Melanocytic Lesions
Adriano Piris
Carlos N. Prieto-Granada
Cecilia Lezcano
Michael J. Imber
Martin C. Mihm Jr.
The spectrum of benign and malignant melanocytic proliferations is both complex and fascinating. An awareness of the normal developmental biology of the human melanocyte provides diagnostic insight into the morphologic diversity of melanocytic disorders. An understanding of the spectrum of benign proliferative patterns is necessary for recognizing the aberrant histologic and cytologic features of malignant melanoma and precursor lesions.
Melanocytes originate from neural crest precursor cells, which also give rise to peripheral neurons and Schwann cells (1). At this early stage, signaling factors of the BMP/WNT family trigger the expression of transcription factors of the Snail, Twist, Fox, Sox, and ETS families, which mediate epithelium-to-mesenchyme transition (EMT), among other functions. After undergoing EMT, neural crest cells destined to become melanocytes migrate via a dorsolateral pathway to the periderm by expressing matrix metalloproteinases (MMPs) and ADAM proteins and using preexisting structures (2,3). The dichotomous fate (sensory neural vs. melanocytic) is mostly determined by several factors such as the neurogenin (neurog) family of proteins that favor neural sensory development, whereas cells ultimately to become the melanocytic lineage (melanoblasts) are mainly influenced by MiTF and SoxE family transcription factors, particularly SOX10, among others (4). Once they reach their destination, they develop the dendritic morphology and the ultrastructural characteristics of mature melanocytes (5,6). The cytokinetic behavior of melanocytes during embryogenesis and their developmental relationship to other cells of the peripheral nervous system likely are basic factors underlying the morphologic spectrum of benign melanocytic lesions (7,8 and 9). The existence of pigmented neurofibromas and melanotic schwannomas, the association of leptomeningeal melanocytosis with giant congenital melanocytic nevi, and the commonly observed phenomenon of schwannian differentiation or neurotization in acquired melanocytic nevi underscore this point (10). In fact, experimental data point out that besides the embryologic origin from the neural crest, melanocytes can potentially arise from Schwann cell precursors (11). In addition, experiments in animals showed that committed Schwann cells are capable of reverting back to a primitive glial-melanocytic precursor when influenced by endothelin 3 peptides (12).
Stromal-epithelial interactions during embryogenesis may influence the development of distinct melanocyte subpopulations that differ in anatomic distribution and the potential to evolve into various types of melanocytic proliferations (13,14,15,16,17,18 and 19), features that underscore the heterogeneous nature of malignant melanoma with respect to the different site-dependent features.
The earlier mentioned properties suggest that the criteria used to distinguish benign from malignant melanocytic proliferations and to identify precursor and borderline lesions must necessarily differ from those applied to epithelial proliferations. The melanocytes are held in place with E-cadherin, which is expressed on both keratinocyte and melanocyte surfaces. This appears to be an extremely important molecule for keratinocyte-melanocyte interactions. In the normal homeostatic events of melanocyte migration and proliferation, especially in childhood, the melanocyte downregulates E-cadherin, desmoglein 1, and connexin 43 under the influence of several growth factors. Once the melanocyte divides and repositions itself in the epidermis, then the adhesion molecules reappear (20). The proliferation and migration of melanocytes are also normally seen in regenerating epidermis and in areas of vitiligo undergoing repigmentation (21).
The migration of melanocytes across the basement membrane zone of the epidermis and into the papillary dermis is part of the “normal” development of many benign melanocytic lesions, including most of the acquired melanocytic nevi. In addition, upward or lateral migration of melanocytes within the epidermis may be seen in several benign lesions (22), including the spindle and epithelioid cell nevus, the pigmented spindle cell nevus (23), and acral nevi (24). Therefore, the isolated histologic observation of invasive behavior does not necessarily denote malignancy in a melanocytic lesion as it would in epithelial tumors (25). Also, the presence of pagetoid spread in a melanocytic lesion does not always imply melanoma.
In summary, the nature of melanocytic lesions is a reflection of the remarkably nomadic embryologic process undergone by neural crest cells and melanoblasts, with participation of a specific molecular milieu, expression of metalloproteinases, EMT, and cell adhesion changes and dissemination using preexisting structures, all of which tend to be recapitulated in malignant melanocytic lesions (26).
BENIGN MELANOCYTIC PROLIFERATIONS
HISTOLOGIC AND CYTOLOGIC CHARACTERISTICS
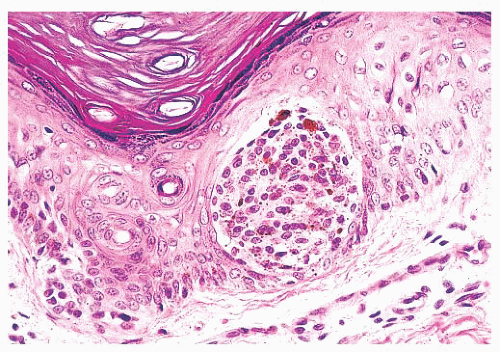
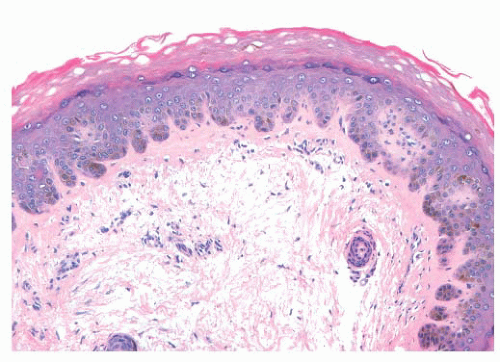
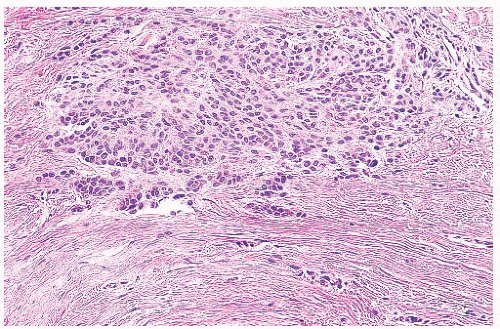
Most benign lesions that originate from epidermal melanocytes display varying combinations of three proliferative patterns that are readily apparent under low-power examination. Lentiginous hyperplasia describes a pattern of crowded single-cell melanocytic growth along the dermal-epidermal junction (Fig. 3.1). This pattern is often seen in compound nevi and lentigines. Nested proliferation describes the clonal growth of numerous microanatomically discrete clusters of melanocytes (Fig. 3.2). The distribution of such nests (tHÈques) of cells within the epidermis or along the junction is often quite characteristic of certain specific types of melanocytic nevi, including the pigmented spindle cell nevus and the spindle and epithelioid cell nevus. Finally, pagetoid proliferation describes a pattern of discohesive single-cell growth throughout the entire epidermis (Fig. 3.3). Although pagetoid spread is commonly associated with melanoma, it may be observed in Spitz nevi and in common acquired nevi occurring at acral locations.
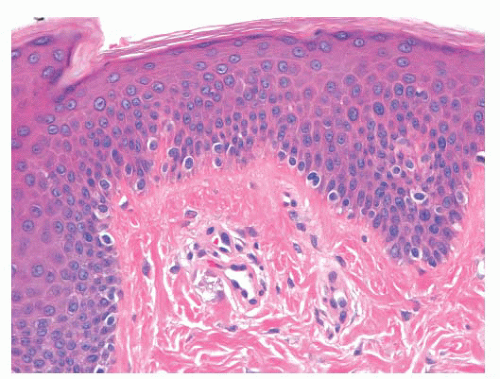 FIGURE 3.1 Lentiginous melanocytic hyperplasia, benign. The melanocytes are moderately increased in number and lie side by side in the basal layer. |
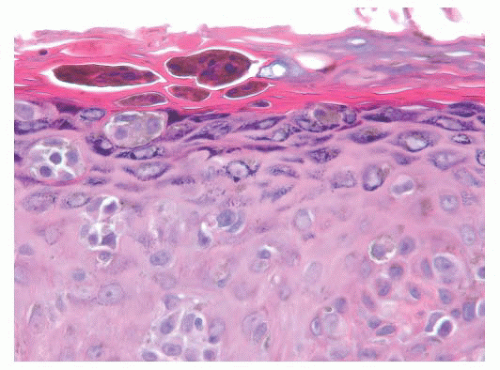
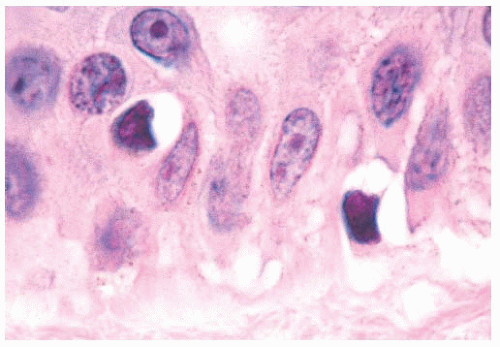
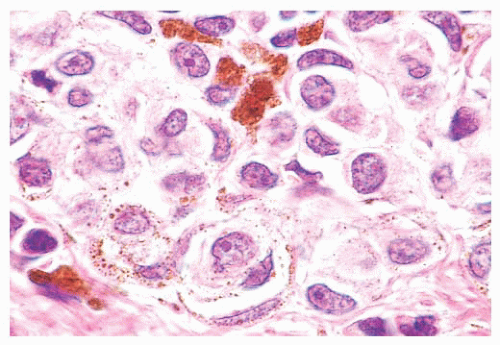
The cytologic characteristics of benign melanocytes or nevus cells are often variable. A comparison of the nuclear features of melanocytes with the features of adjacent normal keratinocyte or endothelial cell nuclei as internal references for nuclear size and detail is often helpful. The nucleus of a normal melanocyte residing within the basal layer of the epidermis typically is somewhat smaller and slightly more hyperchromatic than are the nuclei of nearby keratinocytes (Fig. 3.4) (27). The chromatin pattern is uniform, and no nucleoli are evident. The nuclear contour often appears polygonal or indented, and the cell cytoplasm appears clear as a result of artifactual retraction.
Benign melanocytic lesions do not display anaplastic cytologic characteristics. A wide spectrum of atypical nuclear changes may be seen in such lesions, but these changes generally are reactive, degenerative, or senescent phenomena rather than true anaplastic atypia characteristic of malignant transformation. True hyperchromasia, coarse nuclear membranes, and chromatin clumping are rarely encountered in benign pigment cell lesions. Certain morphologies seem to appear in most benign lesions, whereas other cytologic patterns are typical of specific lesions, such as the Spitz nevus or the pigmented spindle cell nevus.
A symmetric pattern of growth and ultimately of involution is characteristic of benign melanocytic proliferations (28). Histologic symmetry from left to right is readily apparent at low power, and it includes development of the epidermal component that is congruent to that of the dermal component. Simply stated, a junctional melanocytic proliferation synchronously migrates across the dermal-epidermal junction and establishes a dermal component over a lateral dimension that is equal to that of the original epidermal component. The presence of a so-called shoulder of junctional melanocytic hyperplasia lateral
to the bulk of an otherwise benign compound lesion reflects aberrant development. Such apparent histologic asymmetry may reflect the asynchronous migration of the junctional melanocyte population into the papillary dermis or the resumption of junctional melanocytic proliferation in what otherwise was a normally evolving lesion.
to the bulk of an otherwise benign compound lesion reflects aberrant development. Such apparent histologic asymmetry may reflect the asynchronous migration of the junctional melanocyte population into the papillary dermis or the resumption of junctional melanocytic proliferation in what otherwise was a normally evolving lesion.
An apparent vertical gradient of cytologic development that is usually termed maturation is present in benign melanocytic proliferations. Cellular pleomorphism and atypical nuclear features are more evident near the epidermal origin of the tumor, with the deeper cells becoming smaller and more cytologically banal. Architectural maturation is likewise evidenced by the progressively smaller size of nevus cell nests or their replacement by infiltrating discohesive single cells at the base of the lesion.
The subsequent involution is symmetric, proceeding from top to bottom, with the replacement of dermal nevomelanocytes by normal dermal connective tissue. A wide spectrum of histologic patterns of senescence may be seen in benign melanocytic lesions. These include schwannian differentiation or neurotization, lipomatous degeneration, and inflammatory regression. A variety of cytologic patterns of senescence may also be observed, including giant cell transformation, presence of bizarre cells, and balloon cell formation. In all cases, however, such histologic patterns of senescence appear uniform and symmetric.
LENTIGO SIMPLEX
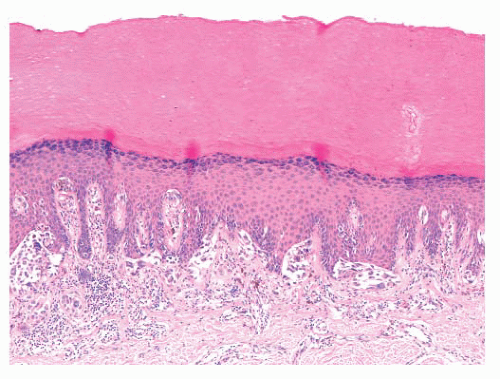
The common lentigo or lentigo simplex is a benign pigmented lesion characterized by accumulation of melanin pigment in basilar keratinocytes and melanocytic hyperplasia. Lentigines are acquired lesions, often occurring during childhood; they appear as small, uniformly dark brown macules. They may occur anywhere on the skin, but they show a predilection for acral sites.
Microscopic examination reveals lentiginous melanocytic hyperplasia that is usually distributed along the tips of rete ridges (Fig. 3.5). The adjacent keratinocytes are usually hyperpigmented, and a slight degree of epidermal rete hyperplasia may be present. The histopathologic features are distinguished from those of acquired melanocytic nevi by the absence of nested melanocytes (29).
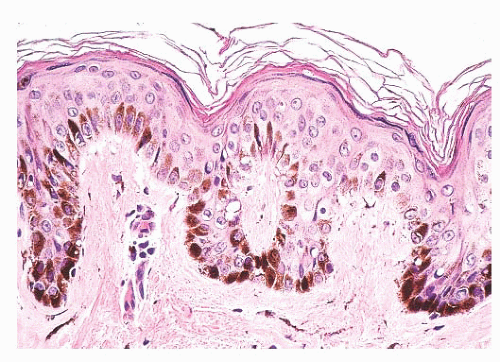 FIGURE 3.5 Lentigo simplex. Dense melanin deposition in basilar keratinocytes is observed primarily along rete ridges. Lentiginous melanocytic hyperplasia may be obscured by the pigment. |
Unlike acquired melanocytic nevi and solar lentigo, lentigo simplex lesions are not associated with mutations of BRAF (V600E), PIK3CA, or FGFR3 (30).
SOLAR LENTIGO
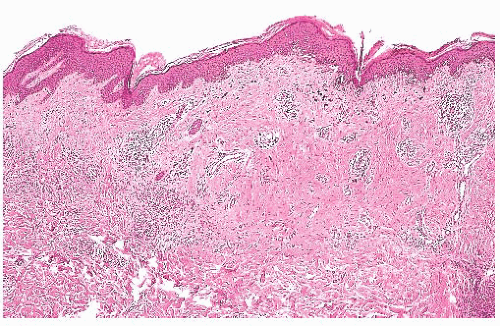
The solar or actinic lentigo is best regarded as a pigmented lesion resulting from chronic sun exposure. There is marked variation in microscopic findings among these lesions (Fig. 3.6). Solar lentigines appear as multiple, often poorly circumscribed areas of macular hyperpigmentation that may exceed 1 cm in diameter. Histologically, besides hyperpigmentation of basilar keratinocytes, lentiginous melanocytic hyperplasia may be observed in association with marked secondary epidermal rete hyperplasia, sometimes simulating the appearance of a reticulated seborrheic keratosis. At times, however, a mixture may be seen in which epidermal atrophy alternates with areas of epidermal hyperplasia. Chronic direct ultraviolet damage may selectively affect the dispersal of melanin pigment among basilar keratinocytes.
The spectrum of changes separating a benign solar lentigo from a pigmented actinic keratosis or lentigo maligna is not always clear. For this reason, caution should be exercised in reporting small biopsy specimens obtained from what otherwise appears to be a large, atypical macular pigmented lesion on chronically sundamaged skin. The observation of cytologically benign changes within a limited tissue sample from such a lesion does not necessarily rule out the presence of adjacent histopathologic atypia.
A variant of solar lentigo is the so-called ink spot or reticulated lentigo, which can potentially be a source of confusion clinically with melanoma. Histologically, it shows lentiginous melanocytic hyperplasia and a “skipped” prominent basilar pigmentation that can focally extend to the stratum spinosus and corneum (31).
Recent studies have shown that solar lentigo and seborrheic keratosis share common mutations in FGFR3, PIK3CA, and RAS (32,33). Interestingly, these same mutations have been detected in a proportion of benign lichenoid keratosis lesions, suggesting that these lesions represent a common pattern of regression of solar lentigo and seborrheic keratosis (34).
NEVUS SPILUS
This entity, which is also known as speckled lentiginous nevus, zosteriform lentiginous nevus, or spots on a spot nevus, is usually a solitary lesion composed of a background, slightly pigmented, macule with multiple hyperpigmented papules most frequently found in the torso and extremities. It can be present at birth or develop at any age. A congenital origin has been suggested (35), but this is controversial (36). Nevus spilus tends to follow Blaschko lines (37) and can present clinically as small/medium size, giant, and zosteriform lesions (38). These lesions can be a component of neurocutaneous syndromes such as phacomatosis pigmentovascularis (var. spilorosea) (39), phacomatosis pigmentokeratotica (40), facial features, cachexia, and eye and skin lesions (FACES) syndrome (41), and the speckled lentiginous nevus syndrome (42).
Histopathologically, these lesions are composed of either lentigo simplex, melanocytic hyperplasia, or a lentiginous junctional nevic component, overlapping a background hyperpigmented macule. The speckles are represented by scattered foci of compound or dermal nevus. As can be seen in benign melanocytic processes, partial regression can sometimes be seen in these lesions.
A multitude of melanocytic lesions can be seen arising in association with nevus spilus, namely Spitz nevi (43,44), blue nevi, and even epidermal tumors such as basal cell carcinomas (45). In a recent report describing an HRAS gain-of-function mutation in a nevus spilus-associated agminated Spitz nevus, the HRAS point mutation was also detected in the background nevus spilus (44). This phenomenon of “background” HRAS mutation was also documented in nevi sebaceous, which are also known to give rise to multiple other types of epidermal tumors which show the same mutation (46). Also, one of the described neurocutaneous syndromes heavily associated with nevus spilus and nevus sebaceous, phakomatosis pigmentokeratotica, was found to be a result of mosaic postzygotic HRAS mutation (47).
ACQUIRED MELANOCYTIC NEVUS
The common acquired melanocytic nevus, or mole, is the most common melanocytic tumor in humans. With increasing age, varying numbers of nevi develop in most persons (53). These usually appear as brown, pigmented lesions that are less than 0.6 cm in diameter, and they may occur anywhere on the skin surface. Common sites include the head and neck, sun-exposed trunk, and extremities. Different patterns have been described in nevi associated with specific anatomic sites. Three major histologic groupings exist that represent stages in the developmental progression of benign nevi, and these often correspond to characteristic gross morphologies. The histopathologic alterations observed in these lesions predominantly affect the epidermis and the papillary dermis.
The junctional nevus is the earliest stage of intraepidermal melanocytic proliferation. Nevomelanocytes are dispersed in multiple discrete nests along the dermal-epidermal junction, although lentiginous melanocytic hyperplasia may also be present. Junctional nevi appear clinically as small, slightly raised, and deeply pigmented lesions. The compound melanocytic nevus includes both a junctional component and the infiltration of the dermis by nevus cells distributed singly and in nests. Clinically, such lesions appear elevated or dome shaped, and they are less intensely pigmented than the junctional nevi. The dermal nevus no longer displays a junctional component; nevomelanocytes are confined to the dermis, and they are often associated with varying degrees of senescent histologic change. Clinically, they appear flesh colored or lightly pigmented, and they are dome shaped or pedunculated.
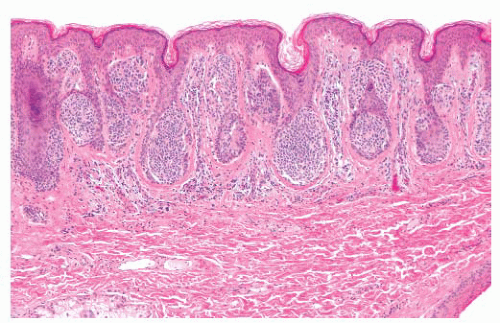 FIGURE 3.7 Acquired compound nevus. The nevus cells are present in the epidermis and papillary dermis in nests with minimal involvement of the superficial reticular dermis. |
The designations junctional, dermal, and compound do not refer to discrete melanocytic entities but rather to the histologically characteristic stages in the natural progression of the common acquired melanocytic nevus. The compulsive search for a rare junctional nest of nevus cells for the sake of labeling a predominantly dermal nevus as compound nevus is wasteful, and it overlooks the significance of a developmental spectrum (54).
Certain cytologic features are characteristic of stages in the evolution of melanocytic nevi (Fig. 3.7). The intraepidermal nevus cell, referred to as the epithelioid melanocyte or type A nevus cell, contains a round to oval nucleus that is slightly smaller than the nuclei of the adjacent keratinocytes (Fig. 3.8). The nucleus contains finely dispersed chromatin that is similar to that of neuroendocrine cells and occasionally a single, inconspicuous
nucleolus. The cytoplasm is prominent, and it often contains moderately coarse melanin granules.
nucleolus. The cytoplasm is prominent, and it often contains moderately coarse melanin granules.
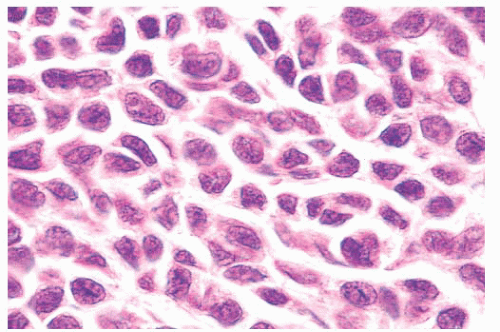
The lymphocyte-like or type B melanocyte is usually part of the dermal component of compound nevi (Fig. 3.9). The nucleus is small and round, and it contains uniformly dispersed chromatin with no apparent nucleoli. Scant, nonpigmented cytoplasm is evident.
The neural or type C nevus cell is often present at the base of melanocytic lesions (Fig. 3.10). This cell is often spindle shaped, and it contains a somewhat smaller oval nucleus with a banal chromatin pattern. These fusiform cells come to rest at, or singly infiltrate, the superficial reticular dermal collagen bundles.
Different patterns of epidermal hyperplasia may be seen in association with melanocytic nevi (55,56). Acanthotic epidermal proliferation with pseudo-horn cyst formation may clinically and histologically mimic a seborrheic keratosis. Extensive reticulated epidermal retiform hyperplasia is common in some pedunculated dermal nevi.
A host inflammatory cell response may represent different phenomena. External trauma to a nevus may result from excoriation or plucking hairs. The presence of focal epidermal necrosis, impetiginization, or a foreign body response to keratinous debris suggests such a cause.
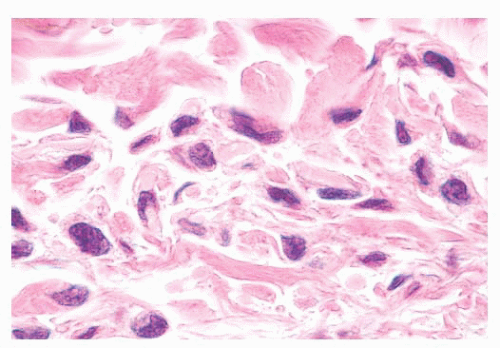 FIGURE 3.10 Type C nevus cells, compound nevus. Fusiform and spindle nevomelanocytes with scant cytoplasm singly infiltrate the superficial reticular dermis at the base of the lesion. |
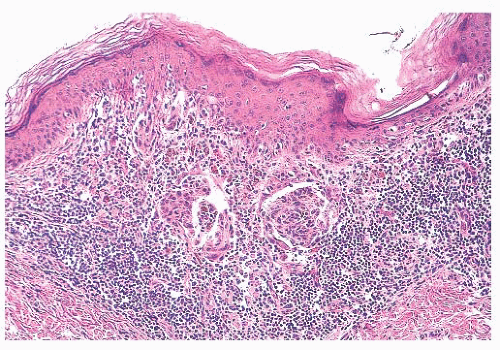 FIGURE 3.11 Halo nevus. A dense lymphocytic infiltrate obscures residual junctional and dermal nests of nevus cells. |
Diffuse infiltration of a nevus by lymphocytes and histiocytes with subsequent destruction of pigment-containing cells occurs in the halo nevus (57,58) (Fig. 3.11), a clinical variant in which an enlarging peripheral rim of hypopigmentation surrounds an acquired nevus (59,60). This uncommon form of symmetric inflammatory regression may be genetically determined. It should not be confused with the inflammatory host response seen in dysplastic or malignant melanocytic proliferations (see the following texts).
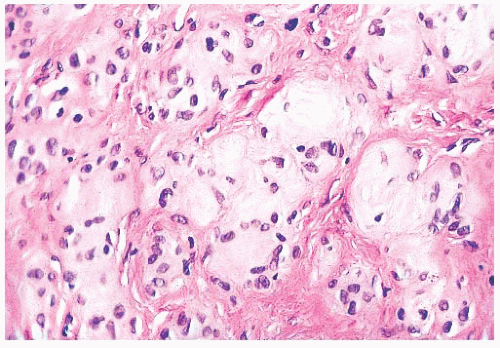
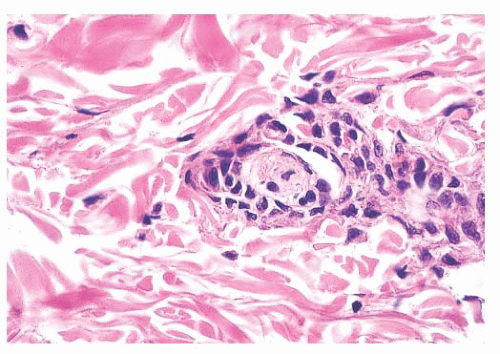
Long-term clinical observation of benign nevi reveals that most lesions undergo gradual involution (61,62). Residual dermal nevus cells are replaced by fibrous stroma. The subsequent fibrous papule or fibroepithelial polyp may contain no histologic evidence of a preexisting pigmented lesion. Melanocytic nevi on occasion display histologic and cytologic variations that reflect senescence. Prominent among the histologic senescent patterns is neurotization, or schwannian differentiation, in which the formation of neuroid structures in loose connective tissue often simulates a neurofibroma (Fig. 3.12). Other less
common patterns include lipomatous degeneration, with infiltration of dermal nevi by fat cells, and osseous metaplasia. Cytologic variants including nevus giant cells or balloon cells can also be seen (63). Such variant histologic and cytologic patterns may also occur in melanoma; therefore, they should never be used as the sole point for discriminating between benign and malignant melanocytic tumors.
common patterns include lipomatous degeneration, with infiltration of dermal nevi by fat cells, and osseous metaplasia. Cytologic variants including nevus giant cells or balloon cells can also be seen (63). Such variant histologic and cytologic patterns may also occur in melanoma; therefore, they should never be used as the sole point for discriminating between benign and malignant melanocytic tumors.
Most common acquired melanocytic nevi and lentigines display symmetry, uniform pigment distribution, and smooth boundaries with adjacent skin. The visual observation of asymmetry, uneven pigment distribution, and irregular or jagged borders within a pigmented lesion is usually a cause for clinical concern. Biopsy or excision is typically performed to rule out the diagnosis of malignant melanoma or a melanoma precursor. In many cases, an atypical-appearing pigmented lesion proves to be microscopically benign.
Histologically benign pigmented lesions may display subtle variations in pigment and melanocyte distribution. These may reflect physiologic or inflammatory characteristics unique to a particular lesion. The observation of such variations is significant if an otherwise histologically benign nevus or lentigo has been removed because of its worrisome clinical appearance. The diagnostic description of such features may provide valuable feedback and reassurance to the clinician.
The most common clinical cause for concern about a pigmented lesion is a reported “changing mole.” In an otherwise previously uniform symmetric nevus, such changes typically include an increase in size, the formation of irregular borders with adjacent normal skin, and a peripheral change in color. Although such clinical changes are characteristic of dysplastic nevi, a substantial percentage displays entirely benign cytologic features. Microscopically, the principal correlate is a so-called shoulder area of lentiginous junctional melanocytic proliferation beyond the lateral border of the underlying dermal component. Careful cytologic assessment of the peripheral lentiginous junctional component in addition to that of the more central domain is essential. The absence of significant anaplastic nuclear changes must be confirmed.
The biologic significance of peripheral changes within previously stable nevi is unclear. The idea that most common acquired nevi begin as junctional nevi is widely accepted. Subsequent dermal migration results in the establishment of a dermal component. The lentiginous junctional pattern of melanocytic hyperplasia is typically associated with an active, growing phase. One possibility is that, in many benign changing moles, radialjunctional proliferation is reactivated. Because the clinical appearance of reactivated radial proliferation in a benign nevus may be indistinguishable from that of many dysplastic nevi and some superficial spreading melanomas, complete excision and careful histologic study are essential.
CONGENITAL MELANOCYTIC NEVUS
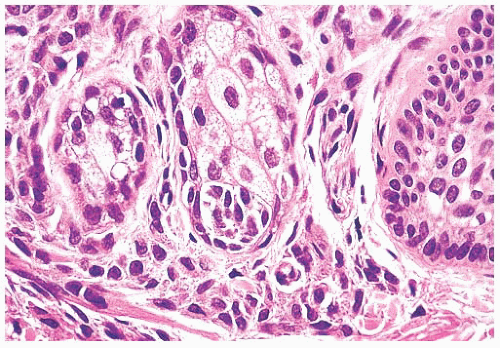
Pigmented lesions occur in approximately 1% of neonates (64). Some of these lesions are lentigines, and others are nevi. Most of the nevi are similar in size to common acquired melanocytic nevi. However, there are distinctive lesions that are quite large. Congenital melanocytic nevi are also more irregular in contour than are acquired nevi, and they are often hair bearing. These have been classified, according to size, as giant, intermediate, and small. The giant nevi can cover large areas of the body and are designated as “bathing trunk nevi” or “garment nevi.” These lesions have a somewhat increased risk for malignancy, estimated at between 5% and 10%, depending on the series (65). Histologically, compound and dermal patterns are seen. Some congenital nevi are microscopically indistinguishable from benign acquired nevi. In the most histologically characteristic lesions, the distribution of nevus cells throughout the dermis is more extensive than that seen in acquired nevi, typically involving the lower two-thirds of the reticular dermis and often infiltrating the subcutaneous fat (Fig. 3.13). The presence of nevus cells within cutaneous structures, including sebaceous lobules, multiple arrector pili muscles, and the perineurium of peripheral nerves, is characteristic of congenital nevi and is not seen in acquired nevi (Figs. 3.14 and 3.15) (66,67). The cytologic features of congenital melanocytic nevus cells and their patterns of maturation and senescence differ little from those previously described for acquired nevi (68,69).
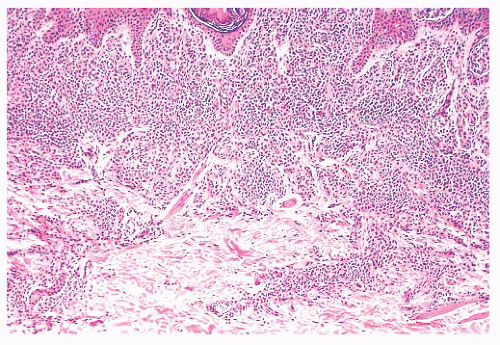 FIGURE 3.13 Congenital compound nevus. Nevus cells are deep within the reticular dermis and in close association with the appendages. |
Congenital melanocytic nevi often display an irregular surface contour and pigmentation. This irregularity may reflect the distribution and density of nevomelanocytes within the underlying appendageal epithelium and arrector pili muscles (Figs. 3.13,3.14 and 3.15). Congenital nevi may appear as a localized collection of individual pigmented lesions in a nonrandom, usually linear array. Microscopically, the individual pigmented lesions comprising the array appear indistinguishable from other congenital nevi. The unusual clinical distribution most likely reflects embryologic
developmental changes occurring along the Blaschko lines (70). Similar developmental changes are thought to underlie other cutaneous disorders characterized by linear and patterned arrays of discrete lesions, including inflammatory linear verrucous epidermal nevi and incontinentia pigmenti.
developmental changes occurring along the Blaschko lines (70). Similar developmental changes are thought to underlie other cutaneous disorders characterized by linear and patterned arrays of discrete lesions, including inflammatory linear verrucous epidermal nevi and incontinentia pigmenti.
In most cases, the history of a pigmented lesion present from birth or the existence of easily recognizable clinical features readily permits a clinical diagnosis. Most congenital nevi are not sampled because they display asymmetric features but because they have changed in appearance. As with most acquired melanocytic nevi in which a change in size or contour is reported, the most common benign microscopic correlate observed is peripheral junctional lentiginous melanocytic hyperplasia. Another common abnormality that would prompt biopsy of a congenital nevus is the emergence of the so-called proliferative nodule. This is an expansile proliferation of epithelioid cells arising within a congenital nevus and morphologically can show benign or atypical features. Recent studies show that immunohistochemically, these lesions may represent a borderline category between nevus and melanoma (71), whereas genetic analysis shows that they have a different aberration pattern than melanomas (72). Most small congenital nevi will show a BRAF mutation; however, larger congenital nevi are more likely to show NRAS mutation (73).
Atypical pigmented lesions that are reported to be of recent or acquired onset in the nonpediatric population sometimes display features of a benign congenital melanocytic nevus. In view of the stages by which embryonic premelanocytes develop from neural crest cells and migrate to cutaneous epithelium, the microscopic observation of “congenital” features in what is clinically reported to be an “acquired” nevus may reflect a socalled latent congenital nevus. In such a congenital nevus, aberrantly migrating premelanocytes may assume a microanatomic dermal distribution characteristic of congenital nevi, without subsequent gestational proliferation. Only later in life, perhaps secondary to hormonal or developmental signals, may the melanocytic proliferation take place, yielding a clinically acquired but histologically congenital nevus.
BALLOON CELL NEVUS
This peculiar change can be found in 1.7% of all melanocytic proliferations (74,75) and can be seen in almost any type of melanocytic proliferation including intranodal nevic nests (76). This change can even be identified clinically via dermoscopy (77).
Microscopically, balloon cell change consists of a partial or complete component of cells exhibiting abundant welldemarcated, clear to foamy cytoplasm containing a small benignappearing hyperchromatic nucleus. Melanocytic pigment can either be absent or abundant. If nuclear atypia is prominent and other features of malignancy are identified, one should suspect the rare balloon cell variant of malignant melanoma (78).
ACRAL MELANOCYTIC NEVUS
Melanocytic proliferations occurring in acral skin (hands and feet) consistently exhibit distinct characteristics that can prompt erroneous diagnosis. Dysplastic nevi and Spitz nevi rarely appear in these anatomic sites. Histologically, acral nevi present as lentiginous proliferations. The junctional nested pattern predominates. The melanocytic nests are discohesive, often irregular in size and shape, sometimes adopt a crescentic shape, and may coalesce (Fig. 3.16). Pagetoid spread is present in up to 61% of all nevi in the palm and soles (22). In the benign lesions, the areas of pagetoid ascent are restricted to the areas of nested proliferation and have benign morphology. In contrast, the pagetoid cells in acral lentiginous melanoma extend well beyond the nests in haphazard array (82,83 and 84).
NEVI OF SPECIAL SITES
A percentage of nevi arising in particular anatomic sites have been recently recognized to show unique characteristics. These nevi must be separated from compound congenital nevi, Spitz nevi, and dysplastic nevi arising at these sites. This particular group of lesions exhibits distinct and, at times, worrisome cytologic and architectural features that separate them from other nevi and are the source of possible diagnostic pitfalls. The regions that are
known to produce these misleading proliferations are the head and neck, especially the periauricular area; the milk line, extending from the axilla to the groin; and the genital region, especially the vulva. The possible causes that determine these peculiar cytologic and architectural features are unknown. Speculation regarding cause has invoked developmental factors or hormonal changes such as occur in adolescence and pregnancy.
known to produce these misleading proliferations are the head and neck, especially the periauricular area; the milk line, extending from the axilla to the groin; and the genital region, especially the vulva. The possible causes that determine these peculiar cytologic and architectural features are unknown. Speculation regarding cause has invoked developmental factors or hormonal changes such as occur in adolescence and pregnancy.
In the head and neck area, these lesions often present diagnostic challenges, especially in the scalp and ear of children and adolescents. These nevi with uncommon characteristics accounted for 10% of scalp nevi in one series (85), whereas in another series of ear and periauricular lesions, they represented 42% of the nevi occurring at this site (86). The lesions showed some common features such as poor circumscription with lateral extension of the junctional component and large irregular nests on the tips, on the sides, and in between the rete ridges. However, ear lesions tend to be symmetric and show stromal fibroplasia, whereas scalp proliferations frequently show a prominent lentiginous component with involvement of follicles and occasional pagetoid spread (87,88). In our experience, scalp lesions have intraepidermal epithelioid cell nests that are finely pigmented and have similar cells in the papillary dermis that are often mistaken for microinvasive melanoma. However, both intraepidermal and dermal cells have atypical but not malignant cytomorphology (87).
Nevi of the milk line, genital nevi, and nevi of flexural sites share several cytologic and architectural features. Nevi of the milk line comprise the axillary folds, the mammary area, the umbilicus, and the inguinal folds. Milk line and genital nevi (Fig. 3.17) share the “nested and discohesive” pattern, characterized by the presence of large, confluent nests at the dermal-epidermal junction, along with diminished melanocytic cohesion (88). Vulvar nests are often oval and lie along the rete ridges. Other distinctive changes of this group of lesions are stromal fibroplasia, which is usually coarser than in dysplastic nevi, and frequent lentiginous proliferation of the nevomelanocytes. Genital lesions, especially those occurring in the vulva, often show more prominent atypical features, such as moderate to severe cytologic atypia (epithelioid cells with prominent nucleoli) and occasional pagetoid spread. Also, the nests in these genital lesions are large, irregular, and frequently exhibit prominent pigmentation (89).
It is important to keep in mind that all these lesions show invariably benign features such as maturation and absence of dermal mitoses. These represent helpful clues to differentiate these sometimes worrisome looking but benign lesions from dysplastic nevi and malignant melanoma.
COMBINED NEVUS
On occasion, elements of two different types of dermal or epidermal melanocytic proliferation may be present in the same pigmented lesion (90). These so-called combined nevi most commonly include components of an acquired melanocytic nevus and the common blue nevus (91). Rarely, Spitz nevus cells may also be present. Combined nevi may display an aberrant histologic pattern of maturation, with pigmented epithelioid type A nevus cells at the base of the lesion instead of at the dermal-epidermal junction. Such variants should be considered in cases that otherwise appear cytologically and architecturally benign. “Inverted” type A cells should be recognized as such, and they should not be mistaken for malignant degeneration simply because they are pigmented and ectopic.
DEEP PENETRATING NEVUS
Clinically, deep penetrating nevi can occur de novo as blue papules or nodules, and often they are mistaken for a blue nevus or nodular melanoma. The more common presentation is in a preexisting acquired or congenital nevus in which a focal bluish discoloration raises the possibility of malignant degeneration. This lesion can be mistaken for malignant melanoma reportedly in as many as 40% of cases (92).
Histologically, this relatively uncommon lesion consists of a symmetric, wedge-shaped proliferation with the base parallel to the long axis of the epidermis and the vertex present in the deep dermis and sometimes in the subcutaneous fat. When it occurs de novo, there is usually an intraepidermal junctional component. When it occurs in a setting of a preexisting nevus, it usually occurs as a focal transformation in the dermal component of the preexisting nevus. The types of cells that comprise the deep penetrating nevus are more commonly the inverted type A nevus cells but sometimes are also spindle cells, type B nevus cells, and blue nevus cells. The inverted type A cells appear pale, epithelioid, and fusiform, occurring in nests often surrounded by melanophages. There is variable but usually slight melanization of the cytoplasm of the nevus cells. The cellular density in the upper dermis rapidly becomes diminished as the cells follow along the follicles or neurovascular bundles into the deeper tissue. Senescent changes often affect the inverted type A cells with nuclear vacuoles and sometimes hyperchromatic nuclei. Mitoses are very rare and, when present, should lead to thorough sectioning to rule out malignant transformation (93).
Efforts to distinguish this nevus from melanoma by molecular means have resulted in the finding of complete loss of dipeptidyl peptidase IV in malignant melanoma, compared with variable but consistent positivity in the deep penetrating nevi. However, there is a higher expression of MMP-2 and integrin beta-3 in the advancing front of deep penetrating nevi compared with that of melanoma. This finding can help explain why the pattern of infiltration resembles malignant melanoma in some cases (94).
INTRANODAL NEVIC NESTS
The finding of nests of nevic cells in lymph nodes was first reported in 1931 by Stewart and Copeland (95). The incidence of such nevic nests, known as nodal nevi, in lymph nodes draining malignancies ranges from 1% to 22% (96). In the majority of cases, nodal nevi are intracapsular or intratrabecular, but cases of intraparenchymal nests have been reported (97). The incidence of nodal nevi in sentinel lymph nodes of patients with cutaneous melanomas is 3.9%, which is higher when compared with the incidence in nonsentinel lymph nodes in melanoma patients (1.01%) (98). The origin of these nevus cells is unclear; a possible “benign metastasis-like” mechanism could be involved. One study reported a higher occurrence of nodal nevi with melanomas arising within congenital nevi (99), supporting the “benign metastasis” theory. However, more studies are needed to validate this concept. It is important to emphasize that the pathologist should be aware of the relatively high incidence of nodal nevi among sentinel lymph nodes in malignant melanoma to avoid erroneous interpretations. Besides the intracapsular and intraseptal location of the nests, perhaps the more important features that will help the pathologist make the right call is the benign cytologic and nuclear features of the nevic cells when compared with the epithelioid melanoma cells with nuclear hyperchromasia and pleomorphism.
RECURRENT MELANOCYTIC NEVUS
For cosmetic reasons, many clinicians prefer to remove acquired melanocytic nevi by a superficial “shave” biopsy and excision technique. Incomplete removal of the nevus may result in the recurrence of a pigmented lesion at the site of prior surgical trauma. Usually, such a recurrence is seen within several months of the procedure (100,101,102,103,104 and 105).
Recurrent melanocytic nevi often appear clinically worrisome because of the association of irregular pigmentation with scarring. When the nevus is re-excised, the specimen typically displays a residual dermal nevus component that is separated from the overlying epidermis by a zone of linear cicatricial fibrosis (Figs. 3.18 and 3.19). The overlying epidermis may display lentiginous melanocytic hyperplasia and basilar keratinocytic hyperpigmentation.
REGRESSING MELANOCYTIC NEVUS
The presence of a dense, superficial lymphoid cell infiltrate in an otherwise cytologically benign melanocytic nevus usually reflects inflammatory regression. The best-known example of such regression is the halo nevus, in which inflammatory regression occurs in a symmetric centripetal fashion (Fig. 3.11). Although the clinical appearance of a halo nevus is quite distinctive, the microscopic features may be no different from those observed in a changing pigmented lesion in which the progressive change in pigmentation is irregular or asymmetric (106).
SPITZ NEVUS AND VARIANTS
SPINDLE AND EPITHELIOID CELL NEVUS (SPITZ NEVUS)
The spindle and epithelioid cell nevus is important because of its histologic similarity to malignant melanoma. This entity was formerly known by the confusing name of benign juvenile melanoma, which nevertheless summarized its clinically benign yet histologically menacing status (107,108). This benign melanocytic lesion often referred to, at present, as the Spitz nevus may occur at all ages, although it is usually observed in children and young adults. Common sites include the head and neck and upper extremities. Clinically, this nevus presents as a small, solitary, dome-shaped dermal nodule. Because of a prominent vascular component in the tumor stroma and a relative lack of melanin pigmentation, it is frequently misdiagnosed clinically as a hemangioma or pyogenic granuloma.
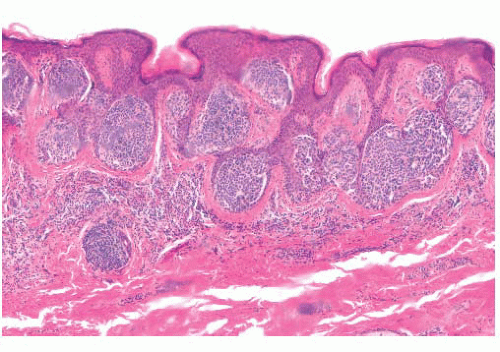
The histologic distribution of nevus cells in the Spitz nevus mirrors that of common acquired nevi, displaying junctional, compound, and dermal forms (Fig. 3.20) (109,110). However, Spitz nevi, like many congenital nevi, usually have a prominent dermal component. The overall architecture of the lesion is symmetric, with abrupt attenuation of the junctional nests at the lateral borders. The nevus is composed of varying proportions of spindle and epithelioid melanocytes. Spindle cells are usually present in fascicles arrayed perpendicular to the epidermis, whereas epithelioid cells are dispersed individually throughout the lesion. The overall histologic pattern is often discohesive and infiltrative, in contrast to the confluent and expansile growth pattern of malignant melanoma. The cells taper
from a broad base superficially to a narrow “point” in the deep dermis, architecturally resembling an inverted imperfect triangle. Also, the nevus cells mature by becoming smaller from the superficial to the deep part of the tumor (Figs. 3.20 and 3.21).
from a broad base superficially to a narrow “point” in the deep dermis, architecturally resembling an inverted imperfect triangle. Also, the nevus cells mature by becoming smaller from the superficial to the deep part of the tumor (Figs. 3.20 and 3.21).
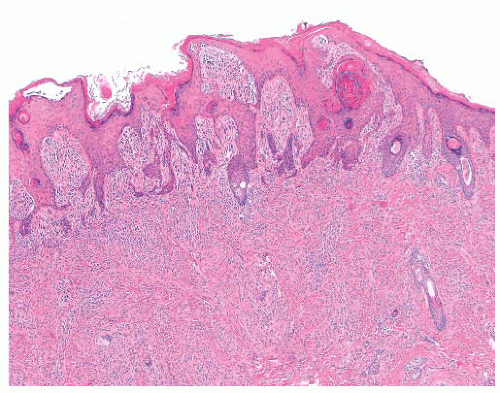 FIGURE 3.20 Spitz nevus. Large superficial nests become smaller in the reticular dermis and break up into single cells. |
Because of its atypical cytologic features, the spindle and epithelioid cell nevus is often confused histopathologically with melanoma (111,112 and 113). An extreme degree of cellular pleomorphism, particularly of epithelioid melanocytes, may be seen (Fig. 3.22). The nuclei of these cells may be quite large and irregular in contour, and they may contain prominent eosinophilic nucleoli; however, they are otherwise open or vesicular in appearance, and they lack the coarse anaplastic features typical of malignant cells. Often present are eosinophilic cytoplasmic invaginations or “nuclear pseudoinclusions.” The presence of mitotic figures and pagetoid epidermal spread of epithelioid melanocytes may also mimic melanoma. The overwhelming majority of the cells, however, have a benign cytologic appearance.
Those nevi that contain predominantly spindled melanocytes arranged in fascicles are usually more readily diagnosed than are those lesions consisting largely of pleomorphic epithelioid cells (Fig. 3.23). Several rather unique histologic features are regularly seen in Spitz nevi. These include the deposition of eosinophilic globules of hyaline-like material near the dermalepidermal junction and the artifactual separation of papillary dermal nests from the overlying epidermis (114,115). The tumor stroma may appear quite vascular and edematous. Lymphocytic infiltration is not uncommon, and long-standing lesions may display a densely sclerotic stroma with scattered individual tumor cells trapped amid dermal collagen (116).
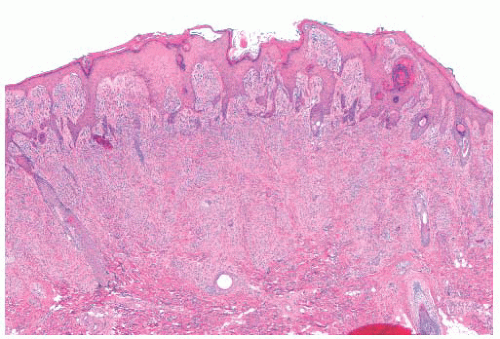 FIGURE 3.21 Spitz nevus. Benign epidermal hyperplasia overlies a proliferation of spindle cells that form an inverted wedge with the apex in the deep dermis. |
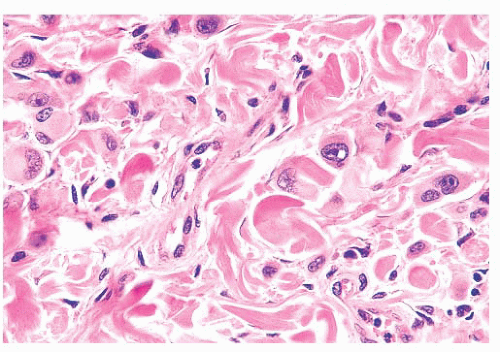 FIGURE 3.22 Spitz nevus. Cytologically atypical epithelioid nevus cells with prominent nucleoli in the reticular dermis are seen in a background of banal-appearing nevus cells. |
Histologic differentiation from melanoma may usually be made based on the overall symmetry of the lesion and the apparent cytologic “maturation” of nevus cells at the base of the lesion. The spindle and epithelioid cell nevus may rarely be a precursor of melanoma. Careful cytologic study for truly anaplastic nuclear features, bizarre mitoses, and aberrant or asymmetric proliferation should be made in borderline cases (117). At a molecular level, most Spitz nevi are composed of diploid cells and not aneuploid cells as seen in melanoma. Furthermore, except for a gain of 11p in a subset of lesions, Spitz nevi do not show significant chromosomal aberrations as seen in melanoma (118,119 and 120).
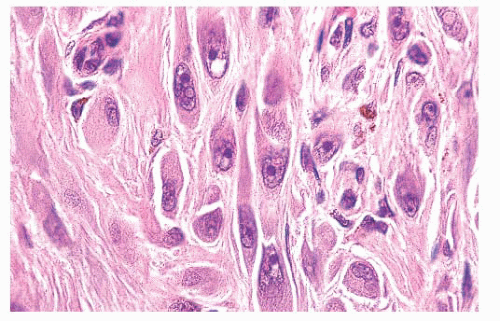 FIGURE 3.23 Spitz nevus. Fascicles of spindled nevomelanocytes with eosinophilic hyalinized cytoplasm and prominent nucleoli infiltrate the reticular dermis in a “raining down” array perpendicular to the skin surface.
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|