Criteria for Acute Myocardial Infarction
The term acute myocardial infarction (MI) should be used when there is evidence of myocardial necrosis in a clinical setting consistent with acute myocardial ischemia. Under these conditions, any one of the following criteria meets the diagnosis for MI:
• Detection of a rise and/or fall of cardiac biomarker values (preferably cardiac troponin [cTn]) with at least one value above the 99th percentile upper reference limit (URL) and with at least one of the following:
• Symptoms of ischemia
• New or presumed new significant ST-segment T-wave (ST-T) changes or new left bundle branch block (LBBB)
• Development of pathologic Q waves in the electrocardiogram (ECG)
• Imaging evidence of new loss of viable myocardium or new regional wall motion abnormality
• Identification of an intracoronary thrombus by angiography or autopsy
• Cardiac death with symptoms suggestive of myocardial ischemia and presumed new ischemic ECG changes of new LBBB, but death occurred before cardiac biomarkers were obtained or before cardiac biomarker values would be increased.
• Percutaneous coronary intervention (PCI)–related MI is arbitrarily defined by elevation of cTn values (>5 × 99th percentile URL) in patients with normal baseline values (≤99th percentile URL) or a rise of cTn values >20% if the baseline values are elevated and are stable or falling. In addition, either (i) symptoms suggestive of myocardial ischemia, or (ii) new ischemic ECG changes, or (iii) angiographic findings consistent with a procedural complication, or (iv) imaging demonstration of new loss of viable myocardium or new regional wall motion abnormality are required.
• Stent thrombosis associated with MI when detected by coronary angiography or autopsy in the setting of myocardial ischemia and with a rise and/or fall of cardiac biomarker values with at least one value above the 99th percentile URL.
• Coronary artery bypass grafting (CABG)–related MI is arbitrarily defined by elevation of cardiac biomarker values (>10 × 99th percentile URL) in patients with normal baseline cTn values (≤99th percentile URL). In addition, either (i) new pathologic Q waves or new LBBB, or (ii) angiographic documented new graft or new native coronary artery occlusion, or (iii) imaging evidence of new loss of viable myocardium or new regional wall motion abnormality.
Source: K Thygesen: Eur Heart J 33:2551, 2012.
CLASSIFICATION OF MYOCARDIAL INFARCTION |
Source: K Thygesen: Eur Heart J 33:2551, 2012.
INITIAL MANAGEMENT
PREHOSPITAL CARE
The prognosis in STEMI is largely related to the occurrence of two general classes of complications: (1) electrical complications (arrhythmias) and (2) mechanical complications (“pump failure”). Most out-of-hospital deaths from STEMI are due to the sudden development of ventricular fibrillation. The vast majority of deaths due to ventricular fibrillation occur within the first 24 h of the onset of symptoms, and of these, over half occur in the first hour. Therefore, the major elements of prehospital care of patients with suspected STEMI include (1) recognition of symptoms by the patient and prompt seeking of medical attention; (2) rapid deployment of an emergency medical team capable of performing resuscitative maneuvers, including defibrillation; (3) expeditious transportation of the patient to a hospital facility that is continuously staffed by physicians and nurses skilled in managing arrhythmias and providing advanced cardiac life support; and (4) expeditious implementation of reperfusion therapy (Fig. 295-3). The greatest delay usually occurs not during transportation to the hospital but, rather, between the onset of pain and the patient’s decision to call for help. This delay can best be reduced by health care professionals educating the public concerning the significance of chest discomfort and the importance of seeking early medical attention. Regular office visits with patients having a history of or who are at risk for ischemic heart disease are important “teachable moments” for clinicians to review the symptoms of STEMI and the appropriate action plan.
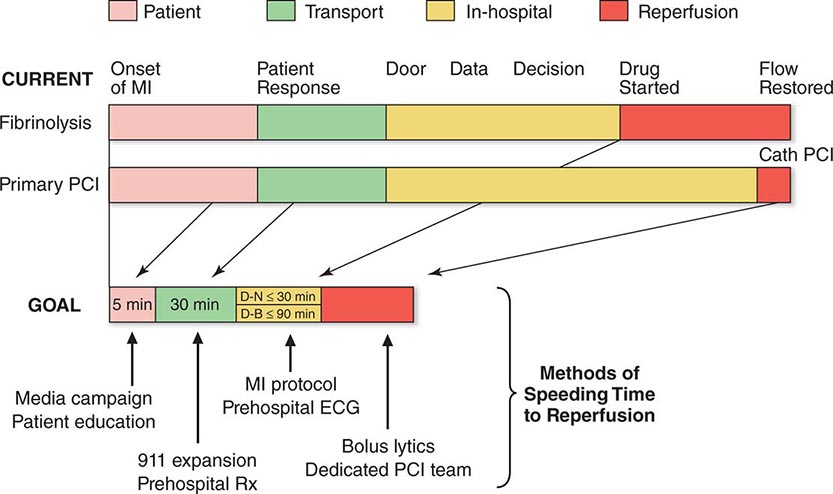
FIGURE 295-3 Major components of time delay between onset of symptoms from ST-segment elevation myocardial infarction and restoration of flow in the infarct-related artery. Plotted sequentially from left to right are the times for patients to recognize symptoms and seek medical attention, transportation to the hospital, in-hospital decision making, implementation of reperfusion strategy, and restoration of flow once the reperfusion strategy has been initiated. The time to initiate fibrinolytic therapy is the “door-to-needle” (D-N) time; this is followed by the period of time required for pharmacologic restoration of flow. More time is required to move the patient to the catheterization laboratory for a percutaneous coronary interventional (PCI) procedure, referred to as the “door-to-balloon” (D-B) time, but restoration of flow in the epicardial infarct–related artery occurs promptly after PCI. At the bottom is a variety of methods for speeding the time to reperfusion along with the goals for the time intervals for the various components of the time delay. (Adapted from CP Cannon et al: J Thromb Thrombol 1:27, 1994.)
Increasingly, monitoring and treatment are carried out by trained personnel in the ambulance, further shortening the time between the onset of the infarction and appropriate treatment. General guidelines for initiation of fibrinolysis in the prehospital setting include the ability to transmit 12-lead ECGs to confirm the diagnosis, the presence of paramedics in the ambulance, training of paramedics in the interpretation of ECGs and management of STEMI, and online medical command and control that can authorize the initiation of treatment in the field.
MANAGEMENT IN THE EMERGENCY DEPARTMENT
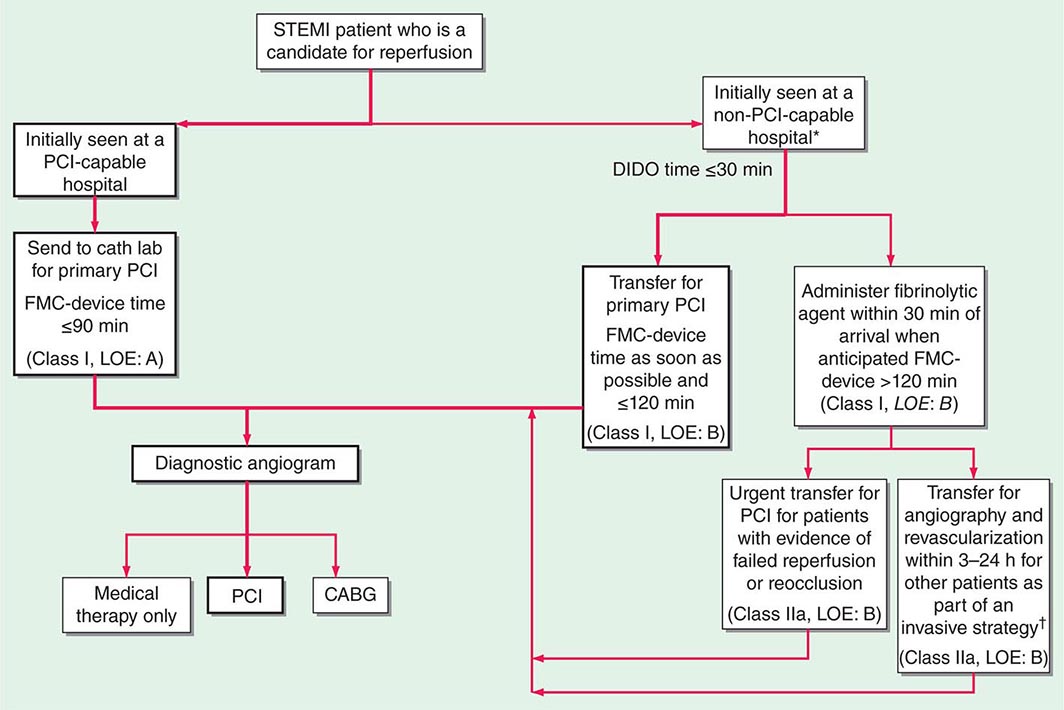
In the Emergency Department, the goals for the management of patients with suspected STEMI include control of cardiac discomfort, rapid identification of patients who are candidates for urgent reperfusion therapy, triage of lower-risk patients to the appropriate location in the hospital, and avoidance of inappropriate discharge of patients with STEMI. Many aspects of the treatment of STEMI are initiated in the Emergency Department and then continued during the in-hospital phase of management (Fig. 295-4). The overarching goal is to minimize the time from first medical contact to initiation of reperfusion therapy. This may involve transfer from a non-PCI hospital to one that is PCI capable, with a goal of initiating PCI within 120 min of first medical contact (Fig. 295-4).
FIGURE 295-4 Reperfusion therapy for patients with ST-segment elevation myocardial infarction (STEMI). The bold arrows and boxes are the preferred strategies. Performance of percutaneous coronary intervention (PCI) is dictated by an anatomically appropriate culprit stenosis. *Patients with cardiogenic shock or severe heart failure initially seen at a non–PCI-capable hospital should be transferred for cardiac catheterization and revascularization as soon as possible, irrespective of time delay from myocardial infarction (MI) onset (Class I, LOE: B). †Angiography and revascularization should not be performed within the first 2 to 3 hours after administration of fibrinolytic therapy. CABG, coronary artery bypass graft; DIDO, door-in–door-out; FMC, first medical contact; LOE, level of evidence; STEMI, ST-elevation myocardial infarction. (Adapted with permission from P O’Gara et al: Circulation 127:e362, 2013.)
Aspirin is essential in the management of patients with suspected STEMI and is effective across the entire spectrum of acute coronary syndromes (Fig. 295-1). Rapid inhibition of cyclooxygenase-1 in platelets followed by a reduction of thromboxane A2 levels is achieved by buccal absorption of a chewed 160–325-mg tablet in the Emergency Department. This measure should be followed by daily oral administration of aspirin in a dose of 75–162 mg.
In patients whose arterial O2 saturation is normal, supplemental O2 is of limited if any clinical benefit and therefore is not cost-effective. However, when hypoxemia is present, O2 should be administered by nasal prongs or face mask (2–4 L/min) for the first 6–12 h after infarction; the patient should then be reassessed to determine if there is a continued need for such treatment.
CONTROL OF DISCOMFORT
Sublingual nitroglycerin can be given safely to most patients with STEMI. Up to three doses of 0.4 mg should be administered at about 5-min intervals. In addition to diminishing or abolishing chest discomfort, nitroglycerin may be capable of both decreasing myocardial oxygen demand (by lowering preload) and increasing myocardial oxygen supply (by dilating infarct-related coronary vessels or collateral vessels). In patients whose initially favorable response to sublingual nitroglycerin is followed by the return of chest discomfort, particularly if accompanied by other evidence of ongoing ischemia such as further ST-segment or T-wave shifts, the use of intravenous nitroglycerin should be considered. Therapy with nitrates should be avoided in patients who present with low systolic arterial pressure (<90 mmHg) or in whom there is clinical suspicion of RV infarction (inferior infarction on ECG, elevated jugular venous pressure, clear lungs, and hypotension). Nitrates should not be administered to patients who have taken a phosphodiesterase-5 inhibitor for erectile dysfunction within the preceding 24 h, because it may potentiate the hypotensive effects of nitrates. An idiosyncratic reaction to nitrates, consisting of sudden marked hypotension, sometimes occurs but can usually be reversed promptly by the rapid administration of intravenous atropine.
Morphine is a very effective analgesic for the pain associated with STEMI. However, it may reduce sympathetically mediated arteriolar and venous constriction, and the resulting venous pooling may reduce cardiac output and arterial pressure. These hemodynamic disturbances usually respond promptly to elevation of the legs, but in some patients, volume expansion with intravenous saline is required. The patient may experience diaphoresis and nausea, but these events usually pass and are replaced by a feeling of well-being associated with the relief of pain. Morphine also has a vagotonic effect and may cause bradycardia or advanced degrees of heart block, particularly in patients with inferior infarction. These side effects usually respond to atropine (0.5 mg intravenously). Morphine is routinely administered by repetitive (every 5 min) intravenous injection of small doses (2–4 mg), rather than by the subcutaneous administration of a larger quantity, because absorption may be unpredictable by the latter route.
Intravenous beta blockers are also useful in the control of the pain of STEMI. These drugs control pain effectively in some patients, presumably by diminishing myocardial O2 demand and hence ischemia. More important, there is evidence that intravenous beta blockers reduce the risks of reinfarction and ventricular fibrillation (see “Beta-Adrenoceptor Blockers” below). However, patient selection is important when considering beta blockers for STEMI. Oral beta blocker therapy should be initiated in the first 24 h for patients who do not have any of the following: (1) signs of heart failure, (2) evidence of a low-output state, (3) increased risk for cardiogenic shock, or (4) other relative contraindications to beta blockade (PR interval greater than 0.24 seconds, second- or third-degree heart block, active asthma, or reactive airway disease). A commonly employed regimen is metoprolol, 5 mg every 2–5 min for a total of three doses, provided the patient has a heart rate >60 beats/min, systolic pressure >100 mmHg, a PR interval <0.24 s, and rales that are no higher than 10 cm up from the diaphragm. Fifteen minutes after the last intravenous dose, an oral regimen is initiated of 50 mg every 6 h for 48 h, followed by 100 mg every 12 h.
Unlike beta blockers, calcium antagonists are of little value in the acute setting, and there is evidence that short-acting dihydropyridines may be associated with an increased mortality risk.
MANAGEMENT STRATEGIES
The primary tool for screening patients and making triage decisions is the initial 12-lead ECG. When ST-segment elevation of at least 2 mm in two contiguous precordial leads and 1 mm in two adjacent limb leads is present, a patient should be considered a candidate for reperfusion therapy (Fig. 295-4). The process of selecting patients for fibrinolysis versus primary PCI (angioplasty or stenting; Chap. 296e) is discussed below. In the absence of ST-segment elevation, fibrinolysis is not helpful, and evidence exists suggesting that it may be harmful.
LIMITATION OF INFARCT SIZE
The quantity of myocardium that becomes necrotic as a consequence of a coronary artery occlusion is determined by factors other than just the site of occlusion. While the central zone of the infarct contains necrotic tissue that is irretrievably lost, the fate of the surrounding ischemic myocardium (ischemic penumbra) may be improved by timely restoration of coronary perfusion, reduction of myocardial O2 demands, prevention of the accumulation of noxious metabolites, and blunting of the impact of mediators of reperfusion injury (e.g., calcium overload and oxygen-derived free radicals). Up to one-third of patients with STEMI may achieve spontaneous reperfusion of the infarct-related coronary artery within 24 h and experience improved healing of infarcted tissue. Reperfusion, either pharmacologically (by fibrinolysis) or by PCI, accelerates the opening of infarct-related arteries in those patients in whom spontaneous fibrinolysis ultimately would have occurred and also greatly increases the number of patients in whom restoration of flow in the infarct-related artery is accomplished. Timely restoration of flow in the epicardial infarct–related artery combined with improved perfusion of the downstream zone of infarcted myocardium results in a limitation of infarct size. Protection of the ischemic myocardium by the maintenance of an optimal balance between myocardial O2 supply and demand through pain control, treatment of congestive heart failure (CHF), and minimization of tachycardia and hypertension extends the “window” of time for the salvage of myocardium by reperfusion strategies.
Glucocorticoids and nonsteroidal anti-inflammatory agents, with the exception of aspirin, should be avoided in patients with STEMI. They can impair infarct healing and increase the risk of myocardial rupture, and their use may result in a larger infarct scar. In addition, they can increase coronary vascular resistance, thereby potentially reducing flow to ischemic myocardium.
PRIMARY PERCUTANEOUS CORONARY INTERVENTION
(See also Chap. 296e) PCI, usually angioplasty and/or stenting without preceding fibrinolysis, referred to as primary PCI, is effective in restoring perfusion in STEMI when carried out on an emergency basis in the first few hours of MI. It has the advantage of being applicable to patients who have contraindications to fibrinolytic therapy (see below) but otherwise are considered appropriate candidates for reperfusion. It appears to be more effective than fibrinolysis in opening occluded coronary arteries and, when performed by experienced operators in dedicated medical centers, is associated with better short-term and long-term clinical outcomes. Compared with fibrinolysis, primary PCI is generally preferred when the diagnosis is in doubt, cardiogenic shock is present, bleeding risk is increased, or symptoms have been present for at least 2–3 h when the clot is more mature and less easily lysed by fibrinolytic drugs. However, PCI is expensive in terms of personnel and facilities, and its applicability is limited by its availability, around the clock, in only a minority of hospitals (Fig. 295-4).
FIBRINOLYSIS
If no contraindications are present (see below), fibrinolytic therapy should ideally be initiated within 30 min of presentation (i.e., door-to-needle time ≤30 min). The principal goal of fibrinolysis is prompt restoration of full coronary arterial patency. The fibrinolytic agents tissue plasminogen activator (tPA), streptokinase, tenecteplase (TNK), and reteplase (rPA) have been approved by the U.S. Food and Drug Administration for intravenous use in patients with STEMI. These drugs all act by promoting the conversion of plasminogen to plasmin, which subsequently lyses fibrin thrombi. Although considerable emphasis was first placed on a distinction between more fibrin-specific agents, such as tPA, and non-fibrin-specific agents, such as streptokinase, it is now recognized that these differences are only relative, as some degree of systemic fibrinolysis occurs with the former agents. TNK and rPA are referred to as bolus fibrinolytics since their administration does not require a prolonged intravenous infusion.
When assessed angiographically, flow in the culprit coronary artery is described by a simple qualitative scale called the Thrombolysis in Myocardial Infarction (TIMI) grading system: grade 0 indicates complete occlusion of the infarct-related artery; grade 1 indicates some penetration of the contrast material beyond the point of obstruction but without perfusion of the distal coronary bed; grade 2 indicates perfusion of the entire infarct vessel into the distal bed, but with flow that is delayed compared with that of a normal artery; and grade 3 indicates full perfusion of the infarct vessel with normal flow. The latter is the goal of reperfusion therapy, because full perfusion of the infarct-related coronary artery yields far better results in terms of limiting infarct size, maintenance of LV function, and reduction of both short- and long-term mortality rates. Additional methods of angiographic assessment of the efficacy of fibrinolysis include counting the number of frames on the cine film required for dye to flow from the origin of the infarct-related artery to a landmark in the distal vascular bed (TIMI frame count) and determining the rate of entry and exit of contrast dye from the microvasculature in the myocardial infarct zone (TIMI myocardial perfusion grade). These methods have an even tighter correlation with outcomes after STEMI than the more commonly employed TIMI flow grade.
tPA and the other relatively fibrin-specific plasminogen activators, rPA and TNK, are more effective than streptokinase at restoring full perfusion—i.e., TIMI grade 3 coronary flow—and have a small edge in improving survival as well. The current recommended regimen of tPA consists of a 15-mg bolus followed by 50 mg intravenously over the first 30 min, followed by 35 mg over the next 60 min. Streptokinase is administered as 1.5 million units (MU) intravenously over 1 h. rPA is administered in a double-bolus regimen consisting of a 10-MU bolus given over 2–3 min, followed by a second 10-MU bolus 30 min later. TNK is given as a single weight-based intravenous bolus of 0.53 mg/kg over 10 s. In addition to the fibrinolytic agents discussed earlier, pharmacologic reperfusion typically involves adjunctive antiplatelet and antithrombotic drugs, as discussed subsequently.
Clear contraindications to the use of fibrinolytic agents include a history of cerebrovascular hemorrhage at any time, a nonhemorrhagic stroke or other cerebrovascular event within the past year, marked hypertension (a reliably determined systolic arterial pressure >180 mmHg and/or a diastolic pressure >110 mmHg) at any time during the acute presentation, suspicion of aortic dissection, and active internal bleeding (excluding menses). While advanced age is associated with an increase in hemorrhagic complications, the benefit of fibrinolytic therapy in the elderly appears to justify its use if no other contraindications are present and the amount of myocardium in jeopardy appears to be substantial.
Relative contraindications to fibrinolytic therapy, which require assessment of the risk-to-benefit ratio, include current use of anticoagulants (international normalized ratio ≥2), a recent (<2 weeks) invasive or surgical procedure or prolonged (>10 min) cardiopulmonary resuscitation, known bleeding diathesis, pregnancy, a hemorrhagic ophthalmic condition (e.g., hemorrhagic diabetic retinopathy), active peptic ulcer disease, and a history of severe hypertension that is currently adequately controlled. Because of the risk of an allergic reaction, patients should not receive streptokinase if that agent had been received within the preceding 5 days to 2 years.
Allergic reactions to streptokinase occur in ∼2% of patients who receive it. While a minor degree of hypotension occurs in 4–10% of patients given this agent, marked hypotension occurs, although rarely, in association with severe allergic reactions.
Hemorrhage is the most frequent and potentially the most serious complication. Because bleeding episodes that require transfusion are more common when patients require invasive procedures, unnecessary venous or arterial interventions should be avoided in patients receiving fibrinolytic agents. Hemorrhagic stroke is the most serious complication and occurs in ∼0.5–0.9% of patients being treated with these agents. This rate increases with advancing age, with patients >70 years experiencing roughly twice the rate of intracranial hemorrhage as those <65 years. Large-scale trials have suggested that the rate of intracranial hemorrhage with tPA or rPA is slightly higher than with streptokinase.
INTEGRATED REPERFUSION STRATEGY
Evidence has emerged that suggests PCI plays an increasingly important role in the management of STEMI. Prior approaches that segregated the pharmacologic and catheter-based approaches to reperfusion have now been replaced with an integrated approach to triage and transfer of STEMI patients to receive PCI (Fig. 295-4). To achieve the degree of integration required to care for a patient with STEMI, all communities should create and maintain a regional system of STEMI care that includes assessment and continuous quality improvement of emergency medical services and hospital-based activities.
Cardiac catheterization and coronary angiography should be carried out after fibrinolytic therapy if there is evidence of either (1) failure of reperfusion (persistent chest pain and ST-segment elevation >90 min), in which case a rescue PCI should be considered; or (2) coronary artery reocclusion (re-elevation of ST segments and/or recurrent chest pain) or the development of recurrent ischemia (such as recurrent angina in the early hospital course or a positive exercise stress test before discharge), in which case an urgent PCI should be considered. Routine angiography and elective PCI even in asymptomatic patients following administration of fibrinolytic therapy are used with less frequency, given the numerous technologic advances that have occurred in the catheterization laboratory and the increasing number of skilled interventionalists. Coronary artery bypass surgery should be reserved for patients whose coronary anatomy is unsuited to PCI but in whom revascularization appears to be advisable because of extensive jeopardized myocardium or recurrent ischemia.
HOSPITAL PHASE MANAGEMENT
CORONARY CARE UNITS
These units are routinely equipped with a system that permits continuous monitoring of the cardiac rhythm of each patient and hemodynamic monitoring in selected patients. Defibrillators, respirators, noninvasive transthoracic pacemakers, and facilities for introducing pacing catheters and flow-directed balloon-tipped catheters are also usually available. Equally important is the organization of a highly trained team of nurses who can recognize arrhythmias; adjust the dosage of antiarrhythmic, vasoactive, and anticoagulant drugs; and perform cardiac resuscitation, including electroshock, when necessary.
Patients should be admitted to a coronary care unit early in their illness when it is expected that they will derive benefit from the sophisticated and expensive care provided. The availability of electrocardiographic monitoring and trained personnel outside the coronary care unit has made it possible to admit lower-risk patients (e.g., those not hemodynamically compromised and without active arrhythmias) to “intermediate care units.”
The duration of stay in the coronary care unit is dictated by the ongoing need for intensive care. If symptoms are controlled with oral therapy, patients may be transferred out of the coronary care unit. Also, patients who have a confirmed STEMI but who are considered to be at low risk (no prior infarction and no persistent chest discomfort, CHF, hypotension, or cardiac arrhythmias) may be safely transferred out of the coronary care unit within 24 h.
Activity Factors that increase the work of the heart during the initial hours of infarction may increase the size of the infarct. Therefore, patients with STEMI should be kept at bed rest for the first 6–12 h. However, in the absence of complications, patients should be encouraged, under supervision, to resume an upright posture by dangling their feet over the side of the bed and sitting in a chair within the first 24 h. This practice is psychologically beneficial and usually results in a reduction in the pulmonary capillary wedge pressure. In the absence of hypotension and other complications, by the second or third day, patients typically are ambulating in their room with increasing duration and frequency, and they may shower or stand at the sink to bathe. By day 3 after infarction, patients should be increasing their ambulation progressively to a goal of 185 m (600 ft) at least three times a day.
Diet Because of the risk of emesis and aspiration soon after STEMI, patients should receive either nothing or only clear liquids by mouth for the first 4–12 h. The typical coronary care unit diet should provide ≤30% of total calories as fat and have a cholesterol content of ≤300 mg/d. Complex carbohydrates should make up 50–55% of total calories. Portions should not be unusually large, and the menu should be enriched with foods that are high in potassium, magnesium, and fiber, but low in sodium. Diabetes mellitus and hypertriglyceridemia are managed by restriction of concentrated sweets in the diet.
Bowel Management Bed rest and the effect of the narcotics used for the relief of pain often lead to constipation. A bedside commode rather than a bedpan, a diet rich in bulk, and the routine use of a stool softener such as dioctyl sodium sulfosuccinate (200 mg/d) are recommended. If the patient remains constipated despite these measures, a laxative can be prescribed. Contrary to prior belief, it is safe to perform a gentle rectal examination on patients with STEMI.
Sedation Many patients require sedation during hospitalization to withstand the period of enforced inactivity with tranquility. Diazepam (5 mg), oxazepam (15–30 mg), or lorazepam (0.5–2 mg), given three to four times daily, is usually effective. An additional dose of any of the above medications may be given at night to ensure adequate sleep. Attention to this problem is especially important during the first few days in the coronary care unit, where the atmosphere of 24-h vigilance may interfere with the patient’s sleep. However, sedation is no substitute for reassuring, quiet surroundings. Many drugs used in the coronary care unit, such as atropine, H2 blockers, and narcotics, can produce delirium, particularly in the elderly. This effect should not be confused with agitation, and it is wise to conduct a thorough review of the patient’s medications before arbitrarily prescribing additional doses of anxiolytics.
PHARMACOTHERAPY
ANTITHROMBOTIC AGENTS
The use of antiplatelet and anticoagulant therapy during the initial phase of STEMI is based on extensive laboratory and clinical evidence that thrombosis plays an important role in the pathogenesis of this condition. The primary goal of treatment with antiplatelet and anticoagulant agents is to maintain patency of the infarct-related artery, in conjunction with reperfusion strategies. A secondary goal is to reduce the patient’s tendency to thrombosis and, thus, the likelihood of mural thrombus formation or deep venous thrombosis, either of which could result in pulmonary embolization. The degree to which antiplatelet and anticoagulant therapy achieves these goals partly determines how effectively it reduces the risk of mortality from STEMI.
As noted previously (see “Management in the Emergency Department” earlier), aspirin is the standard antiplatelet agent for patients with STEMI. The most compelling evidence for the benefits of antiplatelet therapy (mainly with aspirin) in STEMI is found in the comprehensive overview by the Antiplatelet Trialists’ Collaboration. Data from nearly 20,000 patients with MI enrolled in 15 randomized trials were pooled and revealed a relative reduction of 27% in the mortality rate, from 14.2% in control patients to 10.4% in patients receiving antiplatelet agents.
Inhibitors of the P2Y12 ADP receptor prevent activation and aggregation of platelets. The addition of the P2Y12 inhibitor clopidogrel to background treatment with aspirin to STEMI patients reduces the risk of clinical events (death, reinfarction, stroke) and, in patients receiving fibrinolytic therapy, has been shown to prevent reocclusion of a successfully reperfused infarct artery. New P2Y12 ADP receptor antagonists, such as prasugrel and ticagrelor, are more effective than clopidogrel in preventing ischemic complications in STEMI patients undergoing PCI, but are associated with an increased risk of bleeding. Glycoprotein IIb/IIIa receptor inhibitors appear useful for preventing thrombotic complications in patients with STEMI undergoing PCI.
The standard anticoagulant agent used in clinical practice is unfractionated heparin (UFH). The available data suggest that when UFH is added to a regimen of aspirin and a non-fibrin-specific thrombolytic agent such as streptokinase, additional mortality benefit occurs (about 5 lives saved per 1000 patients treated). It appears that the immediate administration of intravenous UFH, in addition to a regimen of aspirin and relatively fibrin-specific fibrinolytic agents (tPA, rPA, or TNK), helps to maintain patency of the infarct-related artery. This effect is achieved at the cost of a small increased risk of bleeding. The recommended dose of UFH is an initial bolus of 60 U/kg (maximum 4000 U) followed by an initial infusion of 12 U/kg per hour (maximum 1000 U/h). The activated partial thromboplastin time during maintenance therapy should be 1.5–2 times the control value.
Alternatives to UFH for anticoagulation of patients with STEMI are the low-molecular-weight heparin (LMWH) preparations, a synthetic version of the critical pentasaccharide sequence (fondaparinux), and the direct antithrombin bivalirudin. Advantages of LMWHs include high bioavailability permitting administration subcutaneously, reliable anticoagulation without monitoring, and greater antiXa:IIa activity. Enoxaparin has been shown to reduce significantly the composite endpoints of death/nonfatal reinfarction and death/nonfatal reinfarction/urgent revascularization compared with UFH in STEMI patients who receive fibrinolysis. Treatment with enoxaparin is associated with higher rates of serious bleeding, but net clinical benefit—a composite endpoint that combines efficacy and safety—still favors enoxaparin over UFH. Interpretation of the data on fondaparinux is difficult because of the complex nature of the pivotal clinical trial evaluating it in STEMI (OASIS-6). Fondaparinux appears superior to placebo in STEMI patients not receiving reperfusion therapy, but its relative efficacy and safety compared with UFH is less certain. Due to the risk of catheter thrombosis, fondaparinux should not be used alone at the time of coronary angiography and PCI but should be combined with another anticoagulant with antithrombin activity such as UFH or bivalirudin. Contemporary trials of bivalirudin used an open-label design to evaluate its efficacy and safety compared with UFH plus a glycoprotein IIb/IIIa inhibitor. Bivalirudin was associated with a lower rate of bleeding, largely driven by reductions in vascular access site hematomas ≥5 cm or the administration of blood transfusions.
Patients with an anterior location of the infarction, severe LV dysfunction, heart failure, a history of embolism, two-dimensional echocardiographic evidence of mural thrombus, or atrial fibrillation are at increased risk of systemic or pulmonary thromboembolism. Such individuals should receive full therapeutic levels of anticoagulant therapy (LMWH or UFH) while hospitalized, followed by at least 3 months of warfarin therapy.
BETA-ADRENOCEPTOR BLOCKERS
The benefits of beta blockers in patients with STEMI can be divided into those that occur immediately when the drug is given acutely and those that accrue over the long term when the drug is given for secondary prevention after an infarction. Acute intravenous beta blockade improves the myocardial O2 supply-demand relationship, decreases pain, reduces infarct size, and decreases the incidence of serious ventricular arrhythmias. In patients who undergo fibrinolysis soon after the onset of chest pain, no incremental reduction in mortality rate is seen with beta blockers, but recurrent ischemia and reinfarction are reduced.
Thus, beta-blocker therapy after STEMI is useful for most patients (including those treated with an angiotensin-converting enzyme [ACE] inhibitor) except those in whom it is specifically contraindicated (patients with heart failure or severely compromised LV function, heart block, orthostatic hypotension, or a history of asthma) and perhaps those whose excellent long-term prognosis (defined as an expected mortality rate of <1% per year, patients <55 years, no previous MI, with normal ventricular function, no complex ventricular ectopy, and no angina) markedly diminishes any potential benefit.
INHIBITION OF THE RENIN-ANGIOTENSIN-ALDOSTERONE SYSTEM
ACE inhibitors reduce the mortality rate after STEMI, and the mortality benefits are additive to those achieved with aspirin and beta blockers. The maximum benefit is seen in high-risk patients (those who are elderly or who have an anterior infarction, a prior infarction, and/or globally depressed LV function), but evidence suggests that a short-term benefit occurs when ACE inhibitors are prescribed unselectively to all hemodynamically stable patients with STEMI (i.e., those with a systolic pressure >100 mmHg). The mechanism involves a reduction in ventricular remodeling after infarction (see “Ventricular Dysfunction” later) with a subsequent reduction in the risk of CHF. The rate of recurrent infarction may also be lower in patients treated chronically with ACE inhibitors after infarction.
Before hospital discharge, LV function should be assessed with an imaging study. ACE inhibitors should be continued indefinitely in patients who have clinically evident CHF, in patients in whom an imaging study shows a reduction in global LV function or a large regional wall motion abnormality, or in those who are hypertensive.
Angiotensin receptor blockers (ARBs) should be administered to STEMI patients who are intolerant of ACE inhibitors and who have either clinical or radiologic signs of heart failure. Long-term aldosterone blockade should be prescribed for STEMI patients without significant renal dysfunction (creatinine ≥2.5 mg/dL in men and ≥2.0 mg/dL in women) or hyperkalemia (potassium ≥5.0 mEq/L) who are already receiving therapeutic doses of an ACE inhibitor, have an LV ejection fraction ≤40%, and have either symptomatic heart failure or diabetes mellitus. A multidrug regimen for inhibiting the renin-angiotensin-aldosterone system has been shown to reduce both heart failure–related and sudden cardiac death–related cardiovascular mortality after STEMI, but has not been as thoroughly explored as ACE inhibitors in STEMI patients.
OTHER AGENTS
Favorable effects on the ischemic process and ventricular remodeling (see below) previously led many physicians to routinely use intravenous nitroglycerin (5–10 μg/min initial dose and up to 200 μg/min as long as hemodynamic stability is maintained) for the first 24–48 h after the onset of infarction. However, the benefits of routine use of intravenous nitroglycerin are less in the contemporary era where beta-adrenoceptor blockers and ACE inhibitors are routinely prescribed for patients with STEMI.
Results of multiple trials of different calcium antagonists have failed to establish a role for these agents in the treatment of most patients with STEMI. Therefore, the routine use of calcium antagonists cannot be recommended. Strict control of blood glucose in diabetic patients with STEMI has been shown to reduce the mortality rate. Serum magnesium should be measured in all patients on admission, and any demonstrated deficits should be corrected to minimize the risk of arrhythmias.
COMPLICATIONS AND THEIR MANAGEMENT
VENTRICULAR DYSFUNCTION
After STEMI, the left ventricle undergoes a series of changes in shape, size, and thickness in both the infarcted and noninfarcted segments. This process is referred to as ventricular remodeling and generally precedes the development of clinically evident CHF in the months to years after infarction. Soon after STEMI, the left ventricle begins to dilate. Acutely, this results from expansion of the infarct, i.e., slippage of muscle bundles, disruption of normal myocardial cells, and tissue loss within the necrotic zone, resulting in disproportionate thinning and elongation of the infarct zone. Later, lengthening of the noninfarcted segments occurs as well. The overall chamber enlargement that occurs is related to the size and location of the infarct, with greater dilation following infarction of the anterior wall and apex of the left ventricle and causing more marked hemodynamic impairment, more frequent heart failure, and a poorer prognosis. Progressive dilation and its clinical consequences may be ameliorated by therapy with ACE inhibitors and other vasodilators (e.g., nitrates). In patients with an ejection fraction <40%, regardless of whether or not heart failure is present, ACE inhibitors or ARBs should be prescribed (see “Inhibition of the Renin-Angiotensin-Aldosterone System” earlier).
HEMODYNAMIC ASSESSMENT
Pump failure is now the primary cause of in-hospital death from STEMI. The extent of infarction correlates well with the degree of pump failure and with mortality, both early (within 10 days of infarction) and later. The most common clinical signs are pulmonary rales and S3 and S4 gallop sounds. Pulmonary congestion is also frequently seen on the chest roentgenogram. Elevated LV filling pressure and elevated pulmonary artery pressure are the characteristic hemodynamic findings, but these findings may result from a reduction of ventricular compliance (diastolic failure) and/or a reduction of stroke volume with secondary cardiac dilation (systolic failure) (Chap. 279).
A classification originally proposed by Killip divides patients into four groups: class I, no signs of pulmonary or venous congestion; class II, moderate heart failure as evidenced by rales at the lung bases, S3 gallop, tachypnea, or signs of failure of the right side of the heart, including venous and hepatic congestion; class III, severe heart failure, pulmonary edema; and class IV, shock with systolic pressure <90 mmHg and evidence of peripheral vasoconstriction, peripheral cyanosis, mental confusion, and oliguria. When this classification was established in 1967, the expected hospital mortality rate of patients in these classes was as follows: class I, 0–5%; class II, 10–20%; class III, 35–45%; and class IV, 85–95%. With advances in management, the mortality rate in each class has fallen, perhaps by as much as one-third to one-half.
Hemodynamic evidence of abnormal global LV function appears when contraction is seriously impaired in 20–25% of the left ventricle. Infarction of ≥40% of the left ventricle usually results in cardiogenic shock (Chap. 326). Positioning of a balloon flotation (Swan-Ganz) catheter in the pulmonary artery permits monitoring of LV filling pressure; this technique is useful in patients who exhibit hypotension and/or clinical evidence of CHF. Cardiac output can also be determined with a pulmonary artery catheter. With the addition of intra-arterial pressure monitoring, systemic vascular resistance can be calculated as a guide to adjusting vasopressor and vasodilator therapy. Some patients with STEMI have markedly elevated LV filling pressures (>22 mmHg) and normal cardiac indices (2.6–3.6 L/[min/m2]), while others have relatively low LV filling pressures (<15 mmHg) and reduced cardiac indices. The former patients usually benefit from diuresis, while the latter may respond to volume expansion.
HYPOVOLEMIA
This is an easily corrected condition that may contribute to the hypotension and vascular collapse associated with STEMI in some patients. It may be secondary to previous diuretic use, to reduced fluid intake during the early stages of the illness, and/or to vomiting associated with pain or medications. Consequently, hypovolemia should be identified and corrected in patients with STEMI and hypotension before more vigorous forms of therapy are begun. Central venous pressure reflects RV rather than LV filling pressure and is an inadequate guide for adjustment of blood volume, because LV function is almost always affected much more adversely than RV function in patients with STEMI. The optimal LV filling or pulmonary artery wedge pressure may vary considerably among patients. Each patient’s ideal level (generally ∼20 mmHg) is reached by cautious fluid administration during careful monitoring of oxygenation and cardiac output. Eventually, the cardiac output level plateaus, and further increases in LV filling pressure only increase congestive symptoms and decrease systemic oxygenation without raising arterial pressure.
CARDIOGENIC SHOCK
Prompt reperfusion, efforts to reduce infarct size and treatment of ongoing ischemia and other complications of MI appear to have reduced the incidence of cardiogenic shock from 20% to about 7%. Only 10% of patients with this condition present with it on admission, while 90% develop it during hospitalization. Typically, patients who develop cardiogenic shock have severe multivessel coronary artery disease with evidence of “piecemeal” necrosis extending outward from the original infarct zone. The evaluation and management of cardiogenic shock and severe power failure after STEMI are discussed in detail in Chap. 326.
RIGHT VENTRICULAR INFARCTION
Approximately one-third of patients with inferior infarction demonstrate at least a minor degree of RV necrosis. An occasional patient with inferoposterior LV infarction also has extensive RV infarction, and rare patients present with infarction limited primarily to the RV. Clinically significant RV infarction causes signs of severe RV failure (jugular venous distention, Kussmaul’s sign, hepatomegaly [Chap. 267]) with or without hypotension. ST-segment elevations of right-sided precordial ECG leads, particularly lead V4R, are frequently present in the first 24 h in patients with RV infarction. Two-dimensional echocardiography is helpful in determining the degree of RV dysfunction. Catheterization of the right side of the heart often reveals a distinctive hemodynamic pattern resembling constrictive pericarditis (steep right atrial “y” descent and an early diastolic dip and plateau in RV waveforms) (Chap. 288). Therapy consists of volume expansion to maintain adequate RV preload and efforts to improve LV performance with attendant reduction in pulmonary capillary wedge and pulmonary arterial pressures.
ARRHYTHMIAS
(See also Chaps. 274 and 276) The incidence of arrhythmias after STEMI is higher in patients seen early after the onset of symptoms. The mechanisms responsible for infarction-related arrhythmias include autonomic nervous system imbalance, electrolyte disturbances, ischemia, and slowed conduction in zones of ischemic myocardium. An arrhythmia can usually be managed successfully if trained personnel and appropriate equipment are available when it develops. Since most deaths from arrhythmia occur during the first few hours after infarction, the effectiveness of treatment relates directly to the speed with which patients come under medical observation. The prompt management of arrhythmias constitutes a significant advance in the treatment of STEMI.
Ventricular Premature Beats Infrequent, sporadic ventricular premature depolarizations occur in almost all patients with STEMI and do not require therapy. Whereas in the past, frequent, multifocal, or early diastolic ventricular extrasystoles (so-called warning arrhythmias) were routinely treated with antiarrhythmic drugs to reduce the risk of development of ventricular tachycardia and ventricular fibrillation, pharmacologic therapy is now reserved for patients with sustained ventricular arrhythmias. Prophylactic antiarrhythmic therapy (either intravenous lidocaine early or oral agents later) is contraindicated for ventricular premature beats in the absence of clinically important ventricular tachyarrhythmias, because such therapy may actually increase the mortality rate. Beta-adrenoceptor blocking agents are effective in abolishing ventricular ectopic activity in patients with STEMI and in the prevention of ventricular fibrillation. As described earlier (see “Beta-Adrenoceptor Blockers”), they should be used routinely in patients without contraindications. In addition, hypokalemia and hypomagnesemia are risk factors for ventricular fibrillation in patients with STEMI; to reduce the risk, the serum potassium concentration should be adjusted to approximately 4.5 mmol/L and magnesium to about 2.0 mmol/L.
Ventricular Tachycardia and Fibrillation Within the first 24 h of STEMI, ventricular tachycardia and fibrillation can occur without prior warning arrhythmias. The occurrence of ventricular fibrillation can be reduced by prophylactic administration of intravenous lidocaine. However, prophylactic use of lidocaine has not been shown to reduce overall mortality from STEMI. In fact, in addition to causing possible noncardiac complications, lidocaine may predispose to an excess risk of bradycardia and asystole. For these reasons, and with earlier treatment of active ischemia, more frequent use of beta-blocking agents, and the nearly universal success of electrical cardioversion or defibrillation, routine prophylactic antiarrhythmic drug therapy is no longer recommended.
Sustained ventricular tachycardia that is well tolerated hemodynamically should be treated with an intravenous regimen of amiodarone (bolus of 150 mg over 10 min, followed by infusion of 1.0 mg/min for 6 h and then 0.5 mg/min) or procainamide (bolus of 15 mg/kg over 20–30 min; infusion of 1–4 mg/min); if it does not stop promptly, electroversion should be used (Chap. 276). An unsynchronized discharge of 200–300 J (monophasic waveform; approximately 50% of these energies with biphasic waveforms) is used immediately in patients with ventricular fibrillation or when ventricular tachycardia causes hemodynamic deterioration. Ventricular tachycardia or fibrillation that is refractory to electroshock may be more responsive after the patient is treated with epinephrine (1 mg intravenously or 10 mL of a 1:10,000 solution via the intracardiac route) or amiodarone (a 75–150-mg bolus).
Ventricular arrhythmias, including the unusual form of ventricular tachycardia known as torsades des pointes (Chaps. 276 and 277), may occur in patients with STEMI as a consequence of other concurrent problems (such as hypoxia, hypokalemia, or other electrolyte disturbances) or of the toxic effects of an agent being administered to the patient (such as digoxin or quinidine). A search for such secondary causes should always be undertaken.
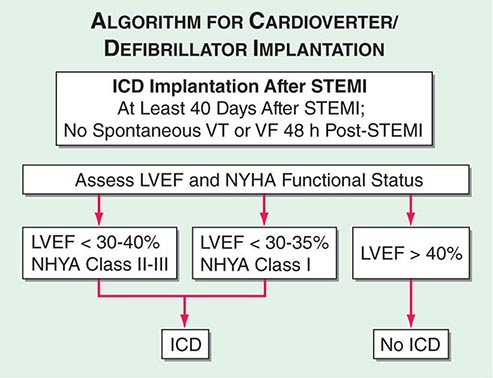
Although the in-hospital mortality rate is increased, the long-term survival is excellent in patients who survive to hospital discharge after primary ventricular fibrillation; i.e., ventricular fibrillation that is a primary response to acute ischemia that occurs during the first 48 h and is not associated with predisposing factors such as CHF, shock, bundle branch block, or ventricular aneurysm. This result is in sharp contrast to the poor prognosis for patients who develop ventricular fibrillation secondary to severe pump failure. For patients who develop ventricular tachycardia or ventricular fibrillation late in their hospital course (i.e., after the first 48 h), the mortality rate is increased both in-hospital and during long-term follow-up. Such patients should be considered for electrophysiologic study and implantation of a cardioverter-defibrillator (ICD) (Chap. 276). A more challenging issue is the prevention of sudden cardiac death from ventricular fibrillation late after STEMI in patients who have not exhibited sustained ventricular tachyarrhythmias during their index hospitalization. An algorithm for selection of patients who warrant prophylactic implantation of an ICD is shown in Fig. 295-5.
FIGURE 295-5 Algorithm for assessment of need for implantation of a cardioverter-defibrillator. The appropriate management is selected based on measurement of left ventricular ejection fraction and assessment of the New York Heart Association (NYHA) functional class. Patients with depressed left ventricular function at least 40 days after ST-segment elevation myocardial infarction (STEMI) are referred for insertion of an implantable cardioverter-defibrillator (ICD) if the left ventricular ejection fraction (LVEF) is <30–40% and they are in NYHA class II–III or if the LVEF is <30–35% and they are in NYHA class I functional status. Patients with preserved left ventricular function (LVEF >40%) do not receive an ICD regardless of NYHA functional class. All patients are treated with medical therapy after STEMI. VF, ventricular fibrillation; VT, ventricular tachycardia. (Adapted from data contained in DP Zipes et al: ACC/AHA/ESC 2006 guidelines for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death; a report of the American College of Cardiology/American Heart Association Task Force and the European Society of Cardiology Committee for Practice Guidelines [Writing Committee to Develop Guidelines for Management of Patients with Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death]. J Am Coll Cardiol 48:1064, 2006.)
Accelerated Idioventricular Rhythm Accelerated idioventricular rhythm (AIVR, “slow ventricular tachycardia”), a ventricular rhythm with a rate of 60–100 beats/min, often occurs transiently during fibrinolytic therapy at the time of reperfusion. For the most part, AIVR, whether it occurs in association with fibrinolytic therapy or spontaneously, is benign and does not presage the development of classic ventricular tachycardia. Most episodes of AIVR do not require treatment if the patient is monitored carefully, as degeneration into a more serious arrhythmia is rare.
Supraventricular Arrhythmias Sinus tachycardia is the most common supraventricular arrhythmia. If it occurs secondary to another cause (such as anemia, fever, heart failure, or a metabolic derangement), the primary problem should be treated first. However, if it appears to be due to sympathetic overstimulation (e.g., as part of a hyperdynamic state), then treatment with a beta blocker is indicated. Other common arrhythmias in this group are atrial flutter and atrial fibrillation, which are often secondary to LV failure. Digoxin is usually the treatment of choice for supraventricular arrhythmias if heart failure is present. If heart failure is absent, beta blockers, verapamil, or diltiazem are suitable alternatives for controlling the ventricular rate, as they may also help to control ischemia. If the abnormal rhythm persists for >2 h with a ventricular rate >120 beats/min, or if tachycardia induces heart failure, shock, or ischemia (as manifested by recurrent pain or ECG changes), a synchronized electroshock (100–200 J monophasic waveform) should be used.
Accelerated junctional rhythms have diverse causes but may occur in patients with inferoposterior infarction. Digitalis excess must be ruled out. In some patients with severely compromised LV function, the loss of appropriately timed atrial systole results in a marked reduction of cardiac output. Right atrial or coronary sinus pacing is indicated in such instances.
Sinus Bradycardia Treatment of sinus bradycardia is indicated if hemodynamic compromise results from the slow heart rate. Atropine is the most useful drug for increasing heart rate and should be given intravenously in doses of 0.5 mg initially. If the rate remains <50–60 beats/min, additional doses of 0.2 mg, up to a total of 2.0 mg, may be given. Persistent bradycardia (<40 beats/min) despite atropine may be treated with electrical pacing. Isoproterenol should be avoided.
Atrioventricular and Intraventricular Conduction Disturbances (See also Chap. 274) Both the in-hospital mortality rate and the postdischarge mortality rate of patients who have complete atrioventricular (AV) block in association with anterior infarction are markedly higher than those of patients who develop AV block with inferior infarction. This difference is related to the fact that heart block in inferior infarction is commonly a result of increased vagal tone and/or the release of adenosine and therefore is transient. In anterior wall infarction, however, heart block is usually related to ischemic malfunction of the conduction system, which is commonly associated with extensive myocardial necrosis.
Temporary electrical pacing provides an effective means of increasing the heart rate of patients with bradycardia due to AV block. However, acceleration of the heart rate may have only a limited impact on prognosis in patients with anterior wall infarction and complete heart block in whom the large size of the infarct is the major factor determining outcome. It should be carried out if it improves hemodynamics. Pacing does appear to be beneficial in patients with inferoposterior infarction who have complete heart block associated with heart failure, hypotension, marked bradycardia, or significant ventricular ectopic activity. A subgroup of these patients, those with RV infarction, often respond poorly to ventricular pacing because of the loss of the atrial contribution to ventricular filling. In such patients, dual-chamber AV sequential pacing may be required.
External noninvasive pacing electrodes should be positioned in a “demand” mode for patients with sinus bradycardia (rate <50 beats/min) that is unresponsive to drug therapy, Mobitz II second-degree AV block, third-degree heart block, or bilateral bundle branch block (e.g., right bundle branch block plus left anterior fascicular block). Retrospective studies suggest that permanent pacing may reduce the long-term risk of sudden death due to bradyarrhythmias in the rare patient who develops combined persistent bifascicular and transient third-degree heart block during the acute phase of MI.
OTHER COMPLICATIONS
Recurrent Chest Discomfort Because recurrent or persistent ischemia often heralds extension of the original infarct or reinfarction in a new myocardial zone and is associated with a near tripling of mortality after STEMI, patients with these symptoms should be referred for prompt coronary arteriography and mechanical revascularization. Administration of a fibrinolytic agent is an alternative to early mechanical revascularization.
Pericarditis (See also Chap. 288) Pericardial friction rubs and/or pericardial pain are frequently encountered in patients with STEMI involving the epicardium. This complication can usually be managed with aspirin (650 mg four times daily). It is important to diagnose the chest pain of pericarditis accurately, because failure to recognize it may lead to the erroneous diagnosis of recurrent ischemic pain and/or infarct extension, with resulting inappropriate use of anticoagulants, nitrates, beta blockers, or coronary arteriography. When it occurs, complaints of pain radiating to either trapezius muscle is helpful, because such a pattern of discomfort is typical of pericarditis but rarely occurs with ischemic discomfort. Anticoagulants potentially could cause tamponade in the presence of acute pericarditis (as manifested by either pain or persistent rub) and therefore should not be used unless there is a compelling indication.
Thromboembolism Clinically apparent thromboembolism complicates STEMI in ∼10% of cases, but embolic lesions are found in 20% of patients in necropsy series, suggesting that thromboembolism is often clinically silent. Thromboembolism is considered to be an important contributing cause of death in 25% of patients with STEMI who die after admission to the hospital. Arterial emboli originate from LV mural thrombi, while most pulmonary emboli arise in the leg veins.
Thromboembolism typically occurs in association with large infarcts (especially anterior), CHF, and an LV thrombus detected by echocardiography. The incidence of arterial embolism from a clot originating in the ventricle at the site of an infarction is small but real. Two-dimensional echocardiography reveals LV thrombi in about one-third of patients with anterior wall infarction but in few patients with inferior or posterior infarction. Arterial embolism often presents as a major complication, such as hemiparesis when the cerebral circulation is involved or hypertension if the renal circulation is compromised. When a thrombus has been clearly demonstrated by echocardiographic or other techniques or when a large area of regional wall motion abnormality is seen even in the absence of a detectable mural thrombus, systemic anticoagulation should be undertaken (in the absence of contraindications), as the incidence of embolic complications appears to be markedly lowered by such therapy. The appropriate duration of therapy is unknown, but 3–6 months is probably prudent.
Left Ventricular Aneurysm The term ventricular aneurysm is usually used to describe dyskinesis or local expansile paradoxical wall motion. Normally functioning myocardial fibers must shorten more if stroke volume and cardiac output are to be maintained in patients with ventricular aneurysm; if they cannot, overall ventricular function is impaired. True aneurysms are composed of scar tissue and neither predispose to nor are associated with cardiac rupture.
The complications of LV aneurysm do not usually occur for weeks to months after STEMI; they include CHF, arterial embolism, and ventricular arrhythmias. Apical aneurysms are the most common and the most easily detected by clinical examination. The physical finding of greatest value is a double, diffuse, or displaced apical impulse. Ventricular aneurysms are readily detected by two-dimensional echocardiography, which may also reveal a mural thrombus in an aneurysm.
Rarely, myocardial rupture may be contained by a local area of pericardium, along with organizing thrombus and hematoma. Over time, this pseudoaneurysm enlarges, maintaining communication with the LV cavity through a narrow neck. Because a pseudoaneurysm often ruptures spontaneously, it should be surgically repaired if recognized.
POSTINFARCTION RISK STRATIFICATION AND MANAGEMENT
Many clinical and laboratory factors have been identified that are associated with an increase in cardiovascular risk after initial recovery from STEMI. Some of the most important factors include persistent ischemia (spontaneous or provoked), depressed LV ejection fraction (<40%), rales above the lung bases on physical examination or congestion on chest radiograph, and symptomatic ventricular arrhythmias. Other features associated with increased risk include a history of previous MI, age >75, diabetes mellitus, prolonged sinus tachycardia, hypotension, ST-segment changes at rest without angina (“silent ischemia”), an abnormal signal-averaged ECG, nonpatency of the infarct-related coronary artery (if angiography is undertaken), and persistent advanced heart block or a new intraventricular conduction abnormality on the ECG. Therapy must be individualized on the basis of the relative importance of the risk(s) present.
The goal of preventing reinfarction and death after recovery from STEMI has led to strategies to evaluate risk after infarction. In stable patients, submaximal exercise stress testing may be carried out before hospital discharge to detect residual ischemia and ventricular ectopy and to provide the patient with a guideline for exercise in the early recovery period. Alternatively, or in addition, a maximal (symptom-limited) exercise stress test may be carried out 4–6 weeks after infarction. Evaluation of LV function is usually warranted as well. Recognition of a depressed LV ejection fraction by echocardiography or radionuclide ventriculography identifies patients who should receive medications to inhibit the renin-angiotensin-aldosterone system. Patients in whom angina is induced at relatively low workloads, those who have a large reversible defect on perfusion imaging or a depressed ejection fraction, those with demonstrable ischemia, and those in whom exercise provokes symptomatic ventricular arrhythmias should be considered at high risk for recurrent MI or death from arrhythmia (Fig. 295-5). Cardiac catheterization with coronary angiography and/or invasive electrophysiologic evaluation is advised.
Exercise tests also aid in formulating an individualized exercise prescription, which can be much more vigorous in patients who tolerate exercise without any of the previously mentioned adverse signs. In addition, predischarge stress testing may provide an important psychological benefit, building the patient’s confidence by demonstrating a reasonable exercise tolerance.
In many hospitals, a cardiac rehabilitation program with progressive exercise is initiated in the hospital and continued after discharge. Ideally, such programs should include an educational component that informs patients about their disease and its risk factors.
The usual duration of hospitalization for an uncomplicated STEMI is about 5 days. The remainder of the convalescent phase may be accomplished at home. During the first 1–2 weeks, the patient should be encouraged to increase activity by walking about the house and outdoors in good weather. Normal sexual activity may be resumed during this period. After 2 weeks, the physician must regulate the patient’s activity on the basis of exercise tolerance. Most patients will be able to return to work within 2–4 weeks.
SECONDARY PREVENTION
Various secondary preventive measures are at least partly responsible for the improvement in the long-term mortality and morbidity rates after STEMI. Long-term treatment with an antiplatelet agent (usually aspirin) after STEMI is associated with a 25% reduction in the risk of recurrent infarction, stroke, or cardiovascular mortality (36 fewer events for every 1000 patients treated). An alternative antiplatelet agent that may be used for secondary prevention in patients intolerant of aspirin is clopidogrel (75 mg orally daily). ACE inhibitors or ARBs and, in appropriate patients, aldosterone antagonists should be used indefinitely by patients with clinically evident heart failure, a moderate decrease in global ejection fraction, or a large regional wall motion abnormality to prevent late ventricular remodeling and recurrent ischemic events.
The chronic routine use of oral beta-adrenoceptor blockers for at least 2 years after STEMI is supported by well-conducted, placebo-controlled trials.
Evidence suggests that warfarin lowers the risk of late mortality and the incidence of reinfarction after STEMI. Most physicians prescribe aspirin routinely for all patients without contraindications and add warfarin for patients at increased risk of embolism (see “Thromboembolism” earlier). Several studies suggest that in patients <75 years old a low dose of aspirin (75–81 mg/d) in combination with warfarin administered to achieve an international normalized ratio >2.0 is more effective than aspirin alone for preventing recurrent MI and embolic cerebrovascular accident. However, there is an increased risk of bleeding and a high rate of discontinuation of warfarin that has limited clinical acceptance of combination antithrombotic therapy. There is increased risk of bleeding when warfarin is added to dual antiplatelet therapy (aspirin and clopidogrel). However, patients who have had a stent implanted and have an indication for anticoagulation should receive dual antiplatelet therapies in combination with warfarin. Such patients should also receive a proton pump inhibitor to minimize the risk of gastrointestinal bleeding and should have regular monitoring of their hemoglobin levels and stool hematest while on combination antithrombotic therapy.
Finally, risk factors for atherosclerosis (Chap. 265e) should be discussed with the patient and, when possible, favorably modified.
296e | Percutaneous Coronary Interventions and Other Interventional Procedures |
Percutaneous transluminal coronary angioplasty (PTCA) was first introduced by Andreas Gruentzig in 1977 as an alternative to coronary bypass surgery. The concept was initially demonstrated by Charles Dotter in 1964 in peripheral vessels. The development of a small inelastic balloon catheter by Gruentzig allowed expansion of the technique into smaller peripheral and coronary vessels. Initial coronary experience was limited to single-vessel coronary disease and discrete proximal lesions due to the technical limitations of the equipment. Advances in technology and greater operator experience allowed the procedure to grow rapidly with expanded use in patients with more complex lesions and multivessel disease. The introduction of coronary stents in 1994 was one of the major advances in the field. These devices reduced acute complications and reduced by half the significant problem of restenosis (or recurrence of the stenosis). Further reductions in restenosis were achieved by the introduction of drug-eluting stents in 2003. These stents slowly release antiproliferative drugs directly into the plaque over a few months. Percutaneous coronary intervention (PCI) is the most common revascularization procedure in the United States and is performed more than twice as often as coronary artery bypass surgery: nearly 600,000 patients a year.
Interventional cardiology is a separate discipline in cardiology that requires a dedicated 1-year interventional cardiology fellowship following a 3-year general cardiology fellowship in order to obtain a separate board certification. The discipline has also expanded to include interventions for structural heart disease including treatment of congenital heart disease and valvular heart disease; it also includes interventions to treat peripheral vascular disease, including atherosclerotic and nonatherosclerotic lesions in the carotid, renal, aortic, and peripheral circulations.
TECHNIQUE
The initial procedure is performed in a similar manner as a diagnostic cardiac catheterization (Chap. 272). Arterial access is obtained via the femoral or radial artery. To prevent thrombotic complications during the procedure, patients who are anticipated to need an angioplasty are given aspirin (325 mg) and may be given clopidogrel (loading dose of 300–600 mg), prasugrel (loading dose of 60 mg), or ticagrelor (loading dose of 180 mg) before the procedure. During the procedure, anticoagulation is achieved by administration of unfractionated heparin, enoxaparin (a low-molecular-weight heparin), or bivalirudin (a direct thrombin inhibitor). The latter has gained popularity due to the significant reduction in bleeding complications. In patients with ST-elevation myocardial infarction, high-risk acute coronary syndrome, or a large thrombus in the coronary artery, an intravenous glycoprotein IIb/IIIa inhibitor (abciximab, tirofiban, or eptifibatide) may also be given.
Following placement of an introducing sheath, preformed guiding catheters are used to cannulate selectively the origins of the coronary arteries. Through the guiding catheter, a flexible, steerable guidewire is negotiated down the coronary artery lumen using fluoroscopic guidance; it is then advanced through the stenosis and into the vessel beyond. This guidewire then serves as a “rail” over which angioplasty balloons, stents, or other therapeutic devices can be advanced to enlarge the narrowed segment of coronary artery. The artery is usually dilated with a balloon catheter followed by placement of a stent. The catheters and introducing sheath are removed and the artery manually held or closed using one of several femoral arterial closure devices to achieve hemostasis. Because PCI is performed under local anesthesia and mild sedation, it requires only a short (1-day) hospitalization or less.
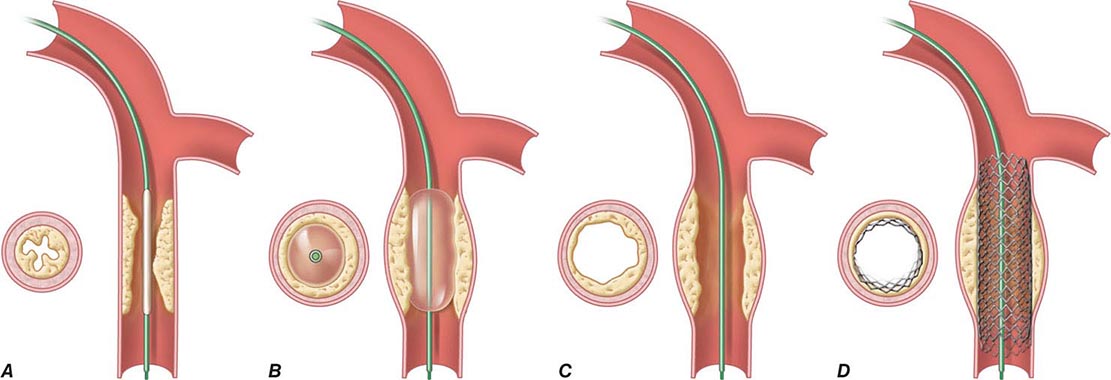
Angioplasty works by stretching the artery and displacing the plaque away from the lumen, enlarging the entire vessel (Figs. 296e-1 and 296e-2). The procedure rarely results in embolization of atherosclerotic material. Owing to inelastic elements in the plaque, the stretching of the vessel by the balloon results in small localized dissections that can protrude into the lumen and be a nidus for acute thrombus formation. If the dissections are severe, then they can obstruct the lumen or induce a thrombotic occlusion of the artery (acute closure). Stents have largely prevented this complication by holding the dissection flaps up against the vessel wall (Fig. 296e-1).
FIGURE 296e-1 Schematic diagram of the primary mechanisms of balloon angioplasty and stenting. A. A balloon angioplasty catheter is positioned into the stenosis over a guidewire under fluoroscopic guidance. B. The balloon is inflated temporarily occluding the vessel. C. The lumen is enlarged primarily by stretching the vessel, often resulting in small dissections in the neointima. D. A stent mounted on a deflated balloon is placed into the lesion and pressed against the vessel wall with balloon inflation (not shown). The balloon is deflated and removed, leaving the stent permanently against the wall acting as a scaffold to hold the dissections against the wall and prevent vessel recoil. (Adapted from EJ Topol: Textbook of Cardiovascular Medicine, 2nd ed. Philadelphia, Lippincott Williams & Wilkins, 2002.)
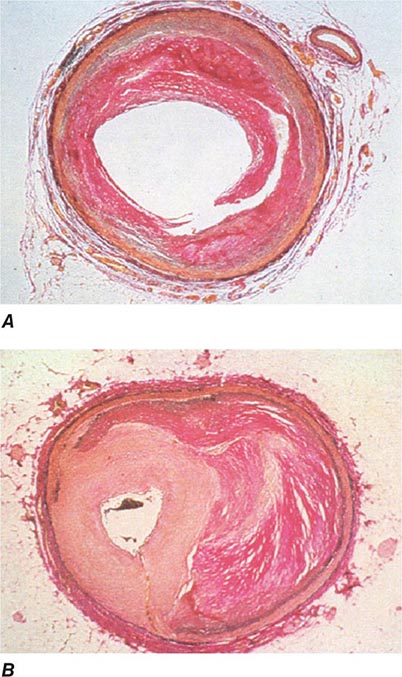
FIGURE 296e-2 Pathology of acute effects of balloon angioplasty with intimal dissection and vessel stretching (A) and an example of neointimal hyperplasia and restenosis showing renarrowing of the vessel (B). (Panel A from M Ueda et al: Eur Heart J 12:937, 1991; with permission. Panel B from CE Essed et al: Br Heart J 49:393, 1983; with permission.)
Stents are currently used in more than 90% of coronary angioplasty procedures. Stents are wire meshes (usually made of stainless steel) that are compressed over a deflated angioplasty balloon. When the balloon is inflated, the stent is enlarged to approximate the “normal” vessel lumen. The balloon is then deflated and removed, leaving the stent behind to provide a permanent scaffold in the artery. Owing to the design of the struts, these devices are flexible, allowing their passage through diseased and tortuous coronary vessels. Stents are rigid enough to prevent elastic recoil of the vessel and have dramatically improved the success and safety of the procedure as a result.
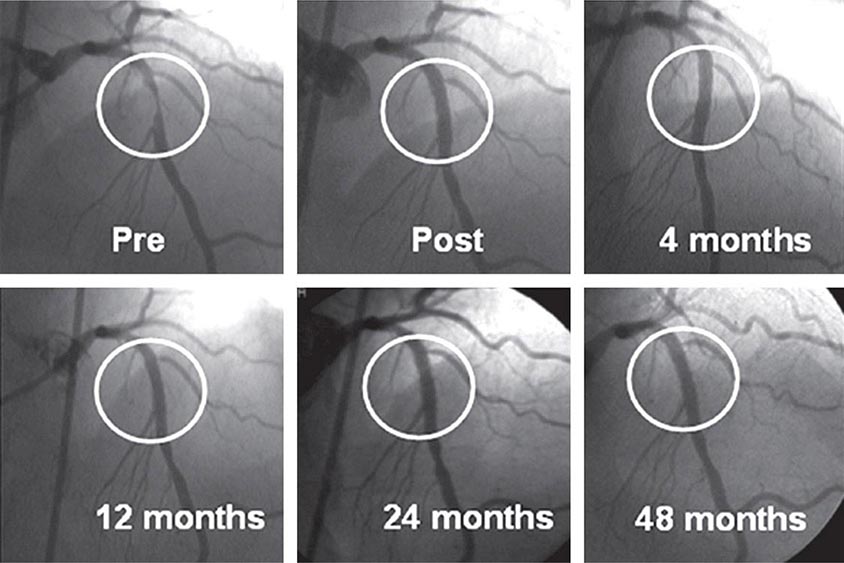
Drug-eluting stents further enhanced the efficacy of PCI. An antiproliferative agent is attached to the metal stent by use of a thin polymer coating. The antiproliferative drug elutes from the stent over a 1- to 3-month period after implantation. Drug-eluting stents have been shown to reduce clinical restenosis by 50%, so that in uncomplicated lesions symptomatic restenosis occurs in 5–10% of patients. Not surprisingly, this led to the rapid acceptance of these devices; currently 80–90% of all stents implanted are drug-eluting. The first-generation devices were coated with either sirolimus or paclitaxel. Second-generation drug-eluting stents use newer agents such as everolimus, biolimus, and zotarolimus. These second-generation drug-eluting stents appear to be more effective with fewer complications, such as early or late stent thrombosis, than the first-generation devices and, therefore, have replaced the first-generation stents. Biodegradable polymers that are used to attach the drugs to the stents may be superior to permanent polymers in preventing late stent thrombosis and are under investigation. In addition, the everolimus-eluting biodegradable vascular scaffold (BVS) stent has been shown to be safe and effective with gradual degradation over several years with return of normal vessel function. It is currently approved in Europe. Additional stents are under investigation. Other interventional devices include atherectomy devices and thrombectomy catheters. These devices are designed to remove atherosclerotic plaque or thrombus and are used in conjunction with balloon dilatation and stent placement. Rotational atherectomy is the most commonly used adjunctive device and is modeled after a dentist’s drill, with small round burrs of 1.25–2.5 mm at the tip of a flexible wire shaft. They are passed over the guidewire up to the stenosis and drill away atherosclerotic material. Because the atherosclerotic particles are ≤25 μm, they pass through the coronary microcirculation and rarely cause problems. The device is particularly useful in heavily calcified plaques that are resistant to balloon dilatation. Given the current advances in stents, rotational atherectomy is infrequently used. Directional atherectomy catheters are not used in the coronaries any longer but are used in peripheral arterial disease. In acute ST-elevation myocardial infarction, specialized catheters without a balloon are used to aspirate thrombus in order to prevent embolization down the coronary vessel and to improve blood flow before angioplasty and stent placement. Some data suggest that manual catheter thrombus aspiration may reduce mortality in addition to improving blood flow in primary PCI.
PCI of degenerated saphenous vein graft lesions has been associated with a significant incidence of distal embolization of atherosclerotic material, unlike PCI of native vessel disease. A number of distal protection devices have been shown to significantly reduce embolization and myocardial infarction in this setting. Most devices work by using a collapsible wire filter at the end of a guidewire that is expanded in the distal vessel before PCI. If atherosclerotic debris is dislodged, the basket captures the material, and at the end of the PCI, the basket is pulled into a delivery catheter and the debris safely removed from the patient.
SUCCESS AND COMPLICATIONS
A successful procedure (angiographic success), defined as a reduction of the stenosis to less than a 20% diameter narrowing, occurs in 95–99% of patients. Lower success rates are seen in patients with tortuous, small, or calcified vessels or chronic total occlusions. Chronic total occlusions have the lowest success rates (60–70%), and their recanalization is usually not attempted unless the occlusion is recent (within 3 months) or there are favorable anatomic features. Improvements in equipment and technique have increased the success rates of recanalization of chronic total occlusions.
Serious complications are rare but include a mortality rate of 0.1–0.3% for elective cases, a large myocardial infarction in less than 3%, and stroke in less than 0.1%. Patients who are elderly (>65 years), undergoing an emergent or urgent procedure, have chronic kidney disease, present with an ST-segment elevation myocardial infarction (STEMI), or are in shock have significantly higher risk. Scoring systems can help to estimate the risk of the procedure. Myocardial infarction during PCI can occur for multiple reasons including an acute occluding thrombus, severe coronary dissection, embolization of thrombus or atherosclerotic material, or closure of a side branch vessel at the site of angioplasty. Most myocardial infarctions are small and only detected by a rise in the creatine phosphokinase (CPK) or troponin level after the procedure. Only those with significant enzyme elevations (more than three to five times the upper limit of normal) are associated with a less favorable long-term outcome. Coronary stents have largely prevented coronary dissections due to the scaffolding effect of the stent.
Metallic stents are also prone to stent thrombosis (1–3%), either acute (<24 h) or subacute (1–30 days), which can be ameliorated by greater attention to full initial stent deployment and the use of dual antiplatelet therapy (DAPT) (aspirin, plus a platelet P2Y12 receptor blocker [clopidogrel, prasugrel, or ticagrelor]). Late (30 days–1 year) and very late stent thromboses (>1 year) occur very infrequently with stents but are slightly more common with first-generation drug-eluting stents, necessitating DAPT for up to 1 year or longer. Use of the second-generation stents is associated with lower rates of late and very late stent thromboses, and shorter durations of DAPT may be possible. Premature discontinuation of DAPT, particularly in the first month after implantation, is associated with a significantly increased risk for stent thrombosis (three- to ninefold greater). Stent thrombosis results in death in 10–20% and myocardial infarction in 30–70% of patients. Elective surgery that requires discontinuation of antiplatelet therapy after drug-eluting stent implantation should be postponed until after 6 months and preferably after 1 year, if at all possible.
Restenosis, or renarrowing of the dilated coronary stenosis, is the most common complication of angioplasty and occurs in 20–50% of patients with balloon angioplasty alone, 10–30% of patients with bare metal stents, and 5–15% of patients with drug-eluting stents within the first year. The fact that stent placement provides a larger acute luminal area than balloon angioplasty alone reduces the incidence of subsequent restenosis. Drug-eluting stents further reduce restenosis through a reduction in excessive neointimal growth over the stent. If restenosis does not occur, the long-term outcome is excellent (Fig. 296e-3). Clinical restenosis is recognized by recurrence of angina or symptoms within 9 months of the procedure. Less frequently, patients with restenosis can present with non-ST-segment elevation myocardial infarction (NSTEMI) (10%) or STEMI (2%) as well. Clinical restenosis requires confirmation of a significant stenosis at the site of the prior PCI. Target lesion revascularization (TLR) or target vessel revascularization (TVR) is defined as angiographic restenosis with repeat PCI or coronary artery bypass grafting (CABG). By angiography, the incidence of restenosis is significantly higher than clinical restenosis (TLR or TVR) because many patients have mild restenosis that does not result in a recurrence of symptoms. The management of clinical restenosis is usually to repeat the PCI with balloon dilatation and placement of a drug-eluting stent. Once a patient has had restenosis, the risk of a second restenosis is further increased. The risk factors for restenosis are diabetes, myocardial infarction, long lesions, small-diameter vessels, and suboptimal initial PCI result.
FIGURE 296e-3 Long-term results from one of the first patients to receive a sirolimus-eluting stent from early Sao Paulo experience. (From: GW Stone, in D Baim [ed]: Cardiac Catheterization, Angiography and Intervention, 7th ed. Philadelphia, Lippincott Williams & Wilkins, 2006; with permission.)
INDICATIONS
The American College of Cardiology (ACC)/American Heart Association (AHA) guidelines extensively review the indications for PCI in patients with stable angina, unstable angina, NSTEMI, and STEMI and should be referred to for a comprehensive discussion of the indications. Briefly, the two principal indications for coronary revascularization in patients with chronic stable angina (Chap. 293) are (1) to improve anginal symptoms in patients who remain symptomatic despite adequate medical therapy and (2) to reduce mortality rates in patients with severe coronary disease. In patients with stable angina who are well controlled on medical therapy, studies such as the Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation (COURAGE) and Bypass Angioplasty Revascularization Investigation 2 Diabetes (BARI 2D) trials have shown that initial revascularization does not lead to better outcomes and can be safely delayed until symptoms worsen or evidence of severe ischemia on noninvasive testing occurs. When revascularization is indicated, the choice of PCI or CABG depends on a number of clinical and anatomic factors (Fig. 296e-4). The Synergy between Percutaneous Coronary Intervention with Taxus and Cardiac Surgery (SYNTAX) trial compared PCI with the paclitaxel drug-eluting stent to CABG in 1800 patients with three-vessel coronary disease or left main disease. The study found no difference in death or myocardial infarction at 1 year, but repeat revascularization was significantly higher in the stent-treated group (13.5 vs. 5.9%), while stroke was significantly higher in the surgical group (2.2 vs. 0.6%). The primary endpoint of death, myocardial infarction, stroke, or revascularization was significantly better with CABG, particularly in those with the most extensive coronary artery disease. The 3-year results confirm these findings. The Future Revascularization Evaluation in Patients With Diabetes Mellitus: Optimal Management of Multivessel Disease (FREEDOM) trial randomized 1900 patients with diabetes and multivessel disease and showed a significantly lower primary endpoint of death, myocardial infarction, or stroke with CABG than PCI. These studies support CABG for those with the most severe left main and three-vessel disease or those with diabetes. Lesser degrees of multivessel disease in patients with or without diabetes have an equal outcome with PCI.
FIGURE 296e-4 In patients requiring revascularization, several factors need to be considered in choosing between bare metal stents, drug-eluting stents, or coronary artery bypass surgery. ACS, acute coronary syndrome; BMS, bare metal stent; CABG, coronary artery bypass grafting; DES, drug-eluting stent; IVUS, intravascular ultrasound; STEMI, ST-segment elevation myocardial infarction. (From AA Bavry, DL Bhatt: Circulation 116:696, 2007; with permission.)
The choice of PCI versus CABG is also related to the anticipated procedural success and complications of PCI and the risks of CABG. For PCI, the characteristics of the coronary anatomy are critically important. The location of the lesion in the vessel (proximal or distal), the degree of tortuosity, and the size of the vessel are considered. In addition, the lesion characteristics, including the degree of the stenosis, the presence of calcium, lesion length, and presence of thrombus, are assessed. The most common reason to decide not to do PCI is that the lesion felt to be responsible for the patient’s symptoms is not treatable. This is most commonly due to the presence of a chronic total occlusion (>3 months in duration). In this setting, the historical success rate has been low (30–70%) and complications are more common. A lesion classification to characterize the likelihood of success or failure of PCI has been developed by the ACC/AHA. Lesions with the highest success are called type A lesions (such as proximal noncalcified subtotal lesions), and those with the lowest success or highest complication rate are type C lesions (such as chronic total occlusions). Intermediate lesions are classified as type B1 or B2 depending on the number of unfavorable characteristics. Approximately 25–30% of patients will not be candidates for PCI due to unfavorable anatomy, whereas only 5% of CABG patients will not be candidates for surgery due to coronary anatomy. The primary reason for being considered inoperable with CABG is the presence of severe comorbidities such as advanced age, frailty, severe chronic obstructive pulmonary disease (COPD), or poor left ventricular function.
Another consideration in choosing a revascularization strategy is the degree of revascularization. In patients with multivessel disease, bypass grafts can usually be placed to all vessels with significant stenosis, whereas PCI may be able to treat only some of the lesions due to the presence of unfavorable anatomy. Assessment of the significance of intermediate lesions using fractional flow reserve (FFR) (Chap. 272) can assist in determining which lesions should be revascularized. The Fractional Flow Reserve versus Angiography for Multivessel Evaluation (FAME) trial showed a 30% reduction in adverse events when revascularization by PCI was restricted to those lesions that were hemodynamically significant (FFR ≤0.80) rather than when guided by angiography alone. Thus, complete revascularization of all functionally significant lesions should be favored and considered when choosing the optimal revascularization strategy. Given the multiple factors that need to be considered in choosing the best revascularization for an individual patient with multivessel disease, it is optimal to have a discussion among the cardiac surgeon, interventional cardiologist, and the physicians caring for the patient (so-called Heart Team) to properly weigh the choices.
Patients with acute coronary syndrome are at excess risk of short- and long-term mortality. Randomized clinical trials have shown that PCI is superior to intensive medical therapy in reducing mortality and myocardial infarction, with the benefit largely confined to those patients who are high risk. High-risk patients are defined as those with any one of the following: refractory ischemia, recurrent angina, positive cardiac-specific enzymes, new ST-segment depression, low ejection fraction, severe arrhythmias, or a recent PCI or CABG. PCI is preferred over surgical therapy in most high-risk patients with acute coronary syndromes unless they have severe multivessel disease or the culprit lesion responsible for the unstable presentation cannot be adequately treated. In STEMI, thrombolysis or PCI (primary PCI) are effective methods to restore coronary blood flow and salvage myocardium within the first 12 h after onset of chest pain. Because PCI is more effective in restoring flow than thrombolysis, it is preferred if readily available. PCI is also performed following thrombolysis to facilitate adequate reperfusion or as a rescue procedure in those who do not achieve reperfusion from thrombolysis or in those who develop cardiogenic shock.
OTHER INTERVENTIONAL TECHNIQUES
STRUCTURAL HEART DISEASE
Interventional treatment for structural heart disease (adult congenital heart disease and valvular heart disease) is a significant and growing component of the field of interventional cardiology.
The most common adult congenital lesion to be treated with percutaneous techniques is closure of atrial septal defects (Chap. 282). The procedure is done as in a diagnostic right heart catheterization with the passage of a catheter up the femoral vein into the right atrium. With echo and fluoroscopic guidance, the size and location of the defect can be accurately defined, and closure is accomplished using one of several approved devices. All devices use a left atrial and right atrial wire mesh or covered disk that are pulled together to capture the atrial septum around the defect and seal it off. The Amplatzer Septal Occluder device (AGA Medical, Minneapolis, Minnesota) is the most commonly used in the United States. The success rate in selected patients is 85–95%, and the device complications are rare and include device embolization, infection, or erosion. Closure of patent foramen ovale (PFO) is done in a similar way. PFO closure may be considered in patients who have had recurrent paradoxical stroke or transient ischemic attack (TIA) despite adequate medical therapy including anticoagulation or antiplatelet therapy. The benefit, however, has not been proven. The CLOSURE I trial randomized 909 patients with cryptogenic stroke or TIA who had a PFO. Closure did not reduce the primary endpoint of death within 30 days or death following a neurologic cause during 2 years of follow-up or stroke/TIA within 2 years. Other trials have confirmed these findings. The use in the treatment of migraine is under clinical investigation and is not an approved indication.
Similar devices can also be used to close patent ductus arteriosus and ventricular septal defects. Other congenital diseases that can be treated percutaneously include coarctation of the aorta, pulmonic stenosis, peripheral pulmonary stenosis, and other abnormal communications between the cardiac chambers or vessels.
The treatment of valvular heart disease is the most rapidly growing area in interventional cardiology. Until recently, the only available techniques were balloon valvuloplasty for the treatment of aortic, mitral, or pulmonic stenosis (Chap. 283). Mitral valvuloplasty is the preferred treatment for symptomatic patients with rheumatic mitral stenosis who have favorable anatomy. The outcome in these patients is equal to that of surgical commissurotomy. The success is highly related to the echocardiographic appearance of the valve. The most favorable setting is commissural fusion without calcification or subchordal fusion and the absence of significant mitral regurgitation. Access is obtained from the femoral vein using a transseptal technique in which a long metal catheter with a needle tip is advanced from the femoral vein through the right atrium and atrial septum at the level of the foramen ovale into the left atrium. A guidewire is advanced into the left ventricle, and a balloon-dilatation catheter is negotiated across the mitral valve and inflated to a predetermined size to enlarge the valve. The most commonly used dilatation catheter is the Inoue balloon. The technique splits the commissural fusion and commonly results in a doubling of the mitral valve area. The success of the procedure in favorable anatomy is 95% and severe complications are rare (1–2%). The most common complications are tamponade due to puncture into the pericardium or the creation of severe mitral regurgitation.
For regurgitant valvular lesions, only severe mitral regurgitation can be effectively treated percutaneously using the MitraClip (Abbott, Abbott Park, Ill) device. The procedure involves the passage of a catheter into the left atrium using the transseptal technique. A special catheter with a metallic clip on the end is passed through the mitral valve and retracted to catch and clip together the mid portion of the anterior and posterior mitral valve leaflet. The clip creates a double opening in the mitral valve and thereby reduces mitral regurgitation similar to the surgical Alfieri repair. In the Endovascular Valve Edge-to-Edge Repair Study (EVEREST II) trial, the device was less effective than surgical repair or replacement but was shown to be safe. It is currently used for patients who are not good candidates for surgical repair, particularly when the regurgitation is due to functional causes.
Severe aortic stenosis can be treated with balloon valvuloplasty as well. In this setting, the valvuloplasty balloon catheter is placed retrograde across the aortic valve from the femoral artery and briefly inflated to stretch open the valve. The success is much less favorable, with only 50% achieving an aortic valve area of >1 cm2 and a restenosis rate of 25–50% after 6–12 months. This poor success rate has limited its use to patients who are not surgical candidates or as a bridge to surgery or transcatheter aortic valve replacement (TAVR). In this setting, the intermediate-term mortality rate of the procedure is high (10%). Repeat aortic valvuloplasty as a treatment for aortic valve restenosis has been reported.
Percutaneous aortic valve replacement (TAVR) has been shown to be an effective treatment for high-risk and inoperable patients with aortic stenosis. Currently, two valve models, the Edwards SAPIEN valve (Edwards Lifescience, Irvine, California) and the CoreValve ReValving system (Medtronic, Minneapolis, Minnesota) are available. In more than 10,000 cases worldwide, follow-up shows no evidence of restenosis or severe prosthetic valve dysfunction in the midterm. The CoreValve is self-expanding, while the Edwards valve is balloon expanded. The cannulas are large (14–22 French), and retrograde access via the femoral artery is most commonly chosen, if possible. In patients with peripheral artery disease, access via the subclavian artery or transapically through a surgical incision can be used. Following balloon valvuloplasty, the valve is positioned across the valve and deployed with postdeployment balloon inflation to ensure full contact with the aortic annulus. The success rate is 80–90%, and the 30-day mortality rate is 10–15%, not unexpectedly as only high-risk patients are undergoing the procedure currently. The Placement of Aortic Transcatheter Valve (PARTNER) randomized trial of the Edwards valve showed a 55% reduction in 1-year mortality and major adverse events in the extreme-risk group randomized to TAVR compared to medical therapy. In a separate randomized trial, high-risk patients had a similar outcome to surgical valve replacement at 1 year. As a result, this valve is approved for both high-risk and extreme-risk patients with severe aortic stenosis.
Pulmonic stenosis can also be effectively treated with balloon valvuloplasty and percutaneously replaced with the Melody stent (Medtronic). Tricuspid valve interventions remain experimental.
PERIPHERAL ARTERY INTERVENTIONS
The use of percutaneous interventions to treat symptomatic patients with arterial obstruction in the carotid, renal, aortic, and peripheral vessels is also part of the field of interventional cardiology. Randomized clinical trial data already support the use of carotid stenting in patients at high risk of complications from carotid endarterectomy (Fig. 296e-5). Recent trials suggest similar outcomes with carotid stenting and carotid endarterectomy in patients at average risk, although depending on the patient’s risk for periprocedural stroke or myocardial infarction, one procedure may be preferred over the other. The success rate of peripheral artery interventional procedures has been improving, including for long segments of occlusive disease historically treated by peripheral bypass surgery (Fig. 296e-6). Peripheral intervention is increasingly part of the training of an interventional cardiologist, and most programs now require an additional year of training after the interventional cardiology training year. The techniques and outcomes are described in detail in the chapter on peripheral vascular disease (Chap. 302).
FIGURE 296e-5 An example of a high-risk patient who requires carotid revascularization, but who is not a candidate for carotid endarterectomy. Carotid artery stenting resulted in an excellent angiographic result. (From M Belkin, DL Bhatt: Circulation 119:2302, 2009; with permission.)
FIGURE 296e-6 Peripheral interventional procedures have become highly effective at treating anatomic lesions previously amenable only to bypass surgery. A. Complete occlusion of the left superficial femoral artery. B. Wire and catheter advanced into subintimal space. C. Intravascular ultrasound positioned in the subintimal space to guide retrograde wire placement through the occluded vessel. D. Balloon dilation of the occlusion. E. Stent placement with excellent angiographic result. (From A Al Mahameed, DL Bhatt: Cleve Clin J Med 73:S45, 2006; with permission.)
CIRCULATORY SUPPORT TECHNIQUES
The use of circulatory support techniques is occasionally needed in order to safely perform PCI on hemodynamically unstable patients. It also can be useful in helping to stabilize patients before surgical interventions. The most commonly used device is the percutaneous intraaortic balloon pump developed in the early 1960s. A 7- to 10-French, 25- to 50-mL balloon catheter is placed retrograde from the femoral artery into the descending aorta between the aortic arch and the abdominal aortic bifurcation. It is connected to a helium gas inflation system that synchronizes the inflation to coincide with early diastole with deflation by mid-diastole. As a result, it increases early diastolic pressure, lowers systolic pressure, and lowers late diastolic pressure through displacement of blood from the descending aorta (counterpulsation). This results in an increase in coronary blood flow and a decrease in afterload. It is contraindicated in patients with aortic regurgitation, aortic dissection, or severe peripheral artery disease. The major complications are vascular and thrombotic. Intravenous heparin is given in order to reduce thrombotic complications.
Another potentially useful tool is the Impella device (Abiomed, Danvers, Massachusetts). The catheter is placed percutaneously from the femoral artery into the left ventricle. The catheter has a small microaxial pump at its tip that can pump up to 2.5–5 L/min from the left ventricle to the aorta. Other support devices include TandemHeart (CardiacAssist, Pittsburgh, Pennsylvania), which involves placement of a large 21-French catheter from the femoral vein through the right atrium into the left atrium using the transseptal technique and a catheter in the femoral artery. A centrifugal pump can deliver 5 L of blood per minute. It may be useful in patients in shock or with STEMI or very-high-risk PCI. Patients can also be placed on peripheral extracorporeal membrane oxygenation using large cannulas placed in the femoral artery and vein.
INTERVENTIONS FOR PULMONARY EMBOLISM
The treatment of deep vein thrombosis is intravenous anticoagulation, with placement of an inferior vena cava filter if recurrent pulmonary emboli occur. Postphlebitic syndrome is a serious condition due to chronic venous obstruction that can lead to chronic leg edema and venous ulcers. Preliminary studies suggest that mechanical treatments may have a role in treatment, and a large trial is ongoing.
Pulmonary emboli (PE) should be treated with fibrinolytic agents if massive and in some cases if submassive. Surgical pulmonary embolectomy is an option for the treatment of massive PE with hemodynamic instability in patients who have contraindications for systemic fibrinolysis or those in whom it has failed. Catheter-based therapies for submassive and massive PEs are still evolving, but studies have shown promise. The techniques employed include the use of aspiration of the clot with a large catheter (10 French), intraclot infusion of a thrombolytic agent followed by aspiration, ultrasound-assisted catheter-directed thrombolysis, and use of rheolytic thrombectomy. Success for these techniques has been reported to be 80–90%, with major complications occurring in 2–4% of patients.
INTERVENTIONS FOR REFRACTORY HYPERTENSION
The recent recognition of the importance of the renal sympathetic nerves in modulating blood pressure has led to a technique to selectively denervate renal sympathetic nerves in patients with refractory hypertension. The procedure involves applying low-power radiofrequency treatment via a catheter along the length of both renal arteries. In the randomized Symplicity HTN-2 trial, renal denervation significantly reduced blood pressure compared with medical therapy. The Symplicity device (Medtronic) is approved in Europe, though the randomized and blinded U.S. Symplicity HTN-3 trial showed no effect.
CONCLUSION
Interventional cardiology continues to expand its borders. Treatment for coronary artery disease, including complex anatomic subsets, continues to advance, encroaching on what has traditionally been treated by CABG. Technological advances such as drug-eluting stents, now already in their second generation, and manual thrombus aspiration devices are improving the results of PCI. In particular, PCI has been shown to prevent future ischemic events in acute coronary syndromes. For patients with stable coronary disease, PCI has an important role in symptom alleviation. Treatment of peripheral and cerebrovascular disease has also benefited from the application of percutaneous techniques. Structural heart disease is increasingly being treated with percutaneous options, with a high likelihood that interventional approaches will compete with open-heart surgery in a significant proportion of cases in years to come.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree