. There were 18 carcinomas, of which 9 were squamous and 6 adenocarcinomas. In 3 of the 18 carcinomas the diagnosis was established by cytology with negative initial biopsies. One squamous cancer was missed by cytology. Geisinger provided a summary of findings from several papers on this topic. The superiority of brush cytology over biopsies in the diagnosis of esophageal cancer was repeatedly confirmed, thus documenting the clinical value of cytologic techniques.
RARE MALIGNANT TUMORS OF THE ESOPHAGUS
Adenoid Cystic Carcinoma
We have seen one example of this lesion which is most unusual in the esophagus. The tumor has been described in reference to salivary glands and the bronchus (see Chaps. 20 and 32). As is the case in other organs where the tumor is more common, the brush smear disclosed clusters of small, monotonous
cancer cells with fragments of homogeneous, hyaline material, corresponding to the basement membrane material contained within the cystic spaces in the tumor.
cancer cells with fragments of homogeneous, hyaline material, corresponding to the basement membrane material contained within the cystic spaces in the tumor.
Carcinoma with Pagetoid Changes
An exceptional event is the presence of pagetoid change that has been observed in the esophageal epithelium adjacent to areas of carcinoma, reported by Yates and Koss (1968). The invasion of the squamous epithelium by large, clear cells of adjacent carcinoma resulted in this readily identifiable histologic pattern, similar to that occurring in the epithelium of the nipple or vulva in Paget’s disease (see Chaps. 15 and 29). In the case illustrated here, the cancer cells shed from the pagetoid area were approximately spherical, had a clear cytoplasm, and were occasionally
arranged in the cell-in-cell pattern, whereas the cells from the main bulk of the tumor were poorly differentiated (Fig. 24-15).
arranged in the cell-in-cell pattern, whereas the cells from the main bulk of the tumor were poorly differentiated (Fig. 24-15).
TABLE 24-1 RESULTS OF CYTOLOGIC EXAMINATION OF THE ESOPHAGUS | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||
TABLE 24-2 COMPARISON OF RESULTS OF CYTOLOGIC EXAMINATION WITH RESULTS OF BIOPSY IN 148 PRIMARY CANCERS OF ESOPHAGUS | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||
Polypoid Carcinoma (Carcinosarcoma)
Histology
Uncommonly, squamous carcinomas of the esophagus (but also of the larynx, pharynx, and rarely in the vagina) develop into polypoid bulky tumors, the surface of which is formed by a well-differentiated epidermoid carcinoma, sometimes in situ. The bulk of the tumor is composed of elongated spindly cells, occasionally accompanied by bizarre giant cells (Enrile et al, 1973). Experience with these lesions suggests that the spindle and giant cell components represent a peculiar metaplasia of squamous carcinoma and not a benign “pseudosarcoma,” as was originally suggested by Stout and Lattes (1957). Still, the latter term is often used to describe this lesion. It must be stressed that, in spite of its ominous appearance, the polypoid carcinoma appears to offer a much better prognosis than ordinary esophageal carcinoma.
Cytology
The cytologic findings in brushings from one of the rare polypoid carcinomas of the esophagus were described by Selvaggi (1992). The smears contained cells of squamous carcinoma and spindly cancer cells, and a few multinucleated giant cells, corresponding exactly to the histologic make-up of the tumor. We observed a similar tumor in the vagina. The smears were composed of malignant squamous cells only (see Chap. 17).
Malignant Melanoma
Several case reports of the very rare primary esophageal melanoma were published (Bullock et al, 1952; Boyd et al, 1954; Broderick et al, 1972; Chaput et al, 1974; Ludwig et al, 1981), occasionally associated with melanosis (De la Pava et al, 1963; Piccone et al, 1970). Summaries of the subject were presented by Mills and Cooper (1983) and by Kanavaros et al (1989). Broderick et al (1972) reported a case of esophageal melanoma with cytologic diagnosis. The malignant cells were clearly pigmented and, accordingly, the accurate diagnosis could be readily established. A similar case was reported by Aldovini et al (1983). One of these very infrequent tumors of the esophagus was diagnosed cytologically by us as cancer but, in the absence of pigment, it was thought to be an epidermoid carcinoma. De la Pava et al (1963) described pigmentation of normal esophageal epithelium as melanosis. This extremely rare condition could be a source of diagnostic error. However, Piccone et al (1970) and Kanavaros et al (1989) described a malignant melanoma developing in melanosis, thus complicating the issue still further.
Sarcomas
Sapi et al (1992) described the cytologic findings in a polypoid tumor of the esophagus, classified as malignant fibrous histiocytoma on the strength of immunocytologic and ultrastructural findings. The smears were characterized by the presence of bizarre tumor giant cells. Clearly, the differential diagnosis in such cases must include polypoid carcinomas, discussed above.
