PREVENTION AND ERADICATION OF POLIOVIRUS
![]() (See also Chap. 148) After a peak of 57,879 cases of poliomyelitis in the United States in 1952, the introduction of IPV in 1955 and of OPV in 1961 ultimately eradicated disease due to wild-type poliovirus in the Western Hemisphere. Such disease has not been documented in the United States since 1979, when cases occurred among religious groups who had declined immunization. In the Western Hemisphere, paralysis due to wild-type poliovirus was last documented in 1991.
(See also Chap. 148) After a peak of 57,879 cases of poliomyelitis in the United States in 1952, the introduction of IPV in 1955 and of OPV in 1961 ultimately eradicated disease due to wild-type poliovirus in the Western Hemisphere. Such disease has not been documented in the United States since 1979, when cases occurred among religious groups who had declined immunization. In the Western Hemisphere, paralysis due to wild-type poliovirus was last documented in 1991.
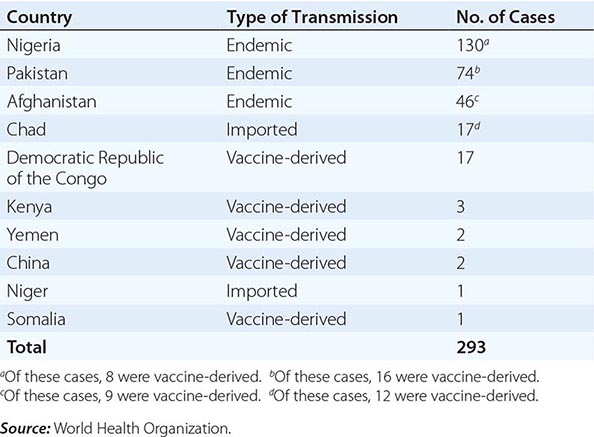
In 1988, the World Health Organization adopted a resolution to eradicate poliomyelitis by the year 2000. From 1988 to 2001, the number of cases worldwide decreased by >99%, with only 496 confirmed cases reported in 2001. Wild-type poliovirus type 2 has not been detected in the world since 1999. The Americas were certified free of indigenous wild-type poliovirus transmission in 1994, the Western Pacific Region in 2000, and the European Region in 2002. However, in 2002, there were 1922 cases of polio, with 1600 cases reported in India. In fact, after the nadir of 496 cases in 2001, 21 countries that had previously been free of polio reported cases imported from 6 polio-endemic countries in 2002–2005. By 2006, polio transmission had been reduced in most of these 21 countries. In 2012, 293 cases of polio were reported (the lowest number ever in a 1-year period); 85% were from Nigeria, Pakistan, and Afghanistan, the only countries where polio remains endemic (Table 228-2). As of November 2013, there had been 390 cases of polio in 2013 compared with 293 cases in 2012. The increase was associated with a marked rise in imported cases, including more than 180 cases in Somalia, more than 10 cases each in Kenya and Syria, and cases in Cameroon and Ethiopia. Also in 2013, wild-type poliovirus was detected in sewage in Israel, prompting a massive vaccination campaign with OPV. As of November 2013, India had not reported a case of polio since January 2011. Polio is a source of concern for unimmunized or partially immunized travelers. Importation of poliovirus accounted for ~50% of cases in 2013. Clearly, global eradication of polio is necessary to eliminate the risk of importation of wild-type virus. Outbreaks are thought to have been facilitated by suboptimal rates of vaccination, isolated pockets of unvaccinated children, poor sanitation and crowding, improper vaccine-storage conditions, and a reduced level of response to one of the serotypes in the vaccine. While the global eradication campaign has markedly reduced the number of cases of endemic polio, doubts have been raised as to whether eradication is a realistic goal, given the large number of asymptomatic infections and the political instability in developing countries.
LABORATORY-CONFIRMED CASES OF POLIOMYELITIS IN 2012 |
The occurrence of outbreaks of poliomyelitis due to circulating vaccine-derived poliovirus of all three types has been increasing, especially in areas with low vaccination rates. In Egypt, 32 cases of vaccine-derived polio occurred in 1983–1993; in the Dominican Republic and Haiti, 21 cases occurred in 2000–2001; in Indonesia, 46 cases were reported in 2005; in Nigeria, 385 cases occurred in 2005–2012; in the Democratic Republic of the Congo, 64 cases were reported in 2008–2012; in Pakistan, 16 cases occurred in 2012, and at least 30 cases occurred in 2013. These OPV-derived viruses reverted to a more neurovirulent phenotype after undetected circulation (probably for >2 years). The epidemic in Hispaniola was rapidly terminated after intensive vaccination with OPV. In 2005, a case of vaccine-derived polio occurred in an unvaccinated U.S. woman returning from a visit to Central and South America. In the same year, an unvaccinated immunocompromised infant in Minnesota was found to be shedding vaccine-derived poliovirus; further investigation identified 4 of 22 infants in the same community who were shedding the virus. All 5 infants were asymptomatic. These outbreaks emphasize the need for maintaining high levels of vaccine coverage and continued surveillance for circulating virus.
IPV is used in most industrialized countries and OPV in most developing countries, including those in which polio still is or recently was endemic. While IM injections of other vaccines (live or attenuated) can be given concurrently with OPV, unnecessary IM injections should be avoided during the first month after OPV vaccination because they increase the risk of vaccine-associated paralysis. Since 1988, an enhanced-potency inactivated poliovirus vaccine has been available in the United States.
After several doses of OPV alone, the seropositivity rate for individual poliovirus serotypes may still be suboptimal for children in developing countries; one or more supplemental doses of IPV can increase the rate of seropositivity for these serotypes. Against a given serotype, monovalent OPV containing only that serotype is more immunogenic than trivalent vaccine because of a lack of interference from other serotypes. With eradication of wild-type poliovirus type 2, bivalent OPV (types 1 and 3), which was shown to be superior to trivalent OPV, has been the vaccine of choice to eliminate polio and has markedly reduced rates of polio in Nigeria. As the frequency of wild-type polio declines and reports of polio associated with circulating vaccine-derived viruses increase, the World Health Organization is investigating whether IPV can be produced from OPV strains that require less biocontainment, ultimately replacing OPV.
OPV and IPV induce antibodies that persist for at least 5 years. Both vaccines induce IgG and IgA antibodies. Compared with recipients of IPV, recipients of OPV shed less virus and less frequently develop reinfection with wild-type virus after exposure to poliovirus. Although IPV is safe and efficacious, OPV offers the advantages of ease of administration, lower cost, and induction of intestinal immunity resulting in a reduction in the risk of community transmission of wild-type virus. Because of progress toward global eradication of polio and the continued occurrence of cases of vaccine-associated polio, an all-IPV regimen was recommended in 2000 for childhood poliovirus vaccination in the United States, with vaccine administration at 2, 4, and 6–18 months and 4–6 years of age. The risk of vaccine-associated polio should be discussed before OPV is administered. Recommendations for vaccination of adults are listed in Table 228-3.
RECOMMENDATIONS FOR POLIOVIRUS VACCINATION OF ADULTS |
Abbreviation: IPV, inactivated poliovirus vaccine.
Source: Modified from Pickering LK, ed. Red Book 2012: Committee on Infectious Diseases, 29th ed.
There are concerns about discontinuing vaccination in the event that endemic spread of poliovirus is eliminated. Among the reasons for these concerns are that poliovirus is shed from some immunocompromised persons for >10 years, that vaccine-derived poliovirus can circulate and cause disease, and that wild-type poliovirus is present in research laboratories.
PARECHOVIRUSES
Human parechoviruses (HPeVs), like enteroviruses, are members of the family Picornaviridae. The 16 serotypes of HPeV commonly cause infections in early childhood. HPeV-1 infections occur throughout the year, while other parechovirus infections occur more commonly in summer and fall. Infections with HPeVs present similarly to those due to enteroviruses and may cause generalized disease of the newborn, aseptic meningitis, encephalitis, transient paralysis, exanthems, respiratory tract disease, and gastroenteritis. While HPeV-1 is the most common serotype and generally causes mild disease, deaths of infants in the United States have been associated with HPeV-1, HPeV-3, and HPeV-6. HPeVs can be isolated from the same sites as enteroviruses, including the nasopharynx, stool, and respiratory tract secretions. PCR using pan-enterovirus primers does not detect HPeVs, and while PCR assays are performed by the CDC and research laboratories, many commercial laboratories do not perform the test.
REOVIRUSES
Reoviruses are double-stranded RNA viruses encompassing three serotypes. Serologic studies indicate that most humans are infected with reoviruses during childhood. Most infections either are asymptomatic or cause mild upper respiratory tract symptoms. Reovirus is considered a rare cause of mild gastroenteritis or meningitis in infants and children. Speculation regarding an association of reovirus type 3 with idiopathic neonatal hepatitis and extrahepatic biliary atresia is based on an elevated prevalence of antibody to reovirus in some affected patients and the detection of viral RNA by PCR in hepatobiliary tissues in some studies. New orthoreoviruses have been associated with human disease—e.g., Melaka and Kampar viruses with fever and acute respiratory disease in Malaysia, and Nelson Bay virus with acute respiratory disease in a traveler from Bali.
229 | Measles (Rubeola) |
DEFINITION
Measles is a highly contagious viral disease that is characterized by a prodromal illness of fever, cough, coryza, and conjunctivitis followed by the appearance of a generalized maculopapular rash. Before the widespread use of measles vaccines, it was estimated that measles caused between 5 million and 8 million deaths worldwide each year.
GLOBAL CONSIDERATIONS
![]() Remarkable progress has been made in reducing global measles incidence and mortality rates through measles vaccination. In the Americas, intensive vaccination and surveillance efforts—based in part on the successful Pan American Health Organization strategy of periodic nationwide measles vaccination campaigns (supplementary immunization activities, or SIAs)—and high levels of routine measles vaccine coverage interrupted endemic transmission of measles virus. In the United States, high-level coverage with two doses of measles vaccine eliminated endemic measles virus transmission in 2000. More recently, progress has been made in reducing measles incidence and mortality rates in sub-Saharan Africa and Asia as a consequence of increasing routine measles vaccine coverage and provision of a second dose of measles vaccine through mass measles vaccination campaigns and childhood immunization programs.
Remarkable progress has been made in reducing global measles incidence and mortality rates through measles vaccination. In the Americas, intensive vaccination and surveillance efforts—based in part on the successful Pan American Health Organization strategy of periodic nationwide measles vaccination campaigns (supplementary immunization activities, or SIAs)—and high levels of routine measles vaccine coverage interrupted endemic transmission of measles virus. In the United States, high-level coverage with two doses of measles vaccine eliminated endemic measles virus transmission in 2000. More recently, progress has been made in reducing measles incidence and mortality rates in sub-Saharan Africa and Asia as a consequence of increasing routine measles vaccine coverage and provision of a second dose of measles vaccine through mass measles vaccination campaigns and childhood immunization programs.
In 2003, the World Health Assembly endorsed a resolution urging member countries to reduce the number of deaths attributed to measles by 50% (compared with 1999 estimates) by the end of 2005. This target was met. Global measles mortality rates were further reduced in 2008; during that year, there were an estimated 164,000 deaths due to measles (uncertainty bounds: 115,000 and 222,000 deaths). These achievements attest to the enormous public-health significance of measles vaccination. However, recent large outbreaks of measles in Europe and Africa illustrate the challenges faced in sustaining measles control: in these outbreaks, measles was imported into countries that had eliminated indigenous transmission of measles virus.
The Measles and Rubella Initiative, a partnership led by the American Red Cross, the United Nations Foundation, UNICEF, the U.S. Centers for Disease Control and Prevention (CDC), and the World Health Organization (WHO), is playing an important role in reducing global measles incidence and mortality rates. Since its inception in 2001, the Initiative has provided governments and communities in more than 80 countries with technical and financial support for routine immunization activities, mass vaccination campaigns, and disease surveillance systems. Through its 2012–2020 Global Measles and Rubella Strategic Plan, the Initiative aims to reduce measles deaths by 95% (compared with year 2000 estimates) by 2015 and to eliminate measles from at least five of the six WHO regions by 2020. As regional goals for measles elimination are set, global measles eradication is likely to become a public health goal in the near future.
ETIOLOGY
Measles virus is a spherical, nonsegmented, single-stranded, negative-sense RNA virus and a member of the Morbillivirus genus in the family Paramyxoviridae. Measles was originally a zoonotic infection, arising from animal-to-human transmission of an ancestral morbillivirus ~10,000 years ago, when human populations had attained sufficient size to sustain virus transmission. Although RNA viruses typically have high mutation rates, measles virus is considered to be an antigenically monotypic virus; i.e., the surface proteins responsible for inducing protective immunity have retained their antigenic structure across time and distance. The public health significance of this stability is that measles vaccines developed decades ago from a single strain of measles virus remain protective worldwide. Measles virus is killed by ultraviolet light and heat, and attenuated measles vaccine viruses retain these characteristics, necessitating a cold chain for vaccine transport and storage.
EPIDEMIOLOGY
Measles virus is one of the most highly contagious directly transmitted pathogens. Outbreaks can occur in populations in which <10% of persons are susceptible. Chains of transmission are common among household contacts, school-age children, and health care workers. There are no latent or persistent measles virus infections that result in prolonged contagiousness, nor are there animal reservoirs for the virus. Thus, measles virus can be maintained in human populations only by an unbroken chain of acute infections, which requires a continuous supply of susceptible individuals. Newborns become susceptible to measles virus infection when passively acquired maternal antibody is lost; when not vaccinated, these infants account for the bulk of new susceptible individuals.
![]() Endemic measles has a typical temporal pattern characterized by yearly seasonal epidemics superimposed on longer epidemic cycles of 2–5 years or more. In temperate climates, annual measles outbreaks typically occur in the late winter and early spring. These annual outbreaks are probably attributable to social networks facilitating transmission (e.g., congregation of children at school) and environmental factors favoring the viability and transmission of measles virus. Measles cases continue to occur during interepidemic periods in large populations, but at low incidence. The longer epidemic cycles occurring every several years result from the accumulation of susceptible persons over successive birth cohorts and the subsequent decline in the number of susceptibles following an outbreak.
Endemic measles has a typical temporal pattern characterized by yearly seasonal epidemics superimposed on longer epidemic cycles of 2–5 years or more. In temperate climates, annual measles outbreaks typically occur in the late winter and early spring. These annual outbreaks are probably attributable to social networks facilitating transmission (e.g., congregation of children at school) and environmental factors favoring the viability and transmission of measles virus. Measles cases continue to occur during interepidemic periods in large populations, but at low incidence. The longer epidemic cycles occurring every several years result from the accumulation of susceptible persons over successive birth cohorts and the subsequent decline in the number of susceptibles following an outbreak.
Secondary attack rates among susceptible household and institutional contacts generally exceed 90%. The average age at which measles occurs depends on rates of contact with infected persons, protective maternal antibody decline, and vaccine coverage. In densely populated urban settings with low-level vaccination coverage, measles is a disease of infants and young children. The cumulative distribution can reach 50% by 1 year of age, with a significant proportion of children acquiring measles before 9 months—the age of routine vaccination in many countries, in line with the schedule recommended by the WHO’s Expanded Programme on Immunization. As measles vaccine coverage increases or population density decreases, the age distribution shifts toward older children. In such situations, measles cases predominate in school-age children. Infants and young children, although susceptible if not protected by vaccination, are not exposed to measles virus at a rate sufficient to cause a large disease burden in this age group. As vaccination coverage increases further, the age distribution of cases may be shifted into adolescence and adulthood; this distribution is seen in measles outbreaks in the United States and necessitates targeted measles vaccination programs for these older age groups.
Persons with measles are infectious for several days before and after the onset of rash, when levels of measles virus in blood and body fluids are highest and when cough, coryza, and sneezing, which facilitate virus spread, are most severe. The contagiousness of measles before the onset of recognizable disease hinders the effectiveness of quarantine measures. Viral shedding by children with impaired cell-mediated immunity can be prolonged.
Medical settings are well-recognized sites of measles virus transmission. Children may present to health care facilities during the prodrome, when the diagnosis is not obvious although the child is infectious and is likely to infect susceptible contacts. Health care workers can acquire measles from infected children and transmit measles virus to others. Nosocomial transmission can be reduced by maintenance of a high index of clinical suspicion, use of appropriate isolation precautions when measles is suspected, administration of measles vaccine to susceptible children and health care workers, and documentation of health care workers’ immunity to measles (i.e., proof of receipt of two doses of measles vaccine or detection of antibodies to measles virus).
As efforts at measles control are increasingly successful, public perceptions of the risk of measles as a disease diminish and are replaced by concerns about possible adverse events associated with measles vaccine. As a consequence, numerous measles outbreaks have occurred because of opposition to vaccination on religious or philosophical grounds or unfounded fears of serious adverse events (see “Active Immunization,” below).
PATHOGENESIS
Measles virus is transmitted primarily by respiratory droplets over short distances and, less commonly, by small-particle aerosols that remain suspended in the air for long periods. Airborne transmission appears to be important in certain settings, including schools, physicians’ offices, hospitals, and enclosed public places. The virus can be transmitted by direct contact with infected secretions but does not survive for long on fomites.
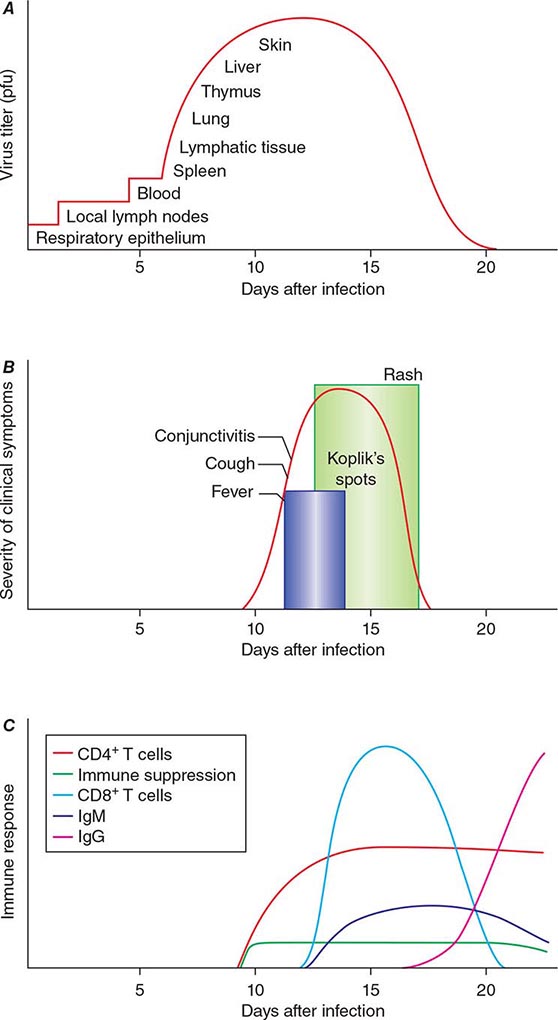
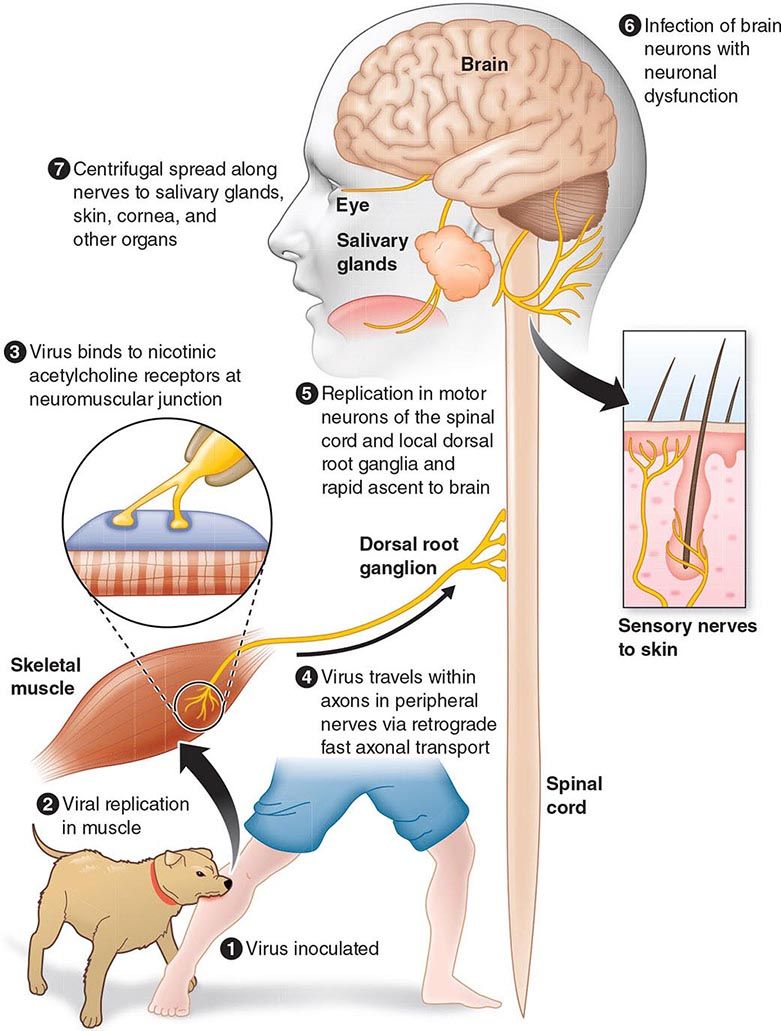
The incubation period for measles is ~10 days to fever onset and 14 days to rash onset. This period may be shorter in infants and longer (up to 3 weeks) in adults. Infection is initiated when measles virus is deposited on epithelial cells in the respiratory tract, oropharynx, or conjunctivae (Fig. 229-1A). During the first 2–4 days after infection, measles virus proliferates locally in the respiratory mucosa and spreads to draining lymph nodes. Virus then enters the bloodstream in infected leukocytes (primarily monocytes), producing the primary viremia that disseminates infection throughout the reticuloendothelial system. Further replication results in secondary viremia that begins 5–7 days after infection and disseminates measles virus throughout the body. Replication of measles virus in these target organs, together with the host’s immune response, is responsible for the signs and symptoms of measles that occur 8–12 days after infection and mark the end of the incubation period (Fig. 229-1B).
FIGURE 229-1 Measles virus infection: pathogenesis, clinical features, and immune responses. A. Spread of measles virus, from initial infection of the respiratory tract through dissemination to the skin. B. Appearance of clinical signs and symptoms, including Koplik’s spots and rash. C. Antibody and T cell responses to measles virus. The signs and symptoms of measles arise coincident with the host immune response. (Source: Modified from WJ Moss, DE Griffin: Nat Rev Microbiol 4:900, 2006.)
IMMUNE RESPONSES
Host immune responses to measles virus are essential for viral clearance, clinical recovery, and the establishment of long-term immunity (Fig. 229-1C). Early nonspecific (innate) immune responses during the prodromal phase include activation of natural killer cells and increased production of antiviral proteins. The adaptive immune responses consist of measles virus–specific antibody and cellular responses. The protective efficacy of antibodies to measles virus is illustrated by the immunity conferred to infants from passively acquired maternal antibodies and the protection of exposed, susceptible individuals after administration of anti–measles virus immunoglobulin. The first measles virus–specific antibodies produced after infection are of the IgM subtype, with a subsequent switch to predominantly IgG1 and IgG4 isotypes. The IgM antibody response is typically absent following reexposure or revaccination and serves as a marker of primary infection.
The importance of cellular immunity to measles virus is demonstrated by the ability of children with agammaglobulinemia (congenital inability to produce antibodies) to recover fully from measles and the contrasting picture for children with severe defects in T lymphocyte function, who often develop severe or fatal disease (Chap. 374). The initial predominant TH1 response (characterized by interferon γ) is essential for viral clearance, and the later TH2 response (characterized by interleukin 4) promotes the development of measles virus–specific antibodies that are critical for protection against reinfection.
The duration of protective immunity following wild-type measles virus infection is generally thought to be lifelong. Immunologic memory to measles virus includes both continued production of measles virus–specific antibodies and circulation of measles virus–specific CD4+ and CD8+ T lymphocytes.
However, the intense immune responses induced by measles virus infection are paradoxically associated with depressed responses to unrelated (non–measles virus) antigens, which persist for several weeks to months beyond resolution of the acute illness. This state of immune suppression enhances susceptibility to secondary infections with bacteria and viruses that cause pneumonia and diarrhea and is responsible for a substantial proportion of measles-related morbidity and deaths. Delayed-type hypersensitivity responses to recall antigens, such as tuberculin, are suppressed, and cellular and humoral responses to new antigens are impaired. Reactivation of tuberculosis and remission of autoimmune diseases after measles have been described and are attributed to this period of immune suppression.
CLINICAL MANIFESTATIONS
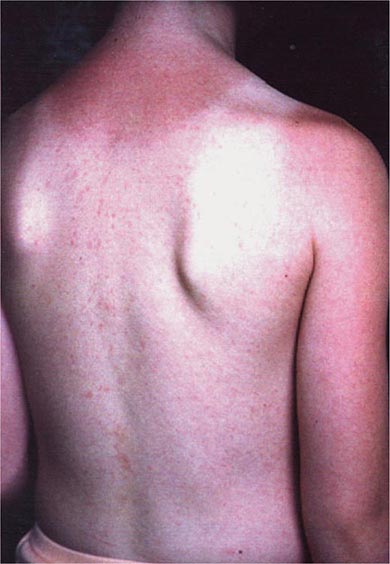
In most persons, the signs and symptoms of measles are highly characteristic (Fig. 229-1B). Fever and malaise beginning ~10 days after exposure are followed by cough, coryza, and conjunctivitis. These signs and symptoms increase in severity over 4 days. Koplik’s spots (see Fig. 25e-2) develop on the buccal mucosa ~2 days before the rash appears. The characteristic rash of measles (see Fig. 25e-3) begins 2 weeks after infection, when the clinical manifestations are most severe, and signal the host’s immune response to the replicating virus. Headache, abdominal pain, vomiting, diarrhea, and myalgia may be present.
Koplik’s spots (see Fig. 25e-2) are pathognomonic of measles and consist of bluish white dots ~1 mm in diameter surrounded by erythema. The lesions appear first on the buccal mucosa opposite the lower molars but rapidly increase in number to involve the entire buccal mucosa. They fade with the onset of rash.
The rash of measles begins as erythematous macules behind the ears and on the neck and hairline. The rash progresses to involve the face, trunk, and arms (see Fig. 25e-3), with involvement of the legs and feet by the end of the second day. Areas of confluent rash appear on the trunk and extremities, and petechiae may be present. The rash fades slowly in the same order of progression as it appeared, usually beginning on the third or fourth day after onset. Resolution of the rash may be followed by desquamation, particularly in undernourished children.
Because the characteristic rash of measles is a consequence of the cellular immune response, it may not develop in persons with impaired cellular immunity (e.g., those with AIDS; Chap. 226). These persons have a high case-fatality rate and frequently develop giant-cell pneumonitis caused by measles virus. T lymphocyte defects due to causes other than HIV-1 infection (e.g., cancer chemotherapy) also are associated with increased severity of measles.
A severe atypical measles syndrome was observed in recipients of a formalin-inactivated measles vaccine (used in the United States from 1963 to 1967 and in Canada until 1970) who were subsequently exposed to wild-type measles virus. The atypical rash began on the palms and soles and spread centripetally to the proximal extremities and trunk, sparing the face. The rash was initially erythematous and maculopapular but frequently progressed to vesicular, petechial, or purpuric lesions (see Fig. 25e-22).
DIFFERENTIAL DIAGNOSIS
The differential diagnosis of measles includes other causes of fever, rash, and conjunctivitis, including rubella, Kawasaki disease, infectious mononucleosis, roseola, scarlet fever, Rocky Mountain spotted fever, enterovirus or adenovirus infection, and drug sensitivity. Rubella is a milder illness without cough and with distinctive lymphadenopathy. The rash of roseola (exanthem subitum) (see Fig. 25e-5) appears after fever has subsided. The atypical lymphocytosis in infectious mononucleosis contrasts with the leukopenia commonly observed in children with measles.
DIAGNOSIS
Measles is readily diagnosed on clinical grounds by clinicians familiar with the disease, particularly during outbreaks. Koplik’s spots (see Fig. 25e-2) are especially helpful because they appear early and are pathognomonic. Clinical diagnosis is more difficult (1) during the prodromal illness; (2) when the rash is attenuated by passively acquired antibodies or prior immunization; (3) when the rash is absent or delayed in immunocompromised children or severely undernourished children with impaired cellular immunity; and (4) in regions where the incidence of measles is low and other pathogens are responsible for the majority of illnesses with fever and rash. The CDC case definition for measles requires (1) a generalized maculopapular rash of at least 3 days’ duration; (2) fever of at least 38.3°C (101°F); and (3) cough, coryza, or conjunctivitis.
Serology is the most common method of laboratory diagnosis. The detection of measles virus–specific IgM in a single specimen of serum or oral fluid is considered diagnostic of acute infection, as is a fourfold or greater increase in measles virus–specific IgG antibody levels between acute- and convalescent-phase serum specimens. Primary infection in the immunocompetent host results in antibodies that are detectable within 1–3 days of rash onset and reach peak levels in 2–4 weeks. Measles virus–specific IgM antibodies may not be detectable until 4–5 days or more after rash onset and usually fall to undetectable levels within 4–8 weeks of rash onset.
Several methods for measurement of antibodies to measles virus are available. Neutralization tests are sensitive and specific, and the results are highly correlated with protective immunity; however, these tests require propagation of measles virus in cell culture and thus are expensive and laborious. Commercially available enzyme immunoassays are most frequently used. Measles can also be diagnosed by isolation of the virus in cell culture from respiratory secretions, nasopharyngeal or conjunctival swabs, blood, or urine. Direct detection of giant cells in respiratory secretions, urine, or tissue obtained by biopsy provides another method of diagnosis.
For detection of measles virus RNA by reverse-transcriptase polymerase chain reaction amplification of RNA extracted from clinical specimens, primers targeted to highly conserved regions of measles virus genes are used. Extremely sensitive and specific, this assay may also permit identification and characterization of measles virus genotypes for molecular epidemiologic studies and can distinguish wild-type from vaccine virus strains.
COMPLICATIONS
Most complications of measles involve the respiratory tract and include the effects of measles virus replication itself and secondary bacterial infections. Acute laryngotracheobronchitis (croup) can occur during measles and may result in airway obstruction, particularly in young children. Giant-cell pneumonitis due to replication of measles virus in the lungs can develop in immunocompromised children, including those with HIV-1 infection. Many children with measles develop diarrhea, which contributes to undernutrition.
Most complications of measles result from secondary bacterial infections of the respiratory tract that are attributable to a state of immune suppression lasting for several weeks to months after acute measles. Otitis media and bronchopneumonia are most common and may be caused by S. pneumoniae, H. influenzae type b, or staphylococci. Recurrence of fever or failure of fever to subside with the rash suggests secondary bacterial infection.
Rare but serious complications of measles involve the central nervous system (CNS). Postmeasles encephalomyelitis complicates ~1 in 1000 cases, affecting mainly older children and adults. Encephalomyelitis occurs within 2 weeks of rash onset and is characterized by fever, seizures, and a variety of neurologic abnormalities. The finding of periventricular demyelination, the induction of immune responses to myelin basic protein, and the absence of measles virus in the brain suggest that postmeasles encephalomyelitis is an autoimmune disorder triggered by measles virus infection. Other CNS complications that occur months to years after acute infection are measles inclusion body encephalitis (MIBE) and subacute sclerosing panencephalitis (SSPE). In contrast to postmeasles encephalomyelitis, MIBE and SSPE are caused by persistent measles virus infection. MIBE is a rare but fatal complication that affects individuals with defective cellular immunity and typically occurs months after infection. SSPE is a slowly progressive disease characterized by seizures and progressive deterioration of cognitive and motor functions, with death occurring 5–15 years after measles virus infection. SSPE most often develops in persons infected with measles virus at <2 years of age.
PROGNOSIS
![]() Most persons with measles recover and develop long-term protective immunity to reinfection. Measles case-fatality proportions vary with the average age of infection, the nutritional and immunologic status of the population, measles vaccine coverage, and access to health care. Among previously vaccinated persons who do become infected, disease is less severe and mortality rates are significantly lower. In developed countries, <1 in 1000 children with measles die. In endemic areas of sub-Saharan Africa, the measles case-fatality proportion may be 5–10% or even higher. Measles is a major cause of childhood deaths in refugee camps and in internally displaced populations, where case-fatality proportions have been as high as 20–30%.
Most persons with measles recover and develop long-term protective immunity to reinfection. Measles case-fatality proportions vary with the average age of infection, the nutritional and immunologic status of the population, measles vaccine coverage, and access to health care. Among previously vaccinated persons who do become infected, disease is less severe and mortality rates are significantly lower. In developed countries, <1 in 1000 children with measles die. In endemic areas of sub-Saharan Africa, the measles case-fatality proportion may be 5–10% or even higher. Measles is a major cause of childhood deaths in refugee camps and in internally displaced populations, where case-fatality proportions have been as high as 20–30%.
PREVENTION
Passive Immunization Human immunoglobulin given shortly after exposure can attenuate the clinical course of measles. In immunocompetent persons, administration of immunoglobulin within 72 h of exposure usually prevents measles virus infection and almost always prevents clinical measles. Administered up to 6 days after exposure, immunoglobulin will still prevent or modify the disease. Prophylaxis with immunoglobulin is recommended for susceptible household and nosocomial contacts who are at risk of developing severe measles, particularly children <1 year of age, immunocompromised persons (including HIV-infected persons previously immunized with live attenuated measles vaccine), and pregnant women. Except for premature infants, children <6 months of age usually will be partially or completely protected by passively acquired maternal antibody. If measles is diagnosed in a household member, all unimmunized children in the household should receive immunoglobulin. The recommended dose is 0.25 mL/kg given intramuscularly. Immunocompromised persons should receive 0.5 mL/kg. The maximum total dose is 15 mL. IV immunoglobulin contains antibodies to measles virus; the usual dose of 100–400 mg/kg generally provides adequate prophylaxis for measles exposures occurring as long as 3 weeks or more after IV immunoglobulin administration.
Active Immunization The first live attenuated measles vaccine was developed by passage of the Edmonston strain in chick embryo fibroblasts to produce the Edmonston B virus, which was licensed in 1963 in the United States. Further passage of Edmonston B virus produced the more attenuated Schwarz vaccine that currently serves as the standard in much of the world. The Moraten (“more attenuated Enders”) strain, which was licensed in 1968 and is used in the United States, is genetically closely related to the Schwarz strain.
Lyophilized measles vaccines are relatively stable, but reconstituted vaccine rapidly loses potency. Live attenuated measles vaccines are inactivated by light and heat and lose about half their potency at 20°C and almost all their potency at 37°C within 1 h after reconstitution. Therefore, a cold chain must be maintained before and after reconstitution. Antibodies first appear 12–15 days after vaccination, and titers peak at 1–3 months. Measles vaccines are often combined with other live attenuated virus vaccines, such as those for mumps and rubella (MMR) and for mumps, rubella, and varicella (MMR-V).
The recommended age of first vaccination varies from 6 to 15 months and represents a balance between the optimal age for seroconversion and the probability of acquiring measles before that age. The proportions of children who develop protective levels of antibody after measles vaccination approximate 85% at 9 months of age and 95% at 12 months. Common childhood illnesses concomitant with vaccination may reduce the level of immune response, but such illness is not a valid reason to withhold vaccination. Measles vaccines have been well tolerated and immunogenic in HIV-1-infected children and adults, although antibody levels may wane. Because of the potential severity of wild-type measles virus infection in HIV-1-infected children, routine measles vaccination is recommended except for those who are severely immunocompromised. Measles vaccination is contraindicated in individuals with other severe deficiencies of cellular immunity because of the possibility of disease due to progressive pulmonary or CNS infection with the vaccine virus.
The duration of vaccine-induced immunity is at least several decades if not longer. Rates of secondary vaccine failure 10–15 years after immunization have been estimated at ~5% but are probably lower when vaccination takes place after 12 months of age. Decreasing antibody concentrations do not necessarily imply a complete loss of protective immunity: a secondary immune response usually develops after reexposure to measles virus, with a rapid rise in antibody titers in the absence of overt clinical disease.
Standard doses of currently licensed measles vaccines are safe for immunocompetent children and adults. Fever to 39.4°C (103°F) occurs in ~5% of seronegative vaccine recipients, and 2% of vaccine recipients develop a transient rash. Mild transient thrombocytopenia has been reported, with an incidence of ~1 case per 40,000 doses of MMR vaccine.
Since the publication of a report in 1998 hypothesizing that MMR vaccine may cause a syndrome of autism and intestinal inflammation, much public attention has focused on this purported association. The events that followed publication of this report led to diminished vaccine coverage in the United Kingdom and provide important lessons in the misinterpretation of epidemiologic evidence and the communication of scientific results to the public. The publication that incited the concern was a case series describing 12 children with a regressive developmental disorder and chronic enterocolitis; 9 of these children had autism. In 8 of the 12 cases, the parents associated onset of the developmental delay with MMR vaccination. This simple temporal association was misinterpreted and misrepresented as a possible causal relationship, first by the lead author of the study and then by elements of the media and the public. Subsequently, several comprehensive reviews and additional epidemiologic studies refuted evidence of a causal relationship between MMR vaccination and autism.
PROSPECTS FOR MEASLES ERADICATION
Progress in global measles control has renewed discussion of measles eradication. In contrast to poliovirus eradication, the eradication of measles virus will not entail challenges posed by prolonged shedding of potentially virulent vaccine viruses and environmental viral reservoirs. However, in comparison with smallpox eradication, higher levels of population immunity will be necessary to interrupt measles virus transmission, more highly skilled health care workers will be required to administer measles vaccines, and containment through case detection and ring vaccination will be more difficult for measles virus because of infectivity before rash onset. New tools, such as aerosol administration of measles vaccines, will facilitate mass vaccination campaigns. Despite enormous progress, measles remains a leading vaccine-preventable cause of childhood mortality worldwide and continues to cause outbreaks in communities with low vaccination coverage rates in industrialized nations.
230e | Rubella (German Measles) |
Rubella was historically viewed as a variant of measles or scarlet fever. Not until 1962 was a separate viral agent for rubella isolated. After an epidemic of rubella in Australia in the early 1940s, the ophthalmologist Norman Gregg noticed the occurrence of congenital cataracts among infants whose mothers had reported rubella infection during early pregnancy, and congenital rubella syndrome (CRS; see “Clinical Manifestations,” below) was first described.
ETIOLOGY
Rubella virus is a member of the Togaviridae family and the only member of the genus Rubivirus. This single-stranded RNA enveloped virus measures 50–70 nm in diameter. Its core protein is surrounded by a single-layer lipoprotein envelope with spike-like projections containing two glycoproteins, E1 and E2. There is only one antigenic type of rubella virus, and humans are its only known reservoir.
PATHOGENESIS AND PATHOLOGY
Although the pathogenesis of postnatal (acquired) rubella has been well documented, data on pathology are limited because of the mildness of the disease. Rubella virus is spread from person to person via respiratory droplets. Primary implantation and replication in the nasopharynx are followed by spread to the lymph nodes. Subsequent viremia occurs, which in pregnant women often results in infection of the placenta. Placental virus replication may lead to infection of fetal organs. The pathology of CRS in the infected fetus is well defined, with almost all organs found to be infected; however, the pathogenesis of CRS is only poorly delineated. In tissue, infections with rubella virus have diverse effects, ranging from no obvious impact to cell destruction. The hallmark of fetal infection is chronicity, with persistence throughout fetal development in utero and for up to 1 year after birth.
Individuals with acquired rubella may shed virus from 7 days before rash onset to ~5–7 days thereafter. Both clinical and subclinical infections are considered contagious. Infants with CRS may shed large quantities of virus from bodily secretions, particularly from the throat and in the urine, up to 1 year of age. Outbreaks of rubella, including some in nosocomial settings, have originated with index cases of CRS. Thus only individuals immune to rubella should have contact with infants who have CRS or who are congenitally infected with rubella virus but are not showing signs of CRS.
EPIDEMIOLOGY
The largest recent rubella epidemic in the United States took place in 1964–1965, when an estimated 12.5 million cases occurred, resulting in ~20,000 cases of CRS. Since the introduction of the routine rubella vaccination program in the United States in 1969, the number of rubella cases reported each year has dropped by >99%; the rate of vaccination coverage with rubella-containing vaccine has been >90% among children 19–35 months old since 1995 and >95% for kindergarten and first-grade entrants since 1980. In 1989 a goal for the elimination of rubella and CRS in the United States was set, and in 2004 a panel of experts agreed unanimously that rubella was no longer an endemic disease in this country. The criteria used to document lack of endemic transmission included low disease incidence, high nationwide rubella antibody seroprevalence, outbreaks that were few and contained (i.e., small numbers of cases), and lack of endemic virus transmission (as assessed by genetic sequencing). In the United States, interruption of endemic transmission of rubella virus has been sustained since 2001; in 2012, however, three cases of CRS were reported in infants whose mothers had acquired rubella infection abroad. Thus health care providers should remain vigilant, considering the possibility of rubella infection in patients emigrating or returning from countries without rubella control programs and the accompanying potential for CRS among their infants.
![]() Although rubella and CRS are no longer endemic in the United States, they remain important public health problems globally. The number of rubella cases reported worldwide in 1999 was ~900,000; this figure declined steadily to 94,030 in 2012. However, numbers of rubella cases are substantially underestimated because cases in many countries are identified through measles surveillance systems that are not specific for rubella. In 2010, it was estimated that 103,000 cases of CRS occur globally.
Although rubella and CRS are no longer endemic in the United States, they remain important public health problems globally. The number of rubella cases reported worldwide in 1999 was ~900,000; this figure declined steadily to 94,030 in 2012. However, numbers of rubella cases are substantially underestimated because cases in many countries are identified through measles surveillance systems that are not specific for rubella. In 2010, it was estimated that 103,000 cases of CRS occur globally.
CLINICAL FEATURES
Acquired Rubella Acquired rubella commonly presents with a generalized maculopapular rash that usually lasts for up to 3 days (Fig. 230e-1), although as many as 50% of cases may be subclinical or without rash. When it occurs, the rash is usually mild and may be difficult to detect in persons with darker skin. In children, rash is usually the first sign of illness. However, in older children and adults, a 1- to 5-day prodrome often precedes the rash and may include low-grade fever, malaise, and upper respiratory symptoms. The incubation period is 14 days (range, 12–23 days).
FIGURE 230e-1 Mild maculopapular rash of rubella in a child.
Lymphadenopathy, particularly occipital and postauricular, may be noted during the second week after exposure. Although acquired rubella is usually thought of as a benign disease, arthralgia and arthritis are common in infected adults, particularly women. Thrombocytopenia and encephalitis are less common complications.
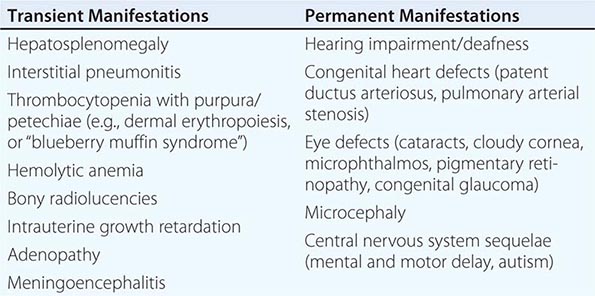
Congenital Rubella Syndrome The most serious consequence of rubella virus infection can develop when a woman becomes infected during pregnancy, particularly during the first trimester. The resulting complications may include miscarriage, fetal death, premature delivery, or live birth with congenital defects. Infants infected with rubella virus in utero may have myriad physical defects (Table 230e-1), which most commonly relate to the eyes, ears, and heart. This constellation of severe birth defects is known as congenital rubella syndrome. In addition to permanent manifestations, there are a host of transient physical manifestations, including thrombocytopenia with purpura/petechiae (e.g., dermal erythropoiesis, “blueberry muffin syndrome”). Some infants may be born with congenital rubella virus infection but have no apparent signs or symptoms of CRS and are referred to as “infants with congenital rubella infection only.”
COMMON TRANSIENT AND PERMANENT MANIFESTATIONS IN INFANTS WITH CONGENITAL RUBELLA SYNDROME |
DIAGNOSIS
Acquired Rubella Clinical diagnosis of acquired rubella is difficult because of the mimicry of many illnesses with rashes, the varied clinical presentations, and the high rates of subclinical and mild disease. Illnesses that may be similar to rubella in presentation include scarlet fever, roseola, toxoplasmosis, fifth disease, measles, and illnesses with suboccipital and postauricular lymphadenopathy. Thus laboratory documentation of rubella virus infection is considered the only reliable way to confirm acute disease.
Laboratory assessment of rubella infection is conducted by serologic and virologic methods. For acquired rubella, serologic diagnosis is most common and depends on the demonstration of IgM antibodies in an acute-phase serum specimen or a fourfold rise in IgG antibody titer between acute- and convalescent-phase specimens. The enzyme-linked immunosorbent assay IgM capture technique is considered most accurate for serologic diagnosis, but the indirect IgM assay also is acceptable. After rubella virus infection, IgM antibody may be detectable for up to 6 weeks. In case of a negative result for IgM in specimens taken earlier than day 5 after rash onset, serologic testing should be repeated. Although uncommon, reinfection with rubella virus is possible, and IgM antibodies may be present. To detect a rise in IgG antibody titer indicative of acute disease, the acute-phase serum specimen should be collected within 7–10 days after onset of illness and the convalescent-phase specimen ~14–21 days after the first specimen.
IgG avidity testing is used in conjunction with IgG testing. Low-avidity antibodies indicate recent infection. Mature (high-avidity) IgG antibodies most likely indicate an infection occurring at least 2 months previously. This test helps distinguish primary infection from reinfection. Avidity testing may be particularly useful in diagnosing rubella in pregnant women and assessing the risk of CRS.
Rubella virus can be isolated from the blood and nasopharynx during the prodromal period and for as long as 2 weeks after rash onset. However, as the secretion of virus in individuals with acquired rubella is maximal just before or up to 4 days after rash onset, this is the optimal time frame for collecting specimens for viral cultures. Rubella can also be diagnosed by viral RNA detection in a reverse-transcriptase polymerase chain reaction (RT-PCR) assay.
Congenital Rubella Syndrome A clinical diagnosis of CRS is reasonable when an infant presents with a combination of cataracts, hearing impairment, and heart defects; this pattern is seen in ~10% of infants with CRS. Infants may present with different combinations of defects depending on when infection occurs during gestation. Hearing impairment is the most common single defect of CRS. However, as with acquired rubella, laboratory diagnosis of congenital infection is highly recommended, particularly because most features of the clinical presentation are nonspecific and may be associated with other intrauterine infections. Early diagnosis of CRS facilitates appropriate medical intervention for specific disabilities and prompts implementation of infection control measures.
Diagnostic tests used to confirm CRS include serologic assays and virus detection. In an infant with congenital infection, serum IgM antibodies are normally present for up to 6 months but may be detectable for up to 1 year after birth. In some instances, IgM may not be detectable until 1 month of age; thus infants who have symptoms consistent with CRS but who test negative shortly after birth should be retested at 1 month. A rubella serum IgG titer persisting beyond the time expected after passive transfer of maternal IgG antibody (i.e., a rubella titer that does not decline at the expected rate of a twofold dilution per month) is another serologic criterion used to confirm CRS.
In congenital infection, rubella virus is isolated most commonly from throat swabs and less commonly from urine and cerebrospinal fluid. Infants with congenital rubella may excrete virus for up to 1 year, but specimens for virus isolation are most likely to be positive if obtained within the first 6 months after birth. Rubella virus in infants with CRS can also be detected by RT-PCR.
Rubella Diagnosis in Pregnant Women In the United States, screening for rubella IgG antibodies is recommended as part of routine prenatal care. Pregnant women with a positive IgG antibody serologic test are considered immune. Susceptible pregnant women should be vaccinated postpartum.
A susceptible pregnant woman exposed to rubella virus should be tested for IgM antibodies and/or a fourfold rise in IgG antibody titer between acute- and convalescent-phase serum specimens to determine whether she was infected during pregnancy. Pregnant women with evidence of acute infection must be clinically monitored, and gestational age at the time of maternal infection must be determined to assess the possibility of risk to the fetus. Of women infected with rubella virus during the first 11 weeks of gestation, up to 90% deliver an infant with CRS; for maternal infection during the first 20 weeks of pregnancy, the CRS rate is 20%.
PREVENTION
![]() After the isolation of rubella virus in the early 1960s and the occurrence of a devastating pandemic, a vaccine for rubella was developed and licensed in 1969. Currently, the majority of rubella-containing vaccines (RCVs) used worldwide are combined measles and rubella (MR) or measles, mumps, and rubella (MMR) formulations. A tetravalent measles, mumps, rubella, and varicella (MMRV) vaccine is available but is not widely used.
After the isolation of rubella virus in the early 1960s and the occurrence of a devastating pandemic, a vaccine for rubella was developed and licensed in 1969. Currently, the majority of rubella-containing vaccines (RCVs) used worldwide are combined measles and rubella (MR) or measles, mumps, and rubella (MMR) formulations. A tetravalent measles, mumps, rubella, and varicella (MMRV) vaccine is available but is not widely used.
The public health burden of rubella infection is measured primarily through the resulting CRS cases. The 1964–1965 rubella epidemic in the United States encompassed >30,000 infections during pregnancy. CRS occurred in ~20,000 infants born alive, including >11,000 infants who were deaf, >3500 infants who were blind, and almost 2000 infants who were mentally retarded. The cost of this epidemic exceeded $1.5 billion. In 1983, the cost per child with CRS was estimated at $200,000.
In most countries, there is little documented evidence to illuminate the epidemiology of CRS. Clusters of CRS cases have been reported in developing countries. Before the introduction of an immunization program, the incidence of CRS is 0.1–0.2 per 1000 live births during endemic periods and 1–4 per 1000 live births during epidemic periods. Where rubella virus is circulating and women of childbearing age are susceptible, CRS cases will continue to occur.
The most effective method of preventing acquired rubella and CRS is through vaccination with an RCV. One dose induces seroconversion in ≥95% of persons ≥1 year of age. Immunity is considered long-term and is probably lifelong. The most commonly used vaccine globally is the RA27/3 virus strain. The current recommendation for routine rubella vaccination in the United States is a first dose of MMR vaccine at 12–15 months of age and a second dose at 4–6 years. Target groups for rubella vaccine include children ≥1 year of age, adolescents and adults without documented evidence of immunity, individuals in congregate settings (e.g., college students, military personnel, child care and health care workers), and susceptible women before and after pregnancy.
Because of the theoretical risk of transmission of live attenuated rubella vaccine virus to the developing fetus, women known to be pregnant should not receive an RCV. In addition, pregnancy should be avoided for 28 days after receipt of an RCV. In follow-up studies of 680 unknowingly pregnant women who received rubella vaccine, no infant was born with CRS. Receipt of an RCV during pregnancy is not ordinarily a reason to consider termination of the pregnancy.
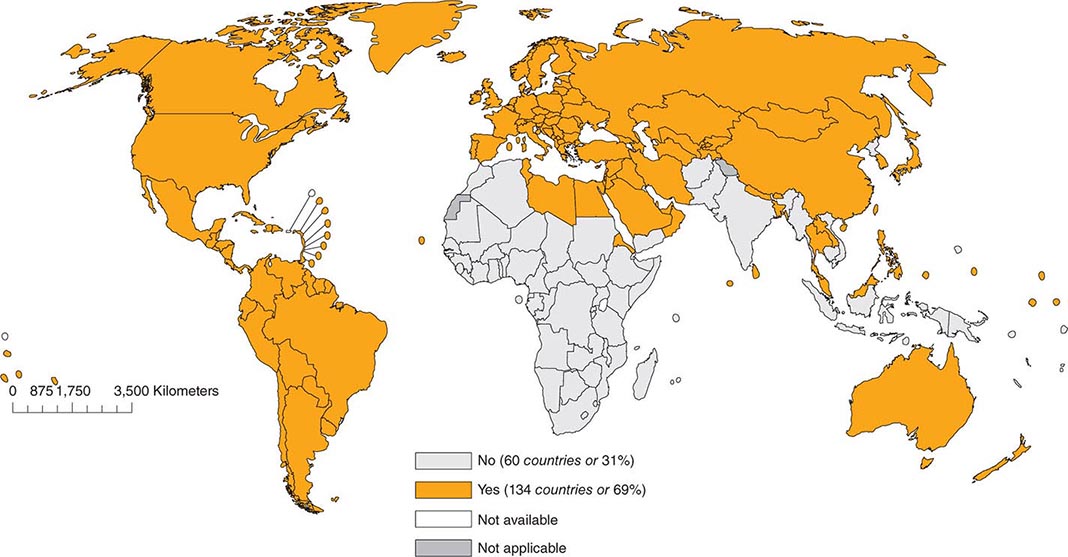
As of 2012, 134 (69%) of the 194 member countries of the World Health Organization recommended inclusion of an RCV in the routine childhood vaccination schedule (Fig. 230e-2). Goals for control or elimination of rubella and CRS have been established in the American Region, the European Region, the South-East Asia Region, and the Western Pacific Region. The other two regions (Eastern Mediterranean and African) have not yet set such goals.
FIGURE 230e-2 Countries using rubella vaccine in their national immunization schedule, 2012. (From the World Health Organization.)
231e | Mumps |
DEFINITION
Mumps is an illness characterized by acute-onset unilateral or bilateral tender, self-limited swelling of the parotid or other salivary gland(s) that lasts at least 2 days and has no other apparent cause.
ETIOLOGIC AGENT
![]() Mumps is caused by a paramyxovirus with a negative-strand, nonsegmented RNA genome of 15,384 bases encoding at least 8 proteins: the nucleo- (N), phospho- (P), V, matrix (M), fusion (F), small hydrophobic (SH), hemagglutinin-neuraminidase (HN), and large (L) proteins. The N, P, and L proteins together provide the polymerase activity responsible for genome transcription and replication. The viral genome is surrounded by a host cell–derived lipid bilayer envelope containing the M, F, SH, and HN proteins. The M protein is involved in viral assembly, whereas the HN and F proteins are responsible for cell attachment and entry and are the major targets of virus-neutralizing antibody. The V and SH proteins are accessory proteins, acting as antagonists of the host antiviral response; the former interferes with the interferon response and the latter with the tumor necrosis factor α (TNF-α)–mediated apoptotic signaling pathway. Because of the hypervariability of the SH gene, its nucleotide sequence is used to “genotype” the virus for molecular epidemiologic purposes. Thus far, 12 mumps virus genotypes have been assigned by SH gene sequence and are designated A–N (with the exclusion of E and M, which have been merged with genotypes C and K, respectively).
Mumps is caused by a paramyxovirus with a negative-strand, nonsegmented RNA genome of 15,384 bases encoding at least 8 proteins: the nucleo- (N), phospho- (P), V, matrix (M), fusion (F), small hydrophobic (SH), hemagglutinin-neuraminidase (HN), and large (L) proteins. The N, P, and L proteins together provide the polymerase activity responsible for genome transcription and replication. The viral genome is surrounded by a host cell–derived lipid bilayer envelope containing the M, F, SH, and HN proteins. The M protein is involved in viral assembly, whereas the HN and F proteins are responsible for cell attachment and entry and are the major targets of virus-neutralizing antibody. The V and SH proteins are accessory proteins, acting as antagonists of the host antiviral response; the former interferes with the interferon response and the latter with the tumor necrosis factor α (TNF-α)–mediated apoptotic signaling pathway. Because of the hypervariability of the SH gene, its nucleotide sequence is used to “genotype” the virus for molecular epidemiologic purposes. Thus far, 12 mumps virus genotypes have been assigned by SH gene sequence and are designated A–N (with the exclusion of E and M, which have been merged with genotypes C and K, respectively).
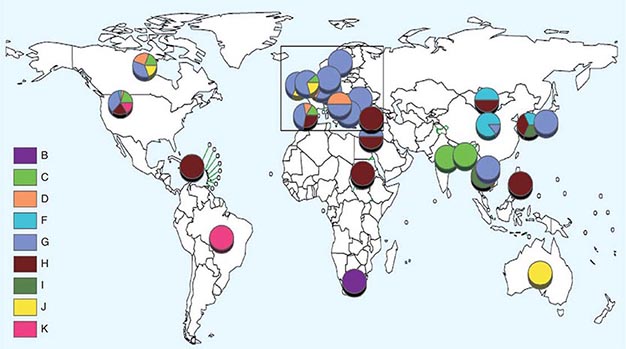
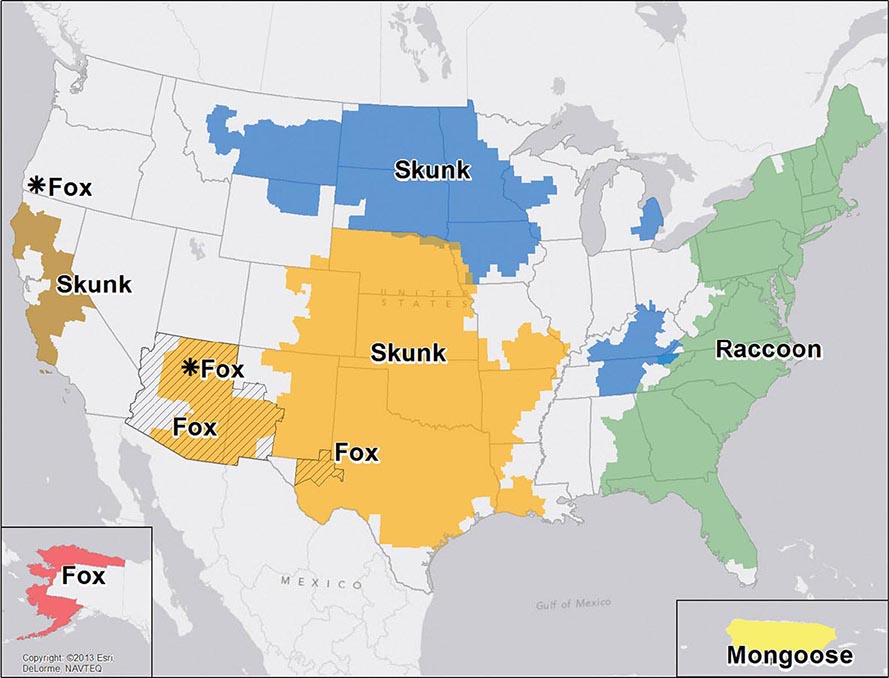
![]() Nucleotide sequencing of clinical isolates shows that virus genotypes D and G circulate predominantly in the Western Hemisphere; genotypes F, C, and I in the Asia–Pacific region; and genotypes B, H, J, and K in the Southern Hemisphere (Fig. 231e-1). Although numerous mumps virus genotypes have been identified and some vary antigenically from others, only one serotype exists, and there is no evidence to suggest that certain circulating virus strains are more virulent or contagious than others.
Nucleotide sequencing of clinical isolates shows that virus genotypes D and G circulate predominantly in the Western Hemisphere; genotypes F, C, and I in the Asia–Pacific region; and genotypes B, H, J, and K in the Southern Hemisphere (Fig. 231e-1). Although numerous mumps virus genotypes have been identified and some vary antigenically from others, only one serotype exists, and there is no evidence to suggest that certain circulating virus strains are more virulent or contagious than others.
FIGURE 231e-1 Distribution of reported mumps genotypes, 2005–2011 (data as of April 20, 2012). Pie-slice size is proportional to the number of years each genotype was reported. (Figure courtesy of WHO, with permission; http://www.who.int/immunization_monitoring/diseases/mumps/en/index.html; accessed September 11, 2012.)
EPIDEMIOLOGY
![]() Mumps is endemic worldwide, with epidemics every 3–5 years in unvaccinated populations. These epidemics typically occur in locations where children and young adults congregate, such as schools, military barracks, and other institutions. In countries without national mumps vaccination programs, the estimated annual global incidence is 100–1000 cases per 100,000 population. After the introduction of mumps vaccine in the United States in 1967, the number of reported cases declined dramatically. By 2001, fewer than 300 cases were reported, representing a 99.8% reduction from prevaccine-era levels. Mumps incidence remained at historic lows in the United States until 2006, when 6584 cases were reported—the largest outbreak since 1987. At the time of the 2006 outbreak, the disease was resurging globally, even in populations with high-level vaccination coverage. The number of reported U.S. cases declined precipitously in the 2 years that followed but then spiked in 2009–2010, with focal outbreaks in New York and New Jersey, and again in 2011, with a focal outbreak in California. A recent study by the Centers for Disease Control and Prevention (CDC) showed that two-dose coverage with measles–mumps–rubella (MMR) vaccine in major U.S. cities (94.8%) remains at or very near the level needed to contain these childhood infections; however, focal areas with inadequate vaccination coverage still leave some children at risk. Sporadic, large-scale mumps outbreaks continue to be reported worldwide, sometimes in countries where the disease was once under control.
Mumps is endemic worldwide, with epidemics every 3–5 years in unvaccinated populations. These epidemics typically occur in locations where children and young adults congregate, such as schools, military barracks, and other institutions. In countries without national mumps vaccination programs, the estimated annual global incidence is 100–1000 cases per 100,000 population. After the introduction of mumps vaccine in the United States in 1967, the number of reported cases declined dramatically. By 2001, fewer than 300 cases were reported, representing a 99.8% reduction from prevaccine-era levels. Mumps incidence remained at historic lows in the United States until 2006, when 6584 cases were reported—the largest outbreak since 1987. At the time of the 2006 outbreak, the disease was resurging globally, even in populations with high-level vaccination coverage. The number of reported U.S. cases declined precipitously in the 2 years that followed but then spiked in 2009–2010, with focal outbreaks in New York and New Jersey, and again in 2011, with a focal outbreak in California. A recent study by the Centers for Disease Control and Prevention (CDC) showed that two-dose coverage with measles–mumps–rubella (MMR) vaccine in major U.S. cities (94.8%) remains at or very near the level needed to contain these childhood infections; however, focal areas with inadequate vaccination coverage still leave some children at risk. Sporadic, large-scale mumps outbreaks continue to be reported worldwide, sometimes in countries where the disease was once under control.
Although historically a disease of unvaccinated children, with the largest proportion of cases occurring in children 5–9 years of age during the prevaccine era, mumps now frequently occurs in older age groups—primarily college students, most of whom were vaccinated in early childhood. This shift in age distribution and the occurrence of mumps in vaccinated populations are probably the result of several coincident circumstances, including (1) situations promoting the spread of respiratory viruses among young adults (e.g., residence in college dormitories), (2) waning of vaccine immunity with time, (3) lack of endemically circulating wild-type virus to periodically boost vaccine-induced immune responses, and (4) continuing global epidemics (due to either lack of mumps vaccination programs or, where such programs do exist, low rates of mumps vaccination). The notable decline in mumps vaccination–induced immunity with time may be due to both declining titers and decreasing avidity of antibodies. The waning of mumps immunity over time is supported by studies suggesting that a third dose of MMR vaccine significantly reduces the mumps attack rate; however, these studies were not adequately controlled to rule out the possibility that the observed declines in mumps incidence were unrelated to the intervention. Therefore, the effectiveness of a third dose of MMR vaccine remains to be demonstrated.
PATHOGENESIS
Humans are the only natural hosts for mumps virus infection. The incubation period of mumps is ~19 days (range, 7–23 days). The virus is transmitted by the respiratory route via droplets, saliva, and fomites. Mumps virus is typically shed from 1 week before to 1 week after symptom onset, although this window appears to be narrower in vaccinated individuals. Persons are most contagious 1–2 days before onset of clinical symptoms. Inference from related respiratory diseases and animal studies indicates that primary replication likely occurs in the nasal mucosa or upper respiratory mucosal epithelium. Mononuclear cells and cells within regional lymph nodes can become infected; such infection facilitates the development of viremia and poses a risk for a wide array of acute inflammatory reactions. Classic sites of mumps virus replication include the salivary glands, testes, pancreas, ovaries, mammary glands, and central nervous system (CNS).
Little is known about the pathology of mumps since the disease is rarely fatal. The virus replicates well in glandular epithelium, but classic parotitis is not a necessary component of mumps infection. Affected glands contain perivascular and interstitial mononuclear cell infiltrates and exhibit hemorrhage with prominent edema. Necrosis of acinar and epithelial duct cells is evident in the salivary glands and in the germinal epithelium of the seminiferous tubules of the testes. The virus probably enters cerebrospinal fluid (CSF) through the choroid plexus or via transiting mononuclear cells during plasma viremia. Although relevant data are limited, typical mumps encephalitis appears to be secondary to respiratory spread and is probably a parainfectious process, as suggested by perivenous demyelination, perivascular mononuclear cell inflammation, and relative sparing of neurons. Although rare, presumed primary encephalitis has been associated with mumps virus isolation from brain tissue. Evidence of placental and intrauterine spread in pregnancy has been found in both early and late gestation.
CLINICAL MANIFESTATIONS
Up to half of mumps virus infections are asymptomatic or lead to nonspecific respiratory symptoms. Inapparent infections are more common in adults than in children. The prodrome of mumps consists of low-grade fever, malaise, myalgia, headache, and anorexia. Mumps parotitis—acute-onset unilateral or bilateral swelling of the parotid or other salivary glands that lasts >2 days and has no other apparent cause—develops in 70–90% of symptomatic infections, usually within 24 h of prodromal symptoms but sometimes as long as 1 week thereafter. Parotitis is generally bilateral, although the two sides may not be involved synchronously. Unilateral involvement is documented in about one-third of cases. Swelling of the parotid is accompanied by tenderness and obliteration of the space between the earlobe and the angle of the mandible (Figs. 231e-2 and 231e-3). The patient frequently reports an earache and finds it difficult to eat, swallow, or talk. The orifice of the parotid duct is commonly red and swollen. The submaxillary and sublingual glands are involved less often than the parotid gland and are almost never involved alone. Glandular swelling increases for a few days and then gradually subsides, disappearing within 1 week. Recurrent sialadenitis is a rare sequela of mumps parotitis. In ~6% of mumps cases, obstruction of lymphatic drainage secondary to bilateral salivary gland swelling may lead to presternal pitting edema, associated often with submandibular adenitis and rarely with the more life-threatening supraglottic edema.
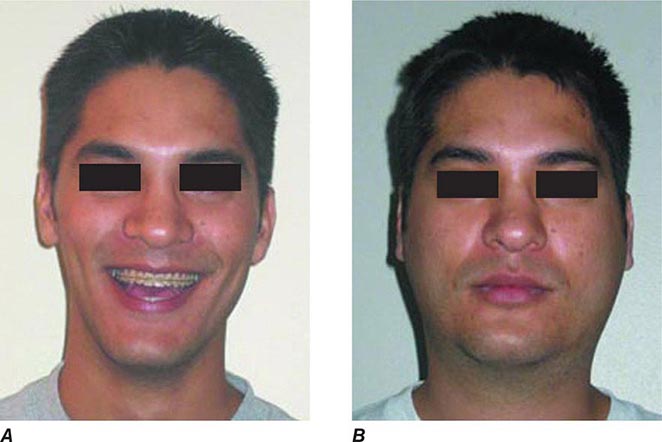
FIGURE 231e-2 A comparison of a person before acquiring mumps (A) and on day 3 (B) of acute bilateral parotitis. (Courtesy of patient C.M. From Shanley JD. The resurgence of mumps in young adults and adolescents. Cleve Clin J Med 2007; 74:42-48. Reprinted with permission. Copyright © 2007 Cleveland Clinic Foundation. All rights reserved.)
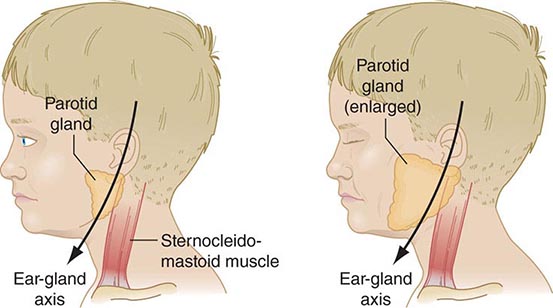
FIGURE 231e-3 Schematic drawing of a parotid gland infected with mumps virus (right) compared with a normal gland (left). An enlarged cervical lymph node is usually posterior to the imaginary line. (Reprinted with permission from Gershon A et al: Mumps, in Krugman’s Infectious Diseases of Children, 11th ed. Philadelphia, Elsevier, 2004, p 392.)
Epididymo-orchitis is the next most common manifestation of mumps, developing in 15–30% of cases in postpubertal males, with bilateral involvement in 10–30% of those cases. Orchitis, accompanied by fever, typically occurs during the first week of parotitis but can develop up to 6 weeks after parotitis or in its absence. The testis is painful and tender and can be enlarged to several times its normal size; this condition usually resolves within 1 week. Testicular atrophy develops in one-half of affected men. Sterility after mumps is rare, although subfertility is estimated to occur in 13% of cases of unilateral orchitis and in 30–87% of cases of bilateral orchitis. Oophoritis occurs in ~5% of women with mumps and may be associated with lower abdominal pain and vomiting but has only rarely been associated with sterility or premature menopause. Mumps infection in postpubertal women may also present with mastitis.
Documented CSF pleocytosis indicates that mumps virus invades the CNS in ~50% of cases; however, symptomatic CNS disease, typically in the form of aseptic meningitis, occurs in <10% of cases, with a male predominance. CNS symptoms of aseptic meningitis (e.g., stiff neck, headache, and drowsiness) appear ~5 days after parotitis and also occur often in the absence of parotid involvement. Within the first 24 h polymorphonuclear leukocytes may predominate in CSF (1000–2000 cells/μL), but by the second day nearly all the cells are lymphocytes. The glucose level in CSF may be low and the protein concentration high, a pattern reminiscent of bacterial meningitis. Mumps meningitis is a self-limited manifestation without significant risk of death or long-term sequelae. Cranial nerve palsies have occasionally led to permanent sequelae, particularly deafness. The reported incidence of mumps-associated hearing loss varies between 1 in 1000 and 1 in 100,000. In ~0.1% of infections, mumps virus may cause encephalitis, which presents as high fever with marked changes in the level of consciousness, seizures, and focal neurologic symptoms. Electroencephalographic abnormalities may be seen. Permanent sequelae are sometimes identified in survivors, and adult infections more commonly have poor outcomes than do pediatric infections. The mortality rate associated with mumps encephalitis is ~1.5%. Other CNS problems occasionally associated with mumps include cerebellar ataxia, facial palsy, transverse myelitis, hydrocephalus, Guillain-Barré syndrome, flaccid paralysis, and behavioral changes.
Mumps pancreatitis, which may present as abdominal pain, occurs in ~4% of infections but is difficult to diagnose because an elevated serum amylase level can be associated with either parotitis or pancreatitis. An etiologic association of mumps virus and juvenile diabetes mellitus remains controversial. Myocarditis and endocardial fibroelastosis are rare and self-limited but may represent severe complications of mumps infection; however, mumps-associated electrocardiographic abnormalities have been reported in up to 15% of cases. Other unusual complications include thyroiditis, nephritis, arthritis, hepatic disease, keratouveitis, and thrombocytopenic purpura. Abnormal renal function is common, but severe, life-threatening nephritis is rare. It remains at issue whether an excessive number of spontaneous abortions are associated with gestational mumps. Mumps in pregnancy does not appear to lead to premature birth, low birth weight, or fetal malformations.
DIFFERENTIAL DIAGNOSIS
During a mumps outbreak, the diagnosis is made easily in patients with parotitis and a history of recent exposure; however, when disease incidence is low, other causes of parotitis should be considered and laboratory testing is required for case confirmation. Infectious causes of parotitis include other viruses (e.g., HIV, coxsackievirus, parainfluenza virus type 3, influenza A virus, Epstein-Barr virus, adenovirus, parvovirus B19, lymphocytic choriomeningitis virus, human herpesvirus 6), gram-positive bacteria, atypical mycobacteria, and Bartonella species. Rarely, other gram-negative or anaerobic bacteria are associated with parotitis. Parotitis can also develop in the setting of sarcoidosis, Sjögren’s syndrome, Mikulicz’s syndrome, Parinaud’s syndrome, uremia, diabetes mellitus, laundry starch ingestion, malnutrition, cirrhosis, and some drug treatments. Unilateral parotitis can be caused by ductal obstruction, cysts, and tumors. In the absence of parotitis or other salivary gland enlargement, symptoms of other visceral organ and/or CNS involvement may predominate, and a laboratory diagnosis is required. Other entities should be considered when manifestations consistent with mumps appear in organs other than the parotid. Testicular torsion may produce a painful scrotal mass resembling that seen in mumps orchitis. Other viruses (e.g., enteroviruses) may cause aseptic meningitis that is clinically indistinguishable from that due to mumps virus.
LABORATORY DIAGNOSIS
Laboratory diagnosis is primarily based on detection of viral RNA by reverse-transcriptase polymerase chain reaction (RT-PCR) or on serology. Detection of viral antigens (e.g., via mumps virus–specific immunofluorescent staining of cultured clinical specimens) is comparatively inefficient and is no longer commonly performed.
For RT-PCR-based testing, viral RNA can be extracted either directly from clinical samples or from cell cultures incubated with clinical samples. Buccal swabs appear to be the best specimens for virus detection, particularly when obtained within 2 days of clinical onset; however, mumps virus can also be detected readily in throat swabs and saliva and, in cases of meningitis, in CSF. Despite the apparent high frequency of viremia during mumps, mumps virus has rarely been detected in blood. The ability to detect viral RNA in clinical samples rapidly diminishes beyond the first week after symptom onset, and in several studies rates of virus detection were substantially lower in recipients of two vaccine doses than in unvaccinated persons or recipients of one dose. The rate of false-negative RT-PCR findings can be quite high, approaching 70% in some studies.
A serologic diagnosis of mumps is typically made by enzyme-linked immunosorbent assay (ELISA). The data must be interpreted with caution. In vaccinated persons with mumps, IgM is typically absent; thus, a negative IgM result in a vaccinated person does not rule out mumps. In addition, regardless of vaccination status, IgM may not be detectable if serum is assayed too early (prior to day 3 of symptom onset) or too late (beyond 6 weeks after symptom onset) in the course of disease. Reliance on a rise in IgG titer in paired acute- and convalescent-phase sera also is problematic: IgG titers in convalescent-phase sera may be only nominally greater than those in acute-phase sera. Thus, at present, the capacity of RNA or viral antigen detection to confirm cases is much greater than that of serologic testing. Traditional and labor-intensive serologic tests such as complement fixation, hemagglutination inhibition, and virus neutralization are now performed only rarely. The main downside to replacement of these functional serologic assays with the more rapid ELISA method is the latter’s detection of all virus-specific antibodies, including those that are nonneutralizing (i.e., nonprotective). Thus, an individual who is seropositive by ELISA may lack protective levels of antibody. While there is a strong association between the presence of mumps virus neutralizing antibody and protection from disease, an absolute antibody titer predictive of serologic protection is lacking; in this respect, mumps differs from other respiratory infections, such as measles.
PREVENTION
![]() Vaccination is the only practical control measure. Nearly all developed countries use mumps-containing vaccines, but in many countries mumps is not a notifiable disease and vaccination is often voluntary. However, where used, mumps vaccination has had a tremendous impact, with reductions in incidence and morbidity typically exceeding 90%. Despite the tremendous success of mumps vaccination programs, large mumps outbreaks continue to occur globally, even in settings of high-level two-dose vaccine coverage. Whereas outbreaks historically involved young (often unvaccinated) children in primary and secondary schools, more recent outbreaks have predominantly involved young adults, particularly on college and university campuses. While primary and secondary (waning-immunity) vaccine failures have been hypothesized to be factors in mumps outbreaks in several countries, in some countries other factors may have played a role, such as lack of compliance with the recommended vaccine schedule, changes to vaccination schedules resulting in missed cohorts, or changes in population demographics, such as large-scale immigration.
Vaccination is the only practical control measure. Nearly all developed countries use mumps-containing vaccines, but in many countries mumps is not a notifiable disease and vaccination is often voluntary. However, where used, mumps vaccination has had a tremendous impact, with reductions in incidence and morbidity typically exceeding 90%. Despite the tremendous success of mumps vaccination programs, large mumps outbreaks continue to occur globally, even in settings of high-level two-dose vaccine coverage. Whereas outbreaks historically involved young (often unvaccinated) children in primary and secondary schools, more recent outbreaks have predominantly involved young adults, particularly on college and university campuses. While primary and secondary (waning-immunity) vaccine failures have been hypothesized to be factors in mumps outbreaks in several countries, in some countries other factors may have played a role, such as lack of compliance with the recommended vaccine schedule, changes to vaccination schedules resulting in missed cohorts, or changes in population demographics, such as large-scale immigration.
In the United States, the benefit-cost ratios for mumps vaccination alone are >13 for direct costs (e.g., medical expenses) and >24 for societal costs (including productivity losses for patients and caregivers). Several mumps virus vaccines are used throughout the world; in the United States, only the live attenuated Jeryl Lynn strain is used. Current recommendations are that mumps vaccine be administered as part of the combined trivalent measles-mumps-rubella vaccine (M-M-R® II) or the quadrivalent measles-mumps-rubella-varicella vaccine (ProQuad®). Monovalent vaccine is no longer produced for the U.S. market but is available in other countries.
Before administering mumps-containing vaccine, physicians should always consult the latest recommendations from the Advisory Committee on Immunization Practices (ACIP). Current recommendations for children specify two doses of mumps-containing vaccine: the first dose given on or after the first birthday and the second dose administered no earlier than 28 days after the first. In the United States, children often receive the second dose between the ages of 4 and 6 years.
In 2009, the ACIP revised its recommendations for evidence of mumps immunity in health care personnel to include (1) documented administration of two doses of a preparation containing live mumps vaccine, (2) laboratory evidence of immunity or laboratory confirmation of disease, or (3) birth date before 1957. For unvaccinated health care personnel born before 1957 who lack laboratory evidence of mumps immunity or laboratory confirmation of mumps, health care facilities should consider two doses of MMR vaccine separated by the appropriate interval; during a mumps outbreak, vaccination of these individuals is recommended.
Mumps vaccine contains live attenuated virus. It is not recommended for pregnant women, for individuals who have had a life-threatening allergic reaction to components of the vaccine, or for people in settings of clinically significant primary or secondary immunosuppression. (For details, see the ACIP guidelines on the CDC’s website [www.cdc.gov/vaccines/pubs/acip-list.htm].) Occasionally, febrile reactions and parotitis have been reported soon after mumps vaccination. Allergic reactions after vaccination (e.g., rash and pruritus) are uncommon and are usually mild and self-limited. More serious complications, such as aseptic meningitis, have been causally associated with certain vaccine strains but not with the Jeryl Lynn strain.
Immunity to mumps is associated with the development of neutralizing antibody, although a specific correlate of protection has not been established. Seroconversion occurs in ~95% of recipients of the Jeryl Lynn strain; however, vaccine efficacy is ~80% for one dose and 90% for two doses. Recent data indicate declining seropositivity rates with time since vaccination. Studies are under way to assess the value of including a third dose in the immunization schedule. Although it is generally accepted that mumps virus is serologically monotypic, antigenic differences between virus isolates have been detected. It is unclear whether such differences can lead to immune escape. The role of the cellular arm of the immune response is unclear, but there is evidence that it may help limit virus spread and complications.
ACKNOWLEDGMENT
The authors thank and acknowledge Dr. Anne Gershon, the author of this chapter in earlier editions.
232 | Rabies and Other Rhabdovirus Infections |
RABIES
Rabies is a rapidly progressive, acute infectious disease of the central nervous system (CNS) in humans and animals that is caused by infection with rabies virus. The infection is normally transmitted from animal vectors. Rabies has encephalitic and paralytic forms that progress to death.
ETIOLOGIC AGENT
Rabies virus is a member of the family Rhabdoviridae. Two genera in this family, Lyssavirus and Vesiculovirus, contain species that cause human disease. Rabies virus is a lyssavirus that infects a broad range of animals and causes serious neurologic disease when transmitted to humans. This single-strand RNA virus has a nonsegmented, negative-sense (antisense) genome that consists of 11,932 nucleotides and encodes 5 proteins: nucleocapsid protein, phosphoprotein, matrix protein, glycoprotein, and a large polymerase protein. Rabies virus variants, which can be characterized by distinctive nucleotide sequences, are associated with specific animal reservoirs. Six other non–rabies virus species in the Lyssavirus genus have been reported to cause a clinical picture similar to rabies. Vesicular stomatitis virus, a vesiculovirus, causes vesiculation and ulceration in cattle, horses, and other animals and causes a self-limited, mild, systemic illness in humans (see “Other Rhabdoviruses,” below).
EPIDEMIOLOGY
![]() Rabies is a zoonotic infection that occurs in a variety of mammals throughout the world except in Antarctica and on some islands. Rabies virus is usually transmitted to humans by the bite of an infected animal. Worldwide, endemic canine rabies is estimated to cause 55,000 human deaths annually. Most of these deaths occur in Asia and Africa, with rural populations and children most frequently affected. Thus, in many resource-poor and resource-limited countries, canine rabies continues to be a threat to humans. However, in Latin America, rabies control efforts in dogs have been quite successful in recent years. Endemic canine rabies has been eliminated from the United States and most other resource-rich countries. Rabies is endemic in wildlife species, and a variety of animal reservoirs have been identified in different countries. Surveillance data from 2012 identified 6162 confirmed animal cases of rabies in the United States (including Puerto Rico). Only 8% of these cases were in domestic animals, including 257 cases in cats, 84 in dogs, and 115 in cattle. In North American wildlife reservoirs, including bats, raccoons, skunks, and foxes, the infection is endemic, with involvement of one or more rabies virus variants in each reservoir species (Fig. 232-1). “Spillover” of rabies to other wildlife species and to domestic animals occurs. Bat rabies virus variants are present in every state except Hawaii and are responsible for most indigenously acquired human rabies cases in the United States. Raccoon rabies is endemic along the entire eastern coast of the United States. Skunk rabies is present in the midwestern states, with another focus in California. Rabies in foxes occurs in Texas, New Mexico, Arizona, and Alaska.
Rabies is a zoonotic infection that occurs in a variety of mammals throughout the world except in Antarctica and on some islands. Rabies virus is usually transmitted to humans by the bite of an infected animal. Worldwide, endemic canine rabies is estimated to cause 55,000 human deaths annually. Most of these deaths occur in Asia and Africa, with rural populations and children most frequently affected. Thus, in many resource-poor and resource-limited countries, canine rabies continues to be a threat to humans. However, in Latin America, rabies control efforts in dogs have been quite successful in recent years. Endemic canine rabies has been eliminated from the United States and most other resource-rich countries. Rabies is endemic in wildlife species, and a variety of animal reservoirs have been identified in different countries. Surveillance data from 2012 identified 6162 confirmed animal cases of rabies in the United States (including Puerto Rico). Only 8% of these cases were in domestic animals, including 257 cases in cats, 84 in dogs, and 115 in cattle. In North American wildlife reservoirs, including bats, raccoons, skunks, and foxes, the infection is endemic, with involvement of one or more rabies virus variants in each reservoir species (Fig. 232-1). “Spillover” of rabies to other wildlife species and to domestic animals occurs. Bat rabies virus variants are present in every state except Hawaii and are responsible for most indigenously acquired human rabies cases in the United States. Raccoon rabies is endemic along the entire eastern coast of the United States. Skunk rabies is present in the midwestern states, with another focus in California. Rabies in foxes occurs in Texas, New Mexico, Arizona, and Alaska.
FIGURE 232-1 Distribution of the major rabies virus variants among wild terrestrial reservoirs in the United States and Puerto Rico, 2008–2012. *Potential host-shift event. (From JL Dyer et al: J Am Vet Med Assoc 243:805, 2013.)
In Canada and Europe, epizootics of rabies in red foxes have been well controlled with the use of baits containing rabies vaccine. A similar approach is used in Canada to control raccoon rabies.
Rabies virus variants isolated from humans or other mammalian species can be identified by reverse-transcription polymerase chain reaction (RT-PCR) amplification and sequencing or by characterization with monoclonal antibodies. These techniques are helpful in human cases with no known history of exposure. Worldwide, most human rabies is transmitted from dogs in countries with endemic canine rabies and dog-to-dog transmission, and human cases can be imported by travelers returning from these regions. In North America, human disease is usually associated with transmission from bats; there may be no known history of bat bite or other bat exposure in these cases. Most human cases are due to a bat rabies virus variant associated with silver-haired and tricolored bats. These are small bats whose bite may not be recognized, and the virus has adapted for replication at skin temperature and in cell types that are present in the skin.
Transmission from nonbite exposures is relatively uncommon. Aerosols generated in the laboratory or in caves containing millions of Brazilian free-tail bats have rarely caused human rabies. Transmission has resulted from corneal transplantation and also from solid organ transplantation and a vascular conduit (for a liver transplant) from undiagnosed donors with rabies in Texas, Florida, and Germany. Human-to-human transmission is extremely rare, although hypothetical concern about transmission to health care workers has prompted the implementation of barrier techniques to prevent exposures.
PATHOGENESIS
The incubation period of rabies (defined as the interval between exposure and the onset of clinical disease) is usually 20–90 days but in rare cases is as short as a few days or >1 year. During most of the incubation period, rabies virus is thought to be present at or close to the site of inoculation (Fig. 232-2). In muscles, the virus is known to bind to nicotinic acetylcholine receptors on postsynaptic membranes at neuromuscular junctions, but the exact details of viral entry into the skin and SC tissues have not yet been clarified. Rabies virus spreads centripetally along peripheral nerves toward the CNS at a rate of up to ~250 mm/d via retrograde fast axonal transport to the spinal cord or brainstem. Once the virus enters the CNS, it rapidly disseminates to other regions of the CNS via fast axonal transport along neuroanatomic connections. Neurons are prominently infected in rabies; infection of astrocytes is unusual. After CNS infection becomes established, there is centrifugal spread along sensory and autonomic nerves to other tissues, including the salivary glands, heart, adrenal glands, and skin. Rabies virus replicates in acinar cells of the salivary glands and is secreted in the saliva of rabid animals that serve as vectors of the disease. There is no well-documented evidence for hematogenous spread of rabies virus.
FIGURE 232-2 Schematic representation of the pathogenetic events following peripheral inoculation of rabies virus by an animal bite. (Adapted from Jackson AC: Human disease, in Rabies: scientific basis of the disease and its management, 3rd ed., AC Jackson [ed], Oxford, UK, Elsevier Academic Press, 2013, pp 269–298; with permission.)
Pathologic studies show mild inflammatory changes in the CNS in rabies, with mononuclear inflammatory infiltration in the leptomeninges, perivascular regions, and parenchyma, including microglial nodules called Babes nodules. Degenerative neuronal changes usually are not prominent, and there is little evidence of neuronal death; neuronophagia is observed occasionally. The pathologic changes are surprisingly mild in light of the clinical severity and fatal outcome of the disease. The most characteristic pathologic finding in rabies is the Negri body (Fig. 232-3). Negri bodies are eosinophilic cytoplasmic inclusions in brain neurons that are composed of rabies virus proteins and viral RNA. These inclusions occur in a minority of infected neurons, are commonly observed in Purkinje cells of the cerebellum and in pyramidal neurons of the hippocampus, and are less frequently seen in cortical and brainstem neurons. Negri bodies are not observed in all cases of rabies. The lack of prominent degenerative neuronal changes has led to the concept that neuronal dysfunction—rather than neuronal death—is responsible for clinical disease in rabies. The basis for behavioral changes, including the aggressive behavior of rabid animals, is not well understood.
FIGURE 232-3 Three large Negri bodies in the cytoplasm of a cerebellar Purkinje cell from an 8-year-old boy who died of rabies after being bitten by a rabid dog in Mexico. (From AC Jackson, E Lopez-Corella: N Engl J Med 335:568, 1996. © Massachusetts Medical Society.)
CLINICAL MANIFESTATIONS
In rabies, the emphasis must be on postexposure prophylaxis (PEP) initiated before any symptoms or signs develop. Rabies should usually be suspected on the basis of the clinical presentation. The disease usually presents as atypical encephalitis with relative preservation of consciousness. Rabies may be difficult to recognize late in the clinical course when progression to coma has occurred. A minority of patients present with acute flaccid paralysis. There are prodromal, acute neurologic, and comatose phases that usually progress to death despite aggressive therapy (Table 232-1).
CLINICAL STAGES OF RABIES |
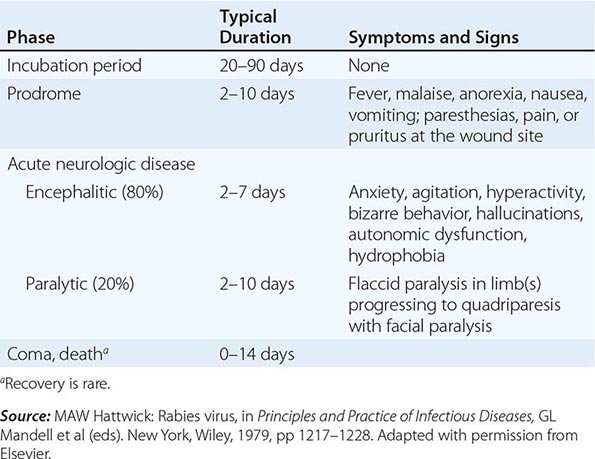
Prodromal Features The clinical features of rabies begin with nonspecific prodromal manifestations, including fever, malaise, headache, nausea, and vomiting. Anxiety or agitation may also occur. The earliest specific neurologic symptoms of rabies include paresthesias, pain, or pruritus near the site of the exposure, one or more of which occur in 50–80% of patients and strongly suggest rabies. The wound has usually healed by this point, and these symptoms probably reflect infection with associated inflammatory changes in local dorsal root or cranial sensory ganglia.
Encephalitic Rabies Two acute neurologic forms of rabies are seen in humans: encephalitic (furious) in 80% and paralytic in 20%. Some of the manifestations of encephalitic rabies, including fever, confusion, hallucinations, combativeness, and seizures, may be seen in other viral encephalitides as well. Autonomic dysfunction is common and may result in hypersalivation, gooseflesh, cardiac arrhythmia, and priapism. In encephalitic rabies, episodes of hyperexcitability are typically followed by periods of complete lucidity that become shorter as the disease progresses. Rabies encephalitis is distinguished by early brainstem involvement, which results in the classic features of hydrophobia (involuntary, painful contraction of the diaphragm and accessory respiratory, laryngeal, and pharyngeal muscles in response to swallowing liquids) and aerophobia (the same features caused by stimulation from a draft of air). These symptoms are probably due to dysfunction of infected brainstem neurons that normally inhibit inspiratory neurons near the nucleus ambiguus, resulting in exaggerated defense reflexes that protect the respiratory tract. The combination of hypersalivation and pharyngeal dysfunction is also responsible for the classic appearance of “foaming at the mouth” (Fig. 232-4). Brainstem dysfunction progresses rapidly, and coma—followed within days by death—is the rule unless the course is prolonged by supportive measures. With such measures, late complications can include cardiac and/or respiratory failure, disturbances of water balance (syndrome of inappropriate antidiuretic hormone secretion or diabetes insipidus), noncardiogenic pulmonary edema, and gastrointestinal hemorrhage. Cardiac arrhythmias may be due to dysfunction affecting vital centers in the brainstem or to myocarditis. Multiple-organ failure is common in patients treated aggressively in critical care units.
FIGURE 232-4 Hydrophobic spasm of inspiratory muscles associated with terror in a patient with encephalitic (furious) rabies who is attempting to swallow water. (Copyright DA Warrell, Oxford, UK; with permission.)
Paralytic Rabies About 20% of patients have paralytic rabies in which muscle weakness predominates and cardinal features of encephalitic rabies (hyperexcitability, hydrophobia, and aerophobia) are lacking. There is early and prominent flaccid muscle weakness, often beginning in the bitten extremity and spreading to produce quadriparesis and facial weakness. Sphincter involvement is common, sensory involvement is usually mild, and these cases are commonly misdiagnosed as Guillain-Barré syndrome. Patients with paralytic rabies generally survive a few days longer than those with encephalitic rabies, but multiple-organ failure nevertheless ensues.
LABORATORY INVESTIGATIONS
Most routine laboratory tests in rabies yield normal results or show nonspecific abnormalities. Complete blood counts are usually normal. Examination of cerebrospinal fluid (CSF) often reveals mild mononuclear cell pleocytosis with a mildly elevated protein level. Severe pleocytosis (>1000 white cells/μL) is unusual and should prompt a search for an alternative diagnosis. CT head scans are usually normal in rabies. MRI brain scans may show signal abnormalities in the brainstem or other gray-matter areas, but these findings are variable and nonspecific. Electroencephalograms show only nonspecific abnormalities. Of course, important tests in suspected cases of rabies include those that may identify an alternative, potentially treatable diagnosis (see “Differential Diagnosis,” below).
DIAGNOSIS
In North America, a diagnosis of rabies often is not considered until relatively late in the clinical course, even with a typical clinical presentation. This diagnosis should be considered in patients presenting with acute atypical encephalitis or acute flaccid paralysis, including those in whom Guillain-Barré syndrome is suspected. The absence of an animal-bite history is common in North America. The lack of hydrophobia is not unusual in rabies. Once rabies is suspected, rabies-specific laboratory tests should be performed to confirm the diagnosis. Diagnostically useful specimens include serum, CSF, fresh saliva, skin biopsy samples from the neck, and brain tissue (rarely obtained before death). Because skin biopsy relies on the demonstration of rabies virus antigen in cutaneous nerves at the base of hair follicles, samples are usually taken from hairy skin at the nape of the neck. Corneal impression smears are of low diagnostic yield and are generally not performed. Negative antemortem rabies-specific laboratory tests never exclude a diagnosis of rabies, and tests may need to be repeated after an interval for diagnostic confirmation.
Rabies Virus–Specific Antibodies In a previously unimmunized patient, serum neutralizing antibodies to rabies virus are diagnostic. However, because rabies virus infects immunologically privileged neuronal tissues, serum antibodies may not develop until late in the disease. Antibodies may be detected within a few days after the onset of symptoms, but some patients die without detectable antibodies. The presence of rabies virus–specific antibodies in the CSF suggests rabies encephalitis, regardless of immunization status. A diagnosis of rabies is questionable in patients who recover from rabies without developing serum neutralizing antibodies to rabies virus.
RT-PCR Amplification Detection of rabies virus RNA by RT-PCR is highly sensitive and specific. This technique can detect virus in fresh saliva samples, skin, CSF, and brain tissues. In addition, RT-PCR with genetic sequencing can distinguish among rabies virus variants, permitting identification of the probable source of an infection.
Direct Fluorescent Antibody Testing Direct fluorescent antibody (DFA) testing with rabies virus antibodies conjugated to fluorescent dyes is highly sensitive and specific and can be performed quickly and applied to skin biopsies and brain tissue. In skin biopsies, rabies virus antigen may be detected in cutaneous nerves at the base of hair follicles.
DIFFERENTIAL DIAGNOSIS
The diagnosis of rabies may be difficult without a history of animal exposure, and no exposure to an animal (e.g., a bat) may be recalled. The presentation of rabies is usually quite different from that of acute viral encephalitis due to most other causes, including herpes simplex encephalitis and arboviral (e.g., West Nile) encephalitis. Early neurologic symptoms may occur at the site of the bite, and there may be early features of brainstem involvement with preservation of consciousness. Anti-N-methyl-D-aspartate receptor (anti-NMDA) encephalitis occurs in young patients (especially females) and is characterized by behavioral changes, autonomic instability, hypoventilation, and seizures. Postinfectious (immune-mediated) encephalomyelitis may follow influenza, measles, mumps, and other infections; it may also occur as a sequela of immunization with rabies vaccine derived from neural tissues, which are used only in resource-limited and resource-poor countries. Rabies may present with unusual neuropsychiatric symptoms and may be misdiagnosed as a psychiatric disorder. Rabies hysteria may occur as a psychological response to the fear of rabies and is often characterized by a shorter incubation period than rabies, aggressive behavior, inability to communicate, and a long course with recovery.
As previously mentioned, paralytic rabies may mimic Guillain-Barré syndrome. In these cases, fever, bladder dysfunction, a normal sensory examination, and CSF pleocytosis favor a diagnosis of rabies. Conversely, Guillain-Barré syndrome may occur as a complication of rabies vaccination with a neural tissue–derived product (e.g., suckling mouse brain vaccine) and may be mistaken for paralytic rabies (i.e., vaccine failure).