Fig. 32.1
Plasmonic bubble generation and detection: a cluster of gold nanoparticles in target cell, b laser pulse (green) nucleates the vapor bubble, c bubble expands and scatters a probe laser beam (purple), d–e bubble collapse
32.2.2 Methods for Plasmonic Nanobubble Detection
PNB detection employs several universal techniques that have to provide sufficient sensitivity, resolution, and speed. First, we consider imaging methods. Optical scattering phenomena allow both PNB imaging and monitoring of its dynamics [77–80, 84–88, 116].
The imaging of a transient PNB can also be realized with much more affordable equipment using slow cameras and pulsed light sources (Fig. 32.2) by using (1) pulsed illumination with a shorter duration than a PNB lifetime within a nano- and pico-second range; (2) sufficient energy of the optical pulse to compensate the very small scattering cross section of the PNB; (3) precise synchronization of the illuminating pulse with the PNB source. These requirements suggest a pulsed laser as the illumination source. The illumination angle and the numerical aperture of the collecting lens further determine the signal-to-noise ratio of this imaging scheme. Forward scattering results in the maximal scattering efficacy but cannot be fully separated from incident light that creates a high background. We achieved the best results with angled side illumination (Fig. 32.2a), providing that the numerical aperture of the imaging (collecting) lens is lower than the angle of incidence of the illuminating radiation. In this case, the lens collects only the light scattered by the PNB. This scheme allows the imaging of a single transient PNB with a lifetime down to 10 ns (Fig. 32.3). The 100 ps illumination pulse of 1 μJ at 576 nm was directed at 45° to the sample cuvette with an optically variable delay relative to the excitation pulse, and the PNB was imaged with 10X–63X microscope objectives. Most of the current CCD- and CMOS-based cameras can do such imaging. We used EMCCD (charged coupled device with electronic multiplication) cameras. In this method, the pixel image amplitude is used as a PNB metric, but it is not very practical: (1) any deviation in the alignment of the probe beam would require the re-calibration of the system, and (2) this metric will not characterize the maximal diameter of a PNB when one fixed time delay is applied. The advantage of this method is in the relatively precise identification of a PNB and its location. It also shows the number of PNBs if multiple events occur.
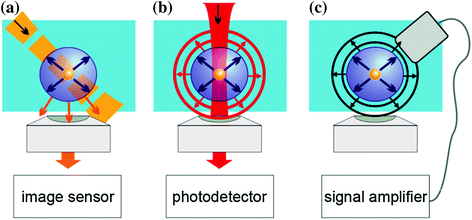
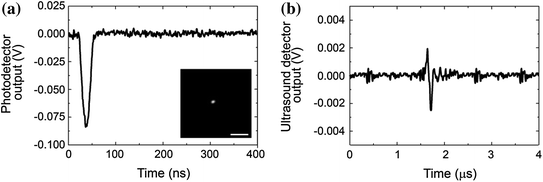

Fig. 32.2
Methods for detecting plasmonic nanobubbles in liquid: a optical time-resolved scattering imaging is obtained with a short probe laser pulse and a slow image detector, b optical scattering (extinction) time response is obtained with a continuous focused probe laser pulse and a fast photodetector, c acoustical time response is obtained with an ultrasound detector. Recreated by kind permission of AIP [116]

Fig. 32.3
Three methods of detecting a single PNB. a Optical scattering (extinction) time response obtained with continuous probe laser shows the expansion and collapse of an PNB (metrics—amplitude and duration) and time-resolved optical scattering imaging (insert) with a pulsed probe laser (metric—a pixel image amplitude). b Acoustical time response obtained with an ultrasound sensor (metric—amplitude). Scale bar is 15 μm. Recreated by kind permission of AIP [116]
The maximal diameter of a PNB is the most important parameter that also characterizes the amount of energy received by the bubble [71–76, 78]. It can be measured with another optical scattering technique that employs the monitoring of the intensity of a continuous probe beam with a broadband high-speed photodetector. The beam is focused at the PNB and its scattering by the PNB changes its axial intensity which is measured as a time response with the photodetector. In one method, the beam is pointed at the low noise detector through the aperture and produces a high base level (Fig. 32.2b). A PNB scatters the light and thus increasingly reduces the axial intensity of the probe beam during its expansion stage and brings it back to the base level during its collapse stage. This results in the bubble-specific shape of the time response (Fig. 32.3a). Technically, the detector measures the optical extinction of the probe beam, but since a PNB does not absorb any optical energy, the extinction becomes equal to its scattering component. Due to the scattering nature of such a signal, we define it as a “scattering”. This definition assumes the absence of any transient optical absorbance developed simultaneously with a PNB. If such absorbance emerges (due to plasma formation or any nonlinear modification of the optical properties of the media), the method may characterize a PNB incorrectly [117, 118]. In another method, the beam is pointed off the detector aperture and produces a low base level at the detector. In this case, only the light scattered by a PNB will reach the detector and will increase its output signal, also producing a time response with a bubble-specific shape.
The most precise way to measure the maximal diameter of a single PNB is to measure the duration of the PNB-specific signal (Fig. 32.3a) which was shown to correlate to its maximal diameter [71–76, 78]. The amplitude of the PNB response also depends upon the maximal diameter of the PNB, but is very sensitive to experimental factors that are difficult to control. The first is the diameter and divergence of the probe beam at the PNB’s plane. The smaller the beam waste, the higher the sensitivity of PNB detection. An increase in the beam’s diameter by 10–15 % and/or a slight deviation of the beam axis may cause a several-fold decrease in the signal amplitude of a dip-shaped PNB signal. Such conditions occur during changing the sample chamber or while scanning a heterogeneous sample across the beam. The simultaneous generation of multiple PNBs around different sources of thermal energy (for example, in suspension around optically absorbing molecules or NPs) instead of a single one, increases the signal amplitude, but such an increase is just an ensemble effect. The simultaneous generation of several PNBs of different maximal sizes influences not only the amplitude but also the shape of the PNB time response. Smaller PNBs form a sharp front and fewer large PNBs form a long tail of lower amplitude. While the generation of PNBs under some settings may typically involve such a multi-PNB pattern, it is still possible to estimate the lifetime of the largest PNBs that represent the maximal deposition of the thermal energy by measuring the duration of the time response at the reduced level instead of at half of maximum. However, the best rule is to avoid such distorted signals because they may lead to misinterpretation of the bubble parameters [117]. In all three considered cases (single PNB, single PNB with misaligned probe beam, multiple PNBs), the durations of the bubble-specific signals could be linked to the maximal diameter of the PNB, while their amplitudes differed significantly, and therefore, their use as PNB metrics may lead to artifacts [118].
The third method of PNB detection employs the pressure waves generated during the bubble expansion and collapse [93, 96] (Fig. 32.2c). In the case of a single inertial bubble, the pressure and rarefaction waves produce a typical signal (Fig. 32.3b). This is definitely the simplest and the least expensive method of the three, because it does not require expensive optical sources, optics, and sensors. In addition, it detects PNBs in optically opaque media, so is probably the most universal of the methods. The most obvious metric of the acoustic signal is its maximal (peak-to-peak) amplitude. However, the correct identification and measurement of a PNB with a standard acoustic detector can be challenging in a single pulse mode. Most detectors have a resonant frequency of 10 MHz and lower where their sensitivity is the maximal. For small PNBs with lifetimes from 5 to 100 ns, the corresponding pressure rise times and associated frequencies seem to be higher than the resonant frequencies of most acoustic sensors. Therefore, the sensor signal does not show the real pressure, but rather represents a response function of the sensor. In this case, the amplitude of the output signal of the acoustic sensor significantly depends upon its resonant frequency and may cause a significant error. For a specific acoustic sensor, the amplitude of its output also depends upon the number of simultaneously generated PNBs. Therefore, a reliable measurement of the PNB diameter through the amplitude of acoustic response assumes that a single PNB is generated.
The next important task is the identification of a PNB and its discrimination from other phenomena such as transient heating or stress waves (Fig. 32.4). The optical scattering response identifies heating through a thermal lens effect that allows the detection and measurement of rapid heating and gradual cooling due to thermal diffusion (Fig. 32.4a). This purely refractive effect does not produce any optical scattering like a PNB does. Next, under high transient temperatures at short time scales, the generation of stress waves (and shock waves) also influences the refractive index of the media and creates a distinct short (relative to the bubble and heating time scale) signal of nanosecond duration that is determined by the speed of sound and the aperture of the probe beam (Fig. 32.4c). Both phenomena can be clearly distinguished from the bubble (Fig. 32.4b). However, all these three different phenomena do not much influence the shape of the acoustic response, but rather its amplitude, which is minimal for heating, increases for a bubble and becomes the maximal for stress waves. Therefore, an acoustic time response is much less bubble specific than an optical scattering time response.
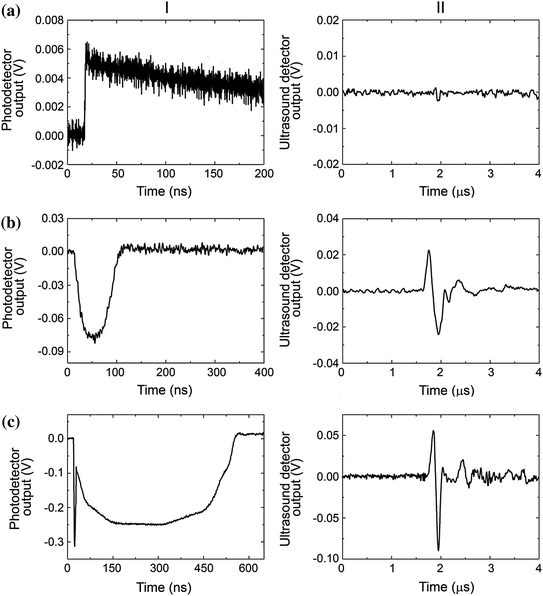

Fig. 32.4
Simultaneously obtained optical scattering (or extinction) (left) and acoustic (right) time responses of three different phenomena. a Transient heating of the gold nanoparticle and the media with a single laser pulse at a fluence below the PNB generation threshold. b Single transient vapor PNB generated around gold nanoparticle. c PNB accompanied by a stress wave that produces a sharp and short optical signal in the very beginning of the time response. Recreated by kind permission of AIP [116]
32.2.3 Tunability of Plasmonic Nanobubbles
The above analysis compares three methods for the detection of PNBs under fixed excitation conditions above the bubble generation fluence. It is important to understand the range of the linear correlation between the PNB maximal diameter and its excitation conditions. We compared the four discussed above metrics, the pixel amplitude of the scattering image, the amplitude and duration of the optical scattering time response, and the amplitude of the acoustic response, as functions of the fluence of the excitation laser pulse, the parameter that, in this model, determines the maximal diameter of the PNB when the fluence exceeds the PNB generation threshold. All bubbles were generated in water around individual identical gold nanospheres.
For the scattering image, we observed the relatively early saturation and nonlinear behavior of the pixel image amplitude compared to the duration of the time response (Fig. 32.5a). Time-resolved scattering images were obtained under a fixed delay between the excitation and probe laser pulses. With an increase in the maximal PNB diameter and lifetime, the moment of illumination moved further from the moment when the PNB reaches its maximal diameter.
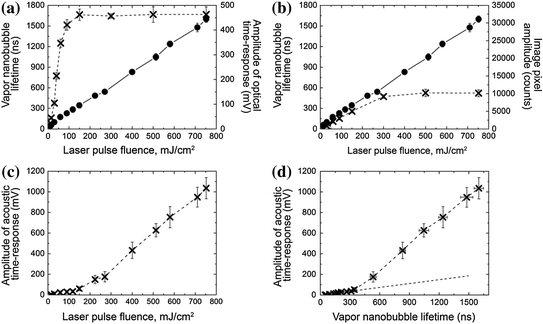

Fig. 32.5
Dependence of the PNB metrics upon the optical excitation fluence that determines the maximal size of PNB. a Lifetime (dots) and amplitude of the optical scattering time response (cross). b Lifetime (dots) of the optical scattering time response and the pixel image amplitude of the time-resolved optical scattering image (cross) at the fixed delay of 10 ns. c Amplitude of acoustic time response. d Amplitude of acoustic time response versus the lifetime of optical time response. Recreated by kind permission of AIP [116]
Next, we analyzed the amplitude of optical scattering time responses (Fig. 32.5b). The dependence of their amplitude and lifetime upon the fluence showed a nonlinear saturation of the amplitude compared to the lifetime. The nonlinear behavior of the response can be explained by the effect of the limited numerical aperture of the collecting lens and the blocking effect of the large bubble on the probe beam. In addition, at high excitation energies, the amplitude of the response can be influenced by the concurrent optical signal associated with the stress wave. Therefore, the use of the optical response amplitude as a metric of the bubble or even of the energy conversion efficacy is not appropriate and may lead to artifact data for the photothermal responses of ensemble of gold NPs [118]. In contrast, the dynamics of the optical response can be much better quantified through the lifetime that correctly describes both the maximal diameter of a PNB and the efficacy of the energy conversion.
The amplitude of the acoustic response demonstrated two quasi-linear zones (Fig. 32.5c). To explain their origin, we detected the acoustical and optical responses simultaneously for single PNBs under a wide range of excitation fluences and plotted the acoustic amplitude as a function of the PNB lifetime (Fig. 32.5d). Two zones for acoustic signals became even more pronounced with an apparent threshold associated with the PNB lifetime around 350 ns that also corresponded to the fluence of 150 mJ/cm2. A detailed analysis of the optical time responses revealed one difference: the optical responses of PNBs larger than 350 ns included an additional component that can be attributed to a stress wave generated due to the high laser-induced temperature in a gold NP (Fig. 32.4c-II). Its additional impact added to the PNB-generated pressure at the acoustic sensor. However, this effect occurs when the PNB lifetime exceeds 300 ns, which corresponds to the maximal diameter above a micrometer, so this is no longer a “nano” bubble. For smaller bubbles of nanosize, both the acoustic amplitude and the duration of the optical scattering time responses provided almost linear signal-energy dependence and showed a good correlation.
The best quantitative analysis of vapor PNBs in the widest range of excitation energies and bubble diameters can be achieved by measuring the duration (lifetime) of the optical scattering time response by using a low-power continuous laser and matching photodetector. In the case of opaque media, the amplitude of the acoustical time response provides good sensitivity and linearity in a range of excitation conditions that are limited by the onset of stress waves that additionally increase the amplitude of the nanobubble signal. The simultaneous use of two or three of these methods provides the best results in the identification, imaging, and measuring of transient nanobubbles in a single event experiment [116].
32.2.4 Spectral Properties of Plasmonic Nanobubbles
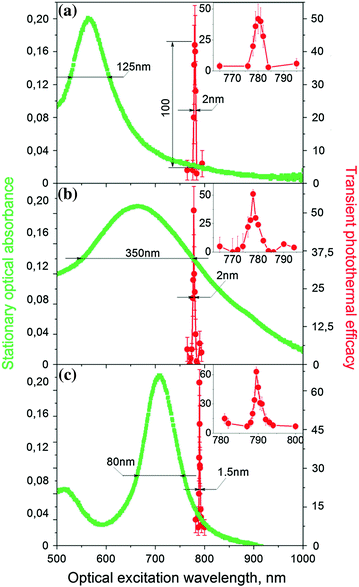
An ability to combine high NIR PT efficacy with gold nanospheres is an attractive alternative to specifically engineered NPs with NIR resonances such as nanorods, nanocages, and nanoshells. We recently observed 20–130-fold amplification of the PNB generation efficacy of solid gold nanospheres in a very narrow (2–3 nm) NIR region at 780 nm (far from their regular plasmon resonance at 530–540 nm), where they are usually considered to have poor PT performance (Fig. 32.6a). The experiments were performed with single 70 ps laser pulses at low fluence levels (10–100 mJ/cm2) associated with the generation of vapor PNBs around gold nanospheres in water. The lifetime of the nanobubble was used as the PT metric because it correlates to the thermal energy released by the NP [119, 120]. NIR amplification of the PT efficacy of nanospheres achieved or exceeded their PT efficacy at the visible wavelength of plasmon resonance. The amplification effect and the NIR peak wavelength of 780 nm were reproduced for a wide range of laser pulse fluence from 10 to 500 mJ/cm2, NP diameters (20–120 nm), aggregation states (single NPs, aggregated NP clusters and suspensions of NPs), and environments including in vitro and in vivo conditions [105, 121]. The observed NIR amplification effect has a low optical fluence threshold (>10 mJ/cm2), and its transient and resonant nature can be associated not with the nanosphere itself, but with a transient nanostructure that creates a high optical absorbance at 780 nm (where a sphere itself does not absorb well). Thus, solid gold nanospheres, which are cheap, easily available, biologically safe, stable, and easily functionalized, can be successfully employed instead of more complex and unstable NIR-engineered NPs.
Similar narrow spectral peaks were also observed for other types of NPs, gold nanorods, and hollow nanoshells (Fig. 32.6b, c) [119]. Though these NPs are specifically engineered for NIR, the observed 1–3-nm-wide peaks were significantly red-shifted and narrowed compared to their preset properties (for nanorods, plasmon resonance wavelength is at 710 nm and spectral width >100 nm, and for hollow nanoshells, plasmon resonance wavelength is at 665 nm and spectral width >160 nm).
Such narrow PT spectral peaks were never observed previously for single NPs and for their random clusters in a wide range of the excitation pulse fluences, not only near the threshold of PNB generation. Therefore, the discovered phenomenon is not a threshold energy-specific effect like in the case of nonlinear spectroscopy methods [122, 123]. On the contrary, the PT output of the NP can be precisely controlled by varying the laser fluence [78, 119, 121]. A wide range of tested NP parameters and conditions revealed the very stable and universal nature of the observed nonstationary PT amplification and spectral narrowing.
Discovered NIR spectral peaks depend on the duration, not energy, of the excitation laser pulse. We observed very strong influence of the laser pulse duration (from 20 ps to 10 ns) on the PNB generation (Fig. 32.7). The shortest pulses delivered the maximal efficacy. For longer pulses of 400 ps and 10 ns, we observed strong suppression of the PNB generation for all types of NPs, spheres, rods, and shells [119]. This suppression effect can be explained by general thermal diffusion and, in addition, by the dynamic modification of the plasmonic structures during NP interaction with a laser pulse. Thus, only short laser pulses (< 400 ps) induced large PNBs and provided the described above spectral narrowing.
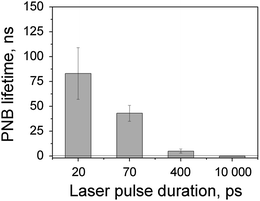

Fig. 32.7
Dependence of the PNB lifetime upon duration of the laser pulse (532 nm, 100 mJ/cm2). PNBs were generated in water around isolated solid gold spheres of 60 nm
To summarize, these results demonstrate the existence of the nonstationary mechanism with unprecedented amplification and spectral narrowing of the PT efficacy of metal NPs.
32.3 Part II Generation and Detection of Plasmonic Nanobubbles in Cells and Tissues
32.3.1 Cellular Specificity of Plasmonic Nanobubbles
The most important feature of the PNB generation mechanism that distinguishes PNBs from all other nanoprobes and nanosources is the optical energy threshold for the bubble generation around gold NP (Fig. 32.8a). Most of other photoinduced phenomena have nonthreshold nature (including fluorescence, photothermal, and photoacoustic phenomena). We have found that this threshold energy radically depends upon the diameter of the NP and the size of the NP clusters: The threshold energy decreases with the size (Fig. 32.8b) [124]. A low laser pulse fluence was sufficient to generate PNBs around large NP clusters but was not sufficient to induce PNBs around single NPs or small NP clusters. This was demonstrated by exposing multiple NP clusters of various sizes to a single laser pulse of low fluence. We observed the selective generation of PNBs only around the largest NP clusters (Fig. 32.8c) whose threshold was lower than the applied fluence [124]. The PNB threshold for smaller clusters was above this fluence, and therefore, such small NP clusters did not return PNBs in response to optical excitation.
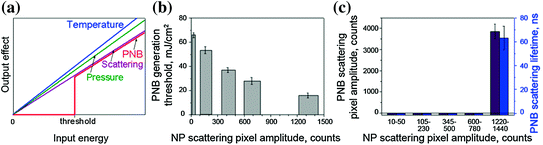

Fig. 32.8
a Threshold nature of the PNB generation compared to nonthreshold nature of other photoactivated phenomena (photoacoustic effect due to thermo-elastic expansion, optical scattering, and photothermal effect). b, c Parameters of PNBs generated around gold NP clusters in water for gold nanoshells [124]. b PNB generation threshold fluence of the excitation laser pulse as function of NP cluster size (measured through optical scattering amplitude of NP cluster image for individual clusters); c PNB lifetime and scattering brightness as function of the NP cluster size (measured through optical scattering amplitude of NP cluster image) at specific fluence of the excitation pulse (778 nm, 22 mJ/cm2)
The dependence of the PNB threshold fluence upon cluster size can be explained through the mechanism of PNB generation around superheated NPs. Merged thermal fields of several tightly aggregated NPs form a common thermal field and vapor layer around the cluster. The initial vapor pressure in such a vapor layer is determined by the fluence of the laser pulse that is converted into heat by each NP in a cluster. Next, the external pressure of surface tension (that needs to be overcome to allow the expansion of the vapor) is inversely proportional to the radius of the vapor–liquid boundary [125–127] and, therefore, decreases with cluster size. We previously analyzed the mechanism of PNB generation around NP clusters versus single NPs [78]. In addition to the above thermal and hydrodynamic factors, NP clustering may enhance their optical absorbance [58, 128], thus additionally increasing the released thermal energy and the initial vapor pressure. All these factors cause the decrease in the PNB generation threshold fluence with cluster size. With the fluence of the excitation pulse below the threshold, the PNB does not emerge and, therefore, creates no impact, unlike NPs (Fig. 32.8c).
This unique property creates the fundamental opportunity to improve the cellular selectivity of PNBs in say, cancer cells, via cancer cell-specific clustering of gold NPs conjugated to cancer-specific vectors, such as antibodies. Target cell-specific formation of the largest gold NP clusters involves several steps (Fig. 32.9a):
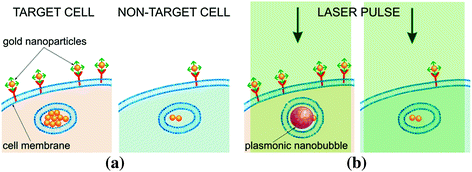

Fig. 32.9
PNBs and NPs in target (left panels) versus nontarget (right panels) cells. a Gold NP conjugates are collected at cellular membranes and are clustered during endocytosis resulting in the largest NP clusters in target cells. b Excitation laser pulse (green) of low fluence induces PNBs only around the largest NP clusters (i.e., only in target cells) because the PNB generation threshold fluence for single NPs and small clusters (nontarget cells) is higher than the fluence of the laser pulse
At the first stage, we used target-specific antibodies to provide higher accumulation of gold NPs at the membranes of target cells compared to the NP accumulation at membranes of nontarget cells. This stage did not provide desired specificity of the targeting but at the same time delivered much more NPs to target cells compared to nontarget cells.
The targeting parameters such as the concentration of NPs and the incubation time were optimized to achieve maximal difference in NP uptake between the target and nontarget cells. Such targeting does not eliminate the nonspecific uptake of NPs by nontarget cells; however, it provides the formation of the largest NP clusters only in target cells for target-specific generation of PNBs. Sphere- and shell-type NP conjugates did not induce any considerable cytotoxicity in either target or nontarget cells. The difference in the NP cluster size between cancer and normal cells create the difference in the PNB generation threshold fluence in cancer (low threshold) and normal (high threshold) cells so that the single laser pulse of specific fluence induces the PNB only in cancer cells (Fig. 32.9b).
Since the largest NP clusters can be selectively formed in target cells through the receptor-mediated endocytosis of NPs [104, 129–136], we further studied the NP cluster–PNB mechanism in living cells in order to compare the cellular specificity of NPs and PNBs under identical conditions of NP targeting and optical excitation. Several different molecular targets were investigated in vitro in cell systems that included cells with a high level of molecular target expression (target cells) and a low level of the expression of the same molecular target (nontarget cells). We studied cell models representing lung (A549), head and neck (HN31), prostate (C4-2B), epithelial (HES, a WISH/HeLa derivative), and blood (Jurkat J32) cancers, and also human T cells that are used for gene therapies of cancer. In all six cases, we observed a higher level of NP signals in target cells, but all nontarget cells also showed a significant level of NP uptake and formation of NP clusters (Figs. 32.10a, 32.11a) so that the ratio of the NP signal for target versus nontarget cells was below 10. However, higher pixel image amplitudes in target cells indicated the formation of the largest NP clusters in target cells. Next, target and nontarget cells were identically treated with single laser pulses within the range of pulse fluences for PNB generation around NP clusters. For each cell model, we experimentally determined the level of excitation pulse fluence that provided the generation of PNBs mainly in target cells and did not induce PNBs in nontarget cells (Fig. 32.10b–d). The optical scattering images and time responses of individual cells were processed to compare the corresponding metrics for NP accumulation (Fig. 32.11a) and PNB generation (Fig. 32.11b, c) in target and nontarget cells. Compared to NP signals, the PNB signals showed a much higher discrimination between target and nontarget cells in all six. Cellular specificity of NPs and PNB was quantitatively shown through the ratios of the target cell signals to the corresponding signals in nontarget cells (shown as colored numbers in each frame of Fig. 32.11). Compared to NPs, the PNBs improved cellular specificity in some models by more than one order of magnitude. While the nontarget cells showed significant uptake of NPs and even their potential aggregation into small clusters, no PNBs, or very small ones, were observed in nontarget cells under identical treatment conditions (Figs. 32.10 and 32.11) [124].
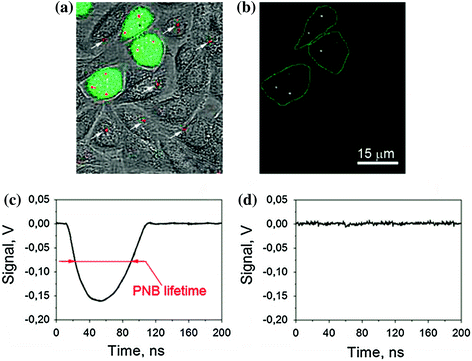
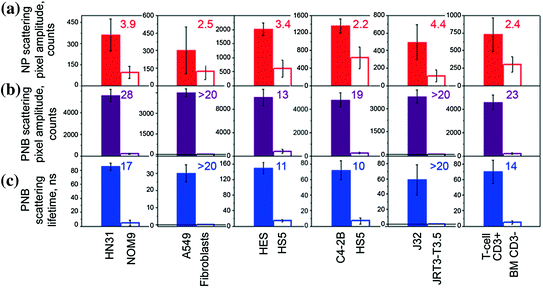

Fig. 32.10
Images and signals of gold NPs and PNBs in co-culture of target (HN31, labeled with green fluorescent protein for identification) and nontarget (NOM9) cells identically treated with 60 nm gold NSP–C225 conjugates (specific to EGFR that is overexpressed in target cells) [124]. a Overlay of bright field, fluorescent, and scattering images shows target cells (green) and gold NPs (red) that can be found in both types of cells (the arrows show NP clusters in nontarget cells); b time-resolved scattering image of the same field shows PNB images (bright white spots) only in target cells; c, d optical scattering time responses of individual target c and nontarget d cells show the PNB-specific signal only for target cell and the definition of the PNB lifetime of PNBs; time is measured from the moment of the exposure to the excitation laser pulse

Fig. 32.11
Cell population-averaged levels of optical scattering signals obtained for individual target (solid bar) and nontarget (hollow bar) cells in six cell models represented by target/nontarget cells/molecular targets [124]: squamous cell carcinoma, HN31/NOM9/EGFR (treated with 50 nm NS–panitumumab conjugates); lung cancer, A549/Fibroblast/EGFR (treated with 60 nm NSP–C225 conjugates); epithelial cancer, HES/HS5/MUC1 (treated with 60 nm NSP–214D4 conjugates); prostate cancer, C2-4B/HS5/PSMA (treated with 60 nm NSP–anti-PSMA conjugates); leukemia, J32/JRT3-T3.5/CD3; and human T cells, T cell/BM/CD3 (treated with 60 nm NSP–OKT3 conjugates) for: row (a) (red): gold NP amplitude of scattering image of gold NPs (a metric for the uptake of NPs by cells); row (b) (purple): time-resolved scattering image amplitudes of PNBs; row (c) (blue): PNB lifetimes. The ratio of the signals for target/nontarget cell is shown for each parameter and cell model and indicates the cellular specificity of NPs (row (a)) and PNBs (rows (b, c))
The difference in cellular specificity of NPs and PNBs can be clearly seen in experiments with a co-culture of target (labeled with green fluorescent protein for identification) and nontarget cells (Fig. 32.10). At a specific fluence of the excitation laser pulse (25 mJ/cm2 at 778 nm), only target cells yielded PNBs while even adjacent nontarget cells with gold NPs did not. Such a difference between NP and PNB signals was observed for all six cell models: adherent (HN31, HES, and A549) and suspension (C4-2B, T cells, and Jurkat) cells, and for all molecular targets: receptors (EGFR, CD3, and PSMA) and glycoproteins (MUC1). These results indicate the universal nature of the high cellular specificity of PNBs compared to that of gold NPs. Therefore, PNB provided better discrimination between target and nontarget cells even when such cells were heterogeneously mixed [124].
