Metastatic Tumors of the Ovary
Tumors of the Pancreas, Biliary Tract, and Liver 703
Tumors of the Urinary Tract 706
Pulmonary and Mediastinal Tumors 707
General Features
The ovaries are involved by metastatic tumors more often than any other organ in the female genital tract, regardless of the location of the primary neoplasm.1 Tumors that extend to the ovary directly from adjacent organs or tissues are also included in the category of secondary tumors; however, most ovarian carcinomas associated with uterine cancers of similar histologic type are independent primary neoplasms (see Chapter 27).
Metastatic tumors to the ovary are common and occur in approximately 30% of women dying of cancer. About 6–7% of all adnexal masses found during physical examination are actually metastatic ovarian tumors, frequently unsuspected by gynecologists.2–4 The metastasis often masquerades as a primary ovarian tumor and may even be the initial manifestation of the patient’s cancer. Pathologists also tend to mistake metastatic tumors for primary ovarian neoplasms, even after microscopic examination, because the existence of a concurrent or prior tumor in another organ is either not known or disregarded. Also, all metastatic tumors may have a functioning ovarian stroma with clinical or pathologic evidence of hyperestrogenism simulating a primary ovarian tumor.
The frequencies of various sites of origin of secondary ovarian tumors differ among different countries according to the incidence of various cancers therein. Carcinomas of the colon, stomach, breast, and endometrium, as well as lymphomas and leukemias, account for the vast majority of cases.5 Rectal or sigmoid colon cancer accounts for 75% of the metastatic intestinal tumors to the ovary, and probably constitutes the most common cause of misdiagnosis.2–5 The Krukenberg tumor is almost always secondary to a gastric adenocarcinoma, but may occasionally originate in the intestine, appendix, breast, or other sites.3,5,6 Rarely, breast cancer metastatic to the ovary presents clinically as an ovarian mass. In fact, in patients with a history of breast cancer, especially BRCA-positive cases, clinically detected ovarian tumors are usually independent primaries and not metastases. In recent years, attention has been drawn to mucinous tumors of the appendix, pancreas, and biliary tract that often spread to the ovary and closely simulate ovarian mucinous borderline tumors or carcinomas.7–11 Resemblance to borderline tumors or even benign cystadenofibromas is due to the so-called maturation phenomenon by which the epithelium of the metastatic carcinoma differentiates and appears flattened and benign. A wide variety of other tumors may metastasize to the ovary.
General features of ovarian metastasis include: bilaterality, multinodularity, involvement of the ovarian surface, extensive extraovarian spread, unusual patterns of dissemination, unusual histologic features, blood vessel and lymphatic invasion, and desmoplastic stromal reaction (Table 30.1).
Mode of Spread
The routes of tumor spread to the ovary are variable. Lymphatic and hematogenous metastasis to the ovaries is the most common form of dissemination.2–4 The importance of hematogenous spread is supported by the higher frequency of ovarian metastases in the well-vascularized ovaries of young patients. Embolic spread often produces multiple nodules within the substance of the ovary and commonly is accompanied by prominent intravascular nests of tumor cells in the ovarian hilum, meso-ovarium, and mesosalpinx.
Direct extension is also a common form of spread from adjacent tumors of the fallopian tube, uterus, and colorectum, for mesotheliomas, and for occasional retroperitoneal sarcomas.5 Transtubal spread provides an explanation for some surface ovarian implants from carcinomas of the uterine corpus,12 but may also account for some cases of spread from the uterine cervix.13,14 Neoplasms may also reach the ovary by the transperitoneal route from abdominal organs, such as the appendix.8 The metastatic tumor is found on the surface of the ovary or in the superficial cortex. Ovulation orifices may possibly represent a portal of entry for tumor cells.
Clinical Features
The circumstances leading to the discovery of the ovarian metastases depend on the site of the primary tumor.1,15 Ovarian metastases are detected before the breast cancer in only 1.5% of cases.15 In patients with a rectal or sigmoid colon cancer, the ovarian metastases and the primary tumor are discovered simultaneously, or more often, the intestinal tumor has been resected months or, years previously (50–75% of cases). Less frequently, the colorectal adenocarcinoma is discovered several months to years after resection of the ovarian metastases (3–20% of cases).3,16 In contrast, in about two-thirds of patients with Krukenberg tumor the diagnosis of the ovarian metastases precedes the discovery of the primary carcinoma.17,18 When a patient presents with abdominopelvic symptoms leading to suspicion of an ovarian tumor, the symptoms are nonspecific and similar to those of ovarian cancer, i.e., pelvic masses, ascites, or vaginal bleeding.17,19 Some patients with ovarian metastases have menstrual abnormalities, postmenopausal bleeding, and virilization, or they deliver a masculinized female fetus. These endocrine manifestations result from hormonally active luteinized stromal cells found in approximately one-third of metastatic tumors. Stromal luteinization occurs most frequently in association with metastatic mucinous carcinomas, particularly of colorectal or gastric origin.
Approximately 80% of patients with a Krukenberg tumor have bilateral ovarian metastases, and 73% of patients with ovarian metastases from breast carcinomas have extraovarian metastases.15,17 Radiologically, patients with a Krukenberg tumor more often have a solid mass with an intratumoral cyst, whereas primary ovarian cancers are predominantly cystic.20,21
Macroscopic Features
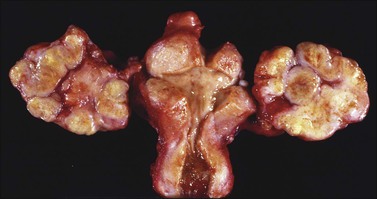
Ovarian metastases are bilateral in over 70% of cases (Figure 30.1).3 In contrast, the ovarian carcinomas most commonly mimicked by metastases (i.e., mucinous, endometrioid, and clear cell carcinomas) are bilateral in less than 15% of cases.22 Metastatic tumors grow as superficial or parenchymatous solid nodules or, frequently, as cysts. The size of ovarian metastases varies even from one side to the other. The ovaries may be only slightly enlarged or measure 10 cm or more. Intraoperative assessment of size and laterality may serve as a helpful guideline in distinguishing primary from metastatic mucinous tumors: bilateral tumors of any size or unilateral tumors under 10 cm have a high probability of being metastatic, whereas unilateral tumors over 10 cm are usually primary.23 However, there are many exceptions,24,25 especially in cases of colorectal and endocervical primaries.26 It is also noteworthy that metastases involving the ovary are often larger than their corresponding primary tumors.
Microscopic Features
The microscopic appearance of the metastases varies depending on the nature of the primary tumor.2–4 The identification of surface implants is extremely helpful in the recognition of secondary ovarian tumors that spread through the abdominal cavity and tubal lumen. Other features more commonly observed in metastatic tumors include multinodular infiltrative growth, single cell infiltration, follicle-like spaces (due to inadequate lymphatic drainage), and intravascular tumor emboli. Presence of a desmoplastic stroma and prominent stromal luteinization around tumor glands are also suspicious for metastasis.
Site of Origin
Intestinal Carcinomas
Most intestinal metastases originate in the large intestine and, much less frequently, in the small bowel. Whereas at autopsy metastases from intestinal carcinomas are less common than those from gastric carcinomas (14% vs 38%, respectively), at the time of operation metastases from intestinal carcinomas are almost five times as frequent as those from gastric carcinomas.27 Up to 45% of colorectal metastases to the ovary are clinically thought to be primary ovarian tumors, and many are misinterpreted as such on microscopic examination.2 Metastasis from the large bowel to the ovary is seen relatively more frequently when this cancer develops in women under 40 years of age. In one large series, one-fourth of patients were less than 40 years old28 and in another about 43% were under 50 years.29 In the latter study, it was found that patients initially presenting with an ovarian mass were significantly younger than those having ovarian spread in the setting of a known colorectal primary (average 48 vs 61 years). Not infrequently, patients have luteinized stromal cells in the ovarian tumor with resultant hormone production and endocrine symptoms.
