Papillary Carcinoma
About 1% to 2% of breast carcinomas can be classified as papillary in women, and a slightly greater percentage of male breast carcinomas are papillary. A distinction should be made between invasive and noninvasive papillary carcinoma (1). Intracystic carcinoma is a variant of in situ papillary carcinoma that may also have an invasive component (2).
Women with solid or cystic papillary carcinoma of the breast are reportedly older than patients with other types of carcinoma, with a mean age ranging from 63 to 67 years (1,3). Nearly 50% of papillary carcinomas arise in the central part of the breast, and, as a consequence, nipple discharge has been described in at least one third of patients. Bleeding from the nipple occurs in a higher percentage of patients with papillary carcinoma than in those with a papilloma. The average size of papillary carcinoma is clinically 2 to 3 cm. Papillary carcinomas are usually rich in estrogen and progesterone receptors (4), and they tend to have a low growth rate when measured by thymidine labeling (5).
Papillary carcinomas often appear as rounded, circumscribed mass lesions on mammography (6,7). If part of the contour on mammography lacks circumscription, an invasive component may be present (7,8). Examination by ultrasound can suggest a papillary tumor when a solid area is detected in a hypoechoic cystic lesion (7,8,9). When present, calcifications tend to be punctate and associated with intraductal papillary carcinoma (9). Coarse, pleomorphic calcifications may develop in areas of sclerosis or resolved hemorrhage. Needle core biopsy can be used effectively for the diagnosis of papillary carcinoma. A series of 26 papillary lesions diagnosed by needle core biopsy included 7 classified as papillary carcinoma in situ (10). Invasive carcinoma was found in three (43%) of subsequent excisional biopsy specimens.
The term papillary applies to carcinomas in which the underlying microscopic pattern is predominantly frond-forming. Many papillary carcinomas have cystic areas, but this is not necessary for the diagnosis. When cyst formation is minimal or absent, separate fronds may be inconspicuous, and the papillary character is appreciated because the epithelium is supported by a network of fibrovascular stroma. Papillary carcinomas that do not form fronds are referred to as solid papillary carcinomas.
The epithelium on the papillae in papillary carcinomas is composed of cells arranged in the patterns typically found in intraductal carcinoma, which include papillary, micropapillary, cribriform, reticular, and solid appearances (Fig. 11.1). In most cases, the epithelial proliferation fills the space between neighboring fibrovascular stromal cores and contributes to the formation of complex patterns of papillae, glandular spaces, and solid areas within the lesion.
Fibrovascular stroma is present, to some extent, in all papillary carcinomas, but it tends to be less evenly distributed in carcinomas than in benign papillary lesions (Figs. 11.1 and 11.2). Rarely, a low-grade papillary carcinoma has a very orderly frond-forming structure with epithelium limited to the surfaces of the fibrovascular stroma. These lesions are very difficult to distinguish from papillomas in hematoxylin and eosin-stained sections, especially in needle core biopsy samples, but they are devoid of myoepithelium when studied with immunostains (Fig. 11.2). Some papillary carcinomas have areas in which there are relatively broad fibrous stalks with extensive sclerosis (Fig. 11.3).
The epithelial cells in papillary carcinomas grow in a less orderly fashion than in papillomas. This is manifested by uneven stratification and loss of epithelial cell polarity with respect to the basement membrane (Fig. 11.4). Epithelial cell nuclei are characteristically hyperchromatic regardless of cytologic grade, and there is usually a high nuclear-to-cytoplasmic ratio. Mitotic figures are variably present and usually more numerous in lesions that exhibit the most severe cytologic atypia. The tumor cells sometimes have secretory “snouts” at the luminal surface (Fig. 11.5). Intracytoplasmic mucin vacuoles may be conspicuous (Fig. 11.6). Mucin may accumulate between papillary fronds (Fig. 11.7). Apocrine areas in a papillary carcinoma exhibit cytologic atypia consistent with the rest of the tumor and therefore differ from the bland foci of apocrine metaplasia sometimes encountered in papillomas. When present, microcalcifications are more often found in glandular portions of the lesion than in the stroma (Fig. 11.8).
Myoepithelial cells, which are distributed relatively uniformly and proportionately with the epithelium of benign papillary lesions, are characteristically absent from papillary carcinoma. Less often they are present as a discontinuous layer, or even hyperplastic, in carcinomatous areas (11,12) (Figs. 11.1, 11.2, 11.5, and 11.9). Although myoepithelial cells can sometimes be visualized in H&E sections of a papillary carcinoma, the most reliable method for detecting them is with immunohistochemistry. Cytoplasmic markers associated with myoepithelium include CD10, smooth muscle actin (SMA), calponin, myosin, and CK5/6. The cytoplasmic markers are sometimes cross reactive with stromal myofibroblasts or vascular structures. The degree to which this cross reactivity will occur is unpredictable and quite variable with respect to individual markers. Thus, there may be considerable stromal reactivity for SMA in a given case and greater myoepithelial specificity for CD10 in the same tissue. The nuclear marker p63 is highly specific for myoepithelial cells, and it exhibits no cross reactivity for stroma, but it can be expressed in the nuclei of epithelial cells in a papillary lesion (13). These p63-positive epithelial cells can be readily distinguished from myoepithelium by their position in the epithelium. Whenever possible, it is prudent to use a panel of several markers when evaluating myoepithelium in a papillary tumor to compensate for issues of variable and cross reactivity. Such a panel should always include p63 and representative cytoplasmic markers, such as CD10, calponin, and SMA. A contemporary H&E recut should be prepared at the same time as the immunostains.
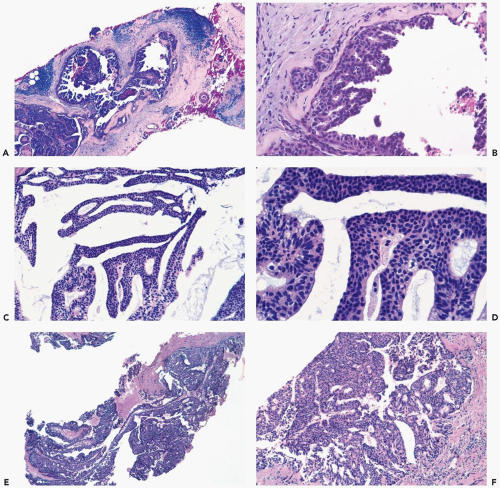 Figure 11.1 Papillary carcinoma. Various growth patterns are represented in these needle core biopsy specimens. A, B. Papillary and micropapillary intraductal carcinoma. Myoepithelial cells represented by oval nuclei arranged parallel to the basement membrane are seen beneath the micropapillary carcinoma in (B). C, D.
Get Clinical Tree app for offline access
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree


|