The Pancreas
Muhammad B. Zaman
Fine-needle aspiration (FNA) biopsy has made a significant contribution to the preoperative and intraoperative diagnosis of a broad variety of space-occupying pancreatic abnormalities that may be benign or malignant. The most common lesions of the pancreas are listed in Table 39-1. Chief among the malignant lesions are ductal adenocarcinomas, which are largely incurable and often rapidly fatal (Gudjonsson, 1987; Warshaw and Swanson 1988; Henne-Bruns et al, 1998). It is not clear at this time whether cytologic techniques will contribute to an improved salvage rate for these patients. However, because of its safety and reliability, FNA of the pancreas has greatly reduced the need for exploratory laparotomies, which are not without the risk of operative morbidity and mortality (Ferrucci et al, 1979). Substantial savings in health care expenditures more than justify the use of the needle aspiration technique (Soudah et al, 1989; Alvarez et al, 1993; Chang et al, 1997). The practicing cytopathologist who interprets the FNA smears of a pancreatic tumor may be rewarded by the diagnosis of a benign condition or tumors that may be curable by surgical resection. Fortunately, these relatively rare tumors, which constitute approximately 10% of pancreatic neoplasms (an estimated 3,000 cases annually in the United States) are recognizable because of their unique cytomorphology.
INDICATIONS AND DIAGNOSTIC TECHNIQUES
The principal reason for investigating the pancreas is to differentiate between inflammatory and neoplastic space-occupying lesions, and, if the lesion is neoplastic, to determine whether it is amenable to effective treatment. The initial step in investigating a patient with a pancreatic lesion is to determine the serum amylase and lipase levels. These levels are usually markedly elevated in acute pancreatitis, but are only slightly elevated in chronic pancreatitis or in the presence of a pancreatic carcinoma. If a satisfactory diagnosis has not been achieved on the basis of clinical presentation and biochemistry, imaging studies of the pancreas are usually the next step in the evaluation of pancreatic disease.
The investigation of pancreatic lesions is based on a variety of imaging techniques, such as computed tomography (CT), ultrasound (US), endoscopic US (EUS), endoscopic
retrograde pancreatography, and angiography, and the use of radioactive tracer substances (e.g., 75 selenomethionine scans). If a space-occupying lesion is observed or suspected, the identity of the lesion must be further established. Cytologic techniques are currently the diagnostic methods of choice.
retrograde pancreatography, and angiography, and the use of radioactive tracer substances (e.g., 75 selenomethionine scans). If a space-occupying lesion is observed or suspected, the identity of the lesion must be further established. Cytologic techniques are currently the diagnostic methods of choice.
TABLE 39-1 COMMON LESIONS OF THE PANCREAS | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||
Methods of Securing Pancreatic Samples
There are four methods available to secure cytologic samples from the pancreas:
Duodenal lavage
Endoscopic cytologic sampling via the common bile duct, using US or retrograde pancreatography
Cytology of pancreatic juice
Transcutaneous, intraoperative, or EUS-guided FNA
The first two methods are described and discussed in Chapter 24, and only the cytology of FNA and pancreatic juice are discussed in this chapter.
Collection of Pancreatic Juice
In several studies, Japanese researchers investigated the diagnostic value of pancreatic juice, which is collected with or without papillotomy of the opening of the pancreatic ducts into the common bile duct. Their results were encouraging. Cytologic studies of the juice allowed for a better classification of mucin-producing tumors of the pancreas (Uehara et al, 1994), and for the occasional discovery of noninvasive intraductal carcinoma (Shimizu et al, 1999).
Techniques of Pancreatic Aspiration
The principal indication for FNA is the identification of a space-occupying solid or cystic lesion of the pancreas. In the latter situation, aspiration may serve as both a diagnostic and a therapeutic procedure. FNA is more likely to provide an accurate diagnosis compared to intraoperative core needle or wedge biopsy of the pancreas (Moossa et al, 1982; Saez et al, 1995).
There are three principal approaches to performing diagnostic FNA of the pancreas:
Percutaneous transabdominal aspiration under US or CT guidance. The success of this method depends on the skill of the operator in guiding the needle to the target area and obtaining an adequate sample. The adequacy of the sampling procedure may be determined at the bedside by rapid microscopic examination of aliquots of the samples by trained personnel. In our experience, adequate samples can be obtained from nearly all pancreatic cancers; however, in some cases the aspiration must be repeated.
EUS-guided FNA. This is a newer approach that can be utilized to sample small pancreatic carcinomas (Chang et al, 1997; Jhala et al, 2003). The procedure is tedious and requires operator experience and collaboration between the cytopathologist and endoscopist. It is now the standard of practice in large medical centers.
Direct visualization or palpation of the pancreas at the time of a laparotomy. This was the initial method used for cytologic investigations of the pancreas before reliable imaging techniques became available (Arnesjo et al, 1972). The advantage of this method is that it can precisely locate the pancreatic lesion with excellent accuracy and very few complications (Hastrup et al, 1977; Stormby, 1979; Alpern and Dekker, 1985; Soudah et al, 1989; Edoute et al, 1991; Blandamura et al, 1995; Saez et al, 1995). The samples are not contaminated by cellular material from serosal, gastric, or intestinal tissues.
Comparison of Intraoperative Aspiration With Tissue Biopsies
Intraoperative aspirations are probably more efficient and accurate than intraoperative wedge or needle core tissue biopsies with intraoperative consultation with frozen sections, which is occasionally done during exploratory laparotomies for pancreatic tumors, particularly when resection of the head of the pancreas is contemplated (Whipple procedure) (Saez et al, 1995; Robins et al, 1995). However, even the small, direct tissue biopsies are not free of the risk of significant morbidity and mortality, and they are not always easy to interpret (Ferucci et al, 1979; Beazley, 1981). The fear of causing acute pancreatitis and peritonitis resulting from the leakage of enzymes, and the difficulty in differentiating chronic pancreatitis from pancreatic carcinoma by gross examination at surgery often results in the acquisition of inadequate tissue samples. Crushing of the small biopsy sample may also create problems in interpretation.
Complications
There are rare reported complications of intraoperative FNA under visual guidance (Simms, 1982), and remarkably few complications of percutaneous FNA, even though the needle passes through a number of viscera, such as the large bowel, before reaching the pancreas. Apparently, the needle puncture sites are rapidly sealed. The complications that
have been reported include cases of acute pancreatitis, needle tract seeding of pancreatic carcinoma, and pancreatic fistula (McLoughlin et al, 1978; Ferrucci et al, 1979; Simms et al, 1982; Rashleigh-Belcher et al, 1986; Bergenfeldt et al, 1988; Fornari et al, 1989; Rosenbaum and Frost, 1990). A case of bile peritonitis was encountered in a patient with carcinoma of the head of pancreas and a distended Courvoisier gall bladder. The perforation was immediately apparent because of the presence of bile in the syringe; however, the patient recovered after laparotomy and treatment with antibiotics (Koss et al, 1992). The frequency of complications from FNA is much lower than that associated with incisional pancreatic biopsies.
have been reported include cases of acute pancreatitis, needle tract seeding of pancreatic carcinoma, and pancreatic fistula (McLoughlin et al, 1978; Ferrucci et al, 1979; Simms et al, 1982; Rashleigh-Belcher et al, 1986; Bergenfeldt et al, 1988; Fornari et al, 1989; Rosenbaum and Frost, 1990). A case of bile peritonitis was encountered in a patient with carcinoma of the head of pancreas and a distended Courvoisier gall bladder. The perforation was immediately apparent because of the presence of bile in the syringe; however, the patient recovered after laparotomy and treatment with antibiotics (Koss et al, 1992). The frequency of complications from FNA is much lower than that associated with incisional pancreatic biopsies.
ANATOMY, HISTOLOGY, AND CYTOLOGY OF THE NORMAL PANCREAS
Anatomy
The pancreas is a catfish-shaped, composite exocrine-endocrine gland that is located transversely in the upper abdomen behind the peritoneum. The gland itself weighs approximately 100 g, measures about 25 × 9 × 4 cm, and is posterior to the body and antrum of the stomach and the transverse colon (see Fig. 24-1). The pancreas is arbitrarily divided into three parts: the head, body, and tail. The head is nestled in the duodenal C loop behind the liver, and the tail extends to the hilus of the spleen (see Chap. 24). The left kidney is immediately posterior to the tail. With so many structures in close proximity, it is clear that when one attempts to reach a pancreatic lesion through the anterior abdominal wall, the needle may sample mesothelial cells from the peritoneum and normal cells from any of the above mentioned organs. Ectopic pancreases may be observed and sometimes may cause diagnostic problems (Sams et al, 1990).
The bulk of the pancreas is formed by the exocrine component, namely the acini, the ducts, and the corresponding blood vessels. The endocrine component, which is comprised of more than a million islets of Langerhans with an aggregate weight of about 1 g, constitutes about 2% of the pancreatic mass. The remaining approximately 10% of the pancreas is formed by the extracellular matrix (Gorelick and Jamieson, 1981).
Histology
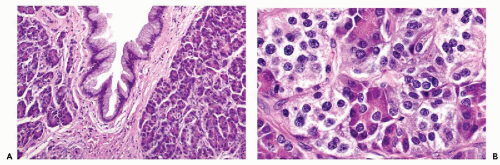
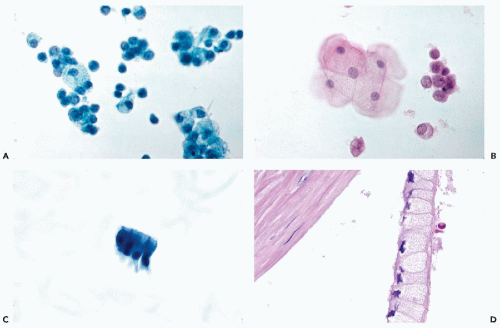
The histology of the exocrine pancreas is similar to that of the salivary glands. The exocrine pancreas is composed of morphologic units called lobules, which are made up of numerous spheroidal acini (Fig. 39-1A). The lobules are separated by a thin fibroconnective and vascular stroma. The acini produce digestive enzymes, such as trypsinogen, chymotrypsin, lipase, and elastase. These enzymes drain into the duodenum by a complex duct system. The smallest intralobular ducts, which are lined by a flattened epithelium, continue into the interlobular ducts with cuboidal epithelium, and then into the main excretory ducts, which are lined by columnar cells with interspersed goblet cells (Fig. 39-1A). The ductal system is embryologically related to the extrahepatic biliary tract, and is morphologically similar to the bile duct and gall bladder epithelium (see Chap. 24).
The pancreatic acini are lined by pyramid-shaped cells surrounding a central small lumen. They have basally placed round nuclei, and the pink to purple cytoplasm contains variable numbers of acidophilic zymogen granules (Fig. 39-1B).
The richly vascularized islets of Langerhans are dispersed throughout the whole organ; however, most are located in the tail of the pancreas. The islets are composed of small polyhedral cells arranged in nests and separated by capillaries. The cytoplasm of the islet cells is transparent; therefore, they are seen in histologic sections as a collection
of small clear cells, approximately five times the size of an acinus (Fig. 39-1B). The islets do not maintain communication with the acinar or ductal system, and they secrete their products directly into the bloodstream. Depending on the hormones produced, the islet cells may be classified as α cells (which produce glucagon), β cells (insulin), δ cells (somatostatin), G1 cells (gastrin), and PP cells (polypeptide). These cell types are identified on the basis of immunohistochemistry or electron microscopy (Mukai et al, 1982). Regardless of type of cell involved, tumors derived from islet cells, known as pancreatic endocrine neoplasms, are morphologically similar to each other.
of small clear cells, approximately five times the size of an acinus (Fig. 39-1B). The islets do not maintain communication with the acinar or ductal system, and they secrete their products directly into the bloodstream. Depending on the hormones produced, the islet cells may be classified as α cells (which produce glucagon), β cells (insulin), δ cells (somatostatin), G1 cells (gastrin), and PP cells (polypeptide). These cell types are identified on the basis of immunohistochemistry or electron microscopy (Mukai et al, 1982). Regardless of type of cell involved, tumors derived from islet cells, known as pancreatic endocrine neoplasms, are morphologically similar to each other.
Cytology
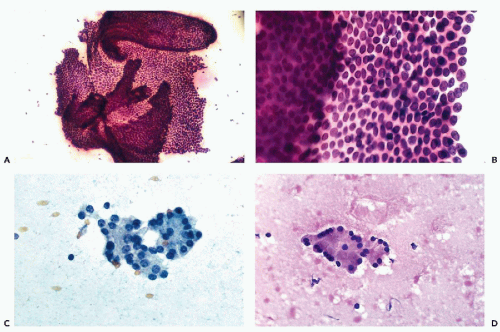
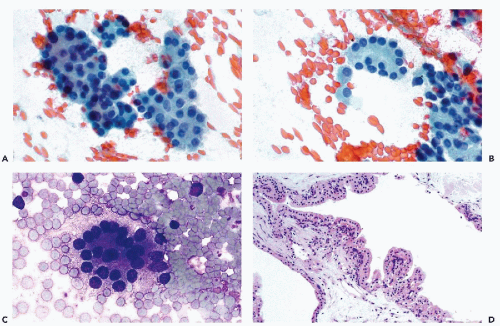
Normal pancreas is never deliberately aspirated, and information regarding its cytologic make-up is obtained mainly from aspirates of very small lesions that have missed the target. Although the normal exocrine pancreas is composed primarily of acini, the dominant cells in FNA smears are ductal epithelial cells. Normal acinar cells and islet cells are rarely seen (see below). The percutaneous transabdominal pancreatic aspirates may also contain mesothelial cells, epithelial cells of gastric or intestinal origin, occasionally hepatocytes, and, very rarely, ganglion cells (Fig. 39-2A-D).
Epithelial Duct Cells
Benign ductal cells of the pancreas appear as sheets of uniform cuboidal cells forming monolayers of various sizes with centrally placed small nuclei (see Fig. 39-1C). At the edge of clusters derived from large ducts, the cells may be columnar in configuration, parallel to each other, with basally placed nuclei. The cytoplasmic borders are well demarcated and the cytoplasm is transparent in fixed smears stained with Papanicolaou stain, accounting for the honeycomb pattern of the flat clusters (Fig. 39-2B). Cytoplasmic vacuoles or goblet cells are very rarely seen. The nuclei are round or slightly ovoid, and equally spaced, and there are no nuclear contour abnormalities. The chromatin is finely granular and uniformly distributed. Tiny nucleoli may be visible.
Occasionally, large areas of the ductal epithelium are aspirated (Fig. 39-2A). The smear may show very large flat sheets of benign glandular cells that may fold at the edges. Occasionally, the ductal cells form thick, multilayered sheets with crowding of nuclei. Such clusters may be a cause for concern because this may represent a well-differentiated ductal adenocarcinoma. Examination of the edges of such thick clusters usually allows for a close examination of the nuclei that fail to show the nucleolar abnormalities characteristic
of cancer. The absence of single cancer cells is another reason to regard such clusters as benign.
of cancer. The absence of single cancer cells is another reason to regard such clusters as benign.
Acinar Cells
The acinar cells usually form small, tightly knit clusters, often arranged in spherical, three-dimensional structures. Higher magnification reveals the pyramidal shape of cells arranged around a central narrow lumen, resembling rosettes, with the nuclei aligned at the periphery of the cells (Fig. 39-2C,D). The cytoplasm is relatively abundant, and in air-dried or well-fixed cells it may show prominent coarse cytoplasmic granularity imparted by the zymogen granules, which are precursors of digestive enzymes. In most cases, however, the granules are lost, and the cytoplasm of the acinar cells appears clear or vacuolated (Hastrup, 1977). The nuclei are uniform and small and round, with a smooth membrane, and are located away from the acinar lumen. The chromatin is granular and evenly distributed, and the nucleoli are inconspicuous.
Islet Cells
In general, the islet cells cannot be recognized in FNA smears without the use of immunohistochemical markers for endocrine function. In a fortuitous case of pancreatic FNA performed in a young male patient with sclerosing cholangitis and total atrophy of the exocrine pancreas, the aspirated islet cells formed loose, approximately spherical or oval aggregates of cells with eosinophilic cytoplasm (Fig. 39-3A). The cell borders were indistinct, and in this material cytoplasmic granules were not visible. The regular round nuclei with minimal pleomorphism were similar in size to the acinar cell nuclei. The nuclear chromatin may show an endocrine “salt and pepper” granularity, with a visible nucleolus. In this particular case, the identity of the cells was confirmed at autopsy (Fig. 39-3B). It must be reemphasized that this presentation of islet cells is exceptional and is not likely to be duplicated in practice.
Other Benign Cells
Large sheets of benign glandular cells may be aspirated from gastric or intestinal (mainly colonic) mucosa. However, benign gastric mucosal cells are identical to pancreatic ductal epithelium (Fig. 39-4A). The origin of the sample can be identified if parietal gastric cells, which have a finely granular and intensely eosinophilic cytoplasm, are present. The presence of goblet cells identifies intestinal epithelium (Fig. 39-4B).
The mesothelial cells of peritoneal origin typically occur in monolayer sheets, in which the cells are separated from each other by wide spaces or windows. These cells have notoriously large, readily visible nucleoli, and therefore may be confused with cancer cells (see Fig. 38-1D; for further discussion and illustrations of these cells, see Chap. 25). Large hepatocytes, occurring singly or in small sheets, may be present in smears if on its way to the pancreas the needle passes through the left liver lobe (see Fig. 39-4C). Their morphology is described in Chapter 38. Capillary vessels, connective tissue cells, and sometimes striated muscle and fat cells may also occur in pancreatic FNA smears. Exceptionally, when the needle penetrates beyond the pancreas and reaches sympathetic ganglia, ganglion cells may be observed (Fig. 39-4D).
INFLAMMATORY LESIONS: PANCREATITIS
The term pancreatitis is used to describe injury to the exocrine component of the pancreas, primarily the acini, with resulting release of digestive enzymes, lipase, elastase, and proteases, leading to the autodigestion of the pancreas and the surrounding tissues. This results in a hemorrhagic necrosis and inflammation of the affected tissues, mainly fat necrosis. The combination of the necrotic fat with calcium salts may result in calcific soap formation, which is readily visible as white granules within the peripancreatic fat.
The known predisposing factors are gallstones with associated obstruction and infection, alcoholism, trauma (including abdominal surgery), and sometimes malignant tumors of the pancreas. Less often, drugs and gastric ulcers
may also be associated with pancreatitis (Marshall, 1993). The pathogenesis of pancreatitis is poorly understood. The formation of pseudocysts may be a consequence of pancreatitis (see below).
may also be associated with pancreatitis (Marshall, 1993). The pathogenesis of pancreatitis is poorly understood. The formation of pseudocysts may be a consequence of pancreatitis (see below).
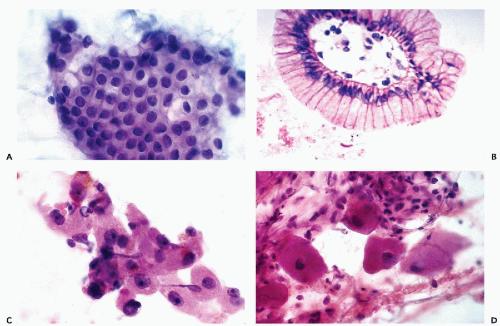 Figure 39-4 Other benign cells observed in pancreatic aspirates. A. Gastric epithelial mucus-producing cells are larger than normal ductal cells and have sharper cytoplasmic borders. Lesions of the body of pancreas, aspirated through the posterior gastric wall by an endoscopic ultrasound (EUS)-guided needle, may show an abundance of such cells, causing diagnostic difficulty. Compare with the illustrations of low-grade mucinous cystic tumors in Figure 39-8. B. Colonic epithelium. A strip of colonic mucosa that is rich in pale goblet cells and may resemble mucinous cystadenoma. C. Hepatocytes are occasionally derived from the left lobe of the liver. They show dense pink cytoplasm and prominent nucleoli. D. Ganglion cells, derived from sympathetic ganglia, may be occasionally seen in aspirations of the pancreas. |
Acute Pancreatitis
Acute pancreatitis may range from a mild and self-limited disorder to a severe, debilitating, even fatal disease. Classically, there is an abrupt onset of severe abdominal pain radiating to the back, with a marked elevation of serum amylase and lipase levels. The episode is often precipitated by excessive alcohol or food consumption. The diagnosis is usually clinically obvious and can be confirmed by biochemical analysis of the serum or, by contrast, enhanced dynamic pancreatography, a refined CT technique that can detect pancreatic necrosis and its extent. This imaging procedure correlates with the severity of the pancreatitis and other complications (Marshall, 1993). Therefore, percutaneous FNA biopsy is usually not needed and is seldom performed.
The limited information available regarding the cytology of acute pancreatitis comes from intraoperative needle aspirations obtained during exploratory laparotomies. Arnejo et al (1972), Hastrup et al (1978), and Frias-Hidvegi (1988) reported the presence of inflammatory cells, macrophages, lipophages, and elements of normal pancreas (mainly ductal cells). The presence of calcific debris from areas of fat necrosis has also been reported.
Subacute and Chronic Pancreatitis
Subacute or chronic pancreatitis is usually the sequela of an acute pancreatitis, but it may also occur as a primary disorder. The patients have chronic recurrent abdominal pain. The levels of serum amylase and lipase may or may not be elevated. Late in the course of the disease, pancreatic insufficiency in the form of steatorrhea, weight loss, and diabetes mellitus may develop. Chronic pancreatitis may also cause bile duct obstruction and jaundice; therefore, it may be mistaken clinically for a pancreatic carcinoma.
Histology
In tissue sections, chronic pancreatitis is characterized by variable degrees of fibrosis, acinar atrophy, and ductal hyperplasia. As a result of atrophy of the exocrine pancreas, the islets of Langerhans are readily visible and appear larger than normal. The stroma shows an infiltrate composed of lymphocytes and plasma cells. The epithelium of the dilated and distorted ducts may show hyperplasia with some atypia, and, rarely, squamous metaplasia in the smaller ducts. The duct lumens may be filled with protein plugs. In the end stage of the disease, the ducts may be distorted by diffuse fibrosis and may show mucinous metaplasia, thus mimicking a well-differentiated adenocarcinoma of the scirrhous type (Fig. 39-5A). Fat necrosis with calcium deposits is commonly observed at the periphery of the pancreatic tissue. At laparotomy, the pancreas may be rock-hard on palpation, grossly mimicking infiltrating carcinoma. It can be a formidable problem to correctly interpret needle core biopsies by frozen sections. Cytologic studies may help in difficult situations.
Cytology
The FNA smears of subacute and chronic pancreatitis vary in cellularity and cell composition, but, contrary to panorcetic cancer, are usually scanty. On screening magnification, the smears typically reveal a polymorphous picture of variably sized and configured clusters of ductal cells, occasional acinar cells, macrophages and other inflammatory cells, connective tissue fragments, granulation tissue, mucus, calcification, and debris (Fig. 39-5B,C). Not all of these components are seen in a given case. The number and type of inflammatory cells present depend on the stage of the disease: In subacute pancreatitis, neutrophils and macrophages predominate. In chronic pancreatitis, lymphocytes, macrophages, and plasma cells predominate. In chronic fibrosing pancreatitis, the FNA smears may have a relatively clean background.
Problems with interpreting pancreatic material in pancreatitis are related to the presence of hyperplastic and atypical ductal cells, which may occur singly or form disorderly clusters (Fig. 39-5D). In some (fortunately very uncommon) cases, nuclear atypia in the form of nuclear enlargement and clearly visible but not very large nucleoli makes it extremely difficult to differentiate chronic pancreatitis from well-differentiated adenocarcinoma. The presence of an inflammatory infiltrate in the background of the smear should strongly suggest the use of caution before interpreting such smears as malignant. The
important factors in the differential diagnosis of inflammatory atypia of ductal cells versus well-differentiated ductal carcinoma are discussed below in reference to carcinoma, and are summarized in Table 39-3 (see below). An additional confounding factor is that ductal carcinoma and chronic fibrosing pancreatitis may coexist and may account for at least some of the false-negative diagnoses on FNA of this organ.
important factors in the differential diagnosis of inflammatory atypia of ductal cells versus well-differentiated ductal carcinoma are discussed below in reference to carcinoma, and are summarized in Table 39-3 (see below). An additional confounding factor is that ductal carcinoma and chronic fibrosing pancreatitis may coexist and may account for at least some of the false-negative diagnoses on FNA of this organ.
NONNEOPLASTIC CYSTIC LESIONS
Pancreatic cysts represent a broad variety of lesions that may be acquired or congenital, benign or malignant. Rarely, some of the otherwise solid tumors, such as pancreatic duct carcinoma or endocrine tumor, may also be wholly or partially cystic. In such cases, the aspiration of the cystic fluid should be supplemented by an aspiration of the cyst wall to reach the correct diagnosis. Several major recent surveys of FNA cytology of various cysts have documented the diversity of the targets and the problems encountered in identifying them (Koss et al, 1992; Laucirica et al, 1992; Centano et al, 1997).
Acquired Cysts
Pancreatic Pseudocysts
The most common type of acquired nonneoplastic cysts is the pancreatic pseudocyst, which follows the destruction and necrosis of pancreatic tissue secondary to pancreatitis. Patients usually present with jaundice, pain, weight loss, nausea, and vomiting. Pseudocysts are usually solitary and unilocular, and while their cystic nature may be determined by CT or US, it is not possible to exclude a cystic neoplasm based on imaging alone. Therefore, the nature of the cyst should be determined by cytologic examination of the cyst contents.
Histology
The thick cyst wall consists of reactive fibrous tissue without epithelial lining (hence the name pseudocyst). Initially, most pseudocysts are small, but continued pancreatic secretion and destruction of tissues may lead to very large symptomatic and even palpable cystic masses containing several hundred cubic centimeters of fluid.
Cytology
Aspiration of fluid from a pseudocyst is often of diagnostic and therapeutic value. After the fluid is aspirated, the cyst may collapse and the patient will experience immediate symptomatic relief. Often there is no reaccumulation of the fluid.
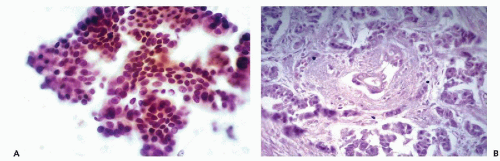
The cyst fluid may appear clear, straw-colored, brown, turbid, or frankly hemorrhagic. The sediment is sometimes acellular but most often contains large numbers of macrophages and some lymphocytes in a background of necrotic debris (Fig. 39-6A). Less often, there are multinucleated macrophages containing ingested hemosiderin and debris, and a very few atypical spindly fibroblasts with enlarged nuclei and prominent nucleoli originating in the capsule of the pseudocyst. The latter can be mistaken for a spindle cell malignant tumor. The small number of such cells, and the typical cystic background of the smear should prevent such errors. Epithelial cells are absent. If epithelial cells are present, the lesion is not likely to be a pseudocyst. A portion of the cyst fluid should always be submitted for analysis of amylase content, that is high in pseudocyst, and carcinoembryonic antigen (CEA) level, which is high in neoplastic cysts but low in pseudocysts (Pinto and Meriano, 1991; Lewandrowski et al, 1993; Centeno et al, 1997).
Lymphoepithelial Cysts
These are uncommon cysts that are similar to branchial cleft cysts in the neck (see Chapter 30) and are often lined by squamous epithelium with lymphocytic deposits in the wall containing pasty, whitish material. There are a few case reports describing the cytologic findings (Centeno et al, 1999; Liu et al, 1999; Mandavilli et al, 1999). The presence of squamous cells, squamous and amorphous debris, and cholesterol crystals has been described as characteristic of the entity (Fig. 39-6B). Centeno et al (1999) reported elevated levels of CEA, CA 125, and amylase in the viscous fluid aspirated from one such cyst.
Congenital Pancreatic Cysts
Congenital pancreatic cysts are rare, true cysts lined by pancreatic ductal epithelium. They can be solitary (thought to result from abnormal development of a duct) or multiple (associated with inherited polycystic diseases). The capsule is thin and lined by a single layer of cuboidal, rarely columnar, or flat epithelial cells, some of which show evidence of mucus formation (Fig. 39-6D).
The aspirated fluid is usually clear and mucoid, and the smears are sparsely cellular. Careful screening usually reveals a few benign cuboidal or columnar epithelial cells from the lining of the cyst (Fig. 39-6C). If such cells are numerous, the possibility of a mucinous cystic tumor cannot be ruled out (see below).
BENIGN AND MALIGNANT NEOPLASTIC CYSTIC LESIONS
Serous Cystadenoma
A serous cystadenoma is a benign epithelial neoplasm composed of numerous small cysts lined by uniform glycogen-rich cuboidal cells, sometimes disposed around a central stellate scar. The lesions are also known as microcystic adenomas or glycogen-rich adenomas (Compagno and Oertel, 1978a; Alpert et al, 1988). They constitute 1% to 2% of the exocrine pancreatic tumors, and are of unknown
etiology and pathogenesis. These tumors occur predominantly in elderly women (mean age of 66 years), but they may also occur in men, and are usually, but not always, located in the body and tail of the pancreas (Compagno and Oertel, 1978; Albores-Saavedra, 1990). A macrocystic variant of this tumor was described by Lewandrowski et al (1992). The tumor can be asymptomatic and incidently found during abdominal examination for unrelated disease, or it may cause nonspecific symptoms related to its bulk, such as abdominal pain, nausea, and vomiting. Grossly, these tumors are well-circumscribed and large, with an average diameter of 10 cm. The cut surface has a characteristic sponge-like appearance due to the presence of numerous cysts of variable size (hence the name microcystic adenoma).
etiology and pathogenesis. These tumors occur predominantly in elderly women (mean age of 66 years), but they may also occur in men, and are usually, but not always, located in the body and tail of the pancreas (Compagno and Oertel, 1978; Albores-Saavedra, 1990). A macrocystic variant of this tumor was described by Lewandrowski et al (1992). The tumor can be asymptomatic and incidently found during abdominal examination for unrelated disease, or it may cause nonspecific symptoms related to its bulk, such as abdominal pain, nausea, and vomiting. Grossly, these tumors are well-circumscribed and large, with an average diameter of 10 cm. The cut surface has a characteristic sponge-like appearance due to the presence of numerous cysts of variable size (hence the name microcystic adenoma).
Histology
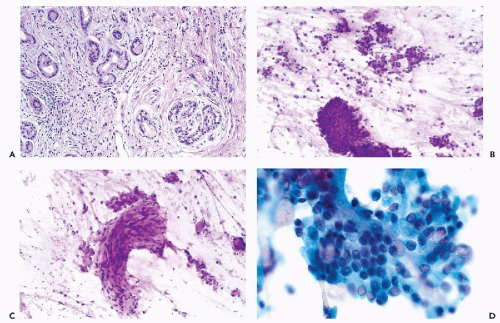
The cysts are lined by a single layer of uniform cuboidal or flattened epithelial cells with clear glycogen-containing cytoplasm, and may show intracystic papillation, usually without a fibrovascular core (Fig. 39-7D). The central stellate scar is formed by fibrous, hyalinized tissue with a few clusters of tiny cysts.
Cytology
The aspirate yields small amounts of clear, watery fluid. The cellularity of the smear is variable and may be abundant in a vigorous aspirate (Fig. 39-7A-C). At low magnification, the smear gives the impression of numerous, tightly cohesive normal acini, which actually represent the microcysts and small intracystic papillae. At high magnification, the small papillary groups are made up of uniform small cells with transparent, well-delineated cytoplasm. Some flat sheets of epithelial cells of variable sizes may be encountered; however, a perfect honeycomb pattern, as found in sheets of benign ductal cells, is not seen. Periodic acid-Schiff (PAS) stain is positive. The nuclei are spherical and show very little, if any, pleomorphism or atypia. Similar findings, with slight variations in cytologic presentation, were reported by Hittmair et al (1991) and Nguyen and Vogelsang (1993).
A malignant counterpart—serous cystadenocarcinoma—has been reported (George et al, 1989). We have
no first-hand experience with such a lesion in aspiration smears.
no first-hand experience with such a lesion in aspiration smears.
Mucinous Cystic Tumors
Clinical Data and Histology
Mucinous cystic tumors are uncommon pancreatic neoplasms, representing approximately 2% to 5% of all exocrine pancreatic tumors (Thompson et al, 1999). More than 500 cases of this tumor have been reported (Compagno and Oertel, 1978b; Zamboni et al, 1999). The tumors are composed of cysts lined by columnar, mucin-producing epithelium supported by ovarian-type stroma. They are subclassified as adenoma, borderline malignant, or noninvasive or invasive carcinoma according to the level of abnormality of the epithelium, which may form single or multiple layers or papillary projections (Fig. 39-8D). There are many similarities between this pancreatic tumor and its ovarian equivalent (see Chap. 15). Mucinous cystic tumors of pancreas occur almost exclusively in middle-aged women (Zamboni, 1999) in the body or tail of the pancreas. The smaller tumors may be an incidental finding, whereas larger tumors may produce nonspecific symptoms of pressure and diabetes mellitus. Malignant mucinous cystic tumors are often slow-growing and have a much better prognosis than pancreatic ductal carcinoma (Warshaw, 1990). No deaths from cancer have been observed in patients classified as benign or borderline malignant (Warshaw et al, 2003). Thus, the recognition and accurate classification of the cystic tumors is of great clinical value.
Cytology
The characteristic feature of fine-needle aspirates of mucinous cystic tumors is the presence of a grossly obvious clear mucoid material that forms the background of the smears. Mucus is much easier to recognize as a stringy, purple substance in air-dried smears, stained with hematologic stains, than as a faintly pink background in fixed smears processed with Papanicolaou’s stain. If the cyst is large, the cell content of the smear may be very low. Smaller cysts yield numerous epithelial cells. In the benign and borderline mucinous cystic tumors, the cells form flat sheets with a “honeycomb” pattern closely resembling normal duct epithelium in a background of mucus (Fig. 38-8A). In other cases, sheets of goblet cells, floating in mucin, may be observed (Fig. 39-8B). As a point of differential diagnosis, occasional goblet cells may occur in aspirates from chronic pancreatitis, but they are never numerous and the smears do not have a mucinous background. In borderline tumors, small clusters and single cells with visible nucleoli are common (Fig. 39-8C). Furthermore,
careful scrutiny of smears from high-grade mucinous cystic tumors reveals papillary or three-dimensional groups with obvious nuclear atypia. Frankly malignant tumors of this category are classified as mucinous adenocarcinoma (see Fig. 39-16A,B) and are cytologically identical to colloid (mucinous noncystic carcinoma; see below). A positive mucin stain excludes serous cystadenoma (Table 39-2). Rubin et al (1994) reported that benign and malignant mucinous cystic neoplasms can be differentiated by the level of the tumor marker CA 15.3 in the cyst fluid.
careful scrutiny of smears from high-grade mucinous cystic tumors reveals papillary or three-dimensional groups with obvious nuclear atypia. Frankly malignant tumors of this category are classified as mucinous adenocarcinoma (see Fig. 39-16A,B) and are cytologically identical to colloid (mucinous noncystic carcinoma; see below). A positive mucin stain excludes serous cystadenoma (Table 39-2). Rubin et al (1994) reported that benign and malignant mucinous cystic neoplasms can be differentiated by the level of the tumor marker CA 15.3 in the cyst fluid.
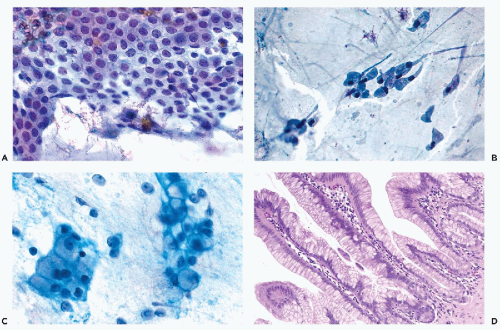 Figure 39-8 Neoplastic cysts of the pancreas. A. Mucinous cystadenoma. A large sheet of epithelial cells in a honeycomb-like arrangement is shown on a background of thick, clear mucus. No nuclear abnormalities or nucleoli are seen. Compare with normal pancreatic ductal cells and gastric mucus cells (Figs. 39-2B and 39-4A). B. Dispersed goblet cells, elongated by the smearing process, in a background of mucus. C. Borderline intraductal papillary mucinous tumor. The smear shows numerous mucus-producing cells. An occasional nucleolus can be noted. D.
Get Clinical Tree app for offline access
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree


|