The ovarian tumors are bilateral in approximately two-thirds of cases (Figure 30.1). Smaller tumors are usually solid, whereas larger tumors are composed of friable gray, yellow, or red tissue with cysts that contain necrotic tumor, mucinous fluid, or blood (Figure 30.2).2,3
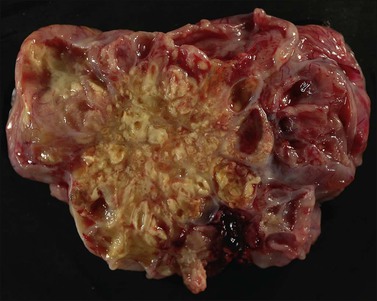
Figure 30.2 Metastatic carcinoma from the colon. Solid and cystic tumor with extensive necrosis and focal hemorrhage.
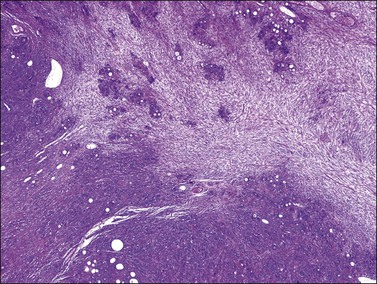
Microscopically, the metastatic tumors may be confused with primary endometrioid or mucinous carcinoma depending on whether the colonic carcinoma is predominantly non-mucinous or mucinous. Features that help distinguish colon cancer from endometrioid carcinoma include luminal necrotic debris (‘dirty necrosis’), focal segmental necrosis of the glands (Figures 30.3–30.5), occasional presence of goblet cells, and absence of müllerian features (squamous differentiation, an adenofibromatous component, or association with endometriosis). Also the nuclei lining the glands of metastatic colon carcinoma exhibit a higher degree of atypia than those of endometrioid carcinoma (Figure 30.5).2,3 The stroma may be desmoplastic (Figure 30.6), edematous, or myxoid, but frequently resembles ovarian stroma. Stromal luteinization is most frequently found in metastatic colorectal carcinomas.30
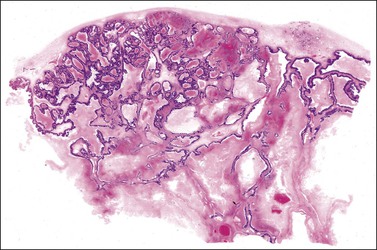
Figure 30.3 Metastatic adenocarcinoma from the colon. Garland-like glandular pattern with focal segmental necrosis of glands and abundant necrotic debris.
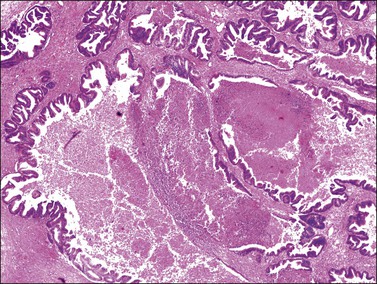
Figure 30.4 Metastatic adenocarcinoma from the colon. Focal segmental necrosis of glands and luminal necrotic debris (‘dirty necrosis’).
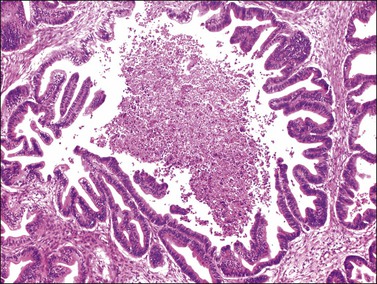
Figure 30.5 Metastatic adenocarcinoma from the colon. The glandular epithelium shows papillary growth and striking nuclear atypia (grade 3).
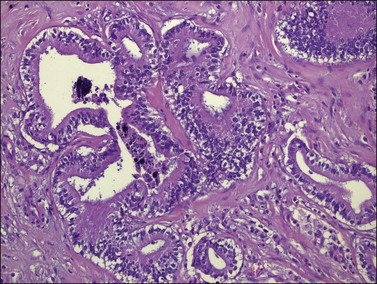
Figure 30.6 Metastatic adenocarcinoma from the colon. Neoplastic glands lying in a desmoplastic stroma.
Metastatic tumors may also resemble closely primary mucinous ovarian tumors both grossly (Figure 30.7) and microscopically. The metastases may be moderately differentiated (Figure 30.8) or so well differentiated that they can be mistaken for mucinous borderline or less often benign ovarian tumors. Generous sampling (at least one block per 1–2 cm of tumor diameter) is recommended. Metastatic mucinous tumors to the ovary can originate in the large intestine, stomach, pancreas, gallbladder, biliary tract, appendix, and even, exceptionally, urachus and lung. Features supportive of the diagnosis of a metastasis already discussed include bilaterality, histologic surface involvement by epithelial cells (surface implants) (Figure 30.9), irregular infiltrative growth with desmoplasia (Figure 30.6), single cell invasion, signet-ring cells, vascular invasion (Figure 30.10), coexistence of benign-appearing mucinous areas with foci showing a high mitotic rate and nuclear hyperchromasia, and histologic surface mucin.31 In some cases, the intestinal metastases contain cells with abundant clear cytoplasm and may simulate either primary clear cell carcinoma or the secretory variant of endometrioid carcinoma (Figure 30.11).32
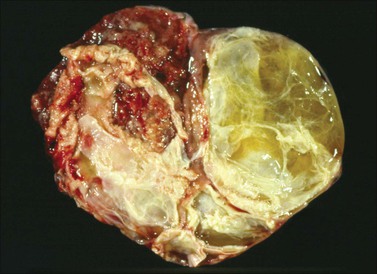
Figure 30.7 Metastatic carcinoma from the cecum. Multilocular mucinous cystic tumor with focal necrosis simulating an ovarian mucinous cystadenocarcinoma.
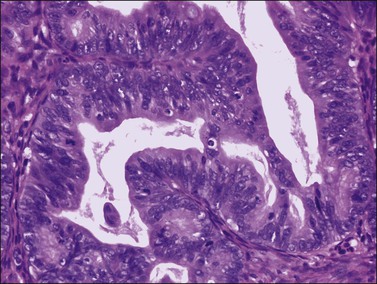
Figure 30.8 Metastatic adenocarcinoma from the colon. The mucinous glands are indistinguishable from those of a primary ovarian mucinous carcinoma. Mitotic figures are seen.
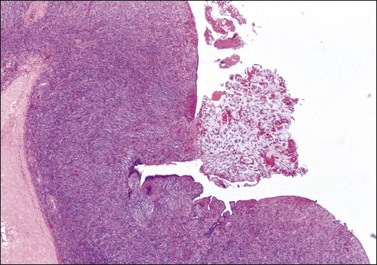
Figure 30.9 Metastatic adenocarcinoma from the colon. Ovarian surface involvement by mucinous epithelial cells (surface implant).
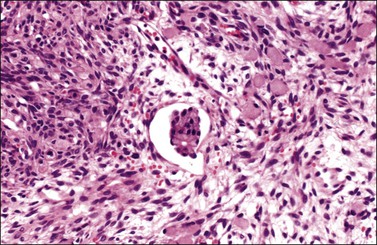
Figure 30.10 Krukenberg tumor. Vascular space invasion. Numerous signet-ring cells lie in the ovarian stroma.
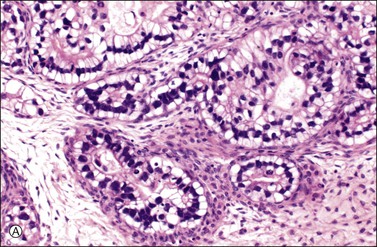
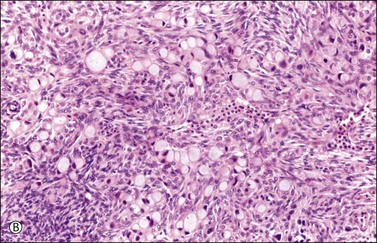
Figure 30.11 Metastatic adenocarcinoma from the small intestine. (A) Tubular glands lined by vacuolated cells resembling those of a secretory endometrioid carcinoma. Note the presence of stromal luteinization. (B) Typical Krukenberg tumor component encountered after additional sampling. Numerous signet-ring cells with eccentric nuclei and pale vacuolated cytoplasm are seen.
Immunohistochemistry
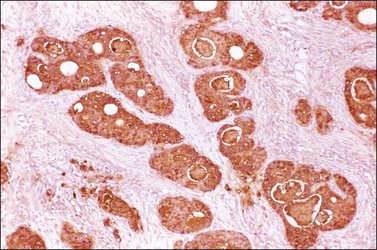
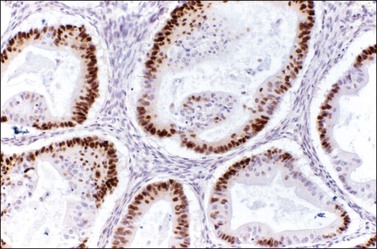
Immunohistochemical stains can be helpful in distinguishing primary adenocarcinoma of the ovary from metastatic colorectal adenocarcinoma (Table 30.2). Cytokeratin (CK) immunostains are the most commonly used. Primary ovarian carcinomas are almost always immunoreactive for CK7 whereas colorectal adenocarcinomas are usually CK7 negative.33,34 Mucinous adenocarcinomas of the ovary may be immunoreactive for CK20, but the reaction is typically weak and focal.33,35 Endometrioid adenocarcinomas are almost invariably CK20 negative.34 In contrast, colorectal adenocarcinomas are diffusely and strongly reactive for CK20. Therefore, a CK7-positive/CK20-negative immunoprofile favors a primary ovarian carcinoma, whereas a CK7-negative/CK20-positive immunoprofile suggests metastatic adenocarcinoma (Table 30.2).35–37 Although the vast majority of colorectal adenocarcinomas express CK20, poorly differentiated and right-sided tumors can be CK20 negative.38 Furthermore, adenocarcinomas of the appendix, small intestine, and stomach can be CK7 positive. Thus, immunostains for CK7 and CK20 should be interpreted with caution, always in the light of all clinical information, and with the understanding that no tumor shows absolute consistency in its staining with these markers.
Table 30.2
Primary Ovarian Carcinomas versus Metastatic Gastrointestinal Carcinomas (Immunophenotypes)
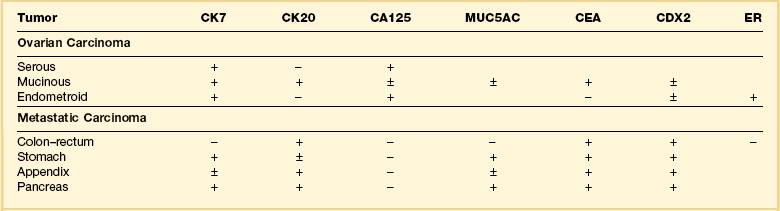
Other immunohistochemical stains have greater overlap in their expressions and should not be used individually in this differential diagnosis. Nevertheless, after taking into account the clinicopathologic findings and the results of the CK immunostains, negative stainings for vimentin39 CA125 and gastric mucin gene MUC5AC,35 and strongly positive staining for carcinoembryonic antigen (CEA) (Figure 30.12)33,34 favor metastatic colorectal cancer over primary ovarian adenocarcinoma (Table 30.2). Likewise, strong immunoreactivity for P53 (Figure 30.13) supports the colonic origin of the neoplasm. CDX2 is often strongly and diffusely positive in colorectal carcinomas, but it can also be positive in ovarian mucinous and endometrioid tumors.40,41 Estrogen receptors (ERs) may be helpful for distinguishing endometrioid adenocarcinomas from metastatic intestinal carcinomas, as the former are usually positive and the latter are negative.42
Krukenberg Tumor
Krukenberg tumors are adenocarcinomas with a distinctive histologic appearance. They consist of mucin-filled signet-ring cells and a striking proliferation of the ovarian stroma. Signet-ring cell carcinomas are associated more often with ovarian metastasis than carcinomas of other histologic types. These tumors originate in the stomach, usually in the pylorus, in the vast majority of cases. It has been demonstrated that gastric signet-ring cell carcinomas metastasize to the ovary much more often than intestinal-type carcinomas of the stomach do.43 Sometimes, the gastric cancer may be small and remains undetected for several years after oophorectomy. Much less frequently, the primary tumor is in the large intestine, breast, gallbladder, uterine cervix, appendix, or urinary bladder. In rare cases, the site of origin of the primary tumor is unknown and a diagnosis of ‘primary’ Krukenberg tumor has been proposed for those cases in which either the patient survives in good health for 10 years or longer or a thorough autopsy fails to reveal an extraovarian primary tumor.44 Possibly, some of the cases reported in the older literature as primary Krukenberg tumors represent mucinous (goblet cell) carcinoids. Patients with Krukenberg tumors tend to be younger than most patients with metastatic carcinoma; most of them are between 40 and 50 years of age. Although the symptoms are usually nonspecific, most frequently abdominal pain and swelling, endocrine manifestations, such as virilization during pregnancy, may result from stromal luteinization.3
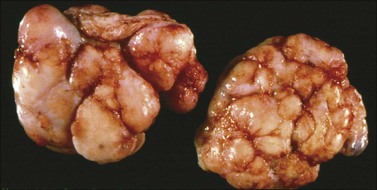
Krukenberg tumors are bilateral in 60–80% of the cases.3 They are typically solid masses with smooth nodular or bosselated outer surfaces (Figure 30.14). The cut surfaces are predominantly white or tan with areas of red or brown discoloration (Figure 30.15); the consistency may be firm, fleshy, or gelatinous. Because of the marked proliferation of the ovarian stroma, the tumors may resemble fibrothecomas on gross examination.
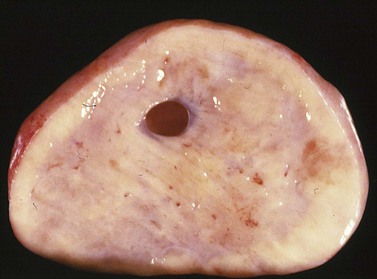
Figure 30.15 Krukenberg tumor. The cut surface shows solid beige-yellow tissue with small foci of hemorrhage.
Microscopically, the plump, rounded carcinoma cells have a signet-ring appearance and are surrounded by a dense cellular stroma (Figure 30.16). The epithelial cells may appear singly or in nests (Figure 30.17) and tend to concentrate in the dense fibroblastic areas. In some cases, the stroma is less cellular, edematous (Figure 30.18), or fibrous. Isolated small glands are usually found45 (Figure 30.19) and, occasionally, there is a predominant tubular architecture (tubular Krukenberg tumor) (Figure 30.20).6 The stromal cells are plump, spindle shaped, and are sometimes extensively luteinized.3,6
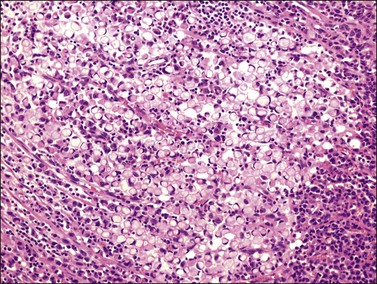
Figure 30.16 Krukenberg tumor. Numerous signet-ring cells with pale cytoplasm are arranged irregularly within a cellular stroma.
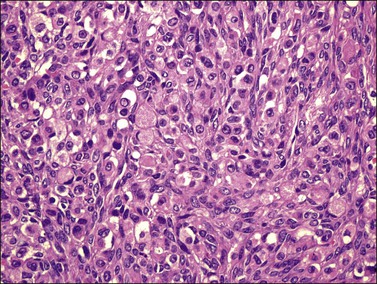
Figure 30.17 Krukenberg tumor. The signet-ring cells have eosinophilic cytoplasm and are distributed singly or in nests.
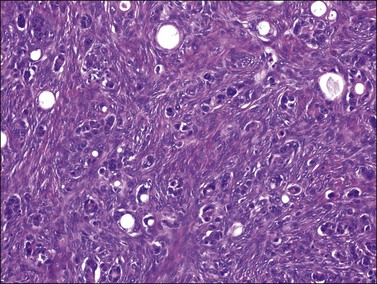
Figure 30.19 Krukenberg tumor. Small glands lined by moderately atypical epithelial cells are admixed with signet-ring cells.
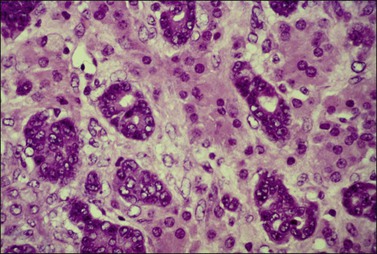
Figure 30.20 Tubular Krukenberg tumor. The tubular glands are lined by markedly atypical cells. The intervening clusters of luteinized stromal cells contribute to the resemblance to a Sertoli–Leydig cell tumor.
Krukenberg tumors must be distinguished from primary and other metastatic ovarian tumors including clear cell adenocarcinoma, mucinous (goblet cell) carcinoid, and a variety of ovarian tumors that contain signet-ring-like cells filled with non-mucinous material. The presence of clear cells may raise the issue of clear cell carcinoma, but the clear cells in the latter contain glycogen; mucin, when present, is typically luminal and extracellular. Rarely, clear cell carcinoma may have a signet-ring cell component that simulates a Krukenberg tumor; however, the signet-ring cells of the former have a characteristic ‘targetoid’ cytoplasm (bull’s eye appearance), containing a large vacuole with a central eosinophilic body. In addition, the identification of a tubulocystic pattern, as well as the presence of hobnail cells, stromal hyalinization, and eosinophilic secretion, is helpful in establishing the diagnosis. In contrast, CK immunostains are not useful, as nearly half of gastric carcinomas metastatic to the ovary are CK7 positive/CK20 negative.38 Mucinous carcinoid, either primary or metastatic, may contain large areas of signet-ring cells; the former neoplasms, however, frequently contain other teratomatous elements, and Grimelius stains as well as immunostains for chromogranin, synaptophysin are usually positive in both.
The tubular variant of Krukenberg tumor, sometimes associated with stromal luteinization, can be confused with a Sertoli–Leydig cell tumor (Figure 30.20); however, signet-ring cells are not a feature of the latter tumor except for the heterologous form that contains mucinous intestinal glands.6 Positive mucicarmine and periodic acid–Schiff (PAS) stains with diastase digestion (Figure 30.21) are of great value in establishing the diagnosis of a Krukenberg tumor.6 Occasional Krukenberg tumors may closely resemble fibromas on macroscopic examination and may have relatively few signet-ring cells. Bilaterality and positive mucin stains facilitate the differential diagnosis.
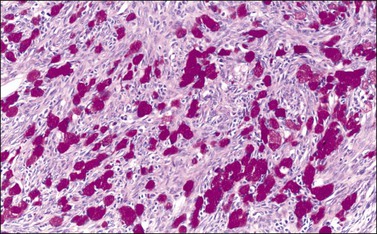
Figure 30.21 Krukenberg tumor. A PAS–diastase stain demonstrates the mucin in the vacuolated tumor cells.
Almost all patients die within a year of the diagnosis of Krukenberg tumor, but rare patients have survived for 5 or more years after gastrectomy and bilateral oophorectomy.46 Although exceptional, this outcome justifies removal of both the stomach and the ovarian metastases in cases in which the tumor appears limited to those organs. Also, in menopausal and postmenopausal women undergoing gastrectomy for carcinoma, the ovaries should be removed routinely to prevent the later development of ovarian metastasis and avoid another operation.
Carcinoid Tumors
Carcinoid tumors of the gastrointestinal tract, pancreas, or bronchus may metastasize to the ovary, and approximately half of the patients have the carcinoid syndrome.47 The primary tumor is usually in the ileum. Whereas mucinous carcinoid tumors of the appendix spread to the ovary in approximately one-third of cases, typical carcinoid tumors of the appendix almost never metastasize to the ovary.3 In the largest series reported, the age of the 35 patients ranged from 21 to 82 years, with a median of 57 years.47 Forty percent of the women had preoperative manifestations of the carcinoid syndrome. Some of them also had clinical evidence of intestinal or ovarian involvement. Extraovarian metastases were found in at least 90% of the cases, in contrast to the rarity of similar spread in cases of primary ovarian carcinoids.
Metastatic carcinoids are typically bilateral solid tumors with smooth or bosselated surfaces. The cut section often shows firm white or yellow confluent nodules, which may simulate fibromas or thecomas (Figure 30.22). Rare carcinoid tumors are predominantly cystic.3 Microscopically, the insular pattern is the most common, but trabecular and mucinous patterns may also be found. The carcinoid glands usually appear scattered in a dense fibrous stroma, which may be hyalinized (Figures 30.23 and 30.24). Focally, metastatic mucinous (goblet cell) carcinoids may resemble Krukenberg tumors. Immunostains for chromogranin and synaptophysin are often positive (Figure 30.25).
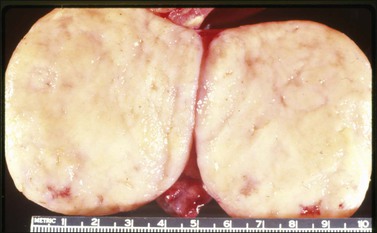
Figure 30.22 Metastatic carcinoid tumor from the pancreas. The sectioned surfaces are solid and white-yellow simulating those of a fibroma or thecoma.
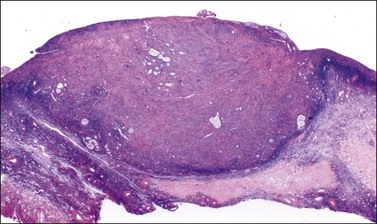
Figure 30.23 Metastatic mucinous carcinoid tumor from the appendix. The ovary is partly replaced by a nodule of dense fibrous tissue containing scattered small glands.
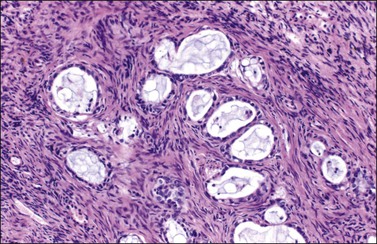
Figure 30.24 Metastatic mucinous carcinoid tumor from the appendix. Small glands lined by flat to cuboidal mucinous epithelium and occasional goblet cells. The intervening stroma appears fibromatous.
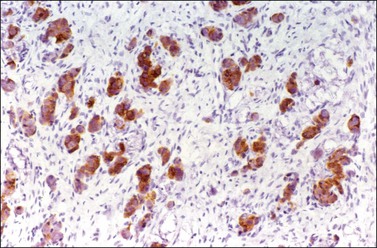
Figure 30.25 Metastatic mucinous carcinoid tumor from the appendix. Positive chromogranin immunoreaction.
Metastatic carcinoids can be confused with primary carcinoids (Table 30.3), granulosa cell tumors, Sertoli–Leydig cell tumors, Brenner tumors, adenofibromas, or endometrioid carcinomas.3,5 If the diagnosis of a carcinoid tumor is difficult in any of the previous situations, more thorough sampling, and immunohistochemical staining for neuroendocrine markers can be performed. Bilaterality, extraovarian extension, and absence of teratomatous elements are important features of metastatic carcinoids. CDX-2 does not distinguish between tumors of intestinal origin and primary ovarian carcinoids.48
Table 30.3
Primary versus Secondary Carcinoid Tumor: Differential Diagnosis
| Metastatic Carcinoid | Primary Carcinoid |
| Clinical Profile | |
| Primary site apparent in 80% | No intestinal tumor |
| Peritoneal seeding and abdominal metastases frequent | Extraovarian spread uncommon |
| Early recurrence and progression | Low rate of recurrence |
| Macroscopic Features | |
| Bilateral almost always | Unilateral always |
| Cut surface nodular and variegated | Cut surface homogeneous |
| Microscopic Features | |
| Teratomatous elements absent | Teratomatous elements usually present |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree