KEY CONCEPTS
![]() Millions of Americans have osteoarthritis (OA). OA prevalence increases with age, with women more commonly affected than men.
Millions of Americans have osteoarthritis (OA). OA prevalence increases with age, with women more commonly affected than men.
![]() Contributors to OA are systemic (age, genetics, hormonal status, obesity, occupational, or recreational activity) and/or local (injury, overloading of joints, muscle weakness, or joint deformity).
Contributors to OA are systemic (age, genetics, hormonal status, obesity, occupational, or recreational activity) and/or local (injury, overloading of joints, muscle weakness, or joint deformity).
![]() OA is primarily a disease of cartilage that reflects a failure of the chondrocyte to maintain proper balance between cartilage formation and destruction. This leads to loss of cartilage in the joint, local inflammation, pathologic changes in underlying bone, and further damage to cartilage triggered by the affected bone.
OA is primarily a disease of cartilage that reflects a failure of the chondrocyte to maintain proper balance between cartilage formation and destruction. This leads to loss of cartilage in the joint, local inflammation, pathologic changes in underlying bone, and further damage to cartilage triggered by the affected bone.
![]() Manifestations of OA are local, affecting one or a few joints; the knees are most commonly affected, as well as the hips and hands. Osteophytes (bony proliferation of affected joints) are often found, in contrast to the soft tissue swelling of rheumatoid arthritis.
Manifestations of OA are local, affecting one or a few joints; the knees are most commonly affected, as well as the hips and hands. Osteophytes (bony proliferation of affected joints) are often found, in contrast to the soft tissue swelling of rheumatoid arthritis.
![]() Nonpharmacologic therapy is the foundation of the treatment plan for all patients with OA. Nonpharmacologic therapy should be initiated before or concurrently with pharmacologic therapy.
Nonpharmacologic therapy is the foundation of the treatment plan for all patients with OA. Nonpharmacologic therapy should be initiated before or concurrently with pharmacologic therapy.
![]() The most common symptom associated with OA is pain, which leads to decreased function and motion. Pain relief is the primary objective of medication therapy.
The most common symptom associated with OA is pain, which leads to decreased function and motion. Pain relief is the primary objective of medication therapy.
![]() Based on efficacy, safety, and cost considerations, scheduled acetaminophen, up to 4 g/day in divided doses, should be tried initially for pain relief in knee and hip OA. If this fails, topical or oral nonsteroidal antiinflammatory drugs (NSAIDs) are recommended, if there are no contraindications.
Based on efficacy, safety, and cost considerations, scheduled acetaminophen, up to 4 g/day in divided doses, should be tried initially for pain relief in knee and hip OA. If this fails, topical or oral nonsteroidal antiinflammatory drugs (NSAIDs) are recommended, if there are no contraindications.
![]() Strategies to reduce NSAID-induced GI toxicity include the use of nonacetylated salicylates, COX-2 selective inhibitors, or the addition of misoprostol or a proton-pump inhibitor. COX-2 selective inhibitors vary in their ability to prevent GI toxicity, and concomitant use of aspirin largely negates their gastroprotective effects.
Strategies to reduce NSAID-induced GI toxicity include the use of nonacetylated salicylates, COX-2 selective inhibitors, or the addition of misoprostol or a proton-pump inhibitor. COX-2 selective inhibitors vary in their ability to prevent GI toxicity, and concomitant use of aspirin largely negates their gastroprotective effects.
![]() COX-2 selective inhibitors can increase the risk of cardiovascular events. This may be a class effect, but the extent of this risk varies among COX-2 selective inhibitors, and traditional NSAIDs may also pose risks. This hazard, in addition to the GI toxicity possible with all NSAIDs, underscores the importance of using NSAIDs only as needed and after assessing the individual patient’s risk.
COX-2 selective inhibitors can increase the risk of cardiovascular events. This may be a class effect, but the extent of this risk varies among COX-2 selective inhibitors, and traditional NSAIDs may also pose risks. This hazard, in addition to the GI toxicity possible with all NSAIDs, underscores the importance of using NSAIDs only as needed and after assessing the individual patient’s risk.
![]() NSAIDs are associated with GI, renal, cardiovascular, liver, and CNS toxicity. Monitoring with complete blood count, serum creatinine, and hepatic transaminase levels can be valuable in detecting potential toxicity.
NSAIDs are associated with GI, renal, cardiovascular, liver, and CNS toxicity. Monitoring with complete blood count, serum creatinine, and hepatic transaminase levels can be valuable in detecting potential toxicity.
![]() Topical NSAIDs are recommended for patients older than 75 years to decrease the risks of systemic toxicity.
Topical NSAIDs are recommended for patients older than 75 years to decrease the risks of systemic toxicity.
![]() Other agents useful in treating knee OA include tramadol, intraarticular injections of corticosteroids, and duloxetine.
Other agents useful in treating knee OA include tramadol, intraarticular injections of corticosteroids, and duloxetine.
Osteoarthritis (OA) is the most common joint disease and is one of the leading causes of disability in the United States.1–3 Knee OA alone is as important a contributor to disability as cardiovascular disease and more important than other comorbidities in this respect. In 2009, OA was ranked fourth as a cause for hospitalization in the United States.3
The progressive destruction of articular cartilage has long been appreciated in OA, but OA involves the entire diarthrodial joint, including articular cartilage, synovium, capsule, and subchondral bone, with surrounding ligaments and muscles also playing important roles. Changes in structure and function of these tissues produce clinical OA, characterized by joint pain and tenderness, with decreased range of motion, weakness, joint instability, and disability.
This chapter will review the epidemiology, etiology, pathogenesis, and diagnosis of OA. It then will focus on nonpharmacologic and pharmacologic treatments for OA, as well as investigational agents. Because millions of persons take medications for OA, the overall risks posed by these medications require serious consideration, particularly by clinicians who treat or advise patients on drug therapy for OA. This chapter examines the risks and benefits of OA treatments, with emphasis on those individuals who have the highest risk for adverse events, to help clinicians maximize benefits and reduce risks to their patients with OA.
EPIDEMIOLOGY
![]() Twenty-seven million adults in the United States were estimated to have clinical OA in 2005, which represented an increase from 21 million in 1995.4 Prevalence of arthritis-related disability is expected to increase to 11.6 million in the United States by 2020, and an estimated 67 million persons will have OA by 2030.3,5
Twenty-seven million adults in the United States were estimated to have clinical OA in 2005, which represented an increase from 21 million in 1995.4 Prevalence of arthritis-related disability is expected to increase to 11.6 million in the United States by 2020, and an estimated 67 million persons will have OA by 2030.3,5
OA imposes a tremendous cost burden, with total medical costs for OA approximately $336 billion in 2004.6 For 2005, out-of-pocket and insurer expenditures were $36 billion and $149 billion, respectively.7 In 2009, more than 900,000 knee and hip replacements were carried out, at a cost of $42 billion, with most of these surgeries necessitated by OA.3
Prevalence by Age, Sex, and Race
Prevalence estimates for OA vary depending on the age group of interest, gender, ethnic group, and the specific joint involved. Estimates also depend on the specific means by which OA is assessed and documented. Clinical OA is based on physical exam and patient history, whereas radiographic OA is determined by X-ray or other imaging, and symptomatic OA is based on history and physical plus X-ray.1,3,5
OA is more prevalent with increasing age. In the United States, prevalence of radiographic knee OA was estimated as 13.8% for all persons over age 25, but 37.4% for persons age 60 and older.4 Prevalence for symptomatic knee OA was estimated as 4.9% for all persons over age 25, but 12.1% for persons age 60 and older.4 Radiologically confirmed hip OA shows clear trends through all age groups, affecting 1.6% of those between ages 30 and 39, up to a prevalence of 14% in those over 85 years of age.8 Radiographic hand OA is found in 5% of those age 40, but in 65% of those over 80 years of age.9
Prevalence of hip OA is 9% in white populations, but is only 4% for Asian, black, and Indian populations. Before age 50, men are more likely to have OA than women, attributed to higher rates of sports and other injuries.10 Women exhibit a higher prevalence of hip and knee OA than men, and are at especially greater risk for hand OA, with 26% of women and 12% of men over age 70 affected.9 Women are also more likely to have inflammatory OA of the proximal and distal interphalangeal joints of the hands, giving rise to the formation of Bouchard’s and Heberden’s nodes, respectively (Fig. 71-1).
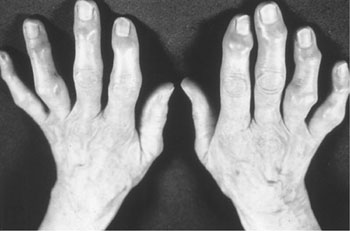
FIGURE 71-1 Heberden’s nodes (distal interphalangeal joint) noted on all fingers and Bouchard’s nodes (proximal interphalangeal joint) noted on most fingers. (From Johnson BE. Arthritis: Osteoarthritis, Gout and Rheumatoid Arthritis. In: South-Paul JE, Matheny SC, Lewis EL, eds. Current Diagnosis and Treatment in Family Medicine. New York: McGraw-Hill, 2004:266, 267.)
Incidence
The incidence of symptomatic OA determined in a large managed care organization was 100 per 100,000 patient-years for hand OA, 88 per 100,000 patient-years for hip OA, and 240 per 100,000 patient-years for knee OA.8 Using a population database of approximately 4 million persons, a recent Canadian study estimated the annual incidence rate of physician-diagnosed OA in men to be 11.6 per 1000 in the period 2003 to 2004, similar to the rate of 11.3 per 1000 estimated for 1996 to 1997.11 For women, the incidence rate significantly increased from 14.7 to 16.7 per 1000 from the period 1996 to 1997 to the period 2003 to 2004. Some of the increase observed for women could have resulted from the aging of the population and women’s longer life expectancy.
ETIOLOGY
![]() The etiology of OA is multifactorial and complex, with development of OA depending on interplay between factors such as genetic predisposition and joint injury.1 Many patients have more than one risk factor for the development of OA. The most common risk factors for the development of OA include age, obesity, gender, occupation, participation in certain sports, history of joint injury or surgery, and genetic predisposition.
The etiology of OA is multifactorial and complex, with development of OA depending on interplay between factors such as genetic predisposition and joint injury.1 Many patients have more than one risk factor for the development of OA. The most common risk factors for the development of OA include age, obesity, gender, occupation, participation in certain sports, history of joint injury or surgery, and genetic predisposition.
Obesity
Obesity is the most important preventable risk factor for OA. This linkage is strongest for knee OA, although hip OA may also be linked with weight. Hand OA does not appear to be linked. As the epidemic of obesity spreads in the United States and in other developed countries, so will the burdens imposed by OA.5 Obesity often precedes OA and contributes to its development, rather than occurring as a result of inactivity from joint pain.12 In an 11-year study of approximately 30,000 Norwegian men and women, obesity significantly increased the risk of developing OA.13 Men who were obese at baseline had a 2.8-fold increased risk of developing knee OA compared with nonobese men, whereas women who were obese at baseline had a 4.4-fold increased risk of developing knee OA compared with nonobese women. Also, there was an increased risk for severe knee OA in obese subjects.
In addition to being a risk factor for OA, obesity is also a predictor for eventual prosthetic joint replacement. In a US study, women who were obese at age 18 were at increased risk of undergoing hip replacement surgery in later life.3 The risk of developing OA increases by approximately 10% with each additional kilogram of weight, and in obese persons without OA, weight loss of even 5 kg (11 pounds) decreases the risk of future knee OA by half.12
Occupation, Sports, and Trauma
OA risk is increased for people in occupations involving excessive mechanical stress. Work that involves prolonged standing, kneeling, squatting, lifting, or moving of heavy objects increases risk of OA. Such occupations include construction, mining, health care assistance, factory work, carpentry, and farming.1,3,14 Repetitive motion also contributes to hand OA, with the dominant hand usually affected.9 Risk for OA depends on the type and intensity of physical activity and whether injury is incurred in the activity. Increased risk of OA is associated with participation in activities such as wrestling, boxing, baseball pitching, cycling, and football, although recreational participants do not have the increased risk seen in the professional athlete.1,3 In the above-referenced study of 30,000 Norwegians, exercise intensity was not associated with any increased risk in the obese subjects compared with those of normal weight.13
Traumatic injury to articular cartilage during sports and other activities or in accidents greatly increases OA risk.1,3,14 Meniscal damage increases the risk of knee OA because of the loss of proper load bearing and shock absorption, increased focal load on cartilage and on subchondral bone. Knee injury in young persons is also an important risk factor for knee OA in old age.1 Quadriceps muscle weakness is also recognized to increase the risk for knee OA, as these muscles are important in maintaining joint stability.10,12 Whether knee malalignment increases risk of developing OA remains unsettled.1 In the person who already has OA, knee malalignment is strongly associated with faster progression of OA.1
Genetic Factors
OA is a complex, polygenic disease. Identification of the genes involved may promote development of agents to prevent OA or to slow or halt its progression.
Genetic influences on OA have been appreciated for many years. Heberden’s nodes are 10 times more prevalent in women than in men, for example, with a twofold higher risk if the woman’s mother had them. Genetic links have been shown with OA of the first metatarsophalangeal joint and with generalized OA. Twin studies indicate that OA can be attributed substantially to genetic factors (39% to 65%, 60%, and 70% for hand, hip, and spine OA, respectively).15 In other twin studies of OA progression, radiographic measurements over 2 years showed that the increased risk for a sibling having radiographic progression if the proband had progression that was threefold for joint space narrowing and 1.5-fold for osteophyte progression.16
One approach OA researchers have used is the candidate gene approach. This hypothesis is based and focuses on genes with known function that could be plausibly linked with the disease. Genome-wide association studies (GWAS) (associating OA with a specific region out of the total human genome, using cases versus controls, offers a powerful approach in seeking the genetic basis for OA.17,18 Using GWAS and candidate gene approaches, possible genetic associations to OA have been found, and some of these appear to code for known proteins that have intriguing connections.18–20 These genes include Col11A1 (extracellular matrix), Chrom 19 (cartilage morphogenesis), MCFL (pain perception), CHST11 (cartilage morphogenesis), GDF5 (TGF-α signaling), and Chrom7Q22. A meta-analysis of GWAS with 6,709 knee OA cases and 44,439 controls revealed that the Chrom7Q22 locus was very highly significantly associated with knee OA. The locus also included six genes that code for proteins known to be expressed in joint tissues.19
For most genes that appear to be linked to OA, the associations have been weak or modest, even if replicable.17 It is quite likely that the genetic risk of developing OA, like many other diseases, may be substantially determined by a combination of modest genetic differences, and this underscores the point that understanding of the genetics and pathology of OA is in its infancy.
PATHOPHYSIOLOGY
OA falls into two major etiologic classes. Primary (idiopathic) OA, the more common type, has no identifiable cause. Secondary OA is that associated with a known cause such as rheumatoid or another inflammatory arthritis, trauma, metabolic or endocrine disorders, and congenital factors.10
The old view of OA as a “wear-and-tear” or degenerative disease, largely focused on joint cartilage, has long been superseded by an appreciation of the dynamic nature of OA and that it represents a failure of the joint and surrounding tissues.21 Some changes in the OA joint may reflect compensatory processes to maintain function in the face of ongoing joint destruction. Not only biomechanical forces but also inflammatory, biochemical, and immunologic factors are involved. An appreciation of the biology and function of normal cartilage can aid in understanding osteoarthritic cartilage and is summarized below.
Normal Cartilage
Function, Structure, and Composition of Cartilage
Articular cartilage possesses viscoelastic properties that provide lubrication with motion, shock absorbency during rapid movements, and load support. In synovial joints, articular cartilage is found between the synovial cavity on one side and a narrow layer of calcified tissue overlying subchondral bone on the other side (Fig. 71-2).22 The layer of cartilage is narrow, with human medial femoral articular cartilage being approximately 2 to 3 mm thick. Despite this, healthy articular cartilage in weight-bearing joints withstands millions of cycles of loading and unloading each year. Cartilage is easily compressed, losing up to 40% of its original height when a load is applied. Compression increases the area of contact and disperses force more evenly to underlying bone, tendons, ligaments, and muscles. In addition, cartilage is almost frictionless, and together with its compressibility, this enables smooth movement in the joint, distributes load across joint tissues to prevent damage, and stabilizes the joint.
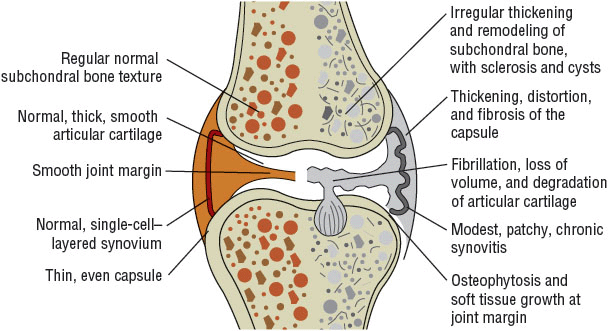
FIGURE 71-2 Characteristics of osteoarthritis in the diarthrodial joint. (Courtesy of Dr. D. Gotlieb.)
Strength, a low coefficient of friction, and compressibility of cartilage derive from its unique structure. Cartilage is a complex, hydrophilic, extracellular matrix (ECM). It is approximately 75% to 85% water and contains 2% to 5% chondrocytes collagen and other proteins, proteoglycans, and long hyaluronic acid (HA) molecules.22 The two major structural components in articular cartilage are type II collagen and aggrecans.23 Type II collagen has a tightly woven triple helical structure, which provides the tensile strength of cartilage. Aggrecan is a proteoglycan linked with HA, providing the long aggrecan molecules a high negative charge. These are squeezed together by surrounding fibrils of type II collagen. The strong electrostatic repulsion of proteoglycans held in close proximity gives cartilage the ability to withstand further compression. Within the cartilage ECM are the chondrocytes, the only cells in cartilage, responsible for laying down all the components of cartilage.
Normal cartilage turnover helps repair and restore cartilage in response to demands of joint loading and during physical activity. In adults, cartilage chondrocyte metabolism is slow and is regulated by growth factors, including bone morphogenetic protein 2, insulin-like growth factor-1, and transforming growth factor, and by catabolism and proteolysis stimulated by matrix metalloproteinases (MMPs), tumor necrosis factor-α (TNF-α), interleukin-1, and other cytokines. Tissue inhibitors of metalloproteinase (TIMP) also contribute to the balance by restraining the catabolic actions of MMPs. If cartilage is injured, chondrocytes react by removing the damaged areas and increasing synthesis of matrix constituents to repair and restore cartilage.23,24
Another component supporting healthy joints are the joint protective mechanisms, such as muscles bridging the joint, sensory receptors in feedback loops to regulate muscle and tendon function, supporting ligaments, and subchondral bone that has shock-absorbent properties.
Finally, it is important to note that adult articular cartilage is avascular, with chondrocytes nourished by synovial fluid. With movement and cyclic loading and unloading of joints, nutrients flow into the cartilage, whereas immobilization reduces nutrient supply. This is one of the reasons that normal physical activity is beneficial for joint health.
Osteoarthritic Cartilage
![]() Important contributors to the development of OA are local mechanical influences, genetic factors, inflammation, and aberrant chondrocyte function leading to loss of articular cartilage.23,24 At a molecular level, OA pathophysiology involves the interplay of dozens, if not hundreds, of extracellular and intracellular molecules with roles including chondrocyte regulation, phenotypic changes, proteolytic degradation of cartilage components, and interactions between articular cartilage, underlying subchondral bone, and the joint synovium.2,5,23–27
Important contributors to the development of OA are local mechanical influences, genetic factors, inflammation, and aberrant chondrocyte function leading to loss of articular cartilage.23,24 At a molecular level, OA pathophysiology involves the interplay of dozens, if not hundreds, of extracellular and intracellular molecules with roles including chondrocyte regulation, phenotypic changes, proteolytic degradation of cartilage components, and interactions between articular cartilage, underlying subchondral bone, and the joint synovium.2,5,23–27
OA most commonly begins with damage to articular cartilage, through trauma or other injury, excess joint loading from obesity or other reasons, or instability or injury of the joint that causes abnormal loading. In response to cartilage damage, chondrocyte activity increases in an attempt to remove and repair the damage. Depending on the degree of damage, the balance between breakdown and resynthesis of cartilage can be lost, and a vicious cycle of increasing breakdown can lead to further cartilage loss and apoptosis of chondrocytes.2,5,23–27 Recent studies have revealed several respects of the very complex nature of OA. For example, expression of hundreds of specific genes are affected by acute experimental injury of human cartilage tissue, that is, injury alters the chondrocyte phenotype.28 Researchers have also shown that within different regions of human OA cartilage obtained at surgery, chondrocyte gene expression from the most damaged areas of cartilage is different from that from less damaged or normal areas.29 Another exciting discovery is that comparative proteomics of articular cartilage from normal persons compared with cartilage from those with OA showed different expression.30
There is increased appreciation of the role of tissues beyond cartilage, within the joint and surrounding it, subchondral bone.24 Subchondral bone undergoes pathologic changes that may precede, coincide with, or follow damage to the articular cartilage. In OA, subchondral bone releases vasoactive peptides and MMPs, and damage to subchondral bone may trigger further damage to articular cartilage.31 Neovascularization and subsequent increased permeability of the adjacent cartilage occur and contributes further to cartilage loss.
Joint space narrowing results from loss of cartilage, which can lead to a painful, deformed joint (Fig. 71-3). Remaining cartilage softens and develops fibrillations (vertical clefts into the cartilage), followed by splitting off of more cartilage and exposure of underlying bone.32 During this time, adjacent subchondral bone undergoes further pathologic changes; cartilage is eroded completely, leaving denuded subchondral bone, which becomes dense, smooth, and glistening (eburnation). A more brittle, stiffer bone results, with decreased weight-bearing ability and development of sclerosis and microfractures. New bone formations, or osteophytes, also appear at joint margins distant from cartilage destruction and are thought to arise from local and humoral factors. There is direct evidence that osteophytes can help stabilize osteoarthritic joints.33
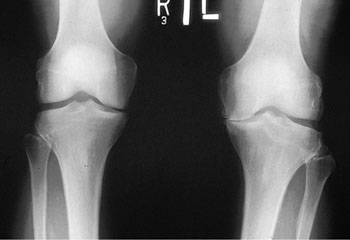
FIGURE 71-3 Plain x-ray films of the knee demonstrating joint space narrowing. (From Johnson BE. Arthritis: Osteoarthritis, Gout and Rheumatoid Arthritis. In: South-Paul JE, Matheny SC, Lewis EL, eds. Current Diagnosis and Treatment in Family Medicine. New York: McGraw-Hill, 2004:267.)
In the joint capsule and synovium, inflammatory changes and pathologic changes can occur.2,22,24–27 Contributors to inflammation may include crystals or cartilage shards in synovial fluid. Other possible players are interleukin-1, prostaglandin E2, TNF-α, and nitric oxide, which are found in synovial fluid. With inflammatory changes in the synovium, effusions and synovial thickening occur.
The pain of OA is not related to the destruction of cartilage but arises from the activation of nociceptive nerve endings within the joint by mechanical and chemical irritants.5 OA pain may result from distension of the synovial capsule by increased joint fluid, microfracture, periosteal irritation, or damage to ligaments, synovium, or the meniscus.
CLINICAL PRESENTATION
Diagnosis
![]() The diagnosis of OA is made through history, physical examination, characteristic radiographic findings, and laboratory testing.10,34,35 The major diagnostic goals are (a) to discriminate between primary and secondary OA and (b) to clarify the joints involved, severity of joint involvement, and response to prior therapies, providing a basis for a treatment plan. The American College of Rheumatology has published traditional diagnostic criteria and “decision trees” for OA diagnosis.35 As with all guidelines, the authors stress these are for assisting the clinician rather than replacing clinical judgment. For example, traditional criteria are as follows: (a) For hip OA, a patient must have pain in the hip and at least two of the following three: an erythrocyte sedimentation rate <20 mm/h (>5.6 μm/s), femoral or acetabular osteophytes on radiography, or joint space narrowing on radiography. This provides a sensitivity of 89% and a specificity of 91%. (b) For a clinical diagnosis of knee OA, a patient must have pain at the knee and osteophytes on radiography plus one of the following: age older than 50 years, morning stiffness no more than 30 minutes, crepitus on motion, bony enlargement, bony tenderness, or palpable warmth. This provides a sensitivity of 95% and a specificity of 69%. The addition of laboratory or radiographic data further improves accuracy of diagnosis. Criteria for hand OA have also been published.36
The diagnosis of OA is made through history, physical examination, characteristic radiographic findings, and laboratory testing.10,34,35 The major diagnostic goals are (a) to discriminate between primary and secondary OA and (b) to clarify the joints involved, severity of joint involvement, and response to prior therapies, providing a basis for a treatment plan. The American College of Rheumatology has published traditional diagnostic criteria and “decision trees” for OA diagnosis.35 As with all guidelines, the authors stress these are for assisting the clinician rather than replacing clinical judgment. For example, traditional criteria are as follows: (a) For hip OA, a patient must have pain in the hip and at least two of the following three: an erythrocyte sedimentation rate <20 mm/h (>5.6 μm/s), femoral or acetabular osteophytes on radiography, or joint space narrowing on radiography. This provides a sensitivity of 89% and a specificity of 91%. (b) For a clinical diagnosis of knee OA, a patient must have pain at the knee and osteophytes on radiography plus one of the following: age older than 50 years, morning stiffness no more than 30 minutes, crepitus on motion, bony enlargement, bony tenderness, or palpable warmth. This provides a sensitivity of 95% and a specificity of 69%. The addition of laboratory or radiographic data further improves accuracy of diagnosis. Criteria for hand OA have also been published.36
CLINICAL PRESENTATION Osteoarthritis
Prognosis
The prognosis for patients with primary OA is variable and depends on the joint involved. If a weight-bearing joint or the spine is involved, considerable morbidity and disability are possible. In the case of secondary OA, the prognosis depends on the underlying cause. Treatment of OA may relieve pain or improve function but does not reverse preexisting damage to the joint.
TREATMENT
Desired Outcome
Management of the patient with OA begins with a diagnosis based on a careful history, physical examination, radiographic findings, and an assessment of the extent of joint involvement. Treatment should be tailored to each individual. Goals are (a) to educate the patient, family members, and caregivers; (b) to relieve pain and stiffness; (c) to maintain or improve joint mobility; (d) to limit functional impairment; and (e) to maintain or improve quality of life.37–39
General Approach to Treatment
Treatment for each OA patient depends on the distribution and severity of joint involvement, comorbid disease states, concomitant medications, and allergies. Management for all individuals with OA should begin with both oral and written patient education, a customized activity and exercise program, and weight loss, if the patient is overweight or obese.37–39
The primary objective of medication is to alleviate pain.37–39 Scheduled acetaminophen, up to 4 g/day in divided doses, should be tried initially (knee, hip), if contraindications are not present. Application of topical NSAIDs over specific joints (knee, hands) and topical capsaicin (hands) are recommended as initial therapy. Nonsteroidal antiinflammatory drugs (NSAIDs) or possibly a cyclooxygenase-2 (COX-2)–selective inhibitor (celecoxib) can be prescribed after careful risk assessment if additional pain control is needed. Intraarticular corticosteroid injections (knee or hip) can relieve pain and are offered concomitantly with oral analgesics or after failed trials of first-line medications, depending on the practitioner’s preference. With centrally acting serotonin reuptake inhibition and analgesic properties, tramadol can also be considered if acetaminophen or topical treatment is ineffective or not tolerated.
Opioid analgesics may be considered if first-line medications are ineffective or pose significant safety concerns in an individual patient. Consideration can also be given to duloxetine or less frequently, HA injections when additional pain control is needed for knee OA. When symptoms are intractable or there is significant loss of function, joint replacement can be appropriate if the patient is a surgical candidate.
There is general agreement that glucosamine and/or chondroitin and topical rubefacients lack uniform efficacy in the treatment of hip and knee OA pain and are not preferred treatment options.
Nonpharmacologic Therapy
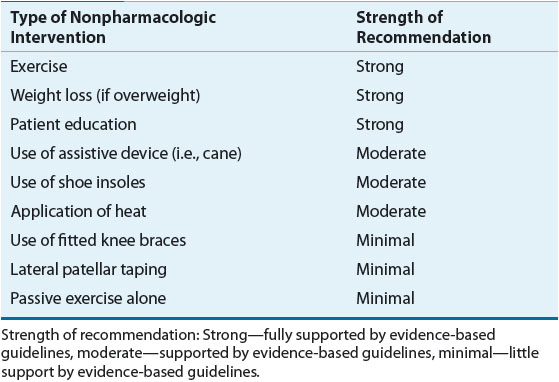
![]() Nonpharmacologic therapy is an integral part of the treatment plan for all patients with OA.37–39 Nonpharmacologic therapy is the only available treatment that has been shown to delay the progression of OA.40 Delaying the progression of OA through active use of nonpharmacologic therapy is critical to prevent future functional impairment. Patient-specific characteristics such as the number and location of affected joints, degree of functional impairment, body mass index, motivation, and overall health status determine which nonpharmacologic therapies should be offered. Nonpharmacologic therapy should be ongoing treatment for all patients, even those who require pharmacologic therapy for pain control (Table 71-1).
Nonpharmacologic therapy is an integral part of the treatment plan for all patients with OA.37–39 Nonpharmacologic therapy is the only available treatment that has been shown to delay the progression of OA.40 Delaying the progression of OA through active use of nonpharmacologic therapy is critical to prevent future functional impairment. Patient-specific characteristics such as the number and location of affected joints, degree of functional impairment, body mass index, motivation, and overall health status determine which nonpharmacologic therapies should be offered. Nonpharmacologic therapy should be ongoing treatment for all patients, even those who require pharmacologic therapy for pain control (Table 71-1).
Patient Education
The first step in OA treatment is patient education about the disease process, the extent of OA, the prognosis, and treatment options. Education is paramount in that OA is often seen as a wear-and-tear disease, an inevitable consequence of aging for which nothing helps. Even worse, patients may resort to the use of alternative but unproven medications or quackery. Organizations such as the Arthritis Foundation provide a wealth of educational information for patients regarding OA, OA medications, local clinics, and agencies offering physical and economic assistance. Exercise, weight loss, and nutritional information are also available. Most educational information is readily available online for patient use.
The benefits of patient education have been documented in a variety of programs.41 These programs are provided across a wide spectrum of delivery methods: from trained volunteers using telephone calls to group sessions for patient support to one-on-one educational sessions with physical therapists or nurse educators. While nearly all of these delivery methods are effective, cost of delivery is highly variable. Long-term cost-effectiveness is very important for sustainability of these patient education programs.
Weight Loss
Excess weight increases the biomechanical load on weight-bearing joints and is the single best predictor of need for eventual joint replacement.42,43 Weight loss of amounts as small as 4% of body weight can lessen OA pain in the knee.44 Greater amounts of weight loss, especially when associated with regular exercise, improve joint function and substantially lessen pain.42,45 At least one randomized controlled trial has demonstrated improvement in pain and self-reported physical function using a combination of modest weight loss (5%) and exercise.46 Patients with appropriate indications for bariatric surgery have significant improvement in joint function and pain associated with the subsequent weight loss.47 A large weight-loss and activity trial is under way, and the Intensive Diet and Exercise for Arthritis trial (IDEA) will address weight-related joint loading and inflammatory biomarkers.48 Weight loss requires a motivated patient, but it should be encouraged and supported for all obese and overweight patients with OA. Effective behavior change strategies should be employed to promote weight loss in patients with OA.40
Exercise
Exercise programs can improve joint function and can decrease disability, pain, and analgesic use by OA patients.40,44,46 Isometric exercise is preferred over isotonic exercise because the latter can aggravate affected joints. Exercises can be taught and then observed before the patient exercises at home. The frequency, types of exercise, and setting of exercise are still uncertain, but patients who exercise at least two to three times per week with a variety of exercises (>8 types) have improved outcomes.49 The patient should be instructed to decrease the number of repetitions if severe pain develops with exercise.
Some regular exercise should be encouraged for all patients with OA.39 With weak or deconditioned muscles, the load is transmitted excessively to the joints; weight-bearing activities can exacerbate symptoms. Many patients fear that exercise will promote further joint damage and avoid exercise as a means to protect the joint. However, avoidance of regular exercise by those with hip or knee OA leads to further deconditioning and/or weight gain. This leads to more pain and impaired joint function, promoting a downward spiral of disability. Current research regarding exercise revolves around effective strategies to promote sustained behavior change in patients.40 A program of patient education, muscle stretching and strengthening, and supervised walking can improve physical function and decrease pain for patients with knee OA.46
Referral to the physical and/or occupational therapist is especially helpful for developing a customized exercise plan for patients with functional disabilities. The therapist can assess muscle strength and joint stability and recommend exercises and assistive and orthotic devices, such as canes, walkers, braces, heel cups, splints, or insoles for use during exercise or daily activities. Heat or cold treatments help to maintain and restore joint range of motion and to reduce pain and muscle spasms. Warm baths or warm water soaks may decrease pain and stiffness. Heating pads should be used with caution, especially in the elderly. Patients should be warned not to fall asleep on the heat source or to lie on it for more than brief periods to avoid burns.
Surgery
Surgery can be recommended for OA patients with functional disability and/or severe pain unresponsive to conservative therapy. Criteria for total joint replacement (arthroplasty) of the knee and hip have been developed, although there is substantial overlap in eligibility criteria.50,51
Few randomized, controlled trials are available comparing total joint arthroplasty with other treatment modalities. Although total knee arthroplasty can decrease pain and improve function for many patients, about 20% experience little or no improvement in pain, disability, and/or quality of life.52 These findings coupled with overlapping indications for the procedure and the expected increase in the number of patients with OA lend some urgency to the need for controlled trials to evaluate the outcome of joint replacement with other treatment modalities.
The MEDIC-study of total knee replacement plus physical and medical therapy or treatment with physical and medical therapy alone is currently enrolling patients in hopes of answering some of these important questions about the role of surgery in the treatment of knee OA.52
Total joint arthroplasty is responsible for a large portion of the direct medical costs associated with OA in the United States. The cost-effectiveness of total knee arthroplasty has been evaluated for a Medicare-age population.53 Calculations were based on Medicare claims data and costs and outcomes data. Cost projections were calculated for lifetime costs as well as quality-adjusted life expectancy (QALE) for different risk populations and across low-volume to high-volume hospitals. Although total knee arthroplasty was found to be cost-effective across hospital settings and patient risk categories, the procedure was found to be most cost-effective when performed in high-volume centers.
Other surgical options are also available. Arthrodesis (joint fusion) can reduce pain but will restrict motion and may be appropriate for smaller joints that are causing intractable pain. For patients with mild knee OA, an osteotomy (removal of bony tissue) may correct the misalignment of genu varum (“bowlegged” knees) or genu valgum (“knock-knees”). In addition, osteotomies of the pelvis or femur can ameliorate joint misalignment in hip OA, subsequently slowing progression of disease. Knee arthroscopy or lavage appear to be equivalent to sham surgery and are not recommended.37 Experimental but potentially restorative approaches involve soft-tissue grafts, chondrocyte transplantation, gene therapy, and use of growth factors or artificial matrices.54 Cartilage-restoration approaches are investigational, and results regarding pain control and joint function have been mixed.
Pharmacologic Therapy
![]() Drug therapy in OA is targeted at relief of pain. OA is commonly seen in older individuals who have other medical conditions, and OA treatment is often long term. As such, a conservative approach to drug treatment, focusing on the needs of the individual patient, is warranted (see Figs. 71-4 and 71-5).37–39 Even when pharmacologic therapy is initiated, appropriate nondrug therapies should be continued and reinforced. Specific drug therapy recommendations depend on which joint(s) are affected, response to previous trials of medication, and patient comorbidities.
Drug therapy in OA is targeted at relief of pain. OA is commonly seen in older individuals who have other medical conditions, and OA treatment is often long term. As such, a conservative approach to drug treatment, focusing on the needs of the individual patient, is warranted (see Figs. 71-4 and 71-5).37–39 Even when pharmacologic therapy is initiated, appropriate nondrug therapies should be continued and reinforced. Specific drug therapy recommendations depend on which joint(s) are affected, response to previous trials of medication, and patient comorbidities.
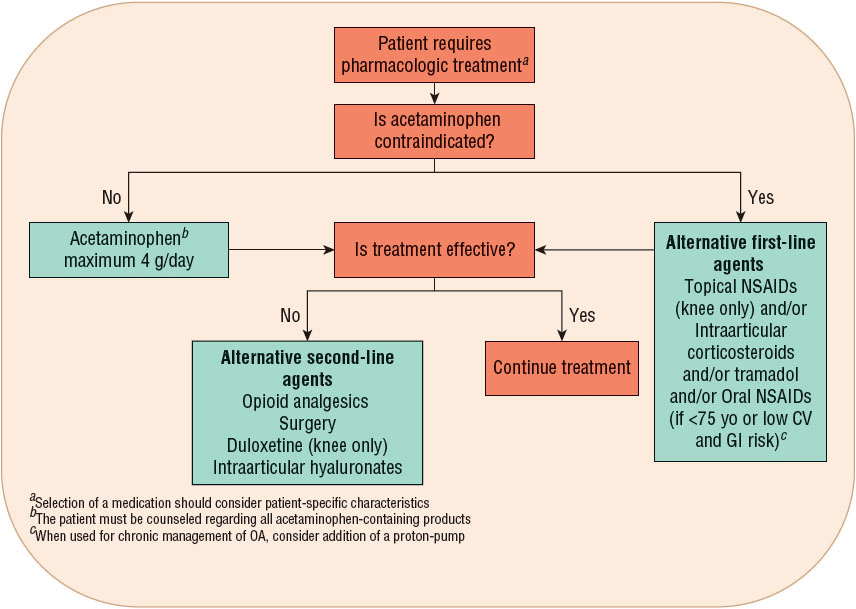
FIGURE 71-4 Treatment recommendations for knee and hip osteoarthritis.37–39 (CV, cardiovascular; NSAID, nonsteroidal antiinflammatory drug.)