As can be seen from Fig. 32.11, the PNB method can better discriminate between target and nontarget cells compared to NPs. Cellular specificity of PNBs was more than one order of magnitude higher than that of NPs (this can be clearly seen by comparing the ratios of the corresponding signals for target and nontarget cells) [124]. Such an effect was achieved through the cluster-threshold mechanism of PNBs that prevents the generation of PNBs around nonspecifically targeted single NPs (and their small clusters).
32.3.2 NP Clustering and Plasmonic Nanobubbles Generation and Detection in Cancer Cells
The mechanism of the formation of mixed gold NP–drug carrier clusters and on-demand generation of PNBs around such clusters was studied in a co-culture of drug-resistant head and neck squamous cell carcinoma (HNSCC) cells and normal cells.
32.3.2.1 Selective Formation of Mixed Intracellular Clusters of Gold NPs and Doxil
The NP clustering and co-localization with Doxil was analyzed with confocal microscopy in scattering (for gold NPs) and fluorescent (for Doxil that contains fluorescent doxorubicin) modes (Fig. 32.12a). A co-culture of HN31 (HNSCC) and NOM9 (normal) cells was treated with C225 conjugates of solid gold 60 nm spheres and Doxil liposomes. The antibody C225 (Erbitux) was clinically proven to target the epidermal growth factor receptor (EGFR) in HNSCC [137–139] and provided intracellular clustering of gold NP–C225 conjugates through the mechanism of EGFR-mediated endocytosis [103, 129, 130]. Individual HN31 cells were identified through the fluorescence of the transfected green fluorescent protein (Fig. 32.10a). Analysis of the confocal images of cancer cells revealed complex clusters with gold NPs (blue in Fig. 32.12a) co-localized with Doxil (red in Fig. 32.12a). Adjacent normal cells also showed occasional NPs due to their nonspecific accumulation, but no large NP–Doxil clusters were detected in normal cells (Fig. 32.12a). Thus, the complex large NP–Doxil clusters were selectively self-assembled only by cancer cells during the separate administration of Doxil and gold NPs [105].
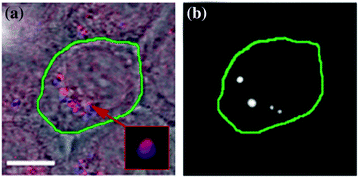

Fig. 32.12
PNB-enhanced endosomal escape in vitro. a Confocal microscopy images of cancer (green boarder) and normal (adjacent) cells show NPs (blue) and Doxil (red) co-localized in the large mixed clusters (inset) only in cancer cell, while adjacent normal cells show nonspecific uptake of NPs and Doxil (scale bar is 5 μm); b time-resolved optical scattering image of the same sample shows PNBs selectively generated with a broad single laser pulse (780 nm, 70 ps, 40 mJ/cm2) only in cancer cell and their co-localization with NP–Doxil cluster. Recreated by kind permission of Ivyspring International Publisher [105]
32.3.2.2 Generation and Detection of Plasmonic Nanobubbles in Individual Cells
We next determined the conditions of the PNB generation in the complex NP–Doxil clusters in cells. PNBs were generated by exposing the co-culture of HN31 and NOM9 cells to single NIR laser pulses (70 ps, 780 nm) of broad diameter (that provided the simultaneous irradiation of cancer and normal cells) and varying laser fluence (the energy per unit of square) in the range of 10–100 mJ/cm2. In this work, we applied for the first time the safest combination of FDA-approved solid gold spheres and NIR laser radiation in a low dose that provides the deepest tissue penetration and the minimal biodamage [140]. Optical scattering time-resolved imaging (Fig. 32.12b) and optical scattering time responses were obtained for individual cancer and normal cells simultaneously with the excitation laser pulses and were employed to image and quantify PNBs in the irradiated cells.
Both images (Fig. 32.12b) and time responses [105] showed the cancer cell-specific generation of PNBs, while normal bystander cells in the co-culture produced no or very small PNBs in a wide range of laser pulse fluences (Fig. 32.12b). This demonstrated the selectivity of the cluster-threshold mechanism of PNB generation. The comparison of the optical scattering images of PNBs (Fig. 32.12b) and NP–Doxil clusters (Fig. 32.12a) revealed their good co-localization in cells. Thus, the mixed NP–Doxil clusters acted as PNB sources, and the mechanical impact of PNBs was co-localized to Doxil liposomes. In this experiment, PNBs provided optical detection of cancer cells with high specificity and sensitivity.
32.3.3 NP Targeting, Plasmonic Nanobubbles Generation, and Detection in HNSCC Tumor In Vivo
In order to translate the achieved result in vivo and to address the impact of tissue on the propagation of light and NPs, we first studied the delivery of NPs, laser radiation, and the generation and detection of PNBs in a xenograft mouse model of HNSCC that used the same cancer cell line, HN31.
32.3.3.1 NP Delivery and Clustering In vivo
The formation of NP clusters in the tumor was imaged and quantified by using TEM microscopy and the PNB generation in the tumor slices. NP–C225 conjugates of solid gold 60 nm spheres were administered locally (intratumoral injection of 1 μl at 9 × 1012 NP/mL (0.8 μg/g)) and systemically (intravenous injection in a tail vein of 200 μL at 4.5 × 1010 NP/mL (0.8 μg/g) at two diameters of NPs, 20 and 60 nm). In 24 h after systemic administering 60 nm gold NP–C225, TEM analysis of the tumor and adjacent normal muscle tissue showed large clusters of gold NPs (an average size 297 ± 135 nm) only in the tumor (Fig. 32.13a), while nonspecific uptake resulted in occasional single NPs in normal tissue (an average size close to that of a single NP, 64 ± 15 nm, Fig. 32.13b). Thus, despite a nonspecific uptake of single gold NPs by normal tissues, the sources of PNBs, larger NP clusters, were observed only in tumors and not in normal tissues [105].
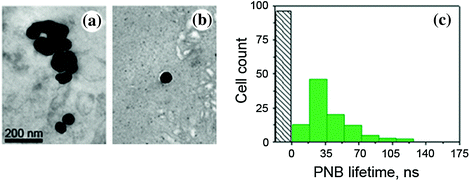

Fig. 32.13
NP–C225, solid gold 60 nm spheres conjugates in HNSCC-bearing mouse: transmission electron microscopy image of a tumor (a) and adjacent muscle tissue (b) in 24 h after systemic injection of NP–C225 to mouse [105]. c Histogram of the PNB lifetime obtained for individual cells in slices of tumor for cancer (green) and normal (gray) cells after being extracted from mice in 24 h after systemic administering of NP–C225. Recreated by kind permission of Ivyspring International Publisher [105]
32.3.3.2 Selectivity of Plasmonic Nanobubble Generation in Tumor
Next, gold NP clusters were analyzed as PNB sources in tumor slices. Each individual cell in a slice was identically exposed to a single laser pulse, and the PNB lifetimes were obtained for cancer (fluorescently identified) and normal cells in a similar way to the above in vitro experiment. At the fluence of 40 mJ/cm2, single pulse excitation resulted in the generation of PNBs only in cancer cells, while normal cells did not generate PNBs under such a low fluence level (Fig. 32.13c). This result indicated that tumor-specific gold NP clusters (not present in normal cells and tissues) provided selective generation of PNBs only in tumor cells. The mechanism of such high selectivity is based on the dependence of the PNB generation threshold upon the size of the NP or its cluster: The threshold fluence decreases with the cluster size [79, 107, 124, 130]. The level of laser pulse fluence applied was below the PNB generation threshold for single NPs but above the PNB generation threshold for large NP clusters. These results correlated well to those obtained previously in vitro and demonstrated selective formation of NP clusters and PNB generation only in tumor, while the nonspecifically taken gold NPs in normal tissue did not form clusters and therefore were unable to generate PNBs [105].
32.3.3.3 Detection of Plasmonic Nanobubbles In vivo in Opaque Tissue
Due to the limited optical transparence of real tissue in animals, we replaced the optical detection of PNBs with the acoustic method (Fig. 32.14a). Briefly, the expansion and collapse of a PNB produce pressure pulses that can be remotely detected in opaque tissue in animals with an ultrasound detector as PNB-specific acoustic time responses (Fig. 32.14b). The amplitude of the acoustic response of a PNB was found to be almost linearly proportional to the optically measured lifetime of the same PNB. Thus, we employed the acoustic signal amplitude as an in vivo metric of PNB [105].
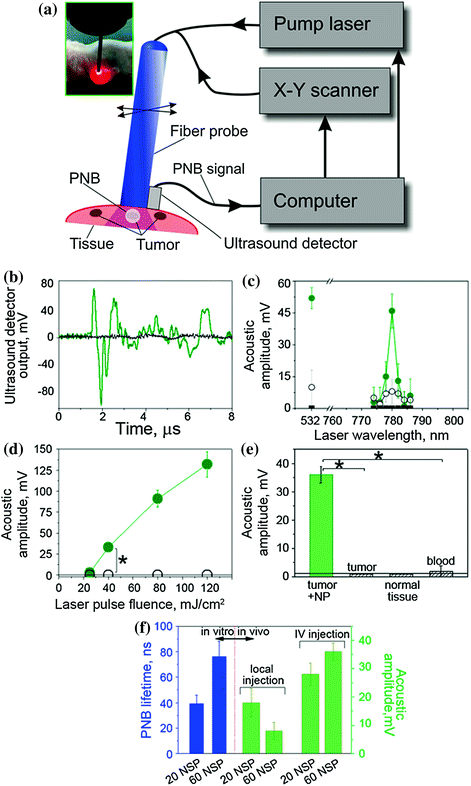

Fig. 32.14
PNBs in HNSCC-bearing mouse. a Functional diagram of the experimental setup. Optical fiber delivers short laser pulse through the surface of the tissue, and PNBs are detected with ultrasound detector, which signals, the scan of the fiber and laser pulse generation are controlled by computer program; b acoustic responses to single laser pulses (70 ps, 780 nm, 40 mJ/cm2) obtained from a tumor (green) and adjacent muscle (black) in mouse systemically treated with 60 nm solid gold sphere conjugates, NP–C225; c spectrum of PNB amplitudes of acoustic responses obtained from a squamous cell carcinoma tumor (solid green) and normal tissue (hollow black circles) of a mouse systemically treated with C225-conjugated 60 nm gold spheres, tumor in intact animal (solid black squares) and (40 mJ/cm2); d dependence of the amplitudes of acoustic responses upon laser pulse fluence (energy per square unit) for the same animals obtained from the tumor (green) and adjacent muscle (black) (*p < 0.001); e acoustic amplitudes obtained during the scans of the tumor, muscle and blood of NP-treated animals and of a tumor of an intact animal (*p < 0.001); f dependence of PNB lifetime (blue, optical response obtained from individual tumor cells in vitro) and the acoustic amplitude of PNB (green, acoustic response obtained from a tumor in animal) as function of NP size (20 nm and 60 nm) and in vivo administration route (local intratumor injection and intravenous injection) under identical laser excitation (single pulse, 70 ps, 780 nm, 40 mJ/cm2). Recreated by kind permission of Ivyspring International Publisher [105]
32.3.3.4 Near-Infrared Pulsed Excitation of Plasmonic Nanobubbles in Tumor
Next, in order to optimize the laser wavelength for NIR excitation, the acoustic amplitudes of PNBs were obtained as the spectra of the excitation laser wavelength in tumor and normal tissue in animals that received intravenous gold NP–C225 conjugates and for an intact tumor in animals that did not receive any gold NPs (Fig. 32.14c). Spectrum of the tumor in the NP-treated animal showed a 3 nm-wide peak at 780 nm. Other NIR wavelengths returned much lower signals comparable to the background level. The normal tissue of the NP-treated animal showed very small acoustic signals that were also comparable to the background level, while intact tumor returned zero signals under identical optical excitation (Fig. 32.14c). We concluded that the detected acoustic signals of the NP-treated tumor were associated with gold NP clusters that were selectively formed in the tumor cells as we found previously (Fig. 32.13a). We therefore employed the NIR wavelength of 780 nm for the experiments in vivo [105].
32.3.3.5 Real-Time Detection of the Tumor with Acoustic Signals of Plasmonic Nanobubbles
The specificity and sensitivity of the tumor detection were studied through the amplitude of acoustic signal of PNBs that was measured as function of tissue type and of the laser excitation fluence. The amplitude of PNB signal increased with the fluence of the excitation laser pulse (Fig. 32.14d). It should be noted that just a single laser pulse was required to detect a tumor-specific acoustic signal. The duration of this diagnostic process was limited by the speed of the ultrasound detector and was below one millisecond.
During the laser scans of a tumor and normal adjacent tissue in the NP-treated animals, the detectable acoustic signal of the PNBs was observed only for the tumor and not for normal muscle tissue and blood (Fig. 32.14d). Tumors in intact animals (that did not receive gold NPs) did not yield any detectable acoustic signal under the identical fluence of laser pulse (Fig. 32.14d). Therefore, the optical fluence of 40 mJ/cm2 was below the bubble generation threshold in blood and other normal tissues for the employed NIR wavelength (780 nm) and duration (70 ps) of laser pulse. The results demonstrated high sensitivity, specificity, safety, and speed of tumor detection in a single NIR laser pulse procedure [105].
32.3.3.6 Optimization of the NP Size and Delivery Route for Plasmonic Nanobubble Generation In Vivo
In addition, we analyzed the influence of the NP diameter and delivery route on PNBs generation in vitro and in vivo. The intravenous systemic delivery of NPs resulted in larger PNBs compared to local intratumor injection of the same NPs under identical optical excitation (Fig. 32.14e). In both cases, the NP–C225 conjugates were administered 24 h prior to the laser treatment. Larger 60 nm NPs generated larger PNBs compared to smaller 20 nm NPs both in vivo and in vitro conditions (Fig. 32.14f). We therefore used systemic intravenous delivery of solid gold 60 nm sphere conjugates, NP–C225, for the next, therapeutic stage.
32.3.4 Plasmonic Nanobubble Diagnostics Versus Nanoparticle-based Diagnostic Methods
The diagnostic part of PNB theranostics in vivo is technically close to photoacoustic methods [141], but PNBs provide higher sensitivity and specificity of tumor detection compared to broadly used gold NPs as photoacoustic probes. Conventional photoacoustics employ acoustic emission from all NPs, instead of cancer cell-specific generation of PNBs only around NP clusters (Fig. 32.10c, d, 32.14b). Under identical optical excitation, PNBs emit stronger pressure pulses compared to those emitted by gold NPs [142]. This explains the high specificity of PNB signals as shown in Fig. 32.14b, c and the high sensitivity that allows to detect even single tumor cells among normal ones [103]. Thus, the PNB method provided high sensitivity, specificity, and speed of tumor detection in a single NIR laser pulse procedure [105] under relatively low optical fluence of 40 mJ/cm2, which is within the medical safety limits for laser radiation [143] and with the safest solid gold spheres.
To summarize, PNBs present excellent diagnostic opportunity via optical scattering detection in vitro and acoustic detection in vivo. In both cases, PNBs provide much higher cancer cell specificity than any nanoparticle-based imaging can provide because the threshold nature of PNBs overcomes the problem of unavoidable nonspecific uptake of imaging probes by nontarget cells.
32.4 Part III Theranostic Applications of Plasmonic Nanobubbles
One of the major limitations of traditional treatments of cancers and other dangerous diseases is the inability to surgically remove a pathology completely without having to remove large areas of normal tissue that serve critical functions. For example, residual tumor cells form the nidus for local recurrences and delayed metastases, which are seen commonly in head and neck, prostate, and other cancers. Not only does this profoundly impact patients’ overall survival, but also their quality of life, due to extensive treatment-related anatomic and functional alterations. The residual cancer cells at the surgical margins often show high resistance to local and systemic adjuvant chemotherapy and radiotherapy [12], thus requiring toxic levels of these treatments. This is of particular concern in HNSCC, the sixth most common cancer diagnosis worldwide, with an estimated 263,000 newly diagnosed cases and over 128,000 deaths occurring yearly [13]. Hence, there is a critical need to develop novel diagnostic and therapeutic modalities that not only complement current therapies to enhance tumor control, but also simultaneously limit the treatment-related morbidity that ensues from treatment intensification, and can be applied as intra- or postoperative treatments.
Recently introduced approaches have employed NPs and external energies to develop putative tumor-targeting capabilities through drug delivery [144–161], hyperthermia [25–28, 100, 162–167], and photodynamic therapy [28, 168, 169]. However, the promises of these new modalities did not provide their fast translation from the laboratory to the clinic for the following reasons: (1) low NP targeting specificity, (2) tumor cell heterogeneity, (3) resistance of phenotypes [12, 144, 145], and (4) NP or energy-delivery toxicities [25–27, 100, 161–166, 170–172]. Therefore, the development of novel technologies that will (a) selectively detect and eliminate drug-resistant residual cancer cells to prevent local and regional recurrence, (b) preserve the functionality of normal tissues and (c) reduce nonspecific toxicity, is highly significant. In addition, an ability to unite both tumor detection and its treatment in one real-time intraoperative theranostic procedure will improve the principal limitations of cancer surgery cited above.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree