Early Lesions
Plasmacytic hyperplasia
Infectious mononucleosis-like
Polymorphic PTLDs
B-Cell Neoplasms :
Diffuse large B-cell lymphoma
Burkitt lymphoma
Plasma cell myeloma
Plasmacytoma-like lesions
T-Cell Neoplasms:
Peripheral T-cell lymphoma, NOS
Hepatosplenic T-cell lymphoma
9.5.1 Early Lesions (Infectious Mononucleosis-Like PTLDs and Plasma Cell Hyperplasia)
Early lesions characteristically show retention of the architecture of involved tissues. Where lymph nodes are involved, the sinuses remain patent, and there is no extension beyond the capsule into adjacent tissues (Fig. 9.1). Reactive or hyperplastic germinal centers may be present. In plasma cell hyperplasia there are sheets of plasma cells but no architectural effacement (Fig. 9.2). Early lesions should be poly- or oligoclonal, and there should be no structural alterations in oncogenes or tumor suppressor genes. The adenotonsillar region is an important localization site for early lesions (Fig. 9.3), which tend to involve younger patients relatively soon after transplantation [9]. Patients with localized adenotonsillar PTLDs are more often females, and the outcome is generally favorable. PTLDs that resemble infectious mononucleosis are characterized by paracortical expansion (see Fig. 9.2) and a mixed population of lymphoid cells and plasma cells as well as immunoblasts, which may be numerous.
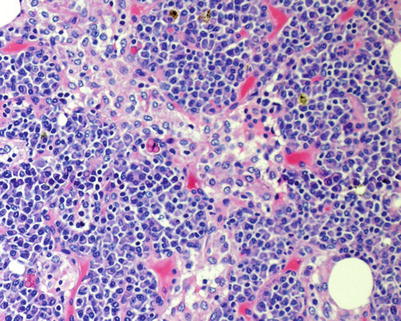
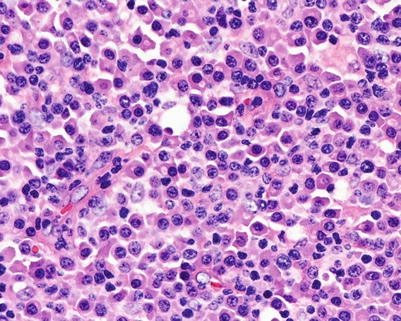
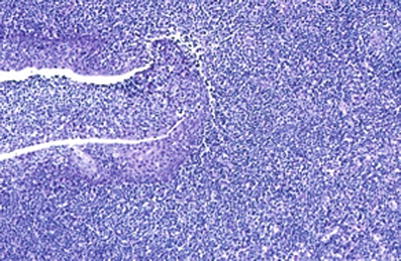

Fig. 9.1
Plasma cell hyperplasia. Section from a cervical lymph node biopsy showing retention of architecture with patent sinuses but uniform plasma cell proliferation. The cells are mature and lack cytologic abnormalities

Fig. 9.2
Plasma cell hyperplasia. There are sheets of mature plasma cells lacking cytologic atypia

Fig. 9.3
Early PTLDs involving the tonsil. A tonsillar crypt is seen with sheets of small lymphoid cells and occasional immunoblasts in this form of early PTLDs that resembles infectious mononucleosis
9.5.2 Polymorphic PTLDs
Polymorphic PTLDs (PL) include a broad spectrum of lymphoid proliferations with variable histologic appearance (Figs. 9.4, 9.5, 9.6, 9.7, 9.8, and 9.9). PL is frequently extranodal, including involvement of the gastrointestinal tract and the transplanted organ. The proliferations tend to occur relatively early after transplantation, overlapping with the early lesions. They may resolve with conservative therapy, but behavior is difficult to predict and dissemination may occur, including involvement of the central nervous system.
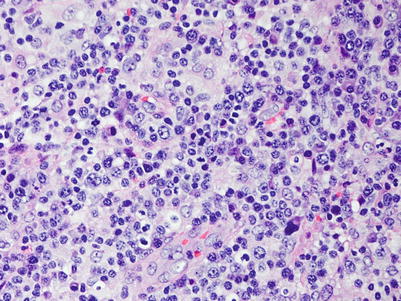
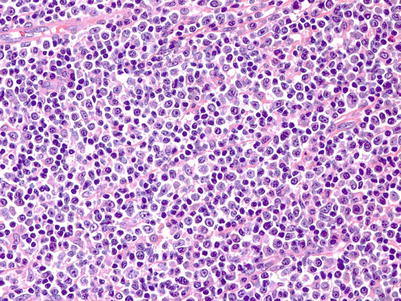
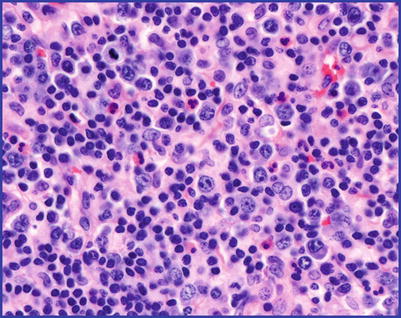
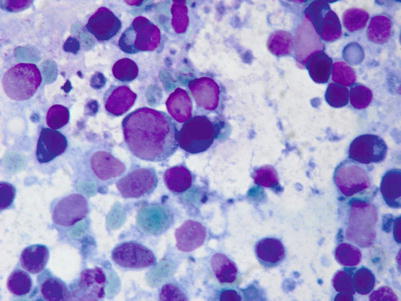
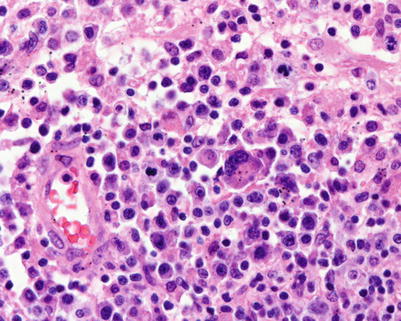
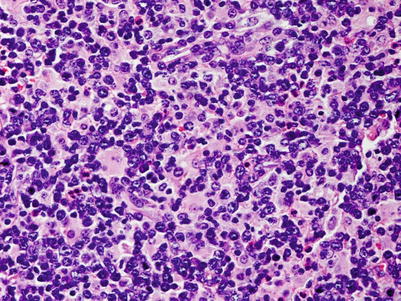

Fig. 9.4
Polymorphic PTLDs. There is a diffuse proliferation that includes small lymphocytes, histiocytes, plasma cells, and immunoblasts

Fig. 9.5
Polymorphic PTLDs. This example is more cellular with sheet-like proliferation, but it retains the mixture of cell types, including plasma cells and immunoblasts

Fig. 9.6
Polymorphic PTLDs. In this example, in addition to scattered immunoblasts there is a mixed inflammatory background, including eosinophils

Fig. 9.7
Fine-needle aspirate from a case of polymorphic PTLDs. There is a spectrum of small- and intermediate-sized lymphoid cells as well as large immunoblasts in a background of “lymphoglandular” bodies

Fig. 9.8
Polymorphic PTLDs. In this example there are markedly dysplastic immunoblastic/plasmablastic cells as well as increased mitotic figures

Fig. 9.9
Polymorphic PTLDs. Immunoblasts are evenly dispersed in a background that includes numerous reactive histiocytes
PL causes effacement of architecture by a proliferation that includes small lymphocytes, plasma cells, histiocytes, and variable numbers of transformed lymphoid cells and immunoblasts. There may be varying degrees of cytologic atypia, including the presence of Hodgkin and Reed Sternberg-like cells (Fig. 9.10). Areas of necrosis are frequently present. In addition to expression of EBV EBER, the large cells are often positive for EBV latent membrane protein (LMP1).
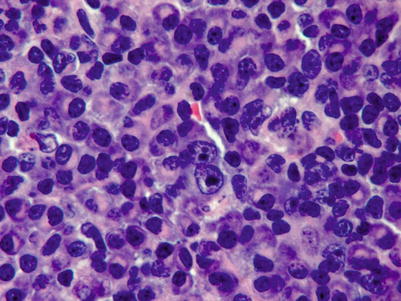

Fig. 9.10
Polymorphic PTLDs. Large immunoblasts resembling H/RS cells are seen
Despite the polymorphous appearance, PL is predominantly monoclonal in terms of immunoglobulin gene rearrangements and light-chain expression (Fig. 9.11) as well as EBV clonality, but lacks structural abnormalities in oncogenes or tumor suppressor genes. Mutations in the BCL6 gene are sometimes found, although they lack BCL6 rearrangement [10, 11].
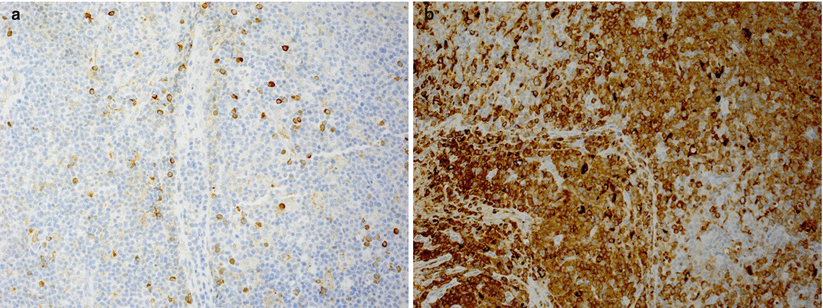

Fig. 9.11
Polymorphic PTLD stained for immunoglobulin light chain. Despite the polymorphic appearance, there is usually a light-chain restriction (in this case cytoplasmic staining for lambda light chain) in this form of PTLD. Kappa (a) and lambda (b) immunoperoxidase stains
9.5.3 Monomorphic PTLDs
Monomorphic PTLDs (ML) usually develop in older patients after a longer interval following transplantation. The lymphomas may be of B- or T-cell origin, and the T-cell types are described below. ML resembles aggressive forms of lymphoma encountered in the general, nontransplant population, particularly diffuse large B-cell lymphoma (DLBCL), and is classified according to the WHO [1]. It may present in lymph nodes and bone marrow as well as extranodal sites and may be disseminated involving the bone marrow. Different clones may be found at different sites of disease in the same patient with multifocal involvement. ML tends to have an aggressive course requiring systemic chemotherapy. It is composed of sheets of large malignant cells that may be centroblastic or immunoblastic in appearance (Figs. 9.12, 9.13, and 9.14). In addition to monoclonal immunoglobulin gene rearrangements, it often contains structural abnormalities in oncogenes or tumor suppressor genes, including MYC, TP53, and RAS.
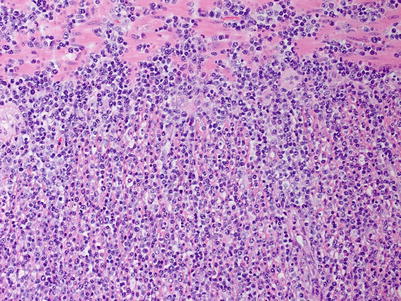
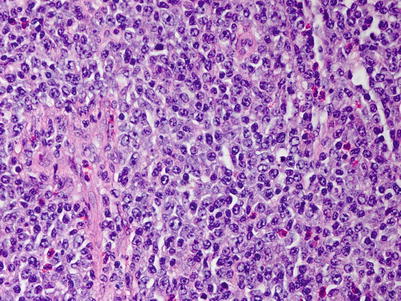
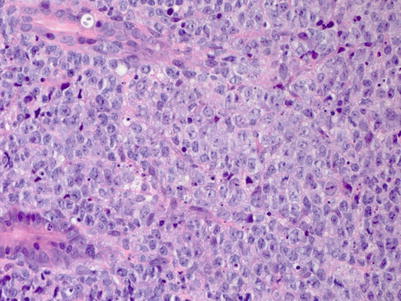

Fig. 9.12
Monomorphic PTLDs with features of diffuse large B-cell lymphoma. There are uniform large lymphoid cells infiltrating skeletal muscle in a soft-tissue mass

Fig. 9.13
Monomorphic PTLDs with features of diffuse large B-cell lymphoma. Higher power view reveals that most of the cells resemble centroblasts with round or oval nuclei and two or more evenly spaced nucleoli aligned at the nuclear membrane

Fig. 9.14
Monomorphic PTLDs with features of diffuse large B-cell lymphoma forming a mass in the wall of the colon. There are sheets of large centroblastic cells with frequent mitoses
9.5.4 Plasmablastic PTLDs
Plasmablastic PTLDs are an unusual form of monomorphic B-cell PTLDs. Most cases have been reported in male patients, and most are associated with EBV [12]. They are composed of sheets of blastic cells with prominent nucleoli and plasmacytoid cytoplasm (Fig. 9.15). They may express CD138 and be negative for CD20, and they are usually positive for EBV (Fig. 9.16). MYC/IGH rearrangements are seen in about one third of cases. Long-term remissions have been achieved, usually in combination with chemotherapy and reduced immunosuppression. Non–EBV-positive cases and those with cytogenetic abnormalities may have a worse prognosis.
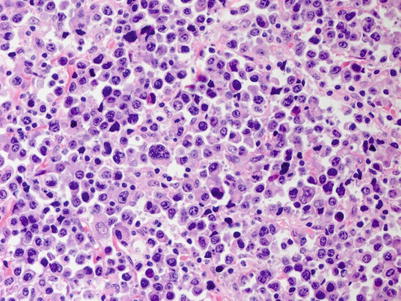

Fig. 9.15




Monomorphic PTLDs, plasmablastic. There are sheets of pleomorphic large cells with plasmablastic appearance and containing abundant amphophilic cytoplasm
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


