Fig. 5.1
The types of neural scaffolds are shown on the left-hand side of the diagram, and the components of tissue-engineered nerve grafts are shown on the right-hand side. Adapted with permission from [1]
Scaffolds should be non-cytotoxic, non-immunogenic, non-allergenic, and non-carcinogenic as well as being sufficiently porous to allow the diffusion of nutrients while inhibiting the invasion of scar tissue [5, 10]. The fabrication technique (cutting holes on the wall, rolling of meshes, fiber spinning, adding a pore-forming agent, or injection molding followed by solvent evaporation) affects the permeability by altering the porous structure of neural scaffolds [11, 12]. Permeability of neural scaffold is also affected by the hydrophilic property of the scaffold material [1]. For a routine clinical application, the neural scaffold has to be easily fabricated, sterilized, and moreover has to satisfy many biomechanical and biological requirements such as biocompatibility and biodegradability [1]. On biomechanical side, a neural scaffold must be flexible to allow bending without kinking; too stiff scaffolds are easily dislocated, while too flexible scaffolds fail to provide sufficient mechanical support for axonal regeneration [1]. On biocompatibility side, the surface properties (including pH and surface charge) are the determining factors in terms of interactions between the neural scaffolds and nerve cells and the ability of the scaffold to blend in the implantation site and promote cell–substrate interaction [1, 10]. For example, the longitudinally oriented surface texture of the neural scaffold has been shown to help directional outgrowth of axons and uniform alignment of Schwann cells in vitro, resulting in improved nerve regeneration [1]. Similarly, multichannel neural scaffolds have greater surface areas for cell attachment and local release of growth factors, thus theoretically can support the nerve regeneration across a larger nerve gap. The data published thus far support the hypothesis that multichannel neural scaffolds reduce the dispersion of regenerating axons through the scaffold lumen; however, they displayed no significant benefit over single-lumen scaffolds [1, 13] probably because a complex multilayer internal structure may at the same time interfere with the permeability and flexibility of the neural scaffold [13].
5.4.1.2 Surface Properties of Nerve Scaffolds
In in vivo, the cells are located in three-dimensional microenvironments where they are surrounded by other cells and by the extracellular matrix, whose components, such as collagen, elastin, and laminin, are organized in nanostructures (i.e., fibers, triple helixes, etc.). This complex tissue network regulates the morphology, migration, and proliferation of Schwann cells, stimulates the release of nerve growth factors from Schwann cells, and also provides binding sites and directionality to the growing axons [14]. It is therefore essential to develop scaffolds that create synthetic microenvironments, providing 3D support, so as to control and direct the cellular behavior and to promote specific cell interactions [5].
Attempts to imitate the natural extracellular matrix by adding macromolecules (proteoglycans, collagen, elastin, laminin, fibronectin) to the internal environment of nerve scaffolds experienced a limited success [14–17]. However, nanomaterials provide a new dimension of interaction with biological systems that takes place on a subcellular level with a high degree of specificity [5, 18, 19]. For example, nanodiamond monolayers provide an excellent growth surface on various materials for functional neuronal networks and bypass the necessity of protein coating [20]. Furthermore, carbon nanotubes enhance nerve regeneration by rendering the scaffold more conductive [89].
The source of the scaffold is another aspect that effects the cell–scaffold and tissue–scaffold interactions. A scaffold may be derived from natural or synthetic materials [21].
5.4.1.3 Natural Nerve Scaffolds
Natural scaffolds are made from tissues that already exist in human body or from materials that exist naturally outside human body [5]. Natural biomaterials are attractive sources for nerve tissue engineering since they constitute cell-friendly matrices that stimulate adhesion, migration, growth, and proliferation of neurogenic cells. These materials also exhibit similar properties to the soft tissues that they are replacing, and they do not elicit foreign body reaction upon implantation and therefore have an excellent biocompatibility [1, 22]. The concerns about natural materials are as follows: the necessity of a careful purification process and inconsistent homogeneity of products between the lots [22].
Acellular Allogeneic or Xenogeneic Tissues
Acellular tissues from same or different species can be obtained by various physical, chemical, and enzymatic decellularization methods [23–25]. These methods aim to remove the immunogenic components and preserve the extracellular matrix components that are essential for the mechanical integrity of the tissue. Acellular tissues are especially useful in the repair of non-critical peripheral nerve gaps with a small length and diameter [1].
Acellular allogeneic and xenogeneic nerve tissues [25–32], tendon [33], and vein and muscle [34, 35] have been used by different groups in order to repair peripheral nerve defects. Avance1 (AxoGen Inc. Alachua, FL) is a commercially available acellular allogeneic nerve graft product which is fabricated from a donated cadaveric nerve through decellularization. Preliminary clinical applications of this product yielded favorable results [28]. However, the major concern about the use of acellular nerve scaffolds is the lack of axonal growth-stimulating bioactive components just like any other synthetic nerve scaffold. Recently, an increasing amount of effort has been directed toward incorporation of support cells or growth factors into the acellular allogeneic or xenogeneic-based neural scaffolds [30–32, 36]. The resultant tissue-engineered nerve grafts allow a better nerve regeneration than the acellular neural scaffolds alone.
Extracellular Matrix Components
Extracellular matrix is a network of proteins and fibers surrounding the cells that together form a complex 3-D structure crucial for proper biological functioning of the cells. Three main components of extracellular matrix, collagen, laminin, and fibronectin, have important roles in nerve regeneration.
Collagen has been extensively investigated as a potential scaffold for neural tissue engineering (Fig. 5.2) [37–41]. The properties of collagen scaffolds can be varied by using different concentrations of collagen or it can be denatured to gelatin that has also been used as a scaffold material [42]. The source of the collagen can be mammals such as rats, bovines, and humans; therefore, there is a risk of immune response if xenogeneic transplantation is used.


Fig. 5.2
SEM views of a collagen nerve scaffold (Revolnerv® tube) with a smooth internal and external texture. Scale bars, 1 mm (a) and 100 μm (b). Adapted with permission from [37]
Chitin, Chitosan, and Other Natural Polysaccharides
Chitin is a natural polysaccharide that is commonly found in the outer shells of crustaceans, insect exoskeletons, and fungal cell walls. It is extensively used in biomedical applications. Chitosan is closely related to chitin and can be obtained from chitin via deactylation [22]. The molecular structure of chitosan has got a strong resemblance with the molecular structure of glycosaminoglycan; therefore, it can interact with the extracellular matrix molecules in a similar way. The favorable biological properties of chitin and chitosan made them useful materials for nerve tissue engineering [1, 43–46].
Silk Fibroin and Other Natural Molecules
5.4.1.4 Synthetic Nerve Scaffolds
Synthetic scaffolds can be made from non-degradable materials such as silicone [50, 51] and poly-2-hydroxyethyl methacrylate-co-methyl methacrylate (PHEMA-MMA) [52] or from biodegradable polymers such as poly-3-hydroxybutyrate [53, 54], polyglycolic acid (PGA) [55, 56], poly L-lactic acid (PLA) [57, 58], poly-lactide-co-glycolide (PLGA) (Fig. 5.3) [59–61], and poly-lactide-co-caprolactone (PLC) [62, 63].
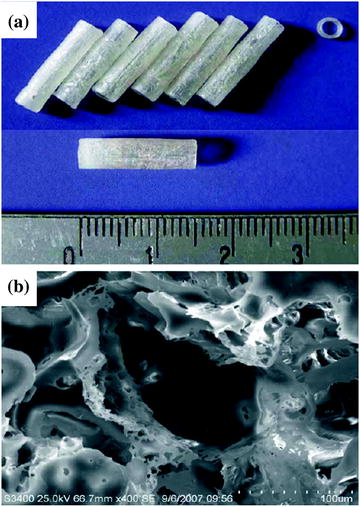

Fig. 5.3
PLGA conduits (a) and structure of the scaffold under scanning electron microscopy (SEM) (b). Scale bar, 100 μm. Adapted with permission from [61]
The main advantage of synthetic nerve scaffolds is their tunable chemical and physical properties. However, their incompatibility with cell adhesion and tissue integration poses a challenge to nerve tissue engineering. Therefore, synthetic materials are often chemically modified to render them more biocompatible [1].
Non-degradable Synthetic Materials
The early neural scaffolds were made of non-degradable synthetic materials such as silicone, rubber, acrylic polymer, polyethylene, elastomer, etc. Even though they achieved variable levels of success, their clinical use is currently limited because of the long-term complications such as chronic nerve compression, chronic foreign body reaction, and the necessity of a second surgical procedure to remove the scaffold [64, 65].
Biodegradable Synthetic Materials
As the concerns rose over the use of non-degradable synthetic materials as nerve scaffolds, biodegradable synthetic scaffolds, which degrade within a reasonable time span after implantation, were developed. Ideally, the neural scaffolds should remain intact throughout the axon regeneration and then gradually degrade with minimal swelling and foreign body reaction [1]. In case of a 10-mm gap of the rat sciatic nerve, the minimum time period that a biodegradable nerve scaffold must remain intact for sufficient nerve regeneration is 3 months [5]. Both the premature and delayed degradation of nerve scaffold might result in increased scar formation that further delays nerve regeneration [5, 66].
Further chemical modifications can be used to increase the biocompatibility of degradable scaffolds via adding sites for cell or extracellular matrix adhesion to allow cells to infiltrate into the scaffold lumen (Fig. 5.4) [19, 67, 68].


Fig. 5.4
SEM image shows Schwann cells on PCL/gelatin nanofibrous scaffolds after 12 days of coculture. Nanofibers encourage the attachment of Schwann cells. Scale bar, 200 μm. Adapted with permission from [19]
5.4.2 Growth Factors
Growth factors act in coordination with extracellular matrix to control the survival, proliferation, migration, and differentiation of various cell types involved in the nerve regeneration [69]. Incorporation of growth factors, such as nerve growth factor, glial cell line-derived neurotrophic factor, and brain-derived neurotrophic factor (BDNF), into the tissue-engineered nerve grafts has been actively attempted in order to avoid the possible side effects and morbidity associated with cellular therapy [70–76]. However, growth factor therapy has fallen short of expectations because of the unpredictable side effects, unknown optimal dosage, and short half-life of the growth factors [5, 74, 75, 77].
5.4.3 Cell Sources for Peripheral Nerve Tissue Engineering
Due to the in vivo inefficiency of growth factors, some researchers implanted cells into the lumen of neural scaffolds in an attempt to provide a continuous supply of growth factors to the nerve defect area [35, 78–81]. Moreover, engineering a complete peripheral nerve is not possible without the in vitro culture and seeding of cells onto the nerve scaffolds. The major cell types used were as follows: Schwann cells, embryonic stem cells (ESCs), neural stem cells (NSCs), mesenchymal stem cells (MSCs), and induced pluripotent stem cells (iPSCs) (Table 5.1).
Table 5.1
Cell lines used as support cells for nerve tissue engineering
Cell type | Advantages | Disadvantages |
|---|---|---|
Schwann cells | Non-immunogenic; major role in myelination; secretes growth factors | Harvest and expansion are time consuming; harvest requires invasive procedures |
Olfactory ensheathing cells | Secrete neurotropic factors; participate in myelination; non-immunogenic | Limited survival in cell cultures; limited donor site |
Neural stem cells | Non-immunogenic; promote axonal regeneration | High risk of tumor formation in vivo |
Bone-marrow-derived stem cells | Easy expansion, non-immunogenic | Harvest requires invasive procedures; low stem cell yield |
Adipose-derived mesenchymal stem cells | Easy harvest and expansion; secret growth factors | Action of mechanism needs to be clarified; |
Skin-derived stem cells | Easy harvest and expansion; secret growth factors; non-immunogenic | Donor site morbidity |
These cell lines could be implanted into the scaffolds via direct injection or coculturing methods before or after in vitro differentiation [33, 82]. The markers for successful neural differentiation are as follows: GFAP, p75NGFR, and S-100 for Schwann cells [83–86] and nestin and NeuN [84, 87] for neural progenitor cells.
Schwann Cells
Schwann cells can be obtained from allogeneic, syngeneic, or autologous sources. Their function is to create a suitable environment for axonal growth by expressing cell adhesion molecules, secreting nerve growth factors, and forming an endoneurial myelin sheath that acts as a guide for the regenerating axons [3, 34]. This central role of Schwann cells in peripheral nerve regeneration made them the most commonly used cell type for experimental nerve tissue engineering applications. Schwann cell-enriched nerve scaffolds improved both the quality and rate of axonal regeneration in rat sciatic nerve defect model [88, 89]. However, among the obstacles in front of their clinical use are; suboptimal attachment of Schwann cells to the nerve scaffolds and the difficulties in obtaining and in vitro expansion of autologous Schwann cells [1, 81].
ESCs
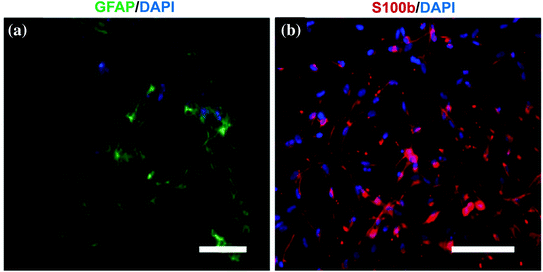
As an alternative to Schwann cells, ESCs can easily be expanded and have a great potential to proliferate and differentiate into neurons under various protocols (Fig. 5.5) [87, 90]. However, the ethical concerns on the use of ESCs for clinical applications limit their use.
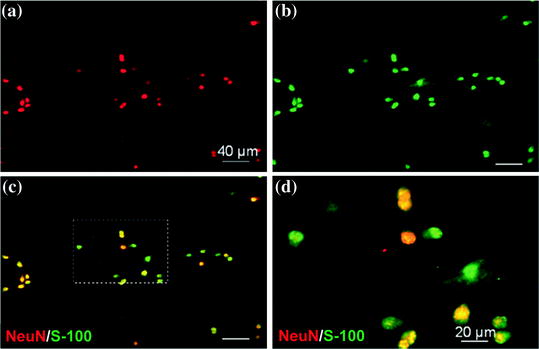

Fig. 5.5
Phenotype characterization of ESCs differentiated into neural progenitors. Confocal fluorescence microscopy imaging shows the cell phenotype 3 days after differentiation induction. Images demonstrate double staining of NeuN (red) (a) and S-100 (green) (b) in neural progenitor cells differentiated from ESCs. Merged image demonstrates the positivity of some of the cells for both NeuN and S-100, implying that these neural progenitors can further differentiate into either neuronal or glia/Schwann cells (c). A close-up view is seen in pane (d). Scale bars, 40 μm (a, b) and 20 μm (c, d). Adapted with permission from [87]
NSCs
Neural stem cells, just like ESCs, are multipotent, highly mobile, and can easily be isolated and cultured in vitro. These properties make NSCs an attractive alternative source of support cells for nerve tissue engineering [91–93].
MSCs
MSCs from various adult tissues (bone marrow, adipose tissue, etc.) became the subject of interest because of various advantages over the other cell lines. It is relatively easily to obtain MSCs through minimally invasive procedures such as the aspiration of the bone marrow or liposuction. MSCs can easily be expanded in a large scale by in vitro culture [94, 95].
Even though they do not possess the wide range of differentiation of ESCs, adult MSCs are multipotent, secrete growth factors and other soluble mediators, and moreover can serve as a vehicle for drug delivery and gene therapy [96]. Several in vitro studies have shown that MSCs can be induced to differentiate into neural lineages including Schwann cell-like cells (Fig. 5.6) [85, 97–101]; however, MSCs promote nerve regeneration not only by direct differentiation but also via spontaneous fusion with host cells and possibly by secreting growth factors [102, 103].
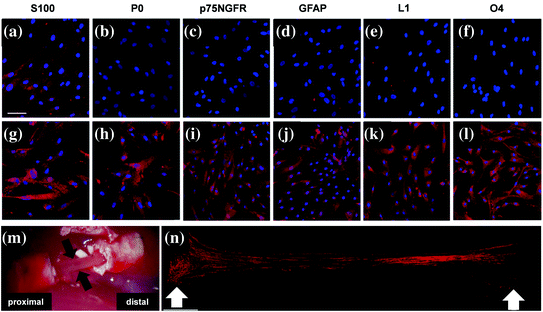

Fig. 5.6
Human MSCs can be differentiated into Schwann cell-like cells. This picture shows immunofluorescence staining for S100, P0, p75NGFR, GFAP, L1, O4 in human MSCs (a–f), and human MSC-derived Schwann cells (g–l). The untreated human MSCs slightly expressed S100 (a) but were negative for other Schwann cell markers before induction (b–f). However, after the induction, cells became positive for all the other Schwann cell markers and exhibited an increased immunoreactivity for S100 (g). Macroscopic view of the tissue within the nerve scaffold seeded with MSC-derived Schwann 3 weeks after transplantation of the scaffold into a rat sciatic nerve defect (black arrows) (m). Neurofilament-positive nerve fibers (red) observed in the newly formed tissue in the scaffold, white arrows mark the coaptation sites (n). Scale bar, 100 μm. Adapted with permission from [101]
iPSCs
The latest and maybe the most significant advancement in stem cell field is the reprogramming of adult somatic cells (e.g., skin fibroblasts) into pluripotent stem cells by the introduction of genes Oct3/4, Sox2, c-Myc, and KLF4 [104]. These cells are named iPSCs, and since they are derived from somatic cells, they bypass the immune system of the host. Similar to ESCs, iPSCs possess an unlimited expansion potential, and they can be differentiated into almost every tissue in human body yet without any of the ethical concerns surrounding ESCs (Fig. 5.7). However, iPSCs require a significantly more genetic manipulation than any other stem cell type during the induction process that subsequently leads to some safety concerns in clinical application [105].