A rare case of rhabdomyosarcoma of the esophagus was described by Shah et al (1995). A case of embryonal rhabdomyosarcoma was described by Willen et al (1989). The cytologic presentation of similar tumors in other organs was described in Chapters 23 and 26.
Choriocarcinoma
A case of an exceedingly rare primary choriocarcinoma of the esophagus was described by Trillo et al (1979). The cytologic features of this tumor are described below in the section on rare gastric tumors, in Chapter 17, discussing rare tumors of the female genital tract, and in Chapter 23, discussing rare tumors of the urinary bladder.
THE STOMACH
ANATOMY
The stomach is a pouch situated between the esophagus and the duodenum, immediately below the diaphragm; it forms a reservoir of variable capacity within which the preliminary stages of digestion take place. The stomach is divided anatomically into several regions, starting at the esophageal end: the cardia (the orifice between the stomach and the esophagus and the adjacent portion of the stomach), a lateral bulge or the fundus, the more distal portion of the stomach or the body, and the most distal pyloric area (antrum), which is separated from the duodenum by a powerful ring of smooth muscle, the pylorus. Obstruction of the gastric lumen may occur at either end of the stomach—the cardia or the pylorus (see Fig. 24-1).
From inside out, the stomach is lined by an epithelium (mucosa), described in detail below, a lamina propria, composed of connective tissue and a thin layer of smooth muscle (muscularis mucosae), a powerful layer of smooth muscle or the muscularis propria, and the outer layer or the serosa, lined by the peritoneum.
HISTOLOGY OF GASTRIC EPITHELIUM (MUCOSA)
Gastric epithelium is by far the most important component of cytologic preparations. The gastric mucosa is composed of simple tubular glands. The lining of the glands of the fundus and the body of the stomach is complex: the surface and the necks of the glands are lined by mucus-producing cells. The deeper portions of the glands contain pepsin-producing chief cells and the eosinophilic parietal cells that produce hydrochloric acid. The hydrochloric acid is excreted through microscopic canaliculi passing between the chief cells. The gastric glands of the pyloric area are fairly uniformly lined by mucus-producing cells (Fig. 24-16A).
Electron microscopic studies revealed distinct differences between the cells lining the gastric surface and those lining the neck of the glands. Both cells are mucus-producing but they differ in the type of secretory granules, thus probably performing somewhat different functions. The ultrastructure of the parietal cell fails to reveal any secretory activity. The pepsin-producing chief cells resemble somewhat the exocrine pancreatic and salivary gland cells inasmuch as they contain cytoplasmic secretory granules and
an abundant rough-surfaced reticulum. Argentaffin cells containing dense cytoplasmic granules may be observed in some of the crypts.
an abundant rough-surfaced reticulum. Argentaffin cells containing dense cytoplasmic granules may be observed in some of the crypts.
CYTOLOGY OF NORMAL STOMACH
The make-up of the specimens varies according to the method used to collect the material. Gastric lavage, extensively used by Schade and his co-workers (1956A,B, 1959, 1960A,B), yields few normal gastric cells. The gastric balloon with a rough surface, devised by Panico et al (1950, 1952) to improve the collection of gastric epithelial cells, yields a richer harvest of cells. The optimal samples of gastric epithelium are obtained by gastric brushings during fiberoptic gastroscopy.
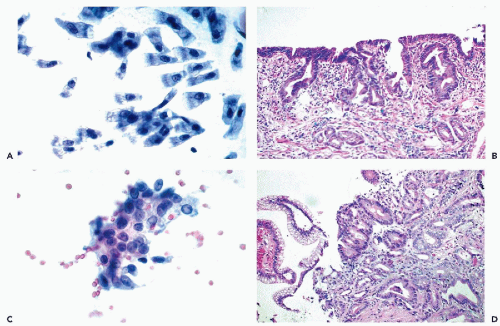
In the absence of disease, normal gastric epithelium in brush specimens is represented by columnar cells, occurring singly or forming cohesive fragments of cells with opaque or clear cytoplasm. The columnar configuration of the component cells is seen at the edge of such clusters, whereas the center of the cluster shows the “honeycomb” pattern (Fig. 24-16B,C). The relatively uncommon, mucus-producing columnar cells display an abundant, clear cytoplasm and have one flattened surface, corresponding to the gastric lumen, whereas the opposite end usually tapers off in the form of a tail (Fig. 24-16D). Raskin et al (1961) pointed out that such cells originate from the area of the pyloric antrum. Such cells are common in intestinal metaplasia of the gastric mucosa (see below).
A “wheel-spokes” arrangement of single cells,




Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree