Diagnostic Considerations in Biliary and Liver Disease
Yuman Fong
Kimberly Moore Dalal
The workup and treatment of biliary and hepatic conditions have undergone major improvements in the last decade. Improvements in ultrasound (US), computed tomography (CT), and magnetic resonance imaging (MRI) scans have allowed for the accurate identification of exceedingly small lesions and precise delineation of the relationship of benign and malignant lesions to major vasculature. A much greater ability to diagnose disease and establish treatment plans, including surgical therapies, now exists. Improvements in invasive procedures such as endoscopic retrograde cholangiopancreatography (ERCP), percutaneous transhepatic cholangiography (PTC), and laparoscopic surgery allow for these minimally invasive therapies to evolve from diagnostic and palliative procedures to a role of definitive therapy. Even difficult tumors, such as cholangiocarcinomas, can now be treated with potential for long-term survival.
Given the large number of imaging modalities available to aid the clinician, the choice of tests and the sequence of testing have become increasingly important. Defining algorithms is essential to streamline workups, arrive expeditiously at a definitive therapeutic plan, and minimize cost. Since patients are being scanned for various reasons, there is an increasing number of incidentally found lesions. Having a reasoned approach at workup of these incidentalomas will be important in diagnosing early cancers for effective treatment, and dismissing benign lesions to avoid morbid invasive procedures and alleviate patient anxiety.
In this chapter, we will introduce clinical algorithms for workup and treatment of common symptoms and signs of hepatic and biliary disease as well as the incidentally discovered asymptomatic gallstone, gallbladder mass, and liver mass. In each section, we will discuss the most important clinical entities responsible, followed by algorithms for workup of particular presentations that combine efficiency with cost considerations. To conclude this chapter, we will also discuss the pregnant patient as well as examine evaluation of liver function as an indicator of surgical risk.
Many patients with biliary and liver disease present for medical attention with nonspecific symptoms such as weight loss, malaise, or indigestion. These patients will then be identified to have biliary or liver pathologic findings by blood tests or diagnostic imaging modalities.
Right Upper Quadrant Pain
The symptom of right upper quadrant (RUQ) pain is not specific for biliary and liver disease, although hepatobiliary disease must be entertained in any patient presenting with pain in this quadrant. Patients presenting with RUQ pain and hemodynamic instability must be considered to have a ruptured liver malignancy, ruptured adenoma, or a ruptured liver from trauma. In parts of the world where hepatitis and cirrhosis are endemic, hepatocellular carcinoma (HCC) can present as rupture and abdominal catastrophe in as many as 10% of patients. Rapid history and physical examination should be directed at eliciting symptoms, signs, or history of hepatitis and cirrhosis. Along with urgent resuscitation, imaging by US or CT should be obtained to look for hemoperitoneum and liver mass. An emergent angiogram can be diagnostic and allow for arterial embolization as the definitive life-saving procedure.
RUQ pain and sepsis can be associated with stone disease complicated by infection. However, the differential diagnosis includes peptic ulcer disease, biliary dyskinesia, colonic diverticulitis, pancreatitis, hepatitis, appendicitis, gallbladder malignancy, or hepatic malignancy. We will discuss the workup and treatment of gallstones, segregating the algorithms according to the presence of jaundice, sepsis, and other symptoms.
Jaundice
Jaundice is the only symptom that is pathognomonic for biliary or hepatic disease. The color of the skin is accompanied usually by acholic stool and dark urine with pruritus occurring in one-third of patients. The causes of jaundice can be prehepatic, hepatic, or posthepatic. Most prehepatic and hepatic causes of jaundice such as hemolysis or acute hepatitis are not surgical diseases and lack signs of biliary dilatation on imaging. The causes of jaundice that are surgical diseases are generally those with biliary obstruction and ductal dilatation. We will discuss in detail the workup of obstructive jaundice.
The majority of patients who present to a surgeon with a hepatobiliary problem have already undergone a partial workup including imaging for their chief complaint. Following are the common clinical scenarios and their workups.
Gallstones in Patients Without Jaundice
These entities may include cholelithiasis, acute cholecystitis, acalculous cholecystitis, and chronic cholecystitis.
Cholelithiasis
Cholelithiasis affects more than 20 million Americans. Common risk factors include the five Fs: fat, female, forty, fecund, and family history. Most patients with gallstones remain asymptomatic, but 20% percent of patients will develop symptoms after 15 years of follow-up. Gallstones may cause biliary colic or acute cholecystitis, obstructive jaundice due to choledocholithiasis, pancreatitis, cholangitis, and gallbladder carcinoma.
Biliary colic is characterized as moderately severe, steady, crescendo-type crampy RUQ pain that can radiate to the right shoulder, subscapular region, or the back, and often follows a meal high in fat content when the gallbladder contracts against a stone, obstructing the cystic duct. The pain can persist from several minutes to several hours, and may be accompanied by nausea, vomiting, and anorexia; if it lasts >4 hours, one should consider the development of acute cholecystitis.
Acute Cholecystitis
Acute cholecystitis most commonly occurs in women ages 30 to 60 years. A diagnosis of acute cholecystitis is based on the history, physical examination, and laboratory tests. Patients complain of fever and RUQ pain, with radiation to the right shoulder or back, associated with nausea, vomiting, and anorexia, often after eating a fatty meal. The pain usually lasts for more than 4 to 6 hours. The most accurate physical finding is a positive Murphy’s sign: inspiratory arrest during RUQ palpation; yet, the sensitivity of this sign may be diminished in the elderly.
In contrast to biliary colic, acute cholecystitis is not fully explained by cystic duct obstruction. Acute cholecystitis may result from irritation of the gallbladder mucosa from lecithin, a normal component of bile, which propagates inflammation through release of additional inflammatory mediators. The majority of patients have infected bile, usually involving Escherichia coli, Enterococcus, Klebsiella, and Enterobacter.
The most common complication of acute cholecystitis is the development of gallbladder gangrene (20%) and perforation (2%). Emphysematous cholecystitis is identified in 1% of cases of acute cholecystitis and is associated with significant morbidity. It more commonly occurs in men in their fifth to seventh decade, and half of these patients are diabetic. This complication is caused by secondary infection of the gallbladder wall with gas-forming Clostridium, E. coli, Pseudomonas, or Klebsiella species. Crepitus in the abdominal wall adjacent to the gallbladder is an important clue to the diagnosis but is rarely detected. There may be mild to moderate unconjugated hyperbilirubinemia. Emphysematous cholecystitis heralds the development of a gangrenous gallbladder and subsequent perforation.
Gangrenous cholecystitis is the most common complication of acute cholecystitis, particularly in older patients, diabetic patients, or those who delay seeking treatment. Perforation occurs after a gangrenous, ischemic region of the gallbladder wall becomes necrotic. The perforation may be walled off by omentum or adjacent organs and be localized as a pericholecystic abscess; at times the perforation occurs into the peritoneum, leading to generalized peritonitis and a high mortality. The inflamed gallbladder may become adherent to the small or large bowel, forming a cholecystoenteric fistula, and leading to gallstone ileus.
Along with fever, RUQ pain, and Murphy’s sign, patients with acute cholecystitis may have a slightly elevated alkaline phosphatase and white blood cell count with a left shift. Elevations in the serum total bilirubin and alkaline phosphatase are not usually seen, and when present should raise suspicion for acute cholangitis, choledocholithiasis, or Mirizzi syndrome, a condition in which a gallstone impacted in the distal cystic duct causes extrinsic compression of the common bile duct (CBD). There may also be an elevation of transaminases.
Acalculous Cholecystitis
If a patient has RUQ pain and fever, but US does not reveal stones or sludge, one must suspect acalculous cholecystitis. Acalculous cholecystitis accounts for 10% of cholecystitis in adults but 30% in children. In the critically ill who may be intubated and sedated, fever, vague abdominal discomfort, and leukocytosis warrant high suspicion for this entity. Risk factors in adults include acquired immunodeficiency syndrome (AIDS), major
trauma or burns, mechanical ventilation, total parenteral nutrition (TPN), recent episode of sepsis, multiple transfusions, childbirth, or hemodynamic instability. Significant vascular disease has been observed in 72% of outpatients diagnosed with acalculous cholecystitis. In children, risk factors include hemolytic disorders such as sickle cell anemia, congenital cystic duct stenosis, acute streptococcal infection, and Borrelia recurrentis. The pathogenesis is multifactorial, including gallbladder stasis and ischemia.
trauma or burns, mechanical ventilation, total parenteral nutrition (TPN), recent episode of sepsis, multiple transfusions, childbirth, or hemodynamic instability. Significant vascular disease has been observed in 72% of outpatients diagnosed with acalculous cholecystitis. In children, risk factors include hemolytic disorders such as sickle cell anemia, congenital cystic duct stenosis, acute streptococcal infection, and Borrelia recurrentis. The pathogenesis is multifactorial, including gallbladder stasis and ischemia.
On physical examination, one may elicit exquisite RUQ tenderness or palpate a mass. Up to 20% of patients will be jaundiced due to partial biliary obstruction induced by inflammation extending into the CBD. Leukocytosis with a left shift and elevated liver function tests are more common than in acute calculous cholecystitis.
Chronic Cholecystitis
Chronic cholecystitis is associated with gallstones and results from the mechanical irritation or recurrent attacks of acute cholecystitis, which leads to fibrosis and gallbladder wall thickening. Symptoms do not correlate with its presence.
Diagnostic Imaging of Patients with Gallstones and Without Jaundice
US is the best initial imaging examination for gallstones due to its availability, low cost, and lack of ionizing radiation. US can be used in pregnant women, patients with contrast allergies, or those in whom MRI is contraindicated. Routine US has a >95% sensitivity for detecting gallstones, and is 88% sensitive and 80% specific for differentiating symptomatic cholelithiasis from acute cholecystitis. Under ideal conditions, stones 3 mm in size may be identified, and intrahepatic ductal dilatation may be ascertained. Characteristic findings of acute cholecystitis include gallbladder wall thickening (>4 to 5 mm) or edema (double-wall sign), pericholecystic fluid, impacted stone or biliary sludge, and a sonographic Murphy’s sign. US may erroneously note the presence of overlying bowel gas, but this gas may be in the gallbladder wall and is worrisome for gangrenous cholecystitis.
US findings that may suggest acalculous cholecystitis include absence of gallstones or sludge, >5-mm thickness of the gallbladder wall with pericholecystic fluid, a positive Murphy’s sign, failure to visualize the gallbladder, emphysematous cholecystitis with gas bubbles in the fundus of the gallbladder (champagne sign), or frank perforation of the gallbladder with associated abscess formation. An edematous gallbladder wall, however, may be visualized on US in patients with congestive heart failure, ascites, hypoalbuminemia, or nephrotic syndrome.
CT of the abdomen is often obtained when a patient presents with abdominal pain and other diagnoses are considered, or if there is evidence of intrahepatic ductal dilatation. CT is unnecessary in the diagnosis of acute cholecystitis, although it can demonstrate gallbladder wall edema, pericholecystic stranding, and high-attenuation bile.
Cholescintigraphy, which uses a technetium-labeled hepatic iminodiacetic acid, is often referred to as a hydroxy iminodiacetic acid (HIDA) scan, and is obtained if the US is indeterminate. HIDA is injected intravenously, taken up by hepatocytes, and secreted into bile. If the cystic duct is patent, the agent leads to visualization of the gallbladder. The contrast should then be visualized in the CBD and small bowel within 30 to 60 minutes. The test is “positive” if the gallbladder does not visualize, which signifies cystic duct obstruction secondary to edema with acute cholecystitis or an obstructing stone. The HIDA scan has 97% sensitivity and 90% specificity rates. False-positive results can result from cystic duct obstruction with a stone or tumor, severe liver disease, fasting patients receiving TPN, biliary sphincterotomy, and hyperbilirubinemia. False-negative results are uncommon and may be due to incomplete cystic duct obstruction. The HIDA scan is not helpful in intensive care unit patients due to bile stasis or poor gallbladder contractility and is also not recommended to assess acalculous cholecystitis as delay in therapy may prove fatal. HIDA scan is also useful in demonstrating biliary dyskinesia, defined as a gallbladder ejection fraction <40%, especially when pain occurs during cholecystokinin injection.
Magnetic resonance cholangiopancreatography (MRCP) is a noninvasive technique for evaluating the intrahepatic and extrahepatic bile ducts. Although the sensitivity of MRCP for detecting choledocholithiasis is 100%, specificity for gallbladder wall thickening is only 69%, compared with US (96%). MRCP is much more expensive than US and should not be used to diagnose acute cholecystitis.
Treatment of Patients with Gallstones and Without Jaundice
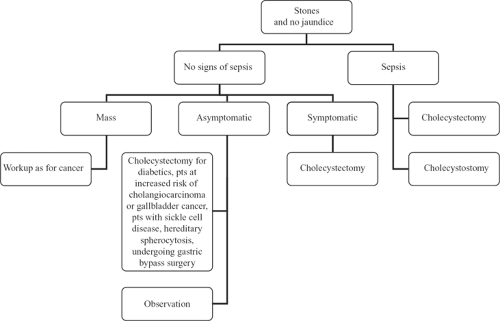
The treatment of these patients is illustrated in Figure 1. In patients with no signs of infection, the most important considerations are whether there is an associated mass or symptoms. Patients with a mass in the gallbladder or liver should be treated as if they have cancer. Ninety percent of patients with gallbladder cancer have associated gallstones. Treating these patients with a definitive operation for gallbladder cancer provides the only chance for long-term survival.
If patients with gallstones have biliary colic, cholecystectomy should be considered. Only a small percentage of asymptomatic patients with gallstones discovered incidentally will ever become symptomatic, and they are not recommended to undergo prophylactic cholecystectomy. However, elective cholecystectomy is reasonable for asymptomatic diabetic patients, who have increased risk of complications from cholecystitis, as well as patients who are at increased risk of developing cholangiocarcinoma or gallbladder cancer: choledochal cysts, Caroli’s disease, anomalous pancreatic ductal drainage, gallbladder adenoma, porcelain gallbladder. Patients with sickle cell disease, hereditary spherocytosis, and those undergoing gastric bypass surgeries are also recommended to undergo incidental cholecystectomy at the time of surgery.
Treatment of patients with acute cholecystitis includes broad-spectrum intravenous antibiotic therapy, as 50% to 70% of patients will have positive bile cultures. Definitive treatment is surgical resection. Laparoscopic cholecystectomy is the standard of care. Optimal timing of laparoscopic cholecystectomy for acute cholecystitis is within 72 hours of the onset of symptoms or after 6 weeks. Most patients with gangrenous cholecystitis will require conversion to an open cholecystectomy. For patients with cholecystitis and related sepsis, cholecystectomy is the definitive treatment. Cholecystostomy may be a temporizing measure for those too sick to have the definitive operation.
Treatment of patients with acalculous cholecystitis includes intravenous broad-spectrum antibiotics after blood cultures are obtained. As the incidence of gangrene and gallbladder perforation is high in patients with acalculous cholecystitis with mortality rates reaching 90% in critically ill patients, one should proceed to the operating room promptly for cholecystectomy, laparoscopic or open, and drainage of an associated abscess. In the unstable patient, a percutaneous cholecystostomy tube placed under US or CT guidance is indicated and may be more than just a temporizing measure. Subsequent cholecystectomy is often unnecessary if the patient recovers and the underlying problem that caused the cholecystitis is gone.
Cirrhotic patients with debilitating symptoms from gallstones and portal hypertension should only be considered for an open cholecystectomy. The cirrhotic liver is difficult to mobilize and retract, limiting one’s ability to provide adequate exposure. Moreover, abdominal wall, variceal, and hepatic bleeding are difficult to control laparoscopically. Puncture of an umbilical vein, which is often patent in patients with portal hypertension, can be life-threatening.
Gallstones in Patients with Jaundice
These entities may include choledocholithiasis and acute cholangitis.
Choledocholithiasis
Choledocholithiasis is present in 10% to 20% of patients with cholelithiasis. Indicators of choledocholithiasis include evidence of CBD stones on imaging, persistent jaundice (total bilirubin >3 mg/dL) especially in the setting of CBD duct dilatation (>6 mm), a history of jaundice or gallstone pancreatitis, or modest elevation of transaminases or alkaline phosphatase. If US detects intrahepatic and extrahepatic ductal dilatation in the setting of biliary colic, the most likely diagnosis is choledocholithiasis.
Cholangitis
Acute cholangitis is a clinical syndrome characterized by fever, RUQ pain, and jaundice, called Charcot’s triad. When associated with altered mental status and hypotension, known as Reynolds’ pentad, it can be life-threatening. In the elderly or those on corticosteroids, hypotension may be the only presenting symptom. In the setting of biliary obstruction and stasis due to biliary calculi or a stricture, high pressure facilitates bacteria (having ascended from the duodenum) to proliferate and migrate into the biliary tract and the circulation causing septicemia.
Biliary stones most commonly cause noniatrogenic cholangitis. Other causes include infected choledochal cysts, primary sclerosing cholangitis (PSC), hemobilia, and Mirizzi syndrome. Mirizzi syndrome refers to common hepatic duct obstruction caused by extrinsic compression from an impacted gallstone in the cystic duct. Iatrogenic conditions account for half of all episodes of acute cholangitis, especially in tertiary referral centers, due to the increasing numbers of biliary stents and complex biliary procedures involving biliary–biliary or biliary–enteric anastomosis, which may lead to biliary strictures.
Laboratory tests reveal elevated white blood count with a left shift and a cholestatic pattern of elevated total bilirubin, transaminases, and alkaline phosphatase. Blood cultures should be performed.
Diagnostic Imaging of Patients with Gallstones and Jaundice
Although intraoperative cholangiography is the “gold standard” for diagnosis, CBD stones can be diagnosed with US, ERCP, or MRCP. US is often the first diagnostic imaging study because of its availability, low cost, and sensitivity rates of 50% and 75% for detecting choledocholithiasis in nondilated and dilated ducts, respectively. ERCP has become “gold standard” for the diagnosis and treatment of intraluminal biliary abnormalities as sphincterotomy and stent placement can provide definitive relief of obstruction. ERCP is invasive, may miss small stones, and may cause pancreatitis. MRCP has a 90% to 95% concordance with ERCP in diagnosing CBD stones over 4 mm in diameter. MRCP may be useful if cholangitis is not severe and the risks of ERCP are high; however, if the patient has Charcot’s triad, therapeutic ERCP should not be delayed.
Endoscopic ultrasound (EUS) has 89% sensitivity and 94% specificity rates for choledocholithiasis. The role of EUS and choledocholithiasis has not been well established; however, it can be considered in patients with an intermediate risk of having a retained CBD stone in which ERCP is
undesirable, such as pregnant women (due to radiation exposure) or patients with gallstone pancreatitis (increased risk of post-ERCP pancreatitis).
undesirable, such as pregnant women (due to radiation exposure) or patients with gallstone pancreatitis (increased risk of post-ERCP pancreatitis).
In patients with no associated sepsis, CT scanning is usually performed in addition to the US to ascertain the level of ductal obstruction and to rule out mass lesions. Patients with a mass in the gallbladder or porta hepatis should be treated as a presumed cancer patient and evaluation for definitive resection performed.
Treatment of Patients with Gallstones and Jaundice
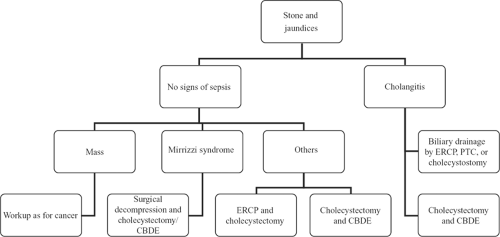
Figure 2 illustrates the algorithm for care of patients with choledocholithiasis and cholangitis.
If diagnosed preoperatively, several treatment options are available but are determined by the patient’s age and condition, the presence of jaundice or cholangitis, the size of the duct and stone, and the availability of a skilled endoscopist. ERCP with sphincterotomy and stone extraction should be considered for the following: choledocholithiasis diagnosed preoperatively, small (<5 mm) CBD, portal hypertension, severe periportal inflammation, and cholangitis with shock. If ERCP is unsuccessful or predicted to be unsuccessful, the obstruction will require relief via laparoscopic or open cholecystectomy and CBD exploration.
For those with common duct stones and no sepsis, both laparoscopic cholecystectomy with CBD exploration and ERCP with subsequent laparoscopic cholecystectomy represent reasonable options for treatment depending on the local expertise in endoscopy and laparoscopic surgery. Intraoperative cholangiograms are performed in the setting of a history of jaundice or gallstone pancreatitis, elevated liver functions tests, dilated CBD (>6 mm), or questionable anatomy. Laparoscopic common bile duct exploration (LCDE) is performed with three techniques: transcystic use of a Fogarty (3 French) catheter, transcystic choledochoscopy, or choledochotomy with choledochoscopy. Choledochotomy with LCDE requires the ability to accurately suture the choledochotomy closed, either primarily or over a T-tube. Laparoscopic cholecystectomy with LCDE offers a safe, effective, and cost-efficient method to care for patients with choledocholithiasis.
For patients with Mirizzi syndrome, surgical treatment is by cholecystectomy and CBD exploration. Laparoscopic technique may be challenging due to dense adhesions and distortion of normal anatomy, which increases the risk for biliary injury. It may become clear at surgery that inflammation at the porta hepatis precludes safe cholecystectomy and common duct exploration. In these cases, opening the gallbladder by a partial cholecystectomy and extracting the stone at the neck of the gallbladder is the safest effective procedure. If a fistula is present, suture of the fistula with absorbable suture or choledochoplasty with the remnant gallbladder is performed. Endoscopic treatment with or without lithotripsy can be a temporizing measure and can be definitive treatment for poor surgical candidates. Since approximately 5% to 10% of all cases of gallbladder cancer are associated with Mirizzi syndrome, the surgeon should be extremely vigilant for signs of cancer on the CT scan or on surgical exploration; it is prudent to obtain an intraoperative frozen section of the gallbladder.
Treatment of cholangitis also includes intravenous fluid resuscitation to maintain urine output, intensive monitoring for sepsis and multiorgan failure, as well as broad-spectrum intravenous antibiotics to cover E. coli, Klebsiella, Enterobacter, and Enterococcus, the most commonly isolated organisms that cause cholangitis. Antifungals and linezolid should be added in patients when the suspicion arises for Candida and vancomycin-resistant enterococcus, respectively. While 80% of patients with acute cholangitis will respond to antibiotic therapy, 20% of patients will require urgent biliary decompression: persistent abdominal pain, hypotension despite aggressive resuscitation, fever greater than 102ºF, or altered mental status. Biliary drainage can be achieved via ERCP, the treatment of choice, with sphincterotomy, stone extraction, and/or stent insertion; stones more than 2 cm in diameter require lithotripsy for fragmentation before removal. Percutaneous drainage can be considered when ERCP is unavailable or unsuccessful. Finally, emergency surgery is associated with a high rate of morbidity and mortality; once the cholangitis is controlled, elective surgery can be entertained. If emergent surgery is the only option for biliary decompression, one can perform a choledochotomy with placement of a large T-tube, which carries a lower mortality compared with cholecystectomy with CBD exploration.
Jaundice Without Gallstones
In patients presenting with jaundice and no gallstones, the most important consideration is to determine if the jaundice is obstructive in nature. Nonobstructive jaundice includes most of the prehepatic and hepatic causes of jaundice, and is generally not a surgical disease. The diseases that cause obstructive jaundice should be segregated according to level of obstruction: only intrahepatic ductal dilatation or both intrahepatic and extrahepatic ductal dilatation. By obtaining fractionated bilirubin levels, hepatitis serologies, and US, the patient with jaundice and without gallstones can be referred to the hepatologist for treatment of medical causes of jaundice, or be sent for appropriate workup according to the algorithms in Figures 3 and 4.
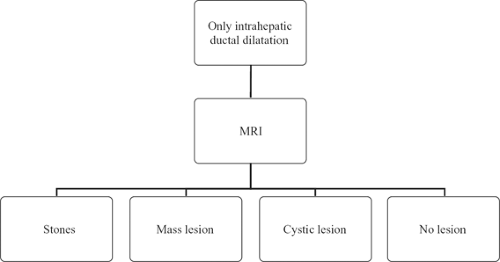
Patients with Only Intrahepatic Ductal Dilatation
Patients with dilated intrahepatic ducts but with no extrahepatic ductal dilatation have obstruction in the liver or high in the porta hepatis. The etiology of high biliary obstruction is stones, mass lesions, cystic lesions, or PSC. Stones can cause common duct obstruction or intrahepatic ductal obstruction. Mass lesions may be identified either as parenchymal lesions in the liver impinging on the bile duct or as masses growing from the biliary tree; the workup of the hepatic parenchymal lesion is discussed further later in this chapter. The most common neoplastic cause of biliary obstruction and jaundice is HCC, which can produce jaundice both by extrinsic compression and by intraductal extension. Jaundice from high biliary obstruction can also result from cholangiocarcinoma, either a small one occurring at the hilus of the liver (Klatskin tumor) or a large one extending to involve the hilus (intrahepatic cholangiocarcinoma). Cystic lesions, both benign and malignant, may also cause jaundice. Benign simple cysts that are large can obstruct the biliary tree, although the most common reason is rapid expansion from a spontaneous bleed into the cyst. Complex cysts resulting from infection (e.g., echinococcal) or malignancy (cystadenocarcinoma) can also cause jaundice. Finally, high biliary stricture can be caused by multifocal or unifocal sclerosing cholangitis. The multifocal form will usually be suspected because of multiple strictures seen on imaging and confirmed by liver biopsy. The unifocal form is often diagnosed only after an extensive liver and biliary resection for presumed cholangiocarcinoma.
Diagnostic Imaging of Patients with Intrahepatic Ductal Dilatation
The imaging test of choice for a high bile duct obstruction is MRCP. This is a very specialized MRI in which images are often taken in 1.5-mm overlapping slices. Images are also acquired at various phases after contrast injection in order to produce detailed depiction of the arterial, portal venous, and hepatic venous vessels. The most important images are those acquired as delayed T2 images that highlight slow-flowing fluid, which appears very bright relative to the low intensity of the surrounding tissues, allowing for precise depiction of the obstructed biliary tree. MRCP can identify larger intrahepatic ducts and extrahepatic ducts in 80% to 100% of nondilated ducts and 90% to 100% in dilated ducts. There is an overall sensitivity and specificity of MRCP of 95% and 97%, respectively, in the diagnosis of biliary obstruction.
Between US and MRCP, most high bile duct obstructions can be diagnosed and even subcentimeter hilar cholangiocarcinomas can be identified with confidence, as shown in Figure 5. These masses can be obscured by indwelling biliary stents. Nondecompressed dilated ducts will often point to the area of obstruction and highlight the mass involved. Ductal dilatation in both hepatic lobes with a contracted gallbladder
or nonunion of the right and left hepatic ducts suggests a Klatskin tumor. In addition to visualization of the tumor, the relationship of the tumor to adjacent vasculature, such as the portal veins and hepatic arteries, can be determined. A most important detail to check in the imaging tests is hepatic lobar atrophy because this usually indicates either long-standing ductal obstruction of that side or portal venous involvement by tumor. Of note, atrophy on one side combined with vascular involvement by tumor on the opposite side indicates unresectable cancer. For patients with PSC, MRCP with MRI can document the segmental extent of ductal involvement in surgical planning, assess for intrahepatic metastases, and identify aberrant ductal anatomy. MRCP has the capability to evaluate the bile ducts above and below a structure while identifying any intrahepatic mass lesions.
or nonunion of the right and left hepatic ducts suggests a Klatskin tumor. In addition to visualization of the tumor, the relationship of the tumor to adjacent vasculature, such as the portal veins and hepatic arteries, can be determined. A most important detail to check in the imaging tests is hepatic lobar atrophy because this usually indicates either long-standing ductal obstruction of that side or portal venous involvement by tumor. Of note, atrophy on one side combined with vascular involvement by tumor on the opposite side indicates unresectable cancer. For patients with PSC, MRCP with MRI can document the segmental extent of ductal involvement in surgical planning, assess for intrahepatic metastases, and identify aberrant ductal anatomy. MRCP has the capability to evaluate the bile ducts above and below a structure while identifying any intrahepatic mass lesions.
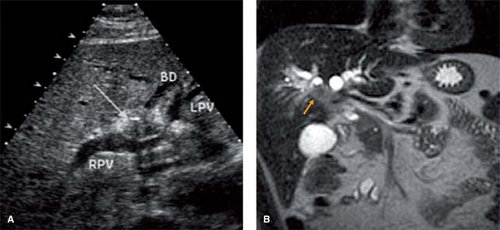 Fig. 5. US (A) and MRCP (B) demonstrating small cholangiocarcinoma (arrow) at hilus of liver and relationship to vasculature. BD, bile duct; LPV, left portal vein; RPV, right portal vein. |
PTC and drainage is preferred over ERCP as palliation for jaundice secondary to high biliary obstruction. Endoscopic decompression is difficult in high biliary obstruction. Additionally, because the liver is often segregated into multiple isolated segments by the obstruction, if ERCP is performed and the liver is incompletely drained, sepsis usually ensues from the contaminated, but incompletely drained segments of liver. PTC is usually performed late because dilated ducts often aid in the diagnosis.
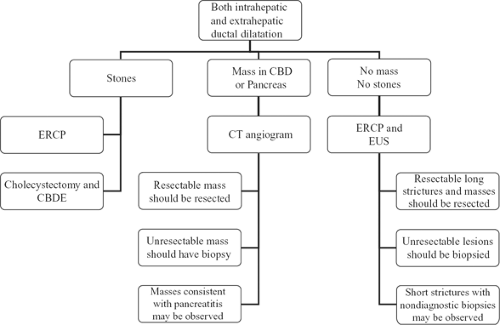
Patients with Both Intra- and Extrahepatic Ductal Dilatation
A finding of extrahepatic ductal dilatation indicates distal bile duct obstruction.
Diagnostic Imaging of Patients with Intra- and Extrahepatic Ductal Dilatation
Patients should be imaged by either ERCP or CT angiography (Fig. 4), a thin-cut CT with images acquired not only at the portal venous phase but also early in the arterial phase. The advantage is that it allows visualization and characterization of small periampullary tumors. If stones are noted by MRCP or US, an ERCP should be performed to extract the stone. If a mass is noted, the enhancement pattern may provide clues for diagnosis. A very hypervascular mass may suggest a neuroendocrine tumor. A small mass at the tip of the dilated duct may suggest an ampullary adenoma or carcinoma. From a practical standpoint, however, the exact diagnosis of a resectable neoplasm is not necessary. Most mass lesions that cause painless jaundice will turn out to be cancerous or precancerous and deserve resection. The major exceptions are lesions that result from pancreatitis. Thus, the scans should be scrutinized for signs of chronic pancreatitis, such as pancreatic calcifications or pseudocysts. If pancreatitis is suspected, observation may be the most appropriate choice of action.
CT angiogram or MRI will also allow for assessment of local vascular invasion by tumor and for signs of liver and peritoneal metastases. If the periampullary cancer is not resectable, a biopsy should be performed to confirm diagnosis and to help plan palliative therapy.
Often, jaundice and extrahepatic biliary obstruction will be found with no appreciable mass lesion. ERCP and EUS should be performed to assess the characteristics of the stricture and to determine if a small mass in the head of the pancreas can be ascertained. Small masses can also be biopsied by EUS guidance. If it is a long stricture, cancer in the form of distal cholangiocarcinoma or pancreatic cancer must be suspected. Short strictures with no appreciable mass may be observed for a time. ERCP will also allow placement of biliary stents for palliation.
Gallbladder Polyps
Most gallbladder masses are benign and are found in up to 5% of patients assessed by US. There is no association between the presence of gallbladder polyps and the typical risk factors for gallstones. They are rarely described in children, and occur either as a primary disorder or in association with Peutz–Jeghers syndrome, metachromatic leukodystrophy, or pancreatobiliary malunion. Benign polyps can be classified into epithelial tumors (adenoma), mesenchymal tumors (fibroma, lipoma, hemangioma), or pseudotumors (cholesterol polyps, inflammatory polyps, and adenomyoma). The majority are cholesterol polyps, which histologically are mucosal deposits of lipid-laden macrophages. These lesions, which make up more than 90% of benign lesions, are characteristically multiple (>3), small, and have an intact mucosa. Adenomyomatosis, characterized by mucosal overgrowth, thickening of the muscle wall, and intramural diverticula, does not involve adenomatous changes in the gallbladder epithelium; therefore, this is not a premalignant condition. The only polyp with malignant potential is the adenoma, which is rare with an incidence of 0.5%. The
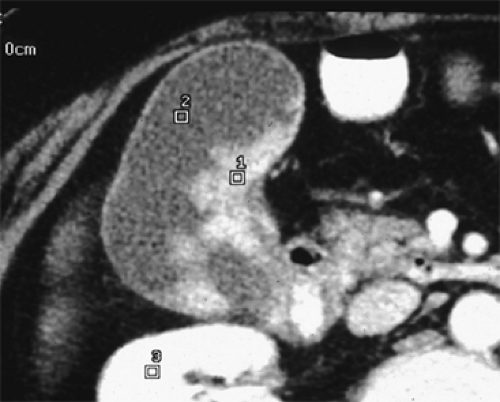
frequency of the adenoma-to-adenocarcinoma sequence is unknown; however, the risk of malignancy is related to polyp size. In general, gallbladder adenocarcinoma is significantly more likely to be found in polyps of older patients (>50 years), solitary and sessile in nature, and more than 1.0 cm in diameter (Fig. 6).
frequency of the adenoma-to-adenocarcinoma sequence is unknown; however, the risk of malignancy is related to polyp size. In general, gallbladder adenocarcinoma is significantly more likely to be found in polyps of older patients (>50 years), solitary and sessile in nature, and more than 1.0 cm in diameter (Fig. 6).
Gallbladder polyps may cause biliary pain, due to prolapse of the polyp into Hartmann’s pouch or from cystic duct obstruction from a detached piece of polyp leading to biliary colic, cholecystitis, or even pancreatitis. Additionally, cholesterolosis and adenomyomatosis have been associated with chronic dyspeptic abdominal pain.
Diagnostic Imaging of Patients with Gallbladder Polyps
Figure 7 illustrates an algorithm for workup and treatment of patients with gallbladder polyps and masses. It is based on longitudinal follow-up studies in which small (<1 cm) gallbladder polyps followed by imaging had a low rate of subsequent diagnosis of cancer. Abdominal US is the study of choice due to its >90% sensitivity and specificity rates in diagnosing small polyps. CT is most useful in patients with suspected gallbladder cancer as it can provide staging information such as liver involvement or metastasis.
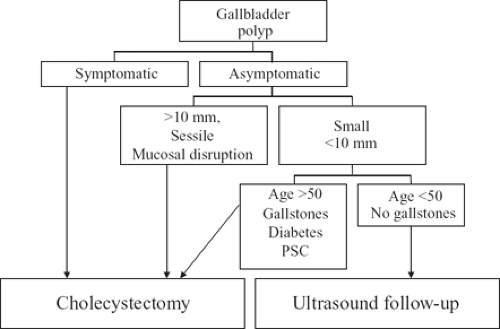 Fig. 7. Algorithm for care of the patient with gallbladder polyp. PSC, primary sclerosing cholangitis.
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|