Perioperative Management: Practical Principles, Molecular Basis of Risk, and Future Directions
Per-Olof Hasselgren
Jeremy W. Cannon
Josef E. Fischer
Introduction
Preparing patients for surgery has grown increasingly complex as the severity of chronic illness within our patients has worsened even as the options for managing these conditions in the perioperative period have expanded. In addition, over the past decade, the process of surgical care in the operating room (OR) and afterwards has been refined in a number of respects aimed at improving patient safety and quality of care. Throughout this perioperative time, the patient’s physiology is taxed to tolerate the surgical insult and then to heal the operative site. This chapter summarizes our perioperative management approach from the time the decision to operate is made through the operative and postoperative course. The most recent evidence on risk minimization is reviewed in order to provide surgeons a practical approach to assuring as safe a surgical course as possible. The physiologic underpinnings of the response to injury are also discussed along with areas for future investigation aimed at reducing the perioperative patient risk.
Perioperative Evaluation and Management
The perioperative period is defined as the time from preoperative workup through the first 30 days of postoperative care. From the patient’s perspective, a surgical procedure and the perioperative period are often a momentous occasion, which involves significant loss of personal control. As such, the surgeon’s responsibility is to engender trust that the decision to operate is sound and that every measure to ensure the patient’s safety throughout the perioperative course is taken.
A careful preoperative history, review of systems, and physical examination will reveal preexisting medical conditions and risk factors known to worsen surgical outcomes. This process can be facilitated by a screening questionnaire structured to trigger the patient’s memory about significant medical illnesses or previous perioperative experiences (Table 1). Findings during this evaluation then guide the array of laboratory studies and additional tests needed to more specifically assess the patient’s risk of an adverse perioperative event. Once all of this information is gathered, a perioperative management plan can be fashioned by the surgeon often with input from the patient’s primary care physician and possibly other specialty consultants in fields such as cardiology, geriatrics, and anesthesiology. The following sections review many of the issues that arise during the perioperative period and provide a recommended approach based on current evidence.
Screening Tests in Generally Healthy Patients
For patients with no or few comorbidities, a selective preoperative testing approach is advised (Table 2). Laboratory testing options include a complete blood count (CBC), electrolyte and renal function tests, serum glucose, liver function tests (LFT), coagulation studies, urinalysis, and pregnancy test. With the exception of pregnancy test, these studies can be obtained within several months of the planned procedure. Patient’s age has been identified as a minor predictor of morbidity and mortality although this seems to be related more to the associated comorbidities that develop with advancing age.
Consequently, age alone should not be used in determining the types of preoperative tests to obtain with the exception of a baseline hemoglobin (Hb) for those over 65 years undergoing major surgery and any patient in whom significant blood loss is anticipated. The cost of an added white blood cell and platelet count is often minimal; so these are often obtained as part of the baseline Hb. Because renal insufficiency strongly correlates with poor perioperative outcomes, identifying patients with occult renal disease is essential. No consensus exists on the indications for such testing, but it has been suggested that a BUN and Cr should be obtained in patients over 50 years of age scheduled for intermediate or high-risk surgery or when perioperative hypotension is considered likely or when nephrotoxic medications are planned. Routine electrolyte, serum glucose, and LFT are not recommended in healthy patients. Patients with a history of a bleeding disorder or an associated illness, which can result in abnormal coagulation function should have coagulation studies performed. Otherwise, routine testing of the partial thromboplastin time, prothrombin time, and international normalized ratio (INR) is not recommended. Routine urinalysis testing is a matter of ongoing debate. On one hand, patients scheduled to have a surgical prosthesis implanted may be at an increased risk for wound or implant infections from a preexisting urinary tract infection (UTI). However, even with treatment, patients with an asymptomatic preoperative UTI develop more postoperative infections, and the cost–benefit ratio of prosthetic infection prevention with routine urinalysis screening does not clearly favor testing the asymptomatic patient. Patients of childbearing age should have a urine or serum pregnancy test, which many institutions require as a matter of policy.
Consequently, age alone should not be used in determining the types of preoperative tests to obtain with the exception of a baseline hemoglobin (Hb) for those over 65 years undergoing major surgery and any patient in whom significant blood loss is anticipated. The cost of an added white blood cell and platelet count is often minimal; so these are often obtained as part of the baseline Hb. Because renal insufficiency strongly correlates with poor perioperative outcomes, identifying patients with occult renal disease is essential. No consensus exists on the indications for such testing, but it has been suggested that a BUN and Cr should be obtained in patients over 50 years of age scheduled for intermediate or high-risk surgery or when perioperative hypotension is considered likely or when nephrotoxic medications are planned. Routine electrolyte, serum glucose, and LFT are not recommended in healthy patients. Patients with a history of a bleeding disorder or an associated illness, which can result in abnormal coagulation function should have coagulation studies performed. Otherwise, routine testing of the partial thromboplastin time, prothrombin time, and international normalized ratio (INR) is not recommended. Routine urinalysis testing is a matter of ongoing debate. On one hand, patients scheduled to have a surgical prosthesis implanted may be at an increased risk for wound or implant infections from a preexisting urinary tract infection (UTI). However, even with treatment, patients with an asymptomatic preoperative UTI develop more postoperative infections, and the cost–benefit ratio of prosthetic infection prevention with routine urinalysis screening does not clearly favor testing the asymptomatic patient. Patients of childbearing age should have a urine or serum pregnancy test, which many institutions require as a matter of policy.
Additional basic testing options include a 12-lead electrocardiogram (EKG), PA and lateral chest x-ray (CXR), and pulmonary function test (PFT). We reserve these tests almost exclusively for patients with prior history of cardiovascular or cardiopulmonary disease. With regards to PFT, these are only obtained in patients with dyspnea in whom a thorough history and physical examination fails to reveal the source of this complaint. Our approach to obtaining these additional tests is also summarized in Table 2.
Table 1 Preoperative Screening Questionnaire | ||
|---|---|---|
|
Table 2 Preoperative Laboratory Testing Indications | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||
Risk Assessment and Management in Patients with Chronic Medical Illness
The most common preexisting medical condition requiring perioperative risk assessment and management is either known or suspected cardiovascular disease. Other common preexisting conditions that are amenable to risk modification include pulmonary diseases, renal insufficiency, liver failure, diabetes mellitus, immunosuppression, and hematologic conditions. The surgeon’s goal should be to minimize the impact of these conditions on the surgical outcome while using a surgical and anesthetic approach, which avoids any further deterioration of the involved organ system and the patient. In each case, communication between the surgeon and the primary care physician or medical specialists involved in the patient’s care is essential while preparing such patients for surgery.
Cardiovascular
Cardiovascular events are responsible for one-third to one-half of perioperative deaths, and of the patients who present for noncardiac surgery, nearly one-third have a known diagnosis of cardiovascular disease. Consequently, cardiovascular risk stratification and modification are fundamental to the perioperative care of many patients.
Patients with a good functional status have a low risk of perioperative cardiovascular complications. This can be assessed by determining the types of daily routines the patient can perform, which translate into multiples of the amount of oxygen consumed while seated at rest (1 MET). Patients
who are unable to walk up two flights of steps or four blocks (>4 METs) have an increased risk of postoperative cardiovascular events. In addition to functional status, cardiovascular risk scoring systems are useful in quantifying the risk of a major perioperative cardiovascular event. The Revised Cardiac Risk Index (RCRI) is the tool we prefer given its simplicity and validation in multiple clinical studies Table 3.
who are unable to walk up two flights of steps or four blocks (>4 METs) have an increased risk of postoperative cardiovascular events. In addition to functional status, cardiovascular risk scoring systems are useful in quantifying the risk of a major perioperative cardiovascular event. The Revised Cardiac Risk Index (RCRI) is the tool we prefer given its simplicity and validation in multiple clinical studies Table 3.
Table 3 Revised Cardiac Risk Index and Associated Rates of Significant Perioperative Cardiovascular Events | ||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||
In addition to basic laboratory studies, patients with cardiovascular disease should have a baseline EKG. Additional testing options include transthoracic echocardiography, exercise or chemical stress testing with or without supplemental echocardiography or radionuclide myocardial perfusion imaging, and coronary angiography. The 2007 American College of Cardiology/American Heart Association (ACC/AHA) guidelines reflect the most current recommended approach to the use of these additional studies (Fig. 1). Alternative algorithms have been proposed by the American College of Physicians (ACP) and by Fleisher and Eagle. In general, if the patient’s cardiovascular disease warrants immediate intervention (i.e., the cardiovascular symptoms are more pressing than those that prompted surgical consultation), additional studies are warranted. Although these algorithms serve to identify and further evaluate patients deemed to be at either intermediate or high risk for adverse perioperative cardiovascular events, their use has, to date, not been shown to improve patient outcomes.
Based on the coronary artery revascularization prophylaxis (CARP) trial and the DECREASE-V pilot study, prophylactic coronary revascularization by percutaneous coronary intervention (PCI) or coronary artery bypass grafting does not appear to alter postoperative outcomes. Accordingly, the current ACC/AHA guidelines recommend preoperative PCI only in patients with an acute coronary syndrome for whom PCI is independently indicated. Patients who undergo coronary revascularization with a bare metal stent should have surgery delayed for 4 to 6 weeks but no more than 12 weeks when the incidence of stent restenosis begins to rise. Conversely, patients who have a drug-eluting stent (DES) placed should have surgery delayed for a year if possible while the patient is on dual antiplatelet therapy. Aspirin should be continued in the perioperative period if at all possible, and thienopyridine therapy (e.g., clopidogrel) should be resumed as soon as possible after surgery to minimize the risk of stent thrombosis.
Patients with unstable angina or a recent MI bear special consideration. Historic studies suggested that a significant and persistent risk of reinfarction or death existed for up to 6 months after an acute MI. However, with improved perioperative monitoring and management, the rates of such complications after subsequent noncardiac surgery have dropped significantly. A stress test after MI or an episode of unstable angina reliably identifies patients who will benefit from revascularization. Those who have no evidence for at-risk myocardium have a low likelihood of reinfarction with noncardiac surgery and can likely be taken for surgery within 4 to 6 weeks.
Preexisting essential hypertension is a common medical problem among patients facing surgery. Good blood pressure control (<140/90 mm Hg in most patients or <130/80 mm Hg in patients with DM or CKD) is ideal. However, national guidelines do not recommend delaying surgery unless the patient’s blood pressure is over 180/110 mm Hg. Patients with poorly controlled hypertension have an increased risk of perioperative blood pressure lability, arrhythmias, and myocardial ischemia. Such patients should have an EKG and renal function testing and should be evaluated for secondary hypertension prior to elective surgery if this workup has not been previously performed. Then, improved blood pressure control should be pursued for 6 to 8 weeks prior to surgery if the urgency of the indicated procedure permits.
Pulmonary
Patients with a known diagnosis of chronic obstructive pulmonary disease (COPD), asthma, upper respiratory tract infections, pneumonia, or other pulmonary conditions warrant special attention. In addition to an assessment of the patient’s smoking status, pulmonary functional baseline, need for supplemental oxygen, and current pulmonary medications, use of a pulmonary risk index can aid in the quantification of the perioperative risk of respiratory failure (Table 4). Patients with pulmonary risk factors should have a preoperative CXR supplemented by PFTs in those with unexplained dyspnea. There is no role for routine preoperative arterial blood gas testing. The benefits of perioperative smoking cessation are discussed below. If the patient has had a recent deterioration in pulmonary function in the recent past due to a reversible cause, elective surgery should be deferred until the patient returns to their prior baseline. Patients with COPD should be managed with an inhaled anticholinergic (e.g., ipratropium) and as needed inhaled beta-agonists. Patients with asthma should be maintained on their home medication regimen unless their current symptom control is poor. In these cases, a step-up in therapy in the perioperative period is warranted. Prophylactic administration of glucocorticoids to asthmatics is not
required unless they are maintained on systemic or high-dose inhaled steroids. In patients at high risk for perioperative pulmonary complications, consideration should be given to use of spinal or epidural anesthesia over general anesthesia. Long-acting neuromuscular blockade (e.g., pancuronium) should be avoided. If a laparoscopic surgical option is available, this should be used over open surgery if possible. For postoperative risk mitigation, an epidural catheter for analgesia should be planned in these patients, and preoperative incentive spirometry teaching should be conducted so the patient is prepared to participate in early and aggressive postoperative lung expansion.
required unless they are maintained on systemic or high-dose inhaled steroids. In patients at high risk for perioperative pulmonary complications, consideration should be given to use of spinal or epidural anesthesia over general anesthesia. Long-acting neuromuscular blockade (e.g., pancuronium) should be avoided. If a laparoscopic surgical option is available, this should be used over open surgery if possible. For postoperative risk mitigation, an epidural catheter for analgesia should be planned in these patients, and preoperative incentive spirometry teaching should be conducted so the patient is prepared to participate in early and aggressive postoperative lung expansion.
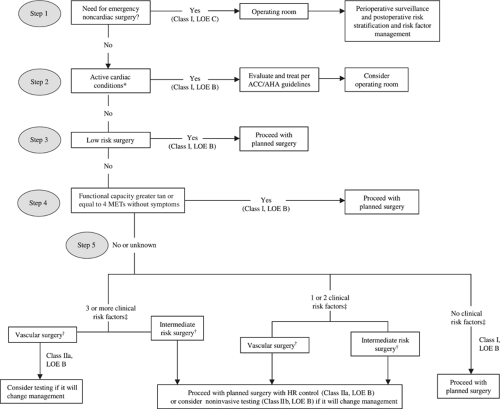 Fig. 1. 2007 ACC/AHA algorithm for the perioperative cardiovascular management of patients aged 50 and above undergoing noncardiac surgery (Adapted from Fleisher et al. 2007.) *Active cardiac conditions include unstable or severe angina, MI between 7 and 30 days prior, decompensated heart failure, significant arrhythmia, severe aortic stenosis, or symptomatic mitral stenosis. †Low risk surgery includes endoscopic procedures, superficial procedures, cataract surgery, breast surgery, and ambulatory surgery; intermediate risk surgery includes intraperitoneal/intrathoracic surgery, carotid endarterectomy, head and neck surgery, orthopedic surgery, and prostate surgery; vascular surgery includes aortic and other major vascular surgery (except carotid endarterectomy) and peripheral vascular surgery. ‡Clinical risk factors are similar to the revised Cardiac Risk Index factors in Table 3 and include history of ischemic heart disease, cerebrovascular disease, compensated or prior heart failure, diabetes mellitus, and renal insufficiency. |
Renal
Chronic renal insufficiency with a serum Cr of ≥2 mg/dL is an independent predictor of postoperative cardiac complications. In addition, the surgical team must take special care to avoid further kidney injury in these patients by maintaining euvolemia, taking appropriate precautions to avoid contrast-induced nephropathy, and by appropriately dosing all medications while minimizing the use of those with potential nephrotoxic effects. In addition, in the perioperative period, intravascular volume status can be more difficult to gauge in this patient population; so we are aggressive with employing all available monitors to assure adequate intravascular volume to include use of a pulmonary artery catheter in some cases.
Patients with end-stage renal disease (ESRD) require coordination of perioperative care between the surgical team, the anesthesia team, and nephrology. Preoperative electrolytes should be obtained in close proximity to the procedure to ensure the serum
potassium is within normal limits and that there are no significant derangements in the other values. If the patient is on hemodialysis or peritoneal dialysis, the timing of pre- and postoperative dialysis should be decided upon in advance. Patients who are on the cusp of requiring renal replacement therapy (RRT) require similar surveillance and coordination in the event that transient RRT is required for postoperative electrolyte management, volume overload, or azotemia.
potassium is within normal limits and that there are no significant derangements in the other values. If the patient is on hemodialysis or peritoneal dialysis, the timing of pre- and postoperative dialysis should be decided upon in advance. Patients who are on the cusp of requiring renal replacement therapy (RRT) require similar surveillance and coordination in the event that transient RRT is required for postoperative electrolyte management, volume overload, or azotemia.
Table 4 Arozullah Respiratory Failure Index and the Associated Risk of Respiratory Failurea | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Liver
Patients with preexisting hepatic failure are at increased risk for complications and death after surgery. The Child–Turcotte and Child–Pugh classification schemes were originally developed to estimate the risk of death after portal-caval shunting and have since been validated as a good estimate for death following a range of other surgeries. More recently, the model for end-stage liver disease (MELD) score has also been used to assess perioperative risk in these patients (Table 5). An online calculator, which uses the patient’s age, ASA class, and MELD score to calculate 7-day, 3-day, 90-day, 1-year, and 5-year predicted mortality is also available at http://www.mayoclinic.org/meld/mayomodel9.html.
In general, patients with mild cirrhosis (Child’s A or MELD < 10) tolerate surgery well while patients with fulminant hepatic failure, severe hepatitis, extrahepatic complications, and advanced cirrhosis (Child’s C or MELD > 15) are likely to have a poor postoperative outcome and should have surgery delayed until their liver function can be optimized or they undergo a liver transplantation if possible. Patients with moderate cirrhosis (Child’s B or MELD 10 to 15) can be considered for surgery after careful preoperative evaluation. Reports of herniorrhaphy outcomes in these patients (both umbilical and inguinal) suggest that these operations can be performed safely. Similarly, cholecystectomy has been reported in patients with cirrhosis for a range of indications, and recent reports indicate that the laparoscopic approach is safe and appears to have improved outcomes when compared with open cholecystectomy. The preoperative preparation of these patients should focus on minimizing ascites, correcting vitamin deficiencies (especially vitamin K), and assessing for and correcting malnutrition.
Table 5 Use of the MELD Score for Preoperative Risk Assessment in Patients with Cirrhosis | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||
Endocrinopathies and Obesity
Diabetes mellitus is a common medical condition present in up to 20% of surgical patients. As shown in Table 3, insulin-requiring diabetes is a marker for increased postoperative cardiac morbidity in the RCRI. Historic glycemic control is a known marker for postoperative infections (pneumonia, wound infection, UTI, and sepsis), and poor perioperative glycemic control has been shown to correlate with surgical complications and death. Thus, careful attention must be paid by the surgical team to cardiac risk modification in diabetic patients and to glucose management throughout the entire perioperative period to assure optimal outcomes.
A reliable measure of historic glycemic control over the previous 3 months is the hemoglobin A1C (Hb A1C). If the Hb A1C is > 7%, postoperative infections are increased while a preoperative glucose level of >200 mg/dL is associated with an increased rate of postoperative deep wound infections. Perioperative management of oral hypoglycemics and insulin is discussed below (see section on “Medication Management”).
Patients with hypothyroidism should continue on their baseline medication regimen throughout the postoperative period. Those who are nil per os (NPO) can have these medications safely held or converted to IV supplementation if a prolonged period of fasting is anticipated. Those with hyperthyroidism undergoing surgery should achieve a euthyroid state before surgical intervention and their antithyroid medications should be continued up until the time of surgery. If urgent surgery is required in a thyrotoxic patient, consultation with an endocrinologist is warranted.
Obesity has been extensively evaluated as a risk factor for poor perioperative outcomes. Recent evidence suggests that, in fact, there is a so-called “obesity paradox” in that such patients have fewer complications than controls. The exceptions to this paradox are wound and thromboembolic complications
including deep venous thrombosis (DVT) and pulmonary embolism (PE).
including deep venous thrombosis (DVT) and pulmonary embolism (PE).
Malnutrition
Preoperative malnutrition has been recognized as an important risk factor for postoperative morbidity and mortality for over 70 years. Quantification of the degree of malnutrition and the correction of severe malnutrition preoperatively remain an important part of surgical management. Assessment of nutritional status begins with a thorough history and physical examination paying careful attention to dietary changes, evidence of malabsorption, and evidence for loss of lean body mass. The Subjective Global Assessment has been used to facilitate this evaluation. Laboratory testing should include albumin, transferrin, and prealbumin to assess the long-term, intermediate-term, and short-term nutritional state of the patient, respectively. If the patient is found to be severely malnourished, surgery should be delayed so that supplemental nutrition can be administered. Enteral supplementation is preferred if the patient can tolerate this route; otherwise, parenteral nutrition (PN) should be initiated. In this population, improvements in nutritional status are assessed at regular intervals until surgery is deemed safe (after 7 to 15 days in some studies). Supplemental nutrition is then continued postoperatively until the patient can meet their caloric needs independently.
Coagulopathy
Patients with inherited coagulopathies and those who are maintained on therapeutic anticoagulation present special challenges with regards to achieving and maintaining postoperative hemostasis. Perioperative management of anticoagulant and antiplatelet medications is discussed below (see section on “Medication Management”). The most common intrinsic coagulopathies in surgical patients are von Willebrand’s disease and the hemophilias. Patients with chronic renal insufficiency also have some baseline degree of platelet dysfunction. The surgical review of systems should specifically focus on a predilection for prolonged epistaxis, easy bruising, and any bleeding complications during previous surgeries. If this evaluation is negative for a bleeding history and the physical examination does not reveal any petechiae or stigmata of chronic renal or liver disease, routine testing of coagulation studies is not indicated. If these studies are obtained and are abnormal, a mixing study is required to determine whether the abnormality is the result of a factor deficiency or an inhibitor (e.g., lupus anticoagulant). If the patient’s personal family history is strongly suggestive of an undiagnosed coagulopathy, consideration should be given to testing for von Willebrand’s disease using the triad of plasma von Willebrand’s factor (VWF) antigen, plasma VWF activity, and factor VIII activity. Patients who carry a diagnosis of von Willebrand’s disease should be pretreated in consultation with a hematologist with either desmopressin (DDAVP) for minor surgery if the patient has previously responded or with VWF concentrate for major surgery. Patients with mild hemophilia A or B can similarly be pretreated with DDAVP while those with severe hemophilia can be treated with specific factor concentrates (Factor VIII or IX) or activated Factor VII in the presence of inhibitors. Patients with thrombocytopenia (e.g., those with inherited thrombocytopenic purpura) should have a preoperative platelet transfusion targeting a minimum of 50,000/μL.
Malignancy and Immunocompromise
Patients with malignancy and those on immunosuppressive medications or with an inherited or acquired immunocompromised state frequently undergo surgery. The preoperative evaluation should proceed as described above guided by the patient’s other medical conditions and nutritional status. For patients on chemotherapy, the timing of the last dose of chemotherapy, the projected cell count nadirs, and planned future therapy should be discussed with the patients and their oncologist. For patients with HIV, a history of an AIDS-defining illness and their current medication regimen should be elicited. Laboratory testing should include a CBC with differential, chemistries, renal function, and liver function studies. If malnutrition is suspected by history and physical examination, nutrition labs should be obtained. Patients with HIV should have a CD4 and a viral load obtained as the former is a surrogate for immunocompetence while the latter has been specifically correlated with increased perioperative complications at a level of 30,000 copies/mL or greater. Patients with neutropenia should have surgery delayed if at all possible. For those with neutropenia in the postoperative state, development of fever should prompt treatment with broad-spectrum antibiotics and, in some cases, an antifungal agent as well. The role of colony stimulating factors in neutropenic patients is limited to those with additional indicators that prolonged neutropenia will be poorly tolerated such as poor functional status, poor nutrition, an open wound, or active infection. It has been shown that although these stimulating factors reverse the neutropenia, they do not reliably reduce hospital length of stay or culture-positive infections.
Rheumatologic
Patients with rheumatologic diseases have a high incidence of associated cardiovascular disease as well as unique pathology, which increases the risk of perioperative complications. Patients with rheumatologic conditions are often maintained on immune-modulating medications such as glucocorticoids, methotrexate, and so-called biologic agents that interfere with the action of TNF and IL-1. The perioperative management of these medications is discussed in section on “Medication Management.” In patients with rheumatoid arthritis, lateral cervical spine films with flexion and extension should be obtained within a year of surgery to assess for atlanto-axial subluxation. Patients with ankylosing spondylitis with severe kyphotic deformities may be difficult to intubate, and thoracic cavity restriction may require postoperative ventilator support. Thus, preoperative anesthesia and critical care consultations should be considered. Likewise, patients with scleroderma can present special anesthetic challenges, including a small oral aperture, difficult intravenous access, a propensity for vasospasm, prolonged response to local anesthetics, and a significant risk of aspiration due to esophageal dysmotility. In addition, preoperative detection of pulmonary or myocardial involvement is essential; so consideration should be given to obtaining PFTs, an arterial blood gas, and echocardiography in addition to a CXR and EKG. Patients with psoriatic arthritis should be advised of the risk for a psoriatic flare at both the surgical and the remote sites. In addition, these patients may be at increased risk for postoperative infection. Patients with systemic lupus erythematosus (SLE) are at increased risk for postoperative wound infection, renal insufficiency, and thrombotic complications, including pulmonary embolism. SLE patients with active disease and imminent vital organ failure can be treated with intravenous immunoglobulin in the perioperative period.
Preoperative Behavioral Modification
In addition to risk modification interventions discussed above, a number of preoperative behavioral modification strategies have been investigated in an attempt to improve surgical outcomes. The most widely published interventions include smoking cessation, preoperative weight loss, and various preoperative exercise regimens (so-called prehabilitation).
Historic evidence suggested that smoking cessation within 8 weeks of surgery actually results in increased pulmonary complications, presumably from bronchorrhea. On the other hand, several smaller studies indicate that some complications such as wound infections and seromas are reduced if smoking cessation occurs as early as 4 weeks prior to surgery although these studies have been inadequately powered to detect differences in pulmonary complications.
Historic evidence suggested that smoking cessation within 8 weeks of surgery actually results in increased pulmonary complications, presumably from bronchorrhea. On the other hand, several smaller studies indicate that some complications such as wound infections and seromas are reduced if smoking cessation occurs as early as 4 weeks prior to surgery although these studies have been inadequately powered to detect differences in pulmonary complications.
Although obesity is associated with an overall increase in cardiovascular disease as well as perioperative wound and thromboembolic complications, the effect of preoperative weight loss on these risks has not been well studied. In patients preparing for bariatric surgery, preoperative weight loss has been correlated with more durable postoperative weight loss. However, improved perioperative surgical outcomes in terms of fewer surgical complications, cardiovascular events, or pulmonary complications have yet to be documented for either bariatric surgery or other surgical procedures in the obese population.
Because functional status correlates strongly with cardiovascular and pulmonary complication rates, several groups have investigated the benefits of specifically targeting improved functionality in the preoperative period. Recent evidence suggests that a simple regimen of daily walking and deep breathing exercises improves exercise capacity in patients awaiting abdominal surgery, an effect that is preserved postoperatively. Similarly, preoperative inspiratory muscle training appears to result in fewer pulmonary complications and a shorter hospital stay.
Medication Management
Adult patients facing surgery are often taking a number of medications for management of their chronic medical conditions. Prior to surgery, a complete list of all medications and herbal supplements must be obtained from the patient and reconciled with the most recent list of medications in their medical record. The most common outpatient medications and their recommended perioperative management are summarized in Table 6. In general, essential medications are continued through surgery with any doses due at the time of surgery taken with a sip of water. Essential medications and those with a significant risk of rebound effects (e.g., beta blockers and clonidine) are continued in an enteral, parenteral, transdermal, or inhaled form during the early postoperative period. As soon as feasible, the patient’s outpatient medication regimen should be resumed or revised in consultation with their primary care physician or medical specialist.
Because of the risk of hemorrhage with surgical intervention, the management of outpatient therapeutic anticoagulation in the perioperative period bears special mention. Patients are maintained on anticoagulation for a range of indications from the management of thromboembolic events to anticoagulation for prosthetic heart valves. The indication for anticoagulation dictates the need for therapeutic “bridge” therapy with a short acting agent while both the surgical procedure and the indication for anticoagulation are used to develop a postoperative anticoagulation plan. For patients with mechanical heart valves, the 2006 ACC/AHA guidelines are the most straightforward to apply. In a patient with a bi-leaflet mechanical aortic valve and no additional risk factors for hypercoagulability (e.g., atrial fibrillation or previous thromboembolism among others), warfarin can be held 48 to 72 hours prior to surgery with an INR checked on the day of surgery targeting less than 1.5. All other patients (e.g., those with mechanical mitral valves and those with additional risk factors for thromboembolism or hypercoagulability) should be managed with bridge therapy. These guidelines recommend the use of therapeutic heparin during this time although therapeutic low-molecular-weight heparin is included in other guidelines. Postoperatively, in patients who do not require bridge therapy, warfarin is resumed 24 hours after surgery. Those on bridge therapy have their anticoagulation resumed as soon as the bleeding risk permits, usually at 24 hours after surgery.
In all other conditions for which patients are on therapeutic anticoagulation, the perioperative management of this regimen requires an estimate of the bleeding risk from surgery and the risk of a perioperative thromboembolic complication. There are no guidelines to inform practice, but some general practice recommendations can be made from the current literature on this topic. Patients with a recent episode of venous or arterial thromboembolism should have surgery delayed for at least 1 month if at all possible. Minor surgery (e.g., outpatient herniorrhaphy or cataract surgery) can be done safely in patients on warfarin so long as the INR is at the low end of the therapeutic range. Those undergoing major surgery should have warfarin therapy withheld approximately 5 days prior to surgery with an INR checked on the day of surgery. Those on the orally available direct thrombin inhibitor dabigatran (Pradaxa) should have this withheld 1 to 2 days before surgery if renal function is normal or 3 to 4 days with a Cr clearance <50 mL/min. Bridging anticoagulation with either intravenous heparin or therapeutic low-molecular-weight heparin should be used in patients at high or intermediate risk for a thromboembolic event. For many indications including atrial fibrillation, individual patient risk stratification should be conducted to determine the need for bridge therapy. Bridge therapy with heparin should be held 4 to 5 hours prior to surgery while low-molecular-weight heparin should be held 24 hours prior. As mentioned above, postoperative resumption of bridge therapy or oral anticoagulants depends on the risk of postoperative bleeding but generally can be considered after 24 hours.
Patients on antiplatelet therapy also commonly face noncardiac surgery. The availability of thienopyridines (e.g., clopidogrel) and the significant increase in DES implantation have also made the scenario of dual antiplatelet therapy in the surgical patient increasingly common. Patients on antiplatelet therapy should have a careful history taken to determine their indication for treatment—primary prevention versus prophylaxis against stent thrombosis. As described above, in patients with coronary stents, the timing of surgery should take into consideration the type of stent and the age of the stent. Similarly, the risk of hemorrhage from the surgery should be considered. Cataract surgery is often performed in the patient on aspirin as is coronary artery bypass grafting. If aspirin is held, this should be 7 to 10 days prior to surgery and then resumed when surgical hemostasis is assured, typically within or at 24 hours of the operation. The use of clopidogrel or another thienopyridine either alone or in combination with aspirin should be elicited as well. Some limited data in the vascular surgery literature suggests that with careful attention to hemostasis, even major vascular operations can be done on dual antiplatelet therapy if necessary with no significant increase in perioperative bleeding complications.
Patients with rheumatologic diseases as well as many other conditions ranging from reactive airway disease to inflammatory bowel disease are maintained on systemic glucocorticoids. Although historically, these patients were given additional steroid doses in the perioperative period—so-called stress dose steroids—recent evidence has called this routine practice into question. Patients who have been on prednisone doses of 20 mg/day (or the equivalent of another agent) for 3 weeks or more or who have a Chushingoid appearance should be presumed to have hypothalamic–pituitary–adrenal (HPA) suppression, which will require supplemental steroid dosing. For patients on lower doses,
our usual approach is to resume their home dose or the equivalent in the postoperative period and observe for hemodynamic instability or significant malaise as a trigger for supplemental steroid dosing. An alternative strategy is to perform specific testing for HPA suppression using either high- or low-dose ACTH stimulation testing. Those who respond normally will likely not need supplemental steroid dosing postoperatively.
our usual approach is to resume their home dose or the equivalent in the postoperative period and observe for hemodynamic instability or significant malaise as a trigger for supplemental steroid dosing. An alternative strategy is to perform specific testing for HPA suppression using either high- or low-dose ACTH stimulation testing. Those who respond normally will likely not need supplemental steroid dosing postoperatively.
Table 6 Perioperative Management of Outpatient Medications and Herbal Supplements | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
There has been some recent interest in perioperative risk reduction through initiating new medications around the time of surgery. Examples include starting a beta blocker or statin in patients with cardiovascular risk factors in the immediate preoperative period. Enthusiasm for initiation of beta blockers around the time of surgery has been recently tempered by the results of the POISE trial and a subsequent meta-analysis, which indicated that although this practice reduces the incidence of perioperative MI, the incidence of perioperative stroke is increased and all-cause mortality is either increased or, at best, unchanged (although the dose of beta blockade in the POISE trial was moderately aggressive at 200 mg of extended release metroprolol daily). In light of these results, the ACC/AHA released a focused update to their perioperative guidelines in 2009, which recommend against the initiation of high-dose beta blockade without dose titration in beta-blocker-naïve patients undergoing surgery. Similarly, the indications for perioperative statin initiation in those without classic indications for lipid lowering therapy have been clarified by recent studies. These indicate that vascular surgery patients likely benefit from this intervention with fewer episodes of myocardial ischemia and a lower perioperative cardiac death rate.
Preoperative Specialty Consultation
Patients with complex or refractory medical problems may benefit from preoperative consultation by a general medical internist, geriatrician, or other medical specialist. Studies evaluating patient outcomes and utilization of medical resources with this practice have generated mixed results. However, provided the consultant’s role is clearly delineated in the initial request and the consultant makes evidence-based recommendations, surgeon satisfaction is high and the surgical patient’s care is likely to improve. It should be evident from the discussion above that asking a medical consultant to “clear” a patient for surgery is a nonsequitur. Instead, the surgeon should ask specific questions relating to risk stratification and perioperative management of particular disease processes or medications. With this approach, the consultant’s input is more likely to be useful to the surgeon and the surgical team in making decisions on surgical timing and perioperative management strategies. Postoperatively, continued involvement of these consultants or a medical hospitalist can, in some cases, improve the care of the patient and should be considered by the surgeon.
Risk Reduction in the Immediate Perioperative Period
Multiple interventions and quality improvement measures have been advanced in recent years to reduce the risk of adverse events in the OR ranging from wound infections to wrong-site surgery. This section reviews some of the processes that now routinely occur as a result of these initiatives and the management decisions that are most often made from the time the patient enters the preoperative area until the surgical intervention commences. In our view, surgeons should continue to take a leading role in directing the surgical team during this time in the patient’s care as it sets the stage for the entire postoperative course.
Surgical Checklist and Preoperative Timeout
Increased awareness of wrong-site surgery led to a summit to address this problem in 2003 and 2007. Attended by leaders from multiple surgical organizations and the Joint Commission on Accreditation of Healthcare Organizations (JCAHO), the 2003 summit resulted in the introduction of the Universal Protocol for Preventing Wrong Site, Wrong Procedure, Wrong Person Surgery in 2004. This protocol consists of three components: preoperative verification of patient information, surgical site marking to prevent ambiguity with unilateral procedures, and conducting a presurgical timeout to review the planned procedure and resolve any concerns. Many hospitals promote use of the final presurgical time out as an opportunity for the entire OR team to review the surgical plan and to confirm that all necessary medications have been given. Despite (or perhaps because of) this increased emphasis on patient safety, the number of reported wrong-site procedures has steadily increased since the introduction of the Universal Protocol. The anticipation by proponents of this culture of safety is that as the number of reports rise and the freedom to raise safety concerns in the OR disseminates, the frequency of major errors will decrease significantly.
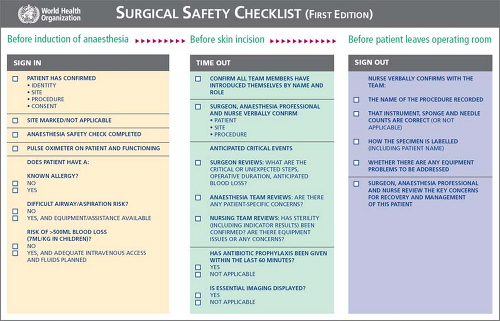
The Safe Surgery Saves Lives Study Group has recently evaluated an intraoperative checklist developed from the World Health Organization (WHO) guidelines for improving perioperative surgical safety. The 19-item checklist used by this group (Fig. 2)
emphasizes communication between the patient and all the various surgical team members in the immediate preoperative period and then focuses the team on critical decisions and communication points in the OR and at the conclusion of the procedure. Use of this checklist in eight different hospitals in eight countries resulted in fewer postoperative complications, including death. This report and others highlight the importance of communication between the surgeon, anesthesiologist, OR nurse, surgical technician, and the patient to ensure that the planned surgical procedure is conducted safely and unplanned intraoperative contingencies are readily identified and well managed.
emphasizes communication between the patient and all the various surgical team members in the immediate preoperative period and then focuses the team on critical decisions and communication points in the OR and at the conclusion of the procedure. Use of this checklist in eight different hospitals in eight countries resulted in fewer postoperative complications, including death. This report and others highlight the importance of communication between the surgeon, anesthesiologist, OR nurse, surgical technician, and the patient to ensure that the planned surgical procedure is conducted safely and unplanned intraoperative contingencies are readily identified and well managed.
Prevention of Deep Venous Thrombosis
Venous thromboembolic (VTE) complications are an all too common perioperative complication. Over half of surgical patients are at moderate risk or greater for VTE events in the postoperative period, and PE is still the most common preventable cause of hospital death. In a recent study of surgical inpatients in 358 hospitals in 32 different countries, although 64.4% of patients were found to be at-risk for VTE, only 58.5% of patients received appropriate VTE prophylaxis. The American College of Chest Physicians Evidence-based Clinical Practice Guidelines (8th edition) on the Prevention of Venous Thromboembolism have been used to establish local policies in many hospitals and serve as a benchmark for best practice in this area (Table 7). These guidelines stratify patients into low-, moderate-, and high-risk categories based principally on the nature of the surgical procedure—an approach that relies on overall group risk assessment as opposed to individual risk assessment. Although some investigators have attempted to develop individual patient risk assessment models, to date, none of these models have been validated. In fact, it appears with few exceptions that the principal predictor of risk is the primary reason for the patient’s hospitalization.
Table 7 Summary of the Level of Thromboembolism Risk and Recommended VTE Prophylaxis in Hospitalized Patients from the American College of Chest Physicians Evidence-Based Clinical Practice Guideline on the Prevention of Venous Thromboembolism (8th Edition) | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||
Mechanical thomboprophylaxis includes intermittent pneumatic compression devices, venous foot pumps, or graduated compression stockings. These modalities have been shown to reduce the risk of DVT in numerous patient populations. However, they have not been demonstrated to reduce the rate of PE or death, and compliance with their use is often poor in the postoperative period. Nonetheless, they are at low risk preventive measure that can be initiated prior to induction of anesthesia in most surgical patients. Those patients at moderate to high risk of VTE should be considered for chemoprophylaxis starting preoperatively within 2 hours of surgery. If the patient is to receive an epidural catheter, local policies should be developed weighing the risk of VTE versus an epidural hematoma as detailed in the American Society of Regional Anesthesia and Pain Medicine (ASRA) evidence-based guideline on regional anesthesia in the patient receiving antithrombotic or thrombolytic therapy. In patients at moderate and greater risk of VTE perioperatively, chemoprophylaxis should be started or resumed as soon as bleeding risk is acceptably low, often shortly postoperatively. This therapy should be continued through the inpatient course in most cases. High-risk general surgical patients, who have undergone major oncologic surgery should also be considered for extended chemoprophylaxis for up to 28 days, even as an outpatient.
Postoperative Ileus Prevention
Patients undergoing bowel resection are at risk for development of a postoperative ileus. The recent availability of peripherally acting mu-opioid receptor antagonists (PAM-OR) such as alvimopan (Entereg) allows surgeons to preemptively treat patients at risk for postoperative ileus. The first dose is given orally from 30 minutes to 5 hours prior to surgery and is then continued during the inpatient course. Use of this preventive strategy appears to result in earlier return of bowel function by multiple measures and shorter inpatient hospital length of stay. Use of chronic narcotics is a contraindication to use of this medication, and hospitals that wish to offer this medication must participate in the ENTEREG Access Support and Education (E.A.S.E.™) Program.
Wound Infection
The Surgical Care Improvement Project (SCIP) sought to reduce postoperative complications (primarily SSI and VTE) by 25% from 2006 to 2010. This broad-based initiative supported by numerous national organizations used a number of quality measures to achieve this goal by promoting evidence-based practice. SCIP quality measures specific to perioperative infectious complications include timely administration of prophylactic antibiotics (within 1 hour prior to the incision), use of appropriate antibiotics for SSI prophylaxis, timely discontinuation of prophylactic antibiotics (within 24 hours of the end of the operation for noncardiac surgery or 48 hours for cardiac surgery), and appropriate hair removal (no hair removal or use of clippers). Additional SCIP measures relating to infectious complications include use of intraoperative temperature management, early removal of indwelling urinary catheters, and glycemic control on the morning after surgery in cardiac patients. Evidence of the impact of compliance with these measures is just now emerging—initial reports suggest that while global compliance may result in a small reduction in SSI, compliance on individual measures results in little or no improvement in SSI rates.
Recommended perioperative antimicrobial prophylaxis regimens are periodically
updated in several publications including Treatment Guidelines from the Medical Letter (2009). The role of several additional SSI reduction strategies have been clarified in the recent literature, including topical antisepsis, the role of mechanical bowel preparation (MBP) for colorectal surgery, fascial closure techniques, and perioperative oxygen supplementation. A recent study comparing skin antisepsis with a chlorhexidine–alcohol preparation versus betadine in a range of clean contaminated surgical cases demonstrated significantly reduced SSI rates with the use of chlorhexidine–alcohol. However, there was no description of whether the betadine was allowed to dry, and a betadine–alcohol preparation was not included in the study. In addition, use of chlorhexidine-containing solutions is not recommended for preparation of exposed mucosal surfaces and alcohol-based preparations are generally considered too risky for use in emergency operations where enough time may not be afforded for the alcohol to dry prior to the use of electrocautery. Nonetheless, chlorhexidine–alcohol preparation appears to be a good choice for a range of surgical procedures.
updated in several publications including Treatment Guidelines from the Medical Letter (2009). The role of several additional SSI reduction strategies have been clarified in the recent literature, including topical antisepsis, the role of mechanical bowel preparation (MBP) for colorectal surgery, fascial closure techniques, and perioperative oxygen supplementation. A recent study comparing skin antisepsis with a chlorhexidine–alcohol preparation versus betadine in a range of clean contaminated surgical cases demonstrated significantly reduced SSI rates with the use of chlorhexidine–alcohol. However, there was no description of whether the betadine was allowed to dry, and a betadine–alcohol preparation was not included in the study. In addition, use of chlorhexidine-containing solutions is not recommended for preparation of exposed mucosal surfaces and alcohol-based preparations are generally considered too risky for use in emergency operations where enough time may not be afforded for the alcohol to dry prior to the use of electrocautery. Nonetheless, chlorhexidine–alcohol preparation appears to be a good choice for a range of surgical procedures.
MBP has been a mainstay of perioperative surgical practice aimed at reducing anastamotic and wound complications for decades. However, systematic study by multiple investigators and subsequent meta-analyses have not convincingly demonstrated any benefit to this practice with respect to either of these complications. In fact, there may be a slight reduction in anastamotic leakage when preoperative MBP is not performed, although, as of 2003, MBP was still widely practiced by colorectal surgeons. Current guidelines leave the use of MBP to the discretion of the surgeon for open low anterior resection and all laparoscopic colonic procedures where the site of the tumor may not be immediately obvious and where intraoperative colonoscopy may be required. For all other colonic resections, preoperative MBP can be safely eliminated.
Fascial closure techniques for abdominal operations have also been evaluated over many years searching for the optimal method, which reestablishes abdominal domain while minimizing the risk of postoperative wound complications ranging from superficial wound infections to complete dehiscence with evisceration. Most studies evaluating fascial closure methods use incisional hernia as the primary endpoint, and until recently, SSI was thought to not be affected by the technique of fascial closure. However, a recent study suggest that when using a running absorbable suture technique, relatively small (5 to 8 mm), closely spaced fascial bites resulting in a suture to wound length ratio of ≥4:1 may reduce the incidence of SSI. Likewise, there is growing interest in using antibiotic-coated suture material that may also reduce the rate of wound infections.
Oxygen supplementation in the immediate postoperative period has also been evaluated by several randomized controlled trials, including the recently published Perioperative Oxygen Fraction (PROXI) study. Although the original US-based study demonstrated increased infections in the oxygen-treated group and the PROXI study showed no benefit to 80% O2 supplementation for 2 hours postoperatively, three other studies have shown a benefit to various types of O2 supplementation. Consequently, pooled analysis of these results still falls in favor of perioperative hyperoxia although the likely benefit is relatively small.
Intraoperative Resuscitation
Inappropriate management of intravenous fluid volumes during surgery can result in a number of postoperative complications ranging from ranging from pulmonary and renal dysfunction to anastamotic failure and sepsis. Achieving the appropriate balance of adequate intravascular volume and oxygen delivery during the surgical procedure has proven difficult, however. This difficulty arises for many reasons, mostly because direct measures of intravascular volume and end-organ perfusion are not readily available while estimates of intraoperative bleeding and insensible losses are notoriously inaccurate. Furthermore, a standard nomenclature for the various fluid administration strategies is lacking, leading to imprecise and variable definitions from study to study. Recognizing these limitations, it has become clear that either too much or too little intravenous fluid administration of any type is harmful. In major abdominal operations where additional monitoring is justified, a “goal-directed” approach based on surrogates for intravascular volume measurement (e.g., esophageal Doppler measurement of changes in peak aortic stroke velocity or arterial waveform variability) while monitoring indicators of oxygen consumption such as ScvO2 is appealing. Combining this approach with a relatively restrictive (but not too restrictive) background of intravenous fluid administration (e.g., 8 to 12 mL/kg/h) appears to balance the various risks of respiratory failure, renal insufficiency, wound infections, congestive heart failure, and postoperative arrhythmia.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree