44
Micronutrients: Vitamins & Minerals
OBJECTIVES
After studying this chapter, you should be able to:
![]() Describe how reference intakes for vitamins and minerals are determined and explain why reference intakes published by different national and international authorities differ.
Describe how reference intakes for vitamins and minerals are determined and explain why reference intakes published by different national and international authorities differ.
![]() Define a vitamin and describe the metabolism, principal functions, deficiency diseases associated with inadequate intake, and the toxicity of excessive intakes of the vitamins.
Define a vitamin and describe the metabolism, principal functions, deficiency diseases associated with inadequate intake, and the toxicity of excessive intakes of the vitamins.
![]() Explain why mineral salts are required in the diet.
Explain why mineral salts are required in the diet.
BIOMEDICAL IMPORTANCE
Vitamins are a group of organic nutrients, required in small quantities for a variety of biochemical functions that, generally, cannot be synthesized by the body and must therefore be supplied in the diet.
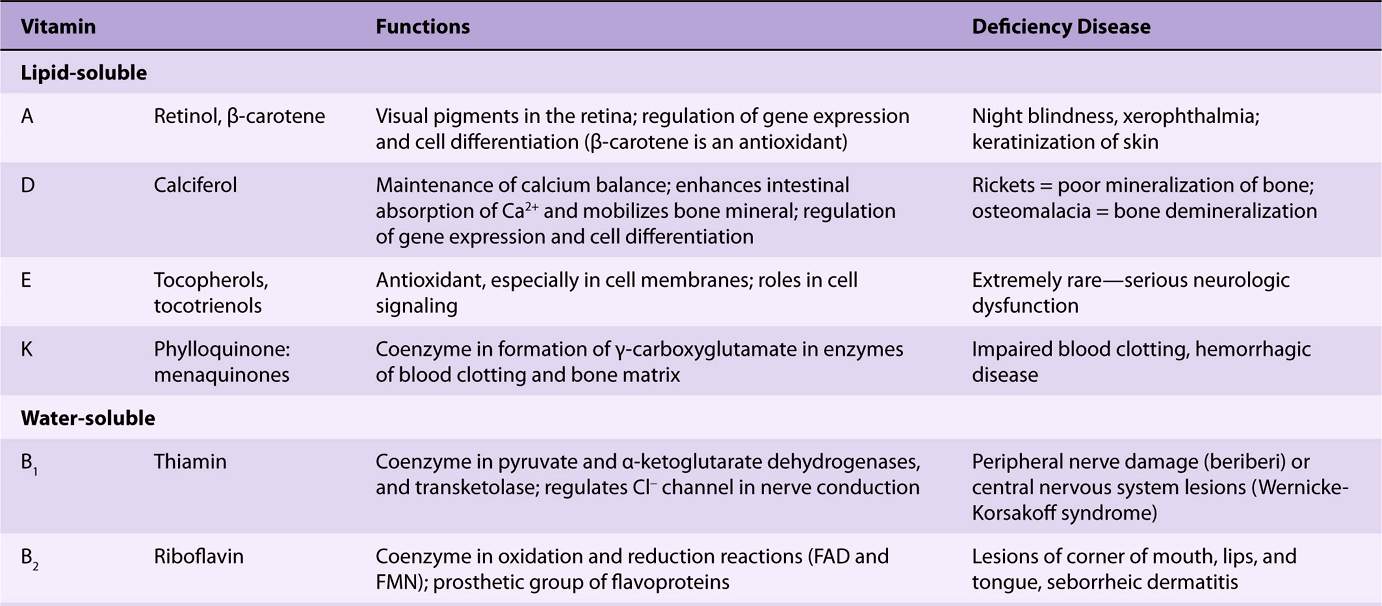
The lipid-soluble vitamins are hydrophobic compounds that can be absorbed efficiently only when there is normal fat absorption. Like other lipids, they are transported in the blood in lipoproteins or attached to specific binding proteins. They have diverse functions—eg, vitamin A, vision and cell differentiation; vitamin D, calcium and phosphate metabolism, and cell differentiation; vitamin E, antioxidant; and vitamin K, blood clotting. As well as dietary inadequacy, conditions affecting the digestion and absorption of the lipid-soluble vitamins, such as a very low fat diet, steatorrhea and disorders of the biliary system, can all lead to deficiency syndromes, including night blindness and xerophthalmia (vitamin A); rickets in young children and osteomalacia in adults (vitamin D); neurologic disorders and hemolytic anemia of the newborn (vitamin E); and hemorrhagic disease of the newborn (vitamin K). Toxicity can result from excessive intake of vitamins A and D. Vitamin A and the carotenes (many of which are precursors of vitamin A), and vitamin E are antioxidants (Chapter 45) and have possible roles in prevention of atherosclerosis and cancer.
The water-soluble vitamins are vitamins B and C; they function mainly as enzyme cofactors. Folic acid acts as a carrier of one-carbon units. Deficiency of a single vitamin of the B complex is rare since poor diets are most often associated with multiple deficiency states. Nevertheless, specific syndromes are characteristic of deficiencies of individual vitamins, eg, beriberi (thiamin); cheilosis, glossitis, seborrhea (riboflavin); pellagra (niacin); megaloblastic anemia, methylmalonic aciduria, and pernicious anemia (vitamin B); megaloblastic anemia (folic acid); and scurvy (vitamin C).
Inorganic mineral elements that have a function in the body must be provided in the diet. When the intake is insufficient, deficiency signs may arise, eg, anemia (iron), and cretinism and goiter (iodine). Excessive intakes may be toxic.
The Determination of Micronutrient Requirements Depends on the Criteria of Adequacy Chosen
For any nutrient, there is a range of intakes between that which is clearly inadequate, leading to clinical deficiency disease, and that which is so much in excess of the body’s metabolic capacity that there may be signs of toxicity. Between these two extremes is a level of intake that is adequate for normal health and the maintenance of metabolic integrity. Individuals do not all have the same requirement for nutrients, even when calculated on the basis of body size or energy expenditure. There is a range of individual requirements of up to 25% around the mean. Therefore, in order to assess the adequacy of diets, it is necessary to set a reference level of intake high enough to ensure that no one either suffers from deficiency or is at risk of toxicity. If it is assumed that individual requirements are distributed in a statistically normal fashion around the observed mean requirement, then a range of ±2 × the standard deviation (SD) around the mean includes the requirements of 95% of the population. Reference or recommended intakes are therefore set at the average requirement plus 2 × SD, and so meet or exceed the requirements of 97.5% of the population.
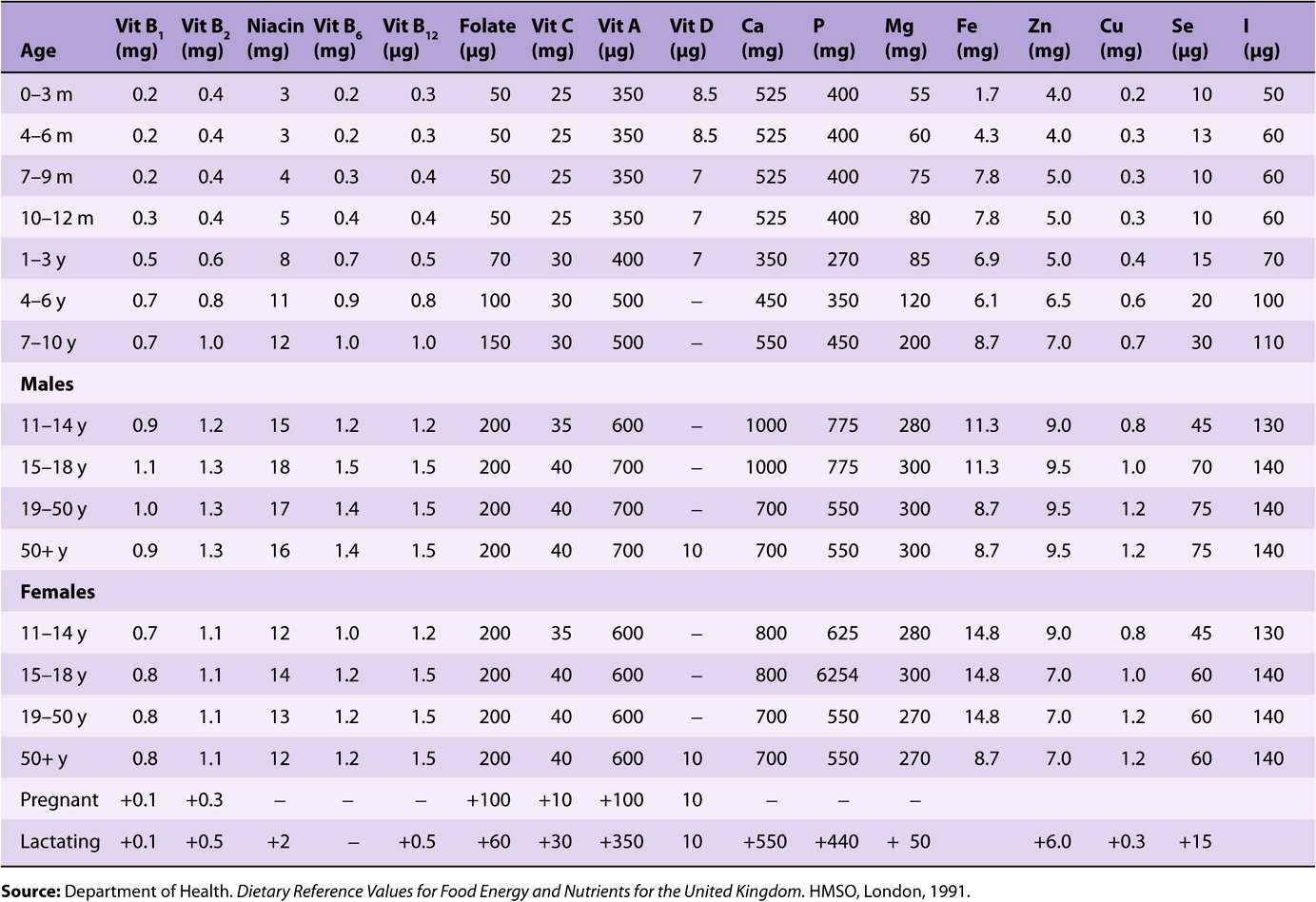
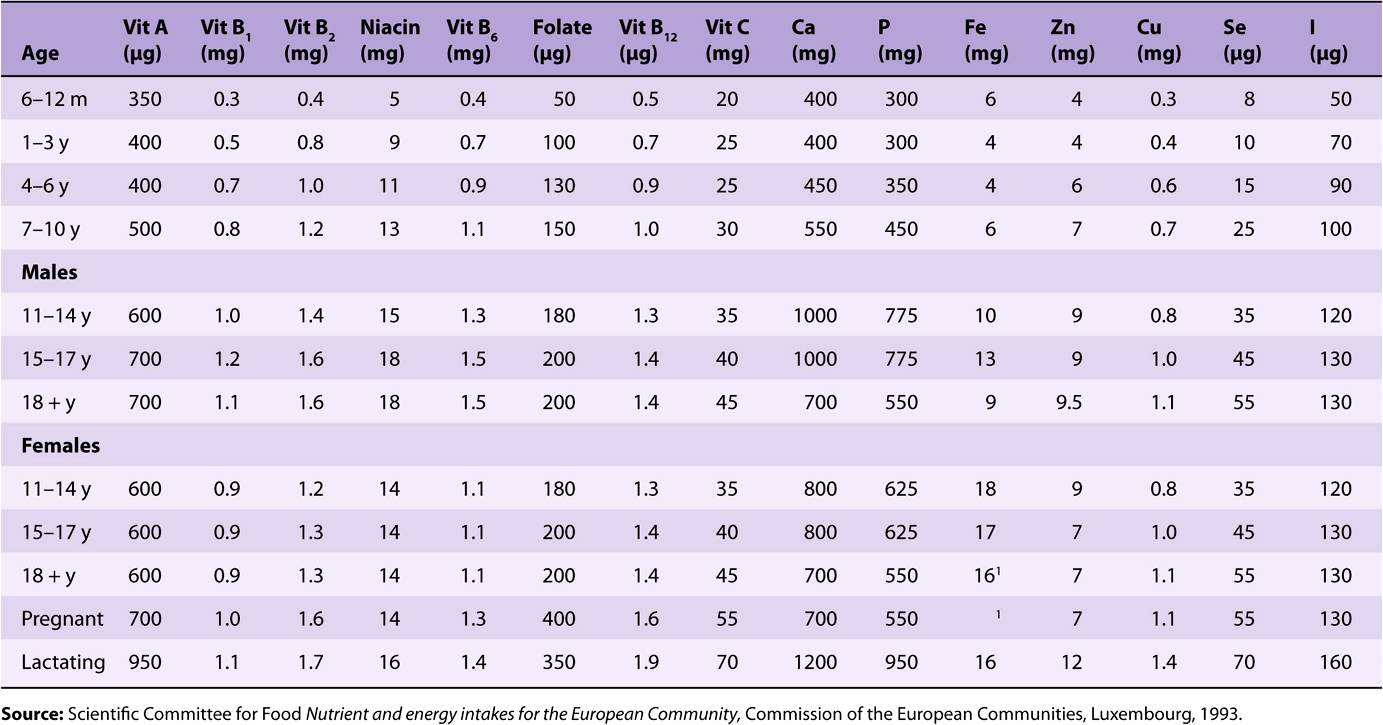
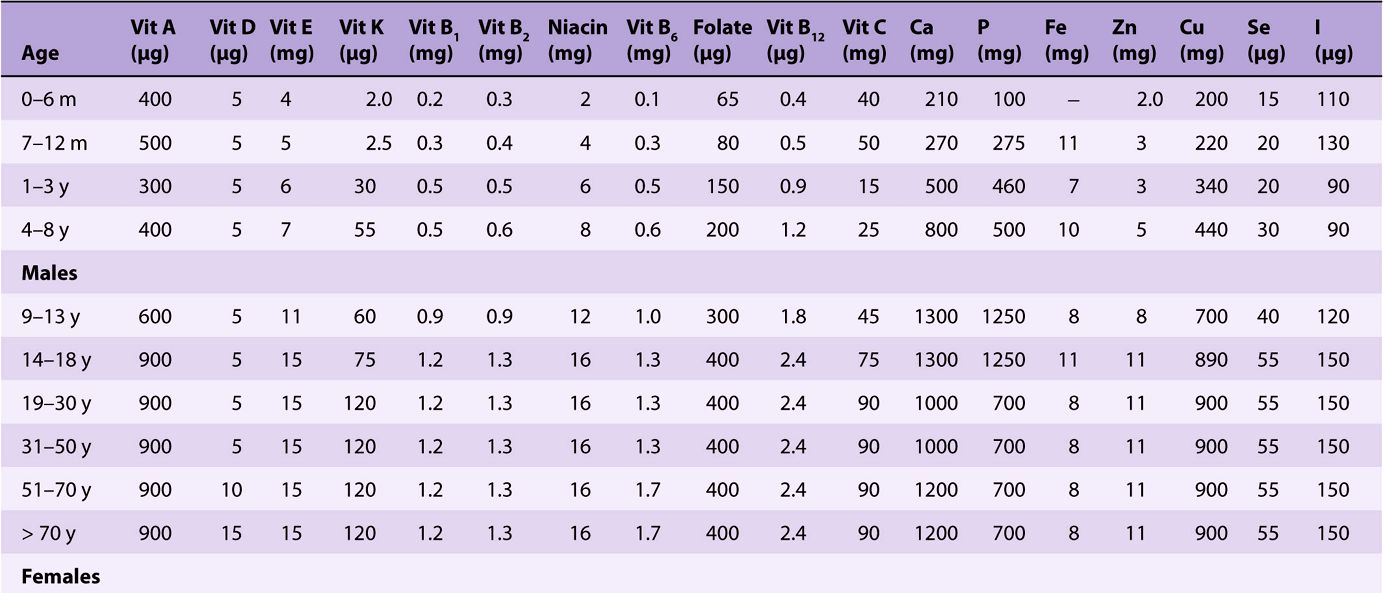
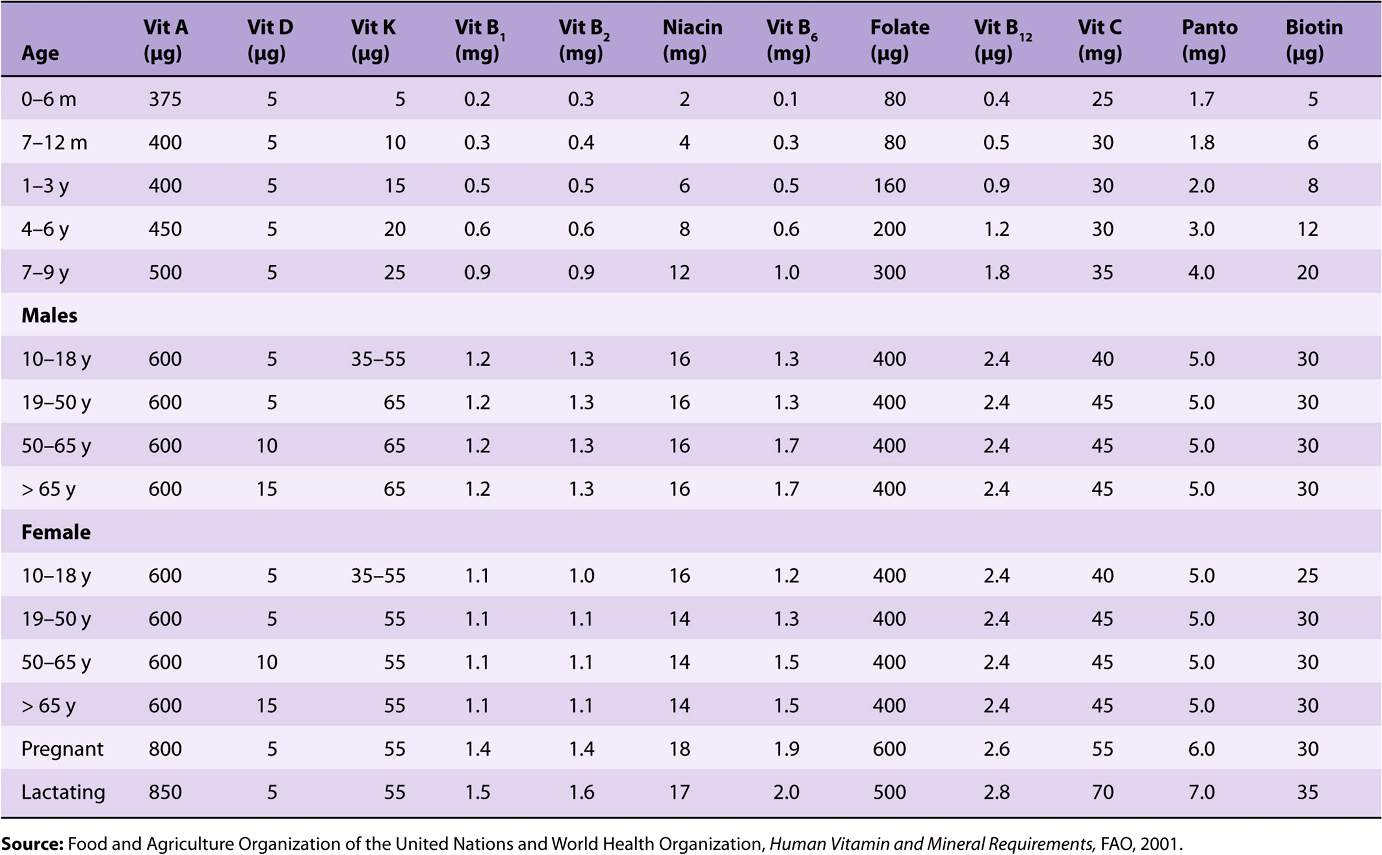
Reference and recommended intakes of vitamins and minerals published by different national and international authorities (Tables 44-1 to 44-4) differ because of different interpretations of the available data, and the availability of new experimental data in more recent publications.
TABLE 44–1 Reference Nutrient Intakes of Vitamins and Minerals, UK 1991
TABLE 44–2 Population Reference Intakes of Vitamins and Minerals, European Union, 1993
TABLE 44–3 Recommended Dietary Allowances and Acceptable Intakes for Vitamins and Minerals, USA and Canada, 1997-2001
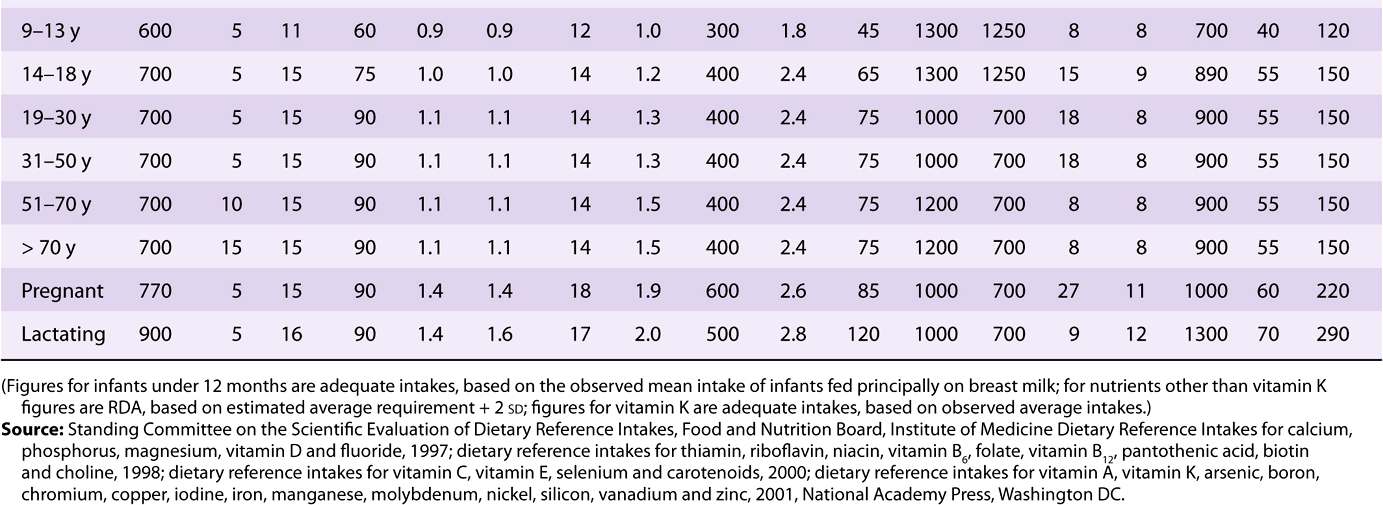
TABLE 44–4 Recommended Nutrient Intakes for Vitamins, FAO 2001
THE VITAMINS ARE A DISPARATE GROUP OF COMPOUNDS WITH A VARIETY OF METABOLIC FUNCTIONS
A vitamin is defined as an organic compound that is required in the diet in small amounts for the maintenance of normal metabolic integrity. Deficiency causes a specific disease, which is cured or prevented only by restoring the vitamin to the diet (Table 44-5). However, vitamin D, which is formed in the skin from 7-dehydrocholesterol on exposure to sunlight, and niacin, which can be formed from the essential amino acid tryptophan, do not strictly comply with this definition.
TABLE 44–5 The Vitamins

LIPID-SOLUBLE VITAMINS
TWO GROUPS OF COMPOUNDS HAVE VITAMIN A ACTIVITY
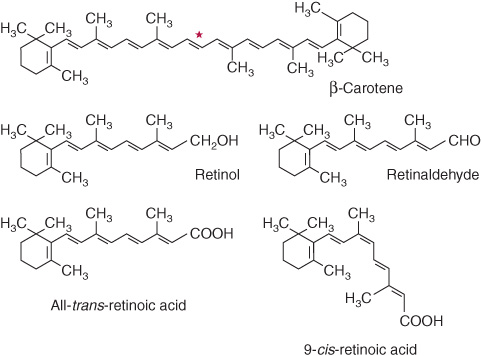
Retinoids comprise retinol, retinaldehyde, and retinoic acid (preformed vitamin A, found only in foods of animal origin); carotenoids, found in plants, are composed of carotenes and related compounds; many are precursors of vitamin A, as they can be cleaved to yield retinaldehyde, then retinol and retinoic acid (Figure 44–1). The α-, β-, and γ-carotenes and cryptoxanthin are quantitatively the most important provitamin A carotenoids. β-Carotene and other provitamin A carotenoids are cleaved in the intestinal mucosa by carotene dioxygenase, yielding retinaldehyde, which is reduced to retinol, esterified and secreted in chylomicrons together with esters formed from dietary retinol. The intestinal activity of carotene dioxygenase is low, so that a relatively large proportion of ingested β-carotene may appear in the circulation unchanged. While the principal site of carotene dioxygenase attack is the central bond of β-carotene, asymmetric cleavage may also occur, leading to the formation of 8′-, 10′-, and 12′-apo-carotenals, which are oxidized to retinoic acid, but cannot be used as sources of retinol or retinaldehyde.
FIGURE 44–1 β-Carotene and the major vitamin A vitamers. Asterisk shows the site of cleavage of β-carotene by carotene dioxygenase, to yield retinaldehyde.
Although it would appear that one molecule of β-carotene should yield two of retinol, this is not so in practice; 6 μg of β-carotene is equivalent to 1 μg of preformed retinol. The total amount of vitamin A in foods is therefore expressed as micrograms of retinol equivalents = μg preformed vitamin A + 1/6 × μg β-carotene + 1/12 × μg other provitamin A carotenoids. Before pure vitamin A was available for chemical analysis, the vitamin A content of foods was determined by biological assay and the results expressed as international units (iu). 1 iu = 0.3 μg retinol; 1 μg retinol = 3.33 iu. Although obsolete, iu is sometimes still used in food labeling. In 2001 The USA/Canadian Dietary Reference Values report introduced the term retinol activity equivalent to take account of the incomplete absorption and metabolism of carotenoids; 1 ![]() μg all-trans-retinol, 12 μg β-carotene, 24 μg α-carotene or β-cryptoxanthin. On this basis, 1 iu of vitamin A activity is equal to 3.6 μg β-carotene or 7.2 μg of other provitamin A carotenoids.
μg all-trans-retinol, 12 μg β-carotene, 24 μg α-carotene or β-cryptoxanthin. On this basis, 1 iu of vitamin A activity is equal to 3.6 μg β-carotene or 7.2 μg of other provitamin A carotenoids.
Vitamin A Has a Function in Vision
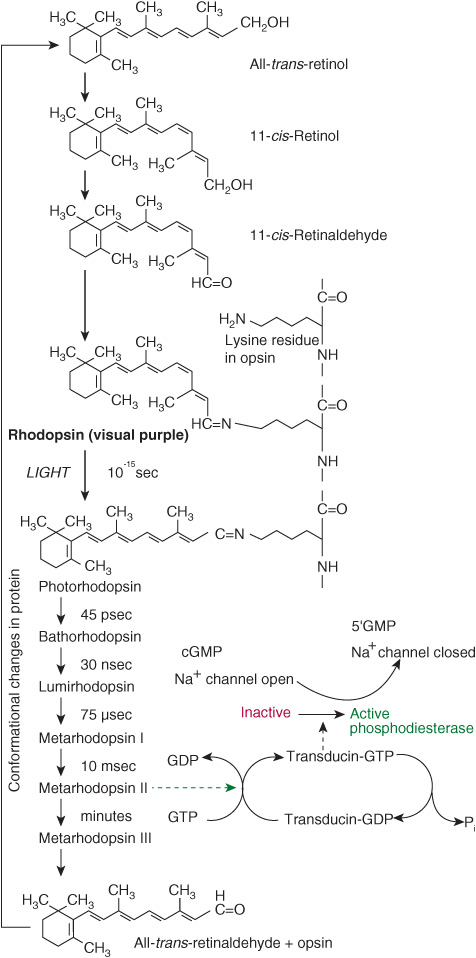
In the retina, retinaldehyde functions as the prosthetic group of the light-sensitive opsin proteins, forming rhodopsin (in rods) and iodopsin (in cones). Any one cone cell contains only one type of opsin and is sensitive to only one color. In the pigment epithelium of the retina, all-trans-retinol is isomerized to 11-cis-retinol and oxidized to 11-cis-retinaldehyde. This reacts with a lysine residue in opsin, forming the holoprotein rhodopsin. As shown in Figure 44–2, the absorption of light by rhodopsin causes isomerization of the retinaldehyde from 11-cis to all-trans, and a conformational change in opsin. This results in the release of retinaldehyde from the protein, and the initiation of a nerve impulse. The formation of the initial excited form of rhodopsin, bathorhodopsin, occurs within picoseconds of illumination. There is then a series of conformational changes leading to the formation of metarhodopsin II, which initiates a guanine nucleotide amplification cascade and then a nerve impulse. The final step is hydrolysis to release all-trans-retinaldehyde and opsin. The key to initiation of the visual cycle is the availability of 11-cis-retinaldehyde, and hence vitamin A. In deficiency, both the time taken to adapt to darkness and the ability to see in poor light are impaired.
FIGURE 44–2 The role of retinaldehyde in the visual cycle.
Retinoic Acid Has a Role in the Regulation of Gene Expression and Tissue Differentiation
A major role of vitamin A is in the control of cell differentiation and turnover. All-trans-retinoic acid and 9-cis-retinoic acid (Figure 44–1) regulate growth, development, and tissue differentiation; they have different actions in different tissues. Like the thyroid and steroid hormones and vitamin D, retinoic acid binds to nuclear receptors that bind to response elements of DNA and regulate the transcription of specific genes. There are two families of nuclear retinoid receptors: the retinoic acid receptors (RAR) bind all-trans-retinoic acid or 9-cis-retinoic acid, and the retinoid X receptors (RXR) bind 9-cis-retinoic acid. Retinoid X receptors also form dimers with vitamin D, thyroid, and other a nuclear acting hormone receptors. Deficiency of vitamin A impairs vitamin D function because of lack of 9-cis-retinoic acid to form receptor dimers, while excessive vitamin A also impairs vitamin D function, because of formation of RXR homodimers, meaning that there are not enough RXR available to form heterodimers with the vitamin D receptor.
Vitamin A Deficiency Is a Major Public Health Problem Worldwide
Vitamin A deficiency is the most important preventable cause of blindness. The earliest sign of deficiency is a loss of sensitivity to green light, followed by impairment to adapt to dim light, followed by night blindness. More prolonged deficiency leads to xerophthalmia: keratinization of the cornea and blindness. Vitamin A also has an important role in differentiation of immune system cells, and even mild deficiency leads to increased susceptibility to infectious diseases. Also, the synthesis of retinol binding protein is reduced in response to infection (it is a negative acute phase protein), decreasing the circulating concentration of the vitamin, and further impairing immune responses.
Vitamin A Is Toxic in Excess
There is only a limited capacity to metabolize vitamin A, and excessive intakes lead to accumulation beyond the capacity of binding proteins, so that unbound vitamin A causes tissue damage. Symptoms of toxicity affect the central nervous system (headache, nausea, ataxia, and anorexia, all associated with increased cerebrospinal fluid pressure); the liver (hepatomegaly with histologic changes and hyperlipidemia); calcium homeostasis (thickening of the long bones, hypercalcemia, and calcification of soft tissues); and the skin (excessive dryness, desquamation, and alopecia).
VITAMIN D IS REALLY A HORMONE
Vitamin D is not strictly a vitamin since it can be synthesized in the skin, and under most conditions that is the major source of the vitamin. Only when sunlight exposure is inadequate is a dietary source required. Its main function is in the regulation of calcium absorption and homeostasis; most of its actions are mediated by way of nuclear receptors that regulate gene expression. It also has a role in regulating cell proliferation and differentiation. There is evidence that intakes considerably higher than are required to maintain calcium homeostasis reduce the risk of insulin resistance, obesity and the metabolic syndrome, as well as various cancers. Deficiency, leading to rickets in children and osteomalacia in adults, continues to be a problem in northern latitudes, where sunlight exposure is inadequate.
Vitamin D Is Synthesized in the Skin
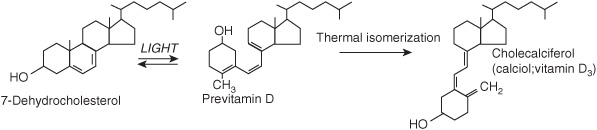
7-Dehydrocholesterol (an intermediate in the synthesis of cholesterol that accumulates in the skin) undergoes a nonenzymic reaction on exposure to ultraviolet light, yielding previtamin D (Figure 44–3). This undergoes a further reaction over a period of hours to form cholecalciferol, which is absorbed into the bloodstream. In temperate climates, the plasma concentration of vitamin D is highest at the end of summer and lowest at the end of winter. Beyond latitudes about 40° north or south, there is very little ultraviolet radiation of the appropriate wavelength in winter.
FIGURE 44–3 The synthesis of vitamin D in the skin.
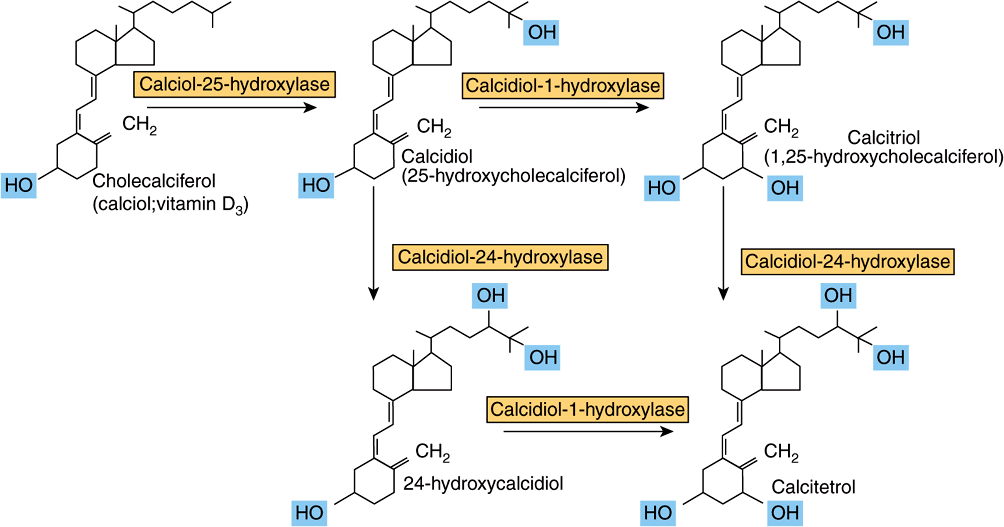
Vitamin D Is Metabolized to the Active Metabolite, Calcitriol, in Liver & Kidney
Cholecalciferol, either synthesized in the skin or from food, undergoes two hydroxylations to yield the active metabolite, 1,25-dihydroxyvitamin D or calcitriol (Figure 44–4). Ergocalciferol from fortified foods undergoes similar hydroxylation to yield ercalcitriol. In the liver, cholecalciferol is hydroxylated to form the 25-hydroxy-derivative, calcidiol. This is released into the circulation bound to a vitamin D binding globulin, which is the main storage form of the vitamin. In the kidney, calcidiol undergoes either 1-hydroxylation to yield the active metabolite 1,25-dihydroxyvitamin D (calcitriol), or 24-hydroxylation to yield a probably inactive metabolite, 24,25-dihydroxyvitamin D (24-hydroxycalcidiol).
FIGURE 44–4 Metabolism of vitamin D.
Vitamin D Metabolism Is Both Regulated by and Regulates Calcium Homeostasis
The main function of vitamin D is in the control of calcium homeostasis, and in turn, vitamin D metabolism is regulated by factors that respond to plasma concentrations of calcium and phosphate. Calcitriol acts to reduce its own synthesis by inducing the 24-hydroxylase and repressing the 1-hydroxylase in the kidney. The principal function of vitamin D is to maintain the plasma calcium concentration. Calcitriol achieves this in three ways: it increases intestinal absorption of calcium; it reduces excretion of calcium (by stimulating resorption in the distal renal tubules); and it mobilizes bone mineral. In addition, calcitriol is involved in insulin secretion, synthesis and secretion of parathyroid and thyroid hormones, inhibition of production of interleukin by activated T-lymphocytes and of immunoglobulin by activated B-lymphocytes, differentiation of monocyte precursor cells, and modulation of cell proliferation. In most of these actions, it acts like a steroid hormone, binding to nuclear receptors and enhancing gene expression, although it also has rapid effects on calcium transporters in the intestinal mucosa. For further details of the role of calcitriol in calcium homeostasis, see Chapter 47.
Higher Intakes of Vitamin D may be Beneficial
There is growing evidence that higher vitamin D status is protective against various cancers, including prostate and colorectal cancer, and also against prediabetes and the metabolic syndrome. Desirable levels of intake may be considerably higher than current reference intakes, and could certainly not be met from unfortified foods. While increased sunlight exposure would meet the need, it carries the risk of developing skin cancer.
Vitamin D Deficiency Affects Children & Adults
In the vitamin D deficiency disease rickets, the bones of children are undermineralized as a result of poor absorption of calcium. Similar problems occur as a result of deficiency during the adolescent growth spurt. Osteomalacia in adults results from the demineralization of bone, especially in women who have little exposure to sunlight, especially after several pregnancies. Although vitamin D is essential for prevention and treatment of osteomalacia in the elderly, there is little evidence that it is beneficial in treating osteoporosis.
Vitamin D Is Toxic in Excess
Some infants are sensitive to intakes of vitamin D as low as 50 μg/day, resulting in an elevated plasma concentration of calcium. This can lead to contraction of blood vessels, high blood pressure, and calcinosis—the calcification of soft tissues. Although excess dietary vitamin D is toxic, excessive exposure to sunlight does not lead to vitamin D poisoning, because there is a limited capacity to form the precursor, 7-dehydrocholesterol, and prolonged exposure of previtamin D to sunlight leads to formation of inactive compounds.
VITAMIN E DOES NOT HAVE A PRECISELY DEFINED METABOLIC FUNCTION
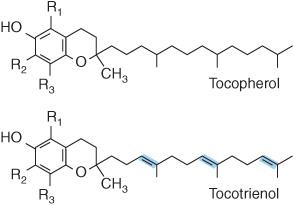
No unequivocal unique function for vitamin E has been defined. It acts as a lipid-soluble antioxidant in cell membranes, where many of its functions can be provided by synthetic antioxidants, and is important in maintaining the fluidity of cell membranes. It also has a (relatively poorly defined) role in cell signaling. Vitamin E is the generic descriptor for two families of compounds, the tocopherols and the tocotrienols (Figure 44–5). The different vitamers have different biologic potency; the most active is D-α-tocopherol, and it is usual to express vitamin E intake in terms of milligrams D-α-tocopherol equivalents. Synthetic DL-a-tocopherol does not have the same biologic potency as the naturally occurring compound.
FIGURE 44–5 Vitamin E vitamers. In α-tocopherol and tocotrienol R1, R2, and R3 are all—CH3 groups. In the β-vitamers R2 is H, in the γ-vitamers R1 is H, and in the δ-vitamers R1 and R2 are both H.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree