Lower Respiratory Tract Infections
KEY CONCEPTS
![]() Respiratory infections remain the major cause of morbidity from acute illness in the United States and likely represent the most common reasons why patients seek medical attention.
Respiratory infections remain the major cause of morbidity from acute illness in the United States and likely represent the most common reasons why patients seek medical attention.
![]() The majority of pulmonary infections follow colonization of the upper respiratory tract with potential pathogens, whereas microbes less commonly gain access to the lungs via the blood from an extrapulmonary source or by inhalation of infected aerosol particles. The competency of a patient’s immune status is an important factor influencing the susceptibility to infection, etiologic cause, and disease severity.
The majority of pulmonary infections follow colonization of the upper respiratory tract with potential pathogens, whereas microbes less commonly gain access to the lungs via the blood from an extrapulmonary source or by inhalation of infected aerosol particles. The competency of a patient’s immune status is an important factor influencing the susceptibility to infection, etiologic cause, and disease severity.
![]() An appropriate treatment regimen for the patient with uncomplicated lower respiratory tract infection can be established by evaluating the patient history, physical examination, chest radiograph, and properly collected sputum for culture and interpreted in light of current knowledge of the most common lung pathogens and their antibiotic susceptibility patterns within the community.
An appropriate treatment regimen for the patient with uncomplicated lower respiratory tract infection can be established by evaluating the patient history, physical examination, chest radiograph, and properly collected sputum for culture and interpreted in light of current knowledge of the most common lung pathogens and their antibiotic susceptibility patterns within the community.
![]() Acute bronchitis is caused most commonly by respiratory viruses and almost always is self-limiting. Therapy targets associated symptoms, such as lethargy, malaise, or fever (ibuprofen or acetaminophen), and fluids for rehydration. Routine use of antibiotics should be avoided and medication to suppress cough is rarely indicated.
Acute bronchitis is caused most commonly by respiratory viruses and almost always is self-limiting. Therapy targets associated symptoms, such as lethargy, malaise, or fever (ibuprofen or acetaminophen), and fluids for rehydration. Routine use of antibiotics should be avoided and medication to suppress cough is rarely indicated.
![]() Chronic bronchitis is caused by several interacting factors, including inhalation of noxious agents (most prominent are cigarette smoke and exposure to occupational dusts, fumes, and environmental pollution) and host factors including genetic factors and bacterial (and possibly viral) infections. The hallmark of this disease is a chronic cough, excessive sputum production, and expectoration with persistent presence of microorganisms in the patient’s sputum.
Chronic bronchitis is caused by several interacting factors, including inhalation of noxious agents (most prominent are cigarette smoke and exposure to occupational dusts, fumes, and environmental pollution) and host factors including genetic factors and bacterial (and possibly viral) infections. The hallmark of this disease is a chronic cough, excessive sputum production, and expectoration with persistent presence of microorganisms in the patient’s sputum.
![]() Treatment of acute exacerbations of chronic bronchitis includes attempts to mobilize and enhance sputum expectoration (chest physiotherapy, humidification of inspired air), oxygen if needed, aerosolized bronchodilators (albuterol) in select patients with demonstrated benefit, and antibiotics.
Treatment of acute exacerbations of chronic bronchitis includes attempts to mobilize and enhance sputum expectoration (chest physiotherapy, humidification of inspired air), oxygen if needed, aerosolized bronchodilators (albuterol) in select patients with demonstrated benefit, and antibiotics.
![]() Respiratory syncytial virus is the most common cause of acute bronchiolitis, an infection that mostly affects infants during their first year of life. In the well infant, bronchiolitis usually is a self-limiting viral illness.
Respiratory syncytial virus is the most common cause of acute bronchiolitis, an infection that mostly affects infants during their first year of life. In the well infant, bronchiolitis usually is a self-limiting viral illness.
![]() The most prominent pathogen causing community-acquired pneumonia in otherwise healthy adults is S. pneumoniae, whereas the most common pathogens