Objectives
- State the Henderson-Hasselbalch equation for the carbon dioxide-bicarbonate buffer system.
- State the major sources for the input of fixed acids and bases into the body, including metabolic processes and activities of the gastrointestinal tract.
- Describe how the input of fixed acids and bases affects body levels of bicarbonate.
- Explain why body levels of carbon dioxide are usually not altered by the input of fixed acids and bases.
- Explain why some low-pH fluids alkalinize the blood after they are metabolized.
- Describe the reabsorption of filtered bicarbonate by the proximal tubule.
- Describe how bicarbonate is excreted in response to an alkaline load.
- Describe how excretion of acid and generation of new bicarbonate are linked.
- Describe how the titration of filtered bases is a means of excreting acid.
- Describe how the conversion of glutamine to ammonium and subsequent excretion of ammonium accomplishes the goal of excreting acid.
- Describe how the kidneys handle ammonium that has been secreted in the proximal tubule.
- State how total acid excretion is related to titratable acidity and ammonium excretion.
- Define the 4 categories of primary acid-base disturbance and the meaning of compensation.
- Describe the renal response to respiratory acid-base disorders.
- Identify the primary types of renal tubular acidosis (RTA).
Overview
A key task of the body is to regulate acid-base balance. Perturbations in acid-base balance are among the most important problems confronting clinicians in a hospital setting. The kidneys are major players in the excretion of acids and bases and in the maintenance of acid-base balance. As explained below, the kidneys work in partnership with the respiratory system to keep plasma acid-base status within normal limits.
It is essential for the body to control the concentration of free protons (hydrogen ions) in the ECF. Although most substances regulated by renal processes exist at plasma levels in the millimolar range or greater, the normal hydrogen ion concentration is a seemingly miniscule 40 nanomolar (1 nanomole is 1 millionth of a millimole). Even though very small, this level is crucial for body function. Proteins contain titratable groups that reversibly bind hydrogen ions. As sites on membrane proteins protonate and deprotonate in response to changes in extracellular pH, the resulting alteration of local charge density affects the shape, and therefore the behavior of those proteins. The plasma levels of hydrogen ions are constantly being altered by a number of processes, including (1) metabolism of ingested food, (2) secretions of the gastrointestinal (GI) tract, (3) de novo generation of acids and bases from metabolism of stored fat and glycogen, and (4) changes in the production of carbon dioxide.
The essence of the physiological response to these changes comes down to 2 processes: (1) matching the excretion of acid-base equivalents to their input, that is, maintaining balance, and (2) regulating the ratio of weak acids to their conjugate bases in buffer systems. Buffer systems limit changes in pH to a small range. The 2 processes of excreting acids and bases, and regulating physiological buffer concentrations are intimately related, but they are not identical. It is possible to be in balance even though buffer ratios are inappropriate.
Acid-Base Fundamentals
Acid-base physiology differs from that of the other substances discussed earlier in the text in a fundamental way. Mineral ions and organic solutes such as urea exist in the body as independent entities. We can describe the concentration of any one of these substances without reference to any others. Acid-base physiology is different. It always involves a set of 3 interacting substances: an acid, a base, and a hydrogen ion (proton). The existence of one implies the existence of the other two, and changes in one always bring about changes in the others. As shown in Equation 9-1, acids dissociate into a conjugate base and a hydrogen ion, and bases combine with hydrogen ions to form an acid.1 At equilibrium the relation between the 3 substances is shown in Equation 9-2, where K is the dissociation constant. Strong acids such as hydrochloric acid fully dissociate and release all their hydrogen ions when dissolved in water. If we dissolve 1 mmol of hydrochloric acid, we produce 1 mmol of free hydrogen ions. On the other hand, weak acids such as acetic acid or lactic acid keep most of the hydrogen ions bound. If we dissolve 1 mmol of a weak acid, the resulting number of free hydrogen ions is only a few percent of 1 mmol. Despite this small fraction, a weak acid present at a millimolar concentration in the blood would dissociate enough hydrogen ions to completely overwhelm the existing nanomolar level of free hydrogen ions if buffer systems did not intervene.
Although the existence of an acid in solution always implies the existence of some of its conjugate base, if we were to place just weak acid in solution there would only be miniscule amounts of the conjugate base because of limited dissociation. However, we can independently increase the conjugate base concentration by adding the salt of the acid. For example, we could mix acetic acid and potassium acetate and have substantial levels of both the acid and the conjugate base. Such a mixture is a buffer system. A buffer system serves the useful purpose of limiting the change in pH upon addition of other acids or bases. When another acid is added, most of the hydrogen ions released by that acid combine with the conjugate base of the buffer system, greatly restricting the increase in free hydrogen ions. Similarly, when another base is added, most of the free hydrogen ions removed by the base are replaced by hydrogen ions that dissociate from the acid of the buffer system.
In any buffer system, the ratio of the acid to its conjugate base fixes the free aqueous concentration of hydrogen ion (which is only a trivial fraction of the concentration of either the acid or base). This relation is shown in Equation 9-2 or in the more familiar pH form (the Henderson-Hasselbalch equation) in Equation 9-3. We should emphasize that buffer systems in the body do not eliminate added acid or base equivalents, but only limit the effect of the equivalents on blood pH. In the face of persistent imbalance between input and output, the acid or base component of the buffer is gradually reduced in concentration as it is converted to the other component. Eventually acid or base equivalents added to the body, even though transiently associated with blood buffers, must be excreted by the kidneys to maintain balance.
Buffer systems exist in the extracellular fluid, the intracellular fluid (the cytosol of the various cells in the body), and the matrix of bone. Although these buffers are in different compartments, they communicate with each other. Phosphate and albumin are important buffers in the ECF. Hemoglobin in red blood cells is an important intracellular buffer, since changes in plasma pH lead to uptake or release of protons from red blood cells. For several reasons, the most important buffer system in the body turns out to be the CO2-bicarbonate buffer system. Fortunately, we can understand the role of buffers in acid-base balance by looking at this single buffer system alone and ignore the others because all buffer systems must have ratios of weak acid to conjugate base that result in the same pH.
![]() One property that sets the CO2-bicarbonate buffer system apart from other buffer systems is that the concentrations of CO2 and bicarbonate are regulated independently. And because the concentrations of both components are regulated, the ratio of their concentrations is also regulated. Therefore, this regulates pH.
One property that sets the CO2-bicarbonate buffer system apart from other buffer systems is that the concentrations of CO2 and bicarbonate are regulated independently. And because the concentrations of both components are regulated, the ratio of their concentrations is also regulated. Therefore, this regulates pH.
In the CO2-bicarbonate buffer system, CO2 is not a weak acid per se, but it acts like a weak acid because it readily combines with water to form carbonic acid (H2CO3). Whenever a solution contains CO2, it always contains a small amount of carbonic acid. (CO2 is often called a volatile acid because it can evaporate. All other acids, for example, sulfuric, lactic, are called fixed acids.) Carbonic acid dissociates, as does any other weak acid, into a proton and its conjugate base, which is bicarbonate (Equation 9-4a). Considered this way, and given the ubiquitous presence of water in our body, it is clear that carbon dioxide is effectively an acid.
The concentration of carbonic acid in our blood is trivial (about 3 μmol/L), and at first glance it appears that this system has little buffering capacity. However, the supply of CO2 is effectively infinite because it is being produced continuously (over 10 mol per day). Any carbonic acid consumed in a reaction is immediately replaced by new generation from existing CO2 as shown in left half of Equation 9-4a.
The reaction on the left side of Equation 9-4 a to form carbonic acid is rather slow, but most tissues express one or several isoforms of the enzyme, carbonic anhydrase, intracellularly, extracellularly, or both. This enzyme greatly speeds the reaction between CO2 and water to form bicarbonate and a hydrogen ion. In doing so it actually skips the step of forming carbonic acid, as shown in Equation 9-4b.2 However, as with all enzyme-catalyzed reactions, the enzyme increases the velocity of the reaction but not the equilibrium concentrations of reactants and products.
Unlike the other buffer systems in the body, where addition or loss of hydrogen ions changes the concentration of the weak acid, in the CO2-bicarbonate system, the concentration of the weak acid (CO2) is held essentially constant. This is because the rate of respiratory excretion is matched to metabolic production. The partial pressure of arterial CO2 (PCO2) is regulated to be about 40 mm Hg. This partial pressure corresponds to a CO2 concentration in blood of 1.2 mmol/L. Any change in PCO2 resulting from the addition or loss of hydrogen ions or change in metabolic production is sensed by arterial chemoreceptors and chemoreceptors in the brainstem, which alter the rate of ventilation to restore the concentration. There are times when the PCO2 does indeed differ from 40 mm Hg, but this reflects changes in the activity of the respiratory system, not a change in PCO2 in response to addition or loss of hydrogen ions.
Although adding or removing hydrogen ions from a source other than CO2 does not change PCO2, such changes do change the concentration of bicarbonate. Adding hydrogen ions drives the reaction in Equations 9-4 to the left and reduces bicarbonate on a nearly mole-for-mole basis. We say nearly because the other blood buffers also take up some of the load. Removing hydrogen ions drives the reaction to the right and increases bicarbonate in the same way. There are many processes that add or remove hydrogen ions, but regardless of the process, the result is to change the concentration of bicarbonate. This is a crucial concept, and therefore warrants repeating. From an acid-base perspective, any metabolic process or reaction that produces hydrogen ions is identical to one that removes bicarbonate (because in both cases the end result is loss of bicarbonate), and any reaction in which a hydrogen ion is a reactant is equivalent to one in which bicarbonate is a product (because in both cases the end result is an increase in bicarbonate).
Adding hydrogen ions = loss of bicarbonate; removing hydrogen ions = addition of bicarbonate. |
From the foregoing, we conclude that the task of maintaining hydrogen ion balance really becomes one of maintaining bicarbonate balance (again assuming that the respiratory system keeps PCO2 constant) because the pH is set by the ratio of bicarbonate to PCO2. When hydrogen ions are added (or bicarbonate is removed), the body has to generate new bicarbonate to replace that which was lost. Analogously, the removal of hydrogen ions (or addition of base) increases bicarbonate, and the extra bicarbonate has to be excreted. Excretion and generation of new bicarbonate is the responsibility of the kidneys.
![]() Before moving on, let us clarify a common misconception. At first glance it might seem that fixed acid equivalents could be converted to CO2 and excreted by exhalation. Fixed acids entering the blood indeed generate CO2. But they also consume bicarbonate, and just exhaling the CO2 does not restore the bicarbonate that disappeared when the acid was added. Without actual renal excretion of those fixed acid equivalents, continuous input would soon reduce plasma bicarbonate to zero. Furthermore, CO2 cannot be converted to fixed acid and excreted in the urine. No more than a few millimoles of CO2 are dissolved in the daily excretion of urine, and far less carbonic acid. If somehow the kidneys could convert metabolic CO2 to fixed acid and add it to the urine, this would generate over 10,000 milliosmoles of solute per day—clearly an impossible load to excrete.
Before moving on, let us clarify a common misconception. At first glance it might seem that fixed acid equivalents could be converted to CO2 and excreted by exhalation. Fixed acids entering the blood indeed generate CO2. But they also consume bicarbonate, and just exhaling the CO2 does not restore the bicarbonate that disappeared when the acid was added. Without actual renal excretion of those fixed acid equivalents, continuous input would soon reduce plasma bicarbonate to zero. Furthermore, CO2 cannot be converted to fixed acid and excreted in the urine. No more than a few millimoles of CO2 are dissolved in the daily excretion of urine, and far less carbonic acid. If somehow the kidneys could convert metabolic CO2 to fixed acid and add it to the urine, this would generate over 10,000 milliosmoles of solute per day—clearly an impossible load to excrete.
1A base is any substance that can bind a hydrogen ion (eg, OH−, lactate−). The term “conjugate base” means the particular base formed when a given acid dissociates a proton.
2The actual reaction involves combining CO2 with a hydroxyl ion (OH−) that is bound to the enzyme, resulting in the immediate formation of bicarbonate (HCO3−). As the bicarbonate dissociates from the enzyme, a water molecule replaces it. The water is then split into a hydrogen ion and a hydroxyl ion. The hydrogen ion dissociates from the enzyme, and the hydroxyl ion stays behind on the enzyme. The end result is that a CO2 molecule and a water molecule are converted to a hydrogen ion and bicarbonate, the same as if they had gone through the slower uncatalyzed reaction of first forming a carbonic acid molecule.
Sources of Acids and Bases
![]() Although the oxidative metabolism of most foodstuff is acid-base neutral, proteins contain some amino acids that contribute acid or base. When sulfur-containing amino acids and those with cationic side chains are metabolized to CO2, water, and urea, the end result is addition of fixed acid. Phosphorylated proteins also contribute to an acid load. Similarly, the oxidative metabolism of amino acids with anionic side chains adds base (consumes hydrogen ions). Depending on whether a person’s diet is high in either meat or fruit and vegetables, the net input can be acid or base. For typical American diets, the input is usually acidic.
Although the oxidative metabolism of most foodstuff is acid-base neutral, proteins contain some amino acids that contribute acid or base. When sulfur-containing amino acids and those with cationic side chains are metabolized to CO2, water, and urea, the end result is addition of fixed acid. Phosphorylated proteins also contribute to an acid load. Similarly, the oxidative metabolism of amino acids with anionic side chains adds base (consumes hydrogen ions). Depending on whether a person’s diet is high in either meat or fruit and vegetables, the net input can be acid or base. For typical American diets, the input is usually acidic.
Fruits and vegetables, particularly citrus fruit, contain many weak acids and the salts of those acids (ie, they contain a weak acid, the conjugate base, a neutral cation such potassium, and a very small amount of free hydrogen ions). We all know that citrus juice is acidic, with some fruit juices having a pH below 4.0. Interestingly, metabolism of these acidic substances alkalinizes the blood, sometimes called the fruit juice paradox. The complete oxidation of the protonated form of an organic acid (eg, citric acid) to CO2 and water is acid-base neutral, no different in principle than the oxidation of glucose. However, the complete oxidation of the base form adds bicarbonate to the body, that is, organic anions are precursors of bicarbonate. One can think of metabolizing an organic base anion as taking a hydrogen ion from the body fluids to protonate the anion, thus converting it to a neutral acid, and then oxidizing the acid. The loss of the hydrogen ion, as emphasized above, adds bicarbonate. This loss of hydrogen ions greatly exceeds the number of free hydrogen ions present in the original fruit juice. Although its pH is low, there is far more base than free hydrogen ions. Before oxidation, the mixture is acidic, but on complete oxidation to CO2 and water, the result is addition of base.
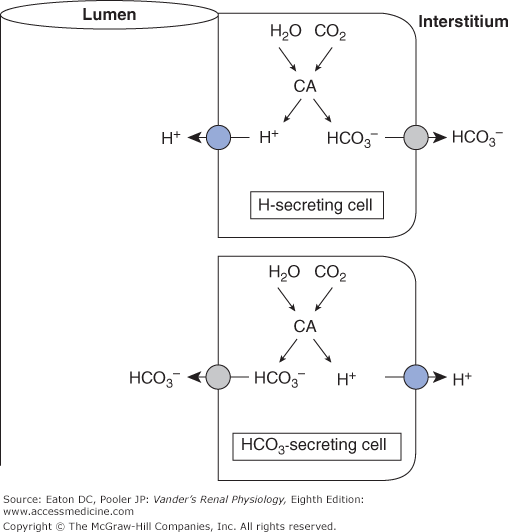
The gastrointestinal (GI) tract, from the salivary glands to the colon, is lined with an epithelium and glands that can secrete hydrogen ions, bicarbonate, or both. In addition, the major exocrine secretions of the pancreas and liver that flow into the duodenum contain large amounts of bicarbonate. To accomplish these tasks, the GI tract (and the kidneys as we discuss later) use the CO2-bicarbonate system in an ingenious way. When we combine CO2 and water to generate bicarbonate and protons within a cell, the result is always acidification of the cytosol because the concentration of protons rises. However, cells of the GI tract then separate the protons from the bicarbonate. They transport protons out of the cell in one direction (eg, into the lumen of the GI tract), and bicarbonate out the other side (eg, into the interstitium bathing the basolateral surface). Therefore the lumen becomes acidified and the surroundings (and therefore the blood leaving the tissue) becomes alkalinized (Figure 9–1). In other regions of the GI tract, the cells reverse the direction of these processes, that is, they transport bicarbonate into the lumen (alkalinizing it) and protons into the surroundings. Thus different regions of the GI acidify or alkalinize the blood. Normally, the sum of GI tract secretions is nearly acid-base neutral, ie, the secretion of acid in one site (eg, the stomach) is balanced by the secretion of bicarbonate elsewhere (eg, the pancreas). Typically, there is a small net secretion of bicarbonate into the lumen of the colon, resulting in addition of protons to the blood. However, in conditions of vomiting or diarrhea, one kind of secretion may vastly exceed the other, resulting in a major loss of acid or base from the body with a major retention of base or acid in the blood.
Figure 9–1.
Generic model of hydrogen ion secretion (upper cell) and bicarbonate secretion (lower cell). The source of secreted ions is CO2 and water. Every hydrogen ion moved out of a cell across one membrane must be accompanied by the transport of a bicarbonate ion out of the cell across the opposite membrane.
The normal oxidative metabolism of carbohydrate and fat is acid-base neutral. Both carbohydrate (glucose) and triglycerides are oxidized to CO2 and water. Although there are intermediates in the metabolism (eg, pyruvate) that are acids or bases, the sum of all the reactions is neutral. However, some conditions lead to production of fixed acids. The anaerobic metabolism of carbohydrate produces a fixed acid (lactic acid). In conditions of poor tissue perfusion or strenuous exercise, this can be a major acidifying factor. The metabolism of triglyceride to β-hydroxybutyrate and acetoacetate also adds fixed acid (ketone bodies). These processes normally do not add much of an acid load but can add a huge acid load in unusual metabolic conditions (eg, severe uncontrolled diabetes mellitus).
Another way in which acid-base loads can enter the body is via intravenous solutions. Hospitalized patients receive a variety of intravenous solutions; a common one being lactated Ringer’s solution, a mixture of salts that contains lactate at a concentration of 28 mEq/L. The pH is approximately 6.5. However, this is an alkalinizing solution for the same reason described as the fruit juice paradox earlier. Lactate is an organic anion, the conjugate base of lactic acid, and when oxidized to CO2 and water, it takes a hydrogen ion from the body fluids, thereby producing bicarbonate. Lactated Ringer’s should not be confused with a lactic acidosis associated with certain forms of shock. In those situations, the body produces equal numbers of hydrogen ions and lactate and the result is to acidify the body fluids.