Paragangliomas
Ernest E. Lack
Extra-adrenal paraganglia make up part of the dispersed neuroendocrine system (DNS) having a centripetal and roughly symmetrical distribution with extension from the base of the skull down to the pelvic floor. There is brief reference to the presence of a structure reminiscent of paraganglia within soft tissues surrounding large femoral vessels, but it is not well documented (1). These paraganglia can be divided into two groups—paraganglia in the head and neck region (including aorticopulmonary paraganglia), which have a close alignment with the parasympathetic nervous system, and the paraganglia of the sympathoadrenal neuroendocrine system.
The amine precursor uptake and decarboxylation (APUD) cell concept was proposed by Pearse (2,3) in an attempt to unify a broad array of endocrine cells that were presumed to have a common embryologic derivation from neural crest. Although no longer widely accepted, this concept accelerated scientific investigation, particularly in the areas of immunology and molecular pathology (4,5). The term neuroendocrine has now come into vogue because it emphasizes the close relationship between the nervous system (e.g., neurons) and endocrine cells (6,7 and 8). A wide variety of hormones and regulatory neuropeptides have been identified in normal paraganglia and the tumors arising from them. Some of these substances have an endocrine function through interaction with receptors at distant sites, whereas others mediate a regulatory effect on neighboring cells (paracrine function) or perhaps even an autoregulatory effect on the same cell (autocrine function).
The terminology of paragangliomas is based on the anatomic site of origin rather than the chromaffin or nonchromaffin status of the tumor. The term pheochromocytoma has been arbitrarily restricted to an adrenal medullary paraganglioma, although there are extra-adrenal tumors that are functionally and morphologically very similar. Because extra-adrenal paragangliomas can arise at such a variety of anatomic sites, one must be aware of other tumors that enter into the differential diagnosis. Accurate recognition of these tumors can be particularly difficult when one is dealing with limited biopsy material or tumor that is partially crushed. Stromal alterations can also cause diagnostic difficulties.
PARAGANGLIA OF THE HEAD AND NECK REGION
EMBRYOLOGY AND PHYSIOLOGY
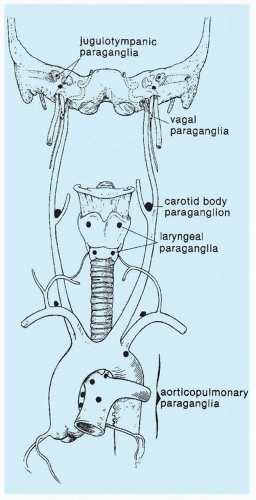
Paraganglia in the head and neck region are closely aligned with the parasympathetic nervous system and often have a close spatial relationship with neural or vascular structures (Fig. 15.1). Using a cytochemical marker system, glomus type I or chief cells have been shown in the avian carotid body to have embryologic origin from neural crest (9), and presumably, chief cells in humans at various other locations in the head and neck region have this same embryogenesis. Carotid body and aorticopulmonary paraganglia have been shown in experimental physiologic studies in animals to have a chemoreceptor role, with modulation of respiratory and cardiovascular function in response to changes in arterial partial pressure of oxygen (PO2), partial pressure of carbon dioxide (PCO2), pH, and other chemical alterations (10,11). de Castro (12) was the first to propose a chemosensory role for carotid bodies based on morphologic observations in animals. The physiologic function of other paraganglia in the head and neck region is not known, but their histologic appearance is very similar, thus suggesting a similar role in chemosensation.
Changes in oxygen (O2) tension are detected by O2-sensitive K+ and Ca2+ channels, and a suprathreshold increase of cytosolic Ca2+ triggers neurotransmitter (i.e., dopamine) release, which activates afferent nerve terminals (13). Despite the immense amount of experimental research on carotid bodies in animals, the precise mechanism(s) of O2 sensing and the
basic chemosensor(s) or transducer(s) is still not entirely clear. Numerous proposals have been put forth. Proposed candidates include reactive oxygen species, mitochondria, AMP-activated kinase, hemoxygenase-2, and succinate dehydrogenase (SDH). No single mechanism has been shown to explain the multitude of accumulated data, and it may be that a cascade involving several mechanisms is required to accomplish the necessary homeostatic response to arterial hypoxemia (14,15 and 16). It has been possible to study dissociated chemoreceptor cells or intact carotid body in culture, and in a recent study, chemoreceptor cells were not sensitive to hypoglycemia (17).
basic chemosensor(s) or transducer(s) is still not entirely clear. Numerous proposals have been put forth. Proposed candidates include reactive oxygen species, mitochondria, AMP-activated kinase, hemoxygenase-2, and succinate dehydrogenase (SDH). No single mechanism has been shown to explain the multitude of accumulated data, and it may be that a cascade involving several mechanisms is required to accomplish the necessary homeostatic response to arterial hypoxemia (14,15 and 16). It has been possible to study dissociated chemoreceptor cells or intact carotid body in culture, and in a recent study, chemoreceptor cells were not sensitive to hypoglycemia (17).
MORPHOLOGY AND ANATOMIC DISTRIBUTION
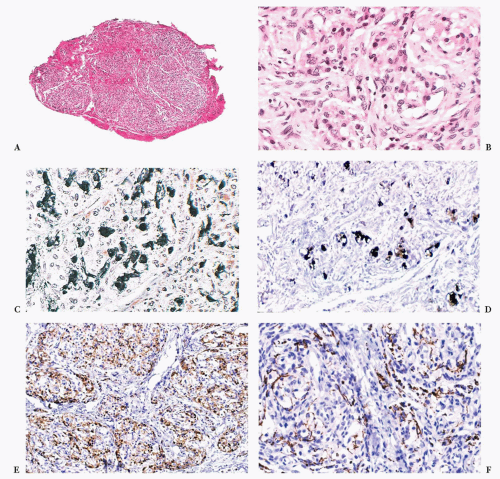
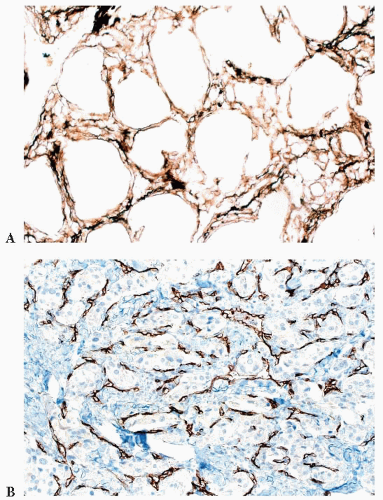
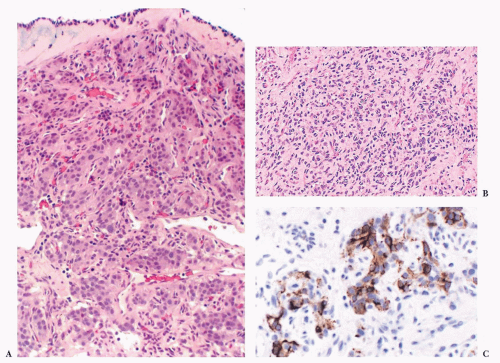
Carotid bodies are the largest compact collection of paraganglia in the head and neck region; they appear as a small ovoid structure on the medial aspect of the carotid bifurcation on each side of the neck (Fig. 15.2). The average combined weight of carotid bodies in adults without chronic hypoxia or systemic hypertension is approximately 12 mg (18,19). Paraganglia located elsewhere in the head and neck region have a nearly identical histomorphologic picture, but they are smaller and usually lack the compact lobular architecture of the carotid bodies (Fig. 15.3A). The basic anatomic unit of the carotid body is the lobule, which contains clusters (“zellballen”) or cords of chief cells (Fig. 15.3B). Within the lobule, a variety of different cells are present, including chief cells and sustentacular or glomus type II cells, as well as pericytes, endothelial cells, and Schwann cells. Chief cells can be vividly depicted by staining for cytoplasmic argyrophilia (Fig. 15.3C) or chromogranin A (Fig. 15.3D), and this organoid arrangement of chief cells is vaguely recapitulated in the neoplasms derived from them. Sustentacular cells are located at the periphery of clusters of chief cells and can be demonstrated by immunostain for S-100 protein (Fig. 15.3E); this immunostain will also highlight the Schwann cells in the normal carotid body and other paraganglia. Sustentacular cells are typically present in paragangliomas at all anatomic sites in the head and neck region and the sympathoadrenal neuroendocrine system. The rich component of neuritic processes (dendritic and/or axonal) can be nicely demonstrated with immunostain for neurofilament protein (Fig. 15.3F).
Carotid body and other paraganglia contain catecholamines, and chief cells have been shown to have enzymes involved in catecholamine synthesis. The immunophenotype of these endocrine cells and tumors derived from them, however, are remarkably diverse. Paraganglia in the head and neck region are located at a variety of sites, including the middle ear, adventitia of the jugular bulb, ganglion nodosum of the vagus nerve, larynx, and base of the heart (Fig. 15.1). This anatomic distribution in some areas parallels the branchial arch derivatives that in aquatic species correspond to gill arches. In some cases, paragangliomas have been reported at sites where normal paraganglionic tissue has not yet been described in humans. Examples include paragangliomas primary in liver (20), ovary (21), bone (22), and parathyroid (23).
HYPERPLASIA OF CHEMORECEPTOR PARAGANGLIA
Carotid body enlargement was reported by Arias-Stella (24) in natives born and living in the Peruvian Andes 14,350 feet above sea level. This was confirmed in other studies (25) and also by Saldana et al. (26), who reported a 10-fold increase in incidence of “chemodectomas” at high altitude. An increased incidence of carotid body paragangliomas (CBPs) has been reported in other locations at high altitude (27,28). Carotid body hypertrophy and hyperplasia have also been observed in humans under normobaric conditions, for example, in patients with chronic obstructive pulmonary disease (18,29), systemic hypertension (18), and some cases of cystic fibrosis (Fig. 15.4A,B) and cyanotic congenital heart disease (19,30). Chronic hypoxia leads to hypersensitivity of the carotid bodies with subsequent morphologic and neurochemical changes in the carotid body, including carotid body enlargement, hyperplasia of glomus cells, and neovascularization (31,32). Hyperplasia of vagal paraganglia (33) and aorticopulmonary paraganglia (34) suggests a similar role in chemosensation. Chemoreceptor hyperplasia in most cases is presumably a compensatory response to prolonged and severe hypoxemia (24). Although on rare occasion paragangliomas associated with hypoxemia related to conditions other than high altitude have been reported (35,36 and 37), the increased risk of developing a paraganglioma appears to be negligible in patients at sea level, with or without hypoxemia.
PARAGANGLIOMAS OF THE HEAD AND NECK REGION
CAROTID BODY PARAGANGLIOMA
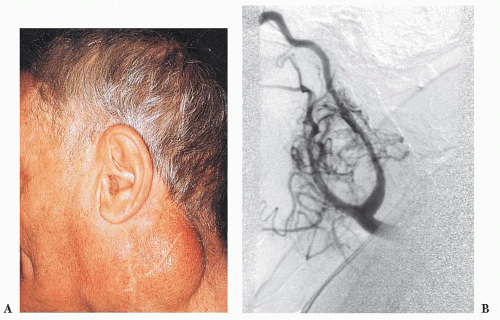
Tumors of this type have been referred to as chemodectomas (chemeia, meaning “infusion”; deschesthai, meaning “to receive”; and oma, meaning “tumor”) (38), and this term has been used synonymously for paragangliomas at other sites in the head and neck region. There is no evidence, however, that any of these tumors have a functional role in chemosensation. The first CBP was reported by Marchand (39) in 1891 in a patient who had been operated on by Riegner in 1880. CBPs typically appear as a slow-growing, painless mass near the angle of the mandible (Fig. 15.5A) (34,40,41,42 and 43). In some patients, there may be signs or symptoms of cranial nerve palsy, such as dysphagia or dysphonia; the carotid sinus syndrome is an unusual feature, with bradycardia and syncopal episodes. On rare occasion, CBPs and other head and neck paragangliomas may be functional, with
signs or symptoms of excess catecholamine secretion (34,44,45). The average age at diagnosis is usually in the fifth decade of life, and the average duration of symptoms is approximately 4 years (43,44). Some series report a roughly equal gender distribution, whereas others show a slightly higher incidence in women (46,47 and 48). It has been estimated in the sporadic setting that 4% to 8% of patients with CBPs have bilateral tumors (49,50). CBPs are more often bilateral when arising in a familial setting. Occasionally, there may be multiple paragangliomas at other sites, such as the vagus nerve, orbit, middle ear, and base
of the heart (51,52). Preoperative selective angiography may be useful for delineating tumor location (Fig. 15.5B), defining blood supply, and providing a route for selective embolization if this is considered before surgery. Preoperative embolization can decrease blood loss in patients with paragangliomas of the head and neck region (48,53) and other sites (54).
signs or symptoms of excess catecholamine secretion (34,44,45). The average age at diagnosis is usually in the fifth decade of life, and the average duration of symptoms is approximately 4 years (43,44). Some series report a roughly equal gender distribution, whereas others show a slightly higher incidence in women (46,47 and 48). It has been estimated in the sporadic setting that 4% to 8% of patients with CBPs have bilateral tumors (49,50). CBPs are more often bilateral when arising in a familial setting. Occasionally, there may be multiple paragangliomas at other sites, such as the vagus nerve, orbit, middle ear, and base
of the heart (51,52). Preoperative selective angiography may be useful for delineating tumor location (Fig. 15.5B), defining blood supply, and providing a route for selective embolization if this is considered before surgery. Preoperative embolization can decrease blood loss in patients with paragangliomas of the head and neck region (48,53) and other sites (54).
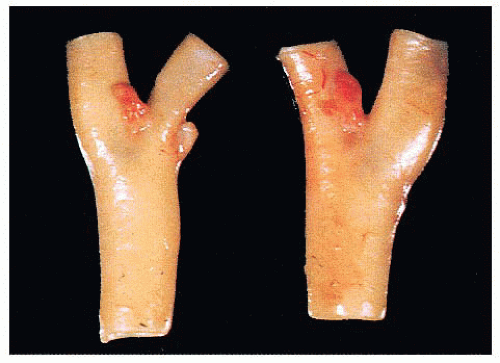
In a large study of CBPs by Shamblin et al. (42), three groups of tumors were identified—group I CBPs which were surgically resectable without significant trauma to the adjacent artery, group II tumors which appeared to partially surround the carotid vessel(s) and were adherent to the adventitia, and group III CBP with a more intimate adherence to the entire
circumference of the carotid bifurcation (42). The tumor first reported by Marchand (39) was a group III CBP (Fig. 15.6).
circumference of the carotid bifurcation (42). The tumor first reported by Marchand (39) was a group III CBP (Fig. 15.6).
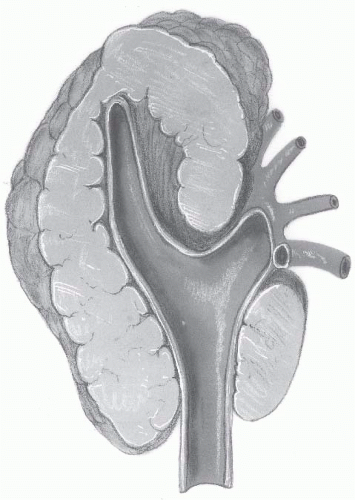 FIGURE 15.6 Cross section of carotid vessels and large carotid body paraganglioma. Tumor encircles the carotid bifurcation and adjacent internal and external branches. This was the first carotid body tumor reported by the German pathologist Felix Jacob Marchand in 1891(39). |
JUGULOTYMPANIC PARAGANGLIOMA
Jugulotympanic paraganglioma (JTP) has a predilection for women, and these tumors arise from microscopic collections of paraganglia, which were described by Guild (55,56) in a study involving serial sectioning of temporal bones; the first case of a patient with JTP was reported by Rosenwasser (57) in 1945. These paraganglia (average of two to three in each temporal bone) may be found along the course of the Jacobsen nerve (tympanic branch of the 9th cranial nerve), the nerve of Arnold (auricular branch of the 10th cranial nerve), the adventitia of the jugular bulb, the osseous canal connecting the jugular fossa to the middle ear cavity, or within the middle ear—usually over the cochlear promontory. The varied locations of these paraganglia and the complex anatomy of this area form the basis for the different clinical presentations of patients with JTP (58). Small tumors arising over the cochlear promontory (tympanic paraganglioma) may arise as an aural polyp, with filling of the middle ear cavity or extension into the external ear canal. JTP can involve the temporal bone, with intracranial extension, or appear as a mass at the base of the skull, with erosion of the jugular foramen. Rarely, these tumors display clinical evidence of malignancy, primarily metastatic disease. The most common site of metastasis includes lymph nodes, skeleton, lungs, and liver (40). The histologic appearance of these tumors is usually not predictive of biologic behavior.
VAGAL PARAGANGLIOMA
These tumors also have a predilection for women in the fourth and fifth decades of life and usually develop as a lateral neck mass with extension up to the base of the skull (43). Vagal paragangliomas (VPs) arise from paraganglia located at the level of or just below the nodose ganglion of the vagus nerve that are usually multiple on either side of the neck (33,59). They have been referred to as vagal body paraganglia, although they are anatomically dispersed microscopic collections of cells. As with CBPs, these tumors may protrude medially with deviation of oropharyngeal structures, such as the palatine tonsil, or extend even higher up into the nasopharynx. They may also demonstrate intracranial extension (60). Vagus nerve palsy may be apparent and may be accompanied by hoarseness and dysphagia (61). VPs arise higher in the neck than CBPs and usually cause anterior bowing of the carotid vessels without direct involvement of the carotid bifurcation. The tumor can also extend into the jugular foramen at the base of the skull (60).
LARYNGEAL PARAGANGLIOMA
Laryngeal paragangliomas (LPs) arise from microscopic collections of paraganglia distributed on either side of the larynx that form a superior and inferior group (62); these tumors usually evolve from superior laryngeal paraganglia above the anterior part of the vocal cords near the aryepiglottic fold, but they can also develop inferiorly and appear in the subglottic or tracheal area. Hoarseness and dysphagia are the most common complaints (63). In the literature, LPs have been associated with a very high rate of malignancy (64), but most of these “alleged” malignant paragangliomas have undoubtedly been confused with laryngeal atypical carcinoid tumor of the larynx (Fig. 15.7) (65,66). At present, it is believed that true LPs have very limited malignant potential (67,68).
AORTICOPULMONARY PARAGANGLIOMA
Aorticopulmonary paragangliomas (APPs) arise from small collections of paraganglia that are located at various sites, both dorsal and ventral, at the base of the heart in relation to the great vessels (69). Some paraganglia can be found above the aortic arch in relation to the subclavian arteries. APPs can cause
symptoms or signs such as hoarseness, dysphagia, chest pain or discomfort, and, rarely, hemoptysis or the superior vena cava syndrome (70). There appears to be a slight predominance among women. The average size of APPs is approximately 7.5 cm (range, 1.2 to 17 cm) (70). Some tumors can involve the pericardium or heart directly (cardiac paragangliomas) (71). A pigmented cardiac paraganglioma has also been reported (72).
symptoms or signs such as hoarseness, dysphagia, chest pain or discomfort, and, rarely, hemoptysis or the superior vena cava syndrome (70). There appears to be a slight predominance among women. The average size of APPs is approximately 7.5 cm (range, 1.2 to 17 cm) (70). Some tumors can involve the pericardium or heart directly (cardiac paragangliomas) (71). A pigmented cardiac paraganglioma has also been reported (72).
OTHER PARAGANGLIOMAS
Paragangliomas have been reported at a variety of other sites in the head and neck region, including the orbit (73,74 and 75), nasal cavity (76), hypopharynx (77), parotid gland (78), cheek (79), palatine tonsil (80), pineal (81), and sellar region (82) including Meckel’s cave (83). The thyroid gland is an uncommon but well-recognized primary site, and recently, a sclerosing paraganglioma was reported (84). They have a histologic appearance similar to CBP. Rare examples of primary pulmonary paragangliomas have also been described (85,86). These tumors may arise from intrapulmonary paraganglia, as described by Blessing and Hora (87), although some may be very difficult to distinguish from a bronchial carcinoid tumor. One example has been reported as multicentric metachronous pulmonary and intravagal paragangliomas with an identical immunophenotype; they showed negative results for epithelial markers, as one would expect for paragangliomas (88). A recent case of Cushing syndrome resulting from an adrenocorticotropic hormone (ACTH)-secreting primary pulmonary paraganglioma has been reported (89). A large primary paraganglioma of the lung was reported measuring 13 cm in diameter (90).
GROSS PATHOLOGY
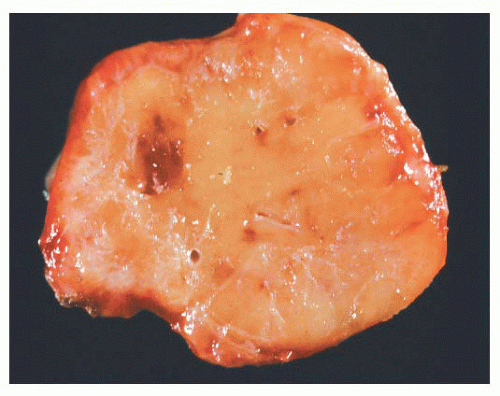
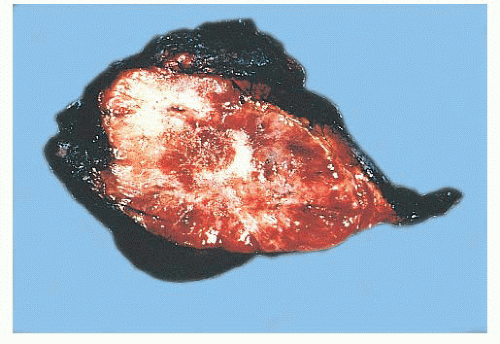
Paragangliomas are rubbery, firm, and usually well demarcated, with expansile borders (Fig. 15.8). The compressed rim of connective tissue surrounding the tumor is best regarded as a fibrous pseudocapsule. Some tumors, such as JTP, may be small and irregular or fragmented, making gross examination difficult. On cross section, these tumors have a meaty to light tan appearance and, if closely scrutinized, may have punctate to linear vascular markings that are often retracted slightly beneath the cut surface. Sometimes, there are intersecting bands of fibrous tissue or intense sclerosis (Fig. 15.9). The average sizes of paragangliomas recorded in one series are as follows: CBPs, 3.8 cm (range, 1.8 to 8.5 cm); VPs, 4.0 cm (range, 2.0 to 6.0 cm); and LP, 2 to 5 cm in diameter (91). The tumor volume was calculated by Nora et al. (92) for CBPs; it ranged from 2.0 to 164 cm3. Larger tumor volume or size correlates with increasing difficulty of surgical resection and longer duration of tumor.
Mechanical manipulation of tumor during surgery may cause a deep red-brown discoloration as a result of congestion and hemorrhage. Areas of degenerative change with necrosis and cystic alteration are very uncommon; however, foci of ischemic necrosis can be seen in some tumors that have been embolized before surgical removal. On rare occasion, a paraganglioma can invade the lumen of the carotid artery or cause total occlusion of a large vessel (93,94).
MICROSCOPIC PATHOLOGY
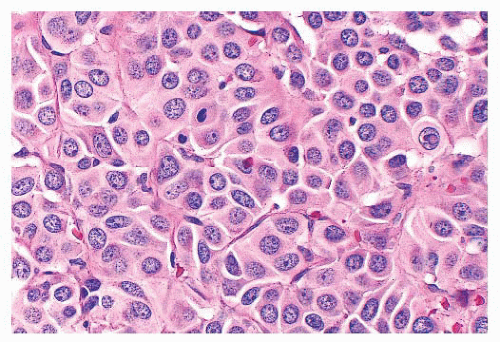
In general, the microscopic patterns encountered in head and neck paragangliomas do not allow one to reliably distinguish between tumors arising at different anatomic sites (34,40). The most typical pattern is a discrete or organoid arrangement of neoplastic chief cells often referred to as zellballen (Fig. 15.10) (95). Several different patterns were recognized by LeCompte (96)—the usual (Fig. 15.10), adenoma-like, and angioma-like. The cell cytoplasm is eosinophilic and faintly granular with indistinct borders. Some tumors may have cells with abundant, deeply eosinophilic cytoplasm, thus imparting an oncocytic appearance (34,40). The orientation of neoplastic chief cells within individual cell clusters tends to be haphazard, without polarization along the fibrovascular septa, and there is no formation of true rosettes or acini, as one might see in other endocrine tumors, such as carcinoids. The nesting pattern can vary in size, and it is most vividly accentuated by staining for reticulum (Fig. 15.11A); the rich vascularity of these tumors can be clearly demonstrated by immunostains for endothelial
markers (Fig. 15.11B). There is a network of supporting cells referred to as sustentacular cells that can be highlighted by immunohistochemical methods with antibodies to S-100 protein. Staining for cytoplasmic argyrophilia can be useful in diagnosis and often shows that the neoplastic chief cells contain myriad pinpoint cytoplasmic granules. This stain is seldom done today.
markers (Fig. 15.11B). There is a network of supporting cells referred to as sustentacular cells that can be highlighted by immunohistochemical methods with antibodies to S-100 protein. Staining for cytoplasmic argyrophilia can be useful in diagnosis and often shows that the neoplastic chief cells contain myriad pinpoint cytoplasmic granules. This stain is seldom done today.
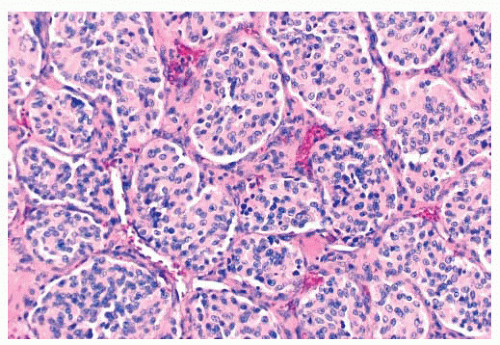 FIGURE 15.10 The typical histologic pattern for head and neck paragangliomas. Organoid clustering of neoplastic chief cells vaguely recalls the architecture of the normal paraganglion (Fig. 15.3B). |
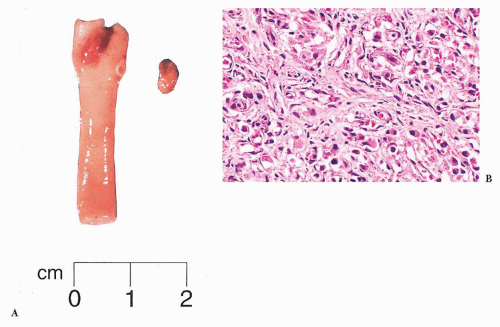
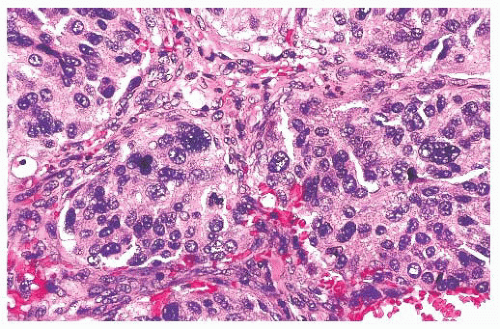
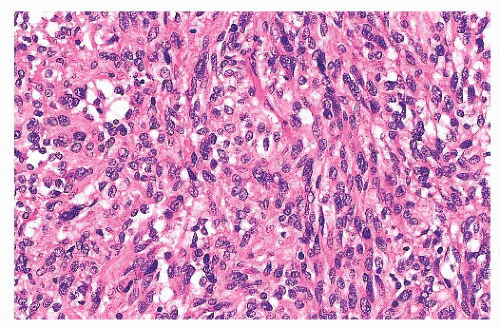
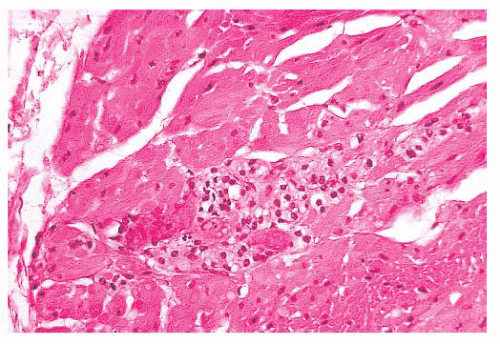
Nuclear hyperchromasia and pleomorphism can be quite prominent, but these are not reliable criteria for judging malignancy (Fig. 15.12) (34,40). Nuclear pseudoinclusions, representing invagination of cell cytoplasm, are occasionally seen (Fig. 15.13). A most unusual architectural pattern is a spindle cell or pseudosarcomatous arrangement of cells that, on careful examination, is usually accompanied by a more typical nesting pattern elsewhere in the tumor (Fig. 15.14) (34,91). Large expansile or locally aggressive tumors, such as CBPs or VPs, may incorporate myelinated nerve bundles, but true intraneural growth is seldom seen (Fig. 15.15). Limited biopsy material can hamper accurate diagnosis; an example is the small tympanic paraganglioma, which can appear as an aural polyp (Fig. 15.16A) or a small, hypercellular biopsy specimen of a skull base tumor without an obvious organoid or nesting pattern (Fig. 15.16B). Immunostain for chromogranin A may greatly aid in diagnosis by demonstrating discrete collections of neoplastic chief cells (Fig. 15.16C). It is well known that biopsies of paragangliomas can be hazardous and lead to brisk hemorrhage.
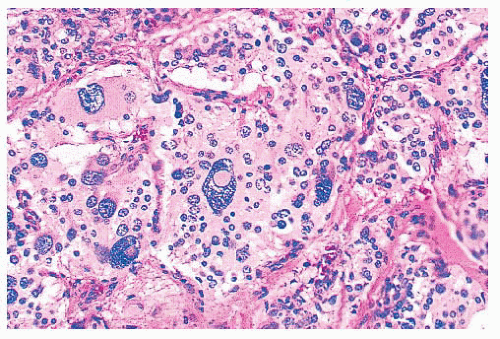 FIGURE 15.13 Several enlarged nuclei in this clinically benign carotid body paraganglioma show round, pink pseudoinclusions of cell cytoplasm. |
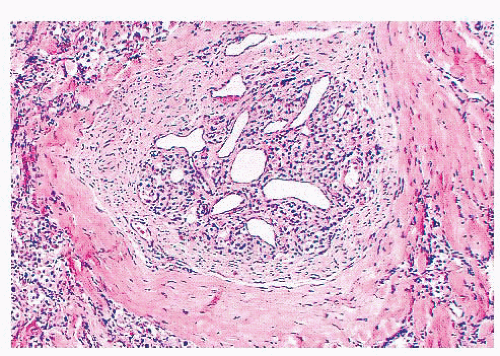
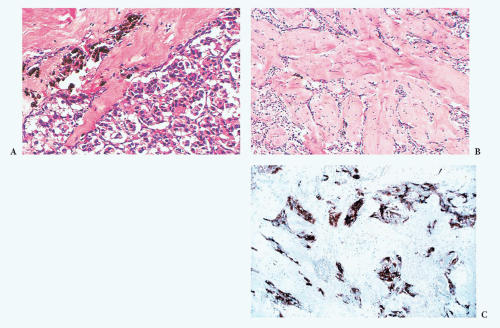
Stromal alterations may be apparent in some paragangliomas and, at times, are so marked as to obscure the true nature of the tumor (34,40). An example is stromal fibrosis with evidence of old hemorrhage (Fig. 15.17A) that may be so extensive that it causes compression and distortion of nests of neoplastic chief cells (Fig. 15.17B). In some cases, one may find sinusoidal sclerosis, which accentuates the organoid arrangement of tumor cells (Fig. 15.18). Congestion and hemorrhage within the tumor can cause wide spacing of individual zellballen (Fig. 15.19), but more typical diagnostic features can usually be found. Interstitial amyloid deposits were reported in two CBPs
by Capella and Solcia (97) on ultrastructural study, but none was illustrated.
by Capella and Solcia (97) on ultrastructural study, but none was illustrated.
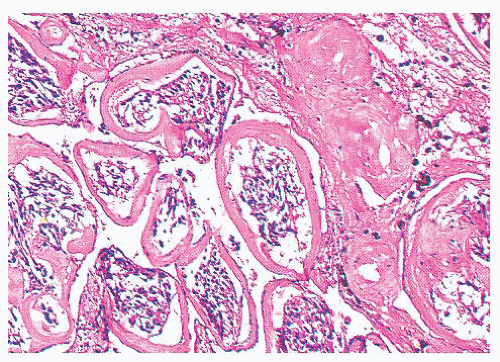 FIGURE 15.18 A carotid body paraganglioma shows encirclement of individual zellballen by fibrous tissue representing sinusoidal sclerosis. There is also evidence of old hemorrhage. |
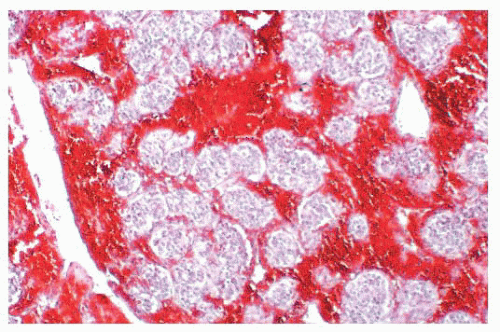 FIGURE 15.19 Marked congestion within a head and neck paraganglioma causes some architectural distortion. |
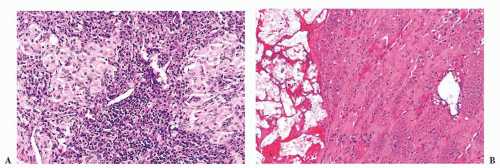
Dysmorphic vessels as a result of symmetric or asymmetric myointimal proliferation or thickening may be present, and in some tumors, vascular ectasia or an arborizing pattern can focally simulate a hemangiopericytoma. Chronic inflammatory infiltrates are uncommon. They may be evident as sparse perivascular lymphocytic infiltration, usually at the periphery of the tumor, but rarely, the inflammatory component can be more extensive, with separation of nests of chief cells. It can also simulate a lymph node metastasis, such as Hürthle cell carcinoma of the thyroid (Fig. 15.20A). Necrosis is an unusual morphologic finding, but it may be extensive and confluent if the tumor has been effectively embolized before surgery (Fig. 15.20B).
SYMPATHOADRENAL NEUROENDOCRINE SYSTEM
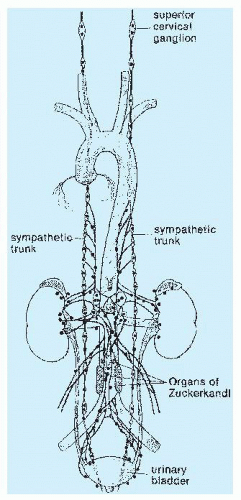
The prototype tissues are the adrenal medullae, which synthesize and secrete catecholamines, causing rapid physiologic changes that are dissipated rather quickly. The distribution of extra-adrenal paraganglia (Fig. 15.21) parallels that of the sympathetic nervous system within the abdomen and urinary bladder, thorax, and neck (98), and some paraganglia are located within viscera such as the gallbladder and urinary bladder (Fig. 15.22). During fetal life, the chromaffin cells of the adrenal gland are usually inconspicuous, but postnatally, they develop rapidly, becoming structurally similar to medullary cells of the adult gland by the end of the first year of life. Epinephrine is the predominant catecholamine in the normal adrenal medulla in a ratio of approximately 4:1 relative to norepinephrine (34).
Most of the chromaffin tissue in the fetus is extra-adrenal in location, with the most prominent collections residing on either side of the aorta near the origin of the inferior mesenteric or renal arteries, down to the aortic bifurcation. These paraganglia were described by Zuckerkandl (99,100) in 1901 and referred to as the “aortic bodies” (Fig. 15.23A,B); on macroscopic
examination, they may be difficult to distinguish from small lymph nodes or sympathetic ganglia (101). The organs of Zuckerkandl involute after birth; by the age of 6 or 7 years, only microscopic collections of chromaffin cells can be detected (Fig. 15.24) (102). The function of fetal chromaffin tissue in utero is not fully known, but it may have some role in maintenance of vascular tone and blood pressure. Norepinephrine predominates in extra-adrenal chromaffin tissue, such as the organs of Zuckerkandl, and catecholamine content is reported to decline with structural involution (103). The microanatomy of sympathetic paraganglia elsewhere in the abdomen, chest, and other locations is less well defined.
examination, they may be difficult to distinguish from small lymph nodes or sympathetic ganglia (101). The organs of Zuckerkandl involute after birth; by the age of 6 or 7 years, only microscopic collections of chromaffin cells can be detected (Fig. 15.24) (102). The function of fetal chromaffin tissue in utero is not fully known, but it may have some role in maintenance of vascular tone and blood pressure. Norepinephrine predominates in extra-adrenal chromaffin tissue, such as the organs of Zuckerkandl, and catecholamine content is reported to decline with structural involution (103). The microanatomy of sympathetic paraganglia elsewhere in the abdomen, chest, and other locations is less well defined.
EXTRA-ADRENAL PARAGANGLIOMAS
These tumors arise predominantly in the retroperitoneum anywhere from the upper abdomen to the pelvic floor. One of the more common sites is the anatomic region corresponding to the
organs of Zuckerkandl. It has been estimated that 5% to 10% of pheochromocytomas are extra-adrenal in location, mainly in the retroperitoneum but also in the posterior thorax and neck (34,40,104). A higher incidence of extra-adrenal tumors has been reported in childhood (20% to 25%) (105), with the most common locations being the retroperitoneum and head and neck region (106). In the literature review of extra-adrenal paragangliomas (mainly sympathoadrenal neuroendocrine system) by Fries and Chamberlin (107), 71% of tumors were located in the superior or inferior para-aortic area, 9.8% arose in the urinary bladder, 12% were intrathoracic, and 1.2% were cervical.
organs of Zuckerkandl. It has been estimated that 5% to 10% of pheochromocytomas are extra-adrenal in location, mainly in the retroperitoneum but also in the posterior thorax and neck (34,40,104). A higher incidence of extra-adrenal tumors has been reported in childhood (20% to 25%) (105), with the most common locations being the retroperitoneum and head and neck region (106). In the literature review of extra-adrenal paragangliomas (mainly sympathoadrenal neuroendocrine system) by Fries and Chamberlin (107), 71% of tumors were located in the superior or inferior para-aortic area, 9.8% arose in the urinary bladder, 12% were intrathoracic, and 1.2% were cervical.
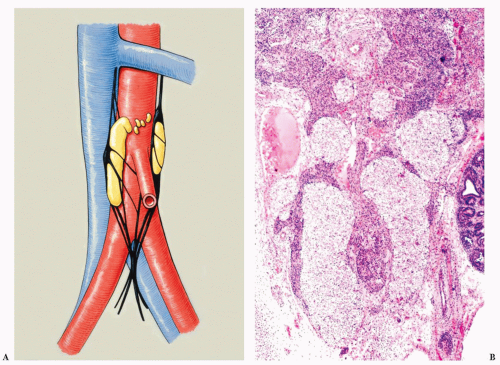 FIGURE 15.23 (A) Artist reproduction of one of two illustrations in the work by Zuckerkandl in 1901 (99). There are multiple discrete collections of paraganglia which was the most common configuration noted. In about 15% of cases, paraganglia (or “aortic bodies”) appeared to be continuous or united just above the inferior mesenteric artery. (B) Coronal section through a human fetus of about 11 weeks gestational age (by dates) shows paraganglia appear as multiple pale-staining structures with largest collections present as elongated structures on either side of the aorta. |
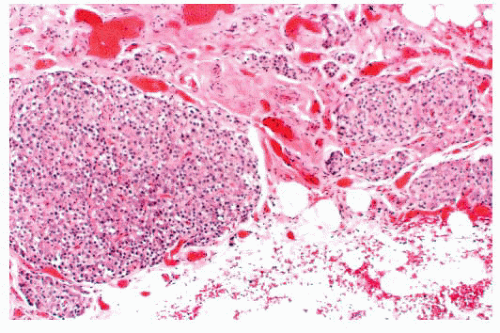 FIGURE 15.24 Small extra-adrenal paraganglionic tissue was discovered incidentally on histologic study of tissue from the retroperitoneum of an adult patient. Multiple dispersed collections of endocrine cells are present and have a delicate lobulated architecture with prominent microvasculature.
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|