3 (Part I) Quality and safety
Introduction
Background
• essential oils are powerful mixtures and have physiological, psychological and pharmacological effects, both desirable and undesirable, when applied to the body;
• in most countries, including the UK, these oils are freely available and there is no restriction on their sale and use;
• the majority of people who buy essential oils are members of the general public, who do not have expert knowledge of their nature and use.
Essential oil quality
Quality variation
Deterpenized oils
Essential oils used in the fragrance industry often have their terpenes partly or wholly removed on account of their insolubility in alcohol, which would result in cloudiness – a distinct commercial and aesthetic disadvantage in a perfume! The deterpenized oil is incomplete and contains in higher proportions the remaining constituents of the oil, for example the deterpenization of peppermint increases the content of the possibly hazardous ketone menthone. In aromatherapy there is no necessity for this and it is imperative not to interfere with the natural balance of the essential oil. Some therapists purchase bergapten-free bergamot oil, as this constituent (a furanocoumarin) can be responsible for phototoxicity of the skin in sunlight, but this is unnecessary (see Ch. 3 Part II).
Contaminants
• a standardized product which can be repeated at any time, which is impossible with naturally grown plants;
• a product at a price compatible with their marketing strategy (often meaning cheap).
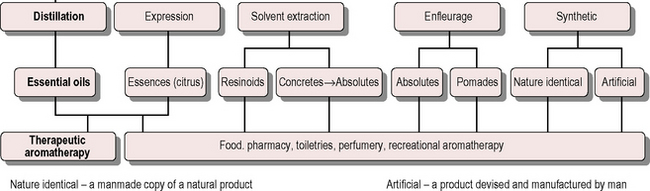
The various types of aromatic product available are shown in Figure 3.1. For therapeutic use aromatherapists use only distilled essential oils and expressed essences, although some occasionally make exception to the rule by using jasmine absolute or benzoin.
Procurement of genuine, authentic essential oils
It is not easy to procure such oils, for many and varied reasons, some of which follow:
1. High quality at the time of harvesting can diminish due to distillation, rectification, fractionation, adulteration, transportation, storage and time.
2. The bulk of essential oils comes from faraway countries, only 3–5% coming from Europe.
3. Availability is influenced by various factors including war (vetiver, patchouli) and changing climate (ambrette seed).
4. The trade has very limited knowledge of scientific names and it is vital for aromatherapy to use the scientific name, because the use of local common names for the plants leads to confusion. More than one name may be given to the same plant, or the same common name can be given to different plants (such as marjoram, which might be Origanum majorana or Thymus mastichina).
Cedarwood oil may be any of the following, since all are traded simply as cedarwood: Cedrus atlantica [Atlas cedarwood], Cedrus deodora [deodar or Himalayan cedarwood], Cedrus libani [cedar of Lebanon], Chamaecyparis lawsoniana [western white cedar], Cryptomeria japonica [Japanese cedar], Juniperus procera [east African cedarwood], Juniperus mexicana [Texas cedarwood], Juniperus virginiana [red cedarwood], Thuja occidentalis [white cedar], Thuja plicata [western red cedar].
The common name oregano is applied to more than 43 different species and 18 hybrids belonging to five different families (Skoula & Harborne 2002).
5. A supply problem exists because the market for herbal medicines is increasing and more oils are being used in aromatherapy.
6. Wild plants are sometimes collected by an unsupervised, unskilled labour force paid by weight of plant collected, and who are not always able to identify the required plant, resulting in indiscriminate harvesting.
Accurate identification
In many cases it is not sufficient merely to specify the genus and species (and the variety if applicable): it is also necessary to designate the chemotype (explained in Ch. 1) and the part of the plant used for extraction. An example is the cinnamon tree, where the oil from the bark consists principally of an aldehyde, whereas the oil from the leaf is mainly a phenol, with different effects and uses. The oil from the thuja or white cedar tree, Thuja occidentalis (responsible for the restriction on cedarwood oils in France), is taken from the leaves, but other ‘cedarwoods’ are taken from the wood. In the Apiaceae family, the seed oils can be significantly different from oils extracted from other parts of the same plant, e.g. in the case of Angelica archangelica the root oil is phototoxic, whereas that from the seed is not. Therapists need to be aware of this; it is their responsibility to ensure that inappropriate treatment is not given.
• Peru balsam from El Salvador and tolu balsam are both standardized or synthesized in Europe.
• Sandalwood from South India (Mysore) is not available yet continues to be sold.
• Rosewood oil from the Amazon is not available because the industry is now finished.
• The same situation exists with Copaiba balsam.
• Tea tree – there is three times more sold in the world than is produced in Australia; tea tree oil is a comparatively simple essential oil comprising only about 30 compounds (cf. ylang ylang, with about 1200) and so is easy to reconstruct; also the ‘natural’ oil is often ‘regulated’ at source.
• Sumatran Patchouli may be relabelled Malaysian Patchouli.
• Barrème lavender (a lavender high in linalyl acetate) has not been produced for many years but unbelievably still appears on some sales lists.
Pharmacopoeia
• stimulate the appetite by increasing salivation
• act as carminatives to relieve gastric discomfort and flatulence
• counteract the griping action of purgatives
• contribute as mild expectorants in cough mixtures and pastilles
• check profuse secretion and relieve congestion of the bronchioles when used in inhalants
• act as counterirritants and rubefacients, for the chest in bronchitis and pleurisy, and for the relief of rheumatic pain, when formulated as ointments, creams and liniments.
Some essential oils listed in the British Pharmacopoeia (BP) are stocked in hospital pharmacies, but oils prepared to BP standards may not be suitable for use in aromatherapy. One reason for this is that many plants, for example thyme, Thymus vulgaris, exist in the wild as many different chemotypes, each chemotype producing quite a different essential oil in makeup and therapeutic action (see Ch. 1 p. 8) and such differences are not always reflected in the pharmacopoeia. Another reason is because the specification is either too broad or incomplete and does not reflect the natural materials currently available; some oils listed are folded, which alters their composition: such essences are not used in aromatherapy.
Storage and shelf life
Factors that affect the storage of aromatherapy products are:
• Air – oxidation can be a problem and essential oils keep best when bottles are full, with as little air as possible. Oxidation – the combining of free oxygen with compounds in the essential oils – affects some oils particularly, altering their therapeutic effects, e.g.
 oils with a significant monoterpenes content can lead to the formation of sensitizing hydroperoxides (e.g. turpentine)
oils with a significant monoterpenes content can lead to the formation of sensitizing hydroperoxides (e.g. turpentine) some oils high in the terpenes limonene and pinene may react with oxygen; the antibacterial activity of lemongrass oil is diminished when oxidized (Orafidiya 1993)
some oils high in the terpenes limonene and pinene may react with oxygen; the antibacterial activity of lemongrass oil is diminished when oxidized (Orafidiya 1993)Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree