Chapter 17 The Female Genital Tract and Breasts
INFECTIONS
5 Name the main viral pathogens causing infections of the lower female genital tract (vulva, vagina, and cervix)
 HSV-2: It causes vesicles, which coalesce and ulcerate. These vesicles appear 3 to 7 days after the infecting intercourse but in only approximately 30% of infected women. Ulcers may persist for 1 to 3 weeks but heal spontaneously without scarring. The virus migrates along the nerves to the lumbar ganglia, where it remains in a content form forever. The virus can be reactivated and descend to the vulva to produce recurrent vesicles.
HSV-2: It causes vesicles, which coalesce and ulcerate. These vesicles appear 3 to 7 days after the infecting intercourse but in only approximately 30% of infected women. Ulcers may persist for 1 to 3 weeks but heal spontaneously without scarring. The virus migrates along the nerves to the lumbar ganglia, where it remains in a content form forever. The virus can be reactivated and descend to the vulva to produce recurrent vesicles.6 What are the most common causes of vaginitis?
 Candida albicans: This fungus is found in the vagina of approximately 10% of women, who are usually unaware of the infection. Pregnancy, oral contraceptives, and diabetes promote fungal growth and cause the appearance of white patches on the mucosal surface and increased vaginal discharge with itching. Diagnosis is best made microscopically on wet mounts or Pap smear.
Candida albicans: This fungus is found in the vagina of approximately 10% of women, who are usually unaware of the infection. Pregnancy, oral contraceptives, and diabetes promote fungal growth and cause the appearance of white patches on the mucosal surface and increased vaginal discharge with itching. Diagnosis is best made microscopically on wet mounts or Pap smear. Trichomonas vaginalis: These flagellated protozoa are best diagnosed microscopically in freshly prepared wet mounts (i.e., smears of unfixed vaginal discharge, in which the protozoa keep moving). T. vaginalis is also visible on routine Pap smear.
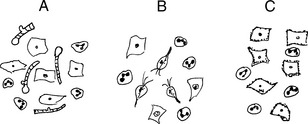
Trichomonas vaginalis: These flagellated protozoa are best diagnosed microscopically in freshly prepared wet mounts (i.e., smears of unfixed vaginal discharge, in which the protozoa keep moving). T. vaginalis is also visible on routine Pap smear. Gardnerella vaginalis: This microbe grows in the vagina. The infection is best recognized on Pap smear by the appearance of “clue cells” (i.e., squamous cells covered with bacilli). Cytologic findings in various forms of vaginitis are illustrated in Fig. 17-1.
Gardnerella vaginalis: This microbe grows in the vagina. The infection is best recognized on Pap smear by the appearance of “clue cells” (i.e., squamous cells covered with bacilli). Cytologic findings in various forms of vaginitis are illustrated in Fig. 17-1.VULVA AND VAGINA
11 What is vulvar leukoplakia?
 Lichen sclerosus, a disease of unknown etiology characterized by atrophy of the epidermis and sclerosing fibrosis of the underlying dermis
Lichen sclerosus, a disease of unknown etiology characterized by atrophy of the epidermis and sclerosing fibrosis of the underlying dermis17 How common is carcinoma of the vagina?
Carcinoma of the vagina is rare, accounting for 1% of all female genital cancers.
CERVIX
28 How does invasive cervical carcinoma present on gross examination?
On gross examination, invasive cervical carcinoma may present in three forms:
BODY OF THE UTERUS
33 What are the causes of dysfunctional uterine bleeding?
 Failure of ovulation: The normal menstrual cycle consists of a proliferative and a nonproliferative stage, separated by the midcyclic point of ovulation. At ovulation, the endometrium ceases to proliferate and enters a nonproliferative secretory phase. If ovulation does not occur, the endometrium will continue to proliferate until it outgrows its own blood supply. At that point, the surface portion of the endometrium becomes ischemic and starts dying off. This leads to bleeding, which typically occurs 2 or 3 weeks after the date of the “missed menstruation.” Anovulatory bleeding occurs most often around puberty and menopause but may be caused by neuroendocrine disturbances (e.g., anxiety and anorexia nervosa), severe malnutrition, or debilitating diseases.
Failure of ovulation: The normal menstrual cycle consists of a proliferative and a nonproliferative stage, separated by the midcyclic point of ovulation. At ovulation, the endometrium ceases to proliferate and enters a nonproliferative secretory phase. If ovulation does not occur, the endometrium will continue to proliferate until it outgrows its own blood supply. At that point, the surface portion of the endometrium becomes ischemic and starts dying off. This leads to bleeding, which typically occurs 2 or 3 weeks after the date of the “missed menstruation.” Anovulatory bleeding occurs most often around puberty and menopause but may be caused by neuroendocrine disturbances (e.g., anxiety and anorexia nervosa), severe malnutrition, or debilitating diseases. Luteal phase inadequacy: If the corpus luteum does not secrete progesterone in adequate amounts, the level of progesterone may not be sufficient to transform fully the proliferative endometrium into a secretory endometrium. Such irregular maturation of the endometrium is usually associated with “spotting,” premature onset of menstrual bleeding, or prolonged bleeding. Curetting of endometrium performed to determine the cause of bleeding often may also help reestablish the normal cycle. In many cases, irregular bleeding recurs; such women may have problems conceiving.
Luteal phase inadequacy: If the corpus luteum does not secrete progesterone in adequate amounts, the level of progesterone may not be sufficient to transform fully the proliferative endometrium into a secretory endometrium. Such irregular maturation of the endometrium is usually associated with “spotting,” premature onset of menstrual bleeding, or prolonged bleeding. Curetting of endometrium performed to determine the cause of bleeding often may also help reestablish the normal cycle. In many cases, irregular bleeding recurs; such women may have problems conceiving.34 What is endometrial hyperplasia?
35 Discuss how endometrial hyperplasia is diagnosed
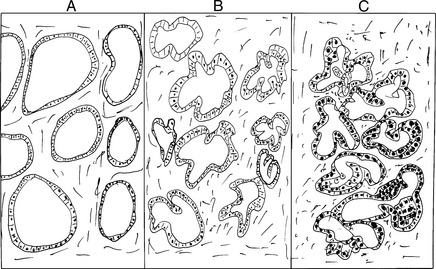
Endometrial hyperplasia is diagnosed microscopically by the pathologist examining the tissue removed by endometrial biopsy or endometrial curettage. Histologically, there are three forms of endometrial hyperplasia, as shown in Fig. 17-2:
 Complex hyperplasia without atypia: The glands appear crowded and are surrounded by relatively scant stroma. The glandular epithelium are lined by uniform cells, which show no nuclear atypia.
Complex hyperplasia without atypia: The glands appear crowded and are surrounded by relatively scant stroma. The glandular epithelium are lined by uniform cells, which show no nuclear atypia. Complex hyperplasia with atypia: The glands appear crowded and have an irregular shape, with stratification of cells that often protrude into the lumen. Nuclei of these glands show atypia, and sometimes it is not possible to distinguish them from well-differentiated adenocarcinoma. Some pathologists call it endometrial intraepithelial neoplasia.
Complex hyperplasia with atypia: The glands appear crowded and have an irregular shape, with stratification of cells that often protrude into the lumen. Nuclei of these glands show atypia, and sometimes it is not possible to distinguish them from well-differentiated adenocarcinoma. Some pathologists call it endometrial intraepithelial neoplasia.< div class='tao-gold-member'>
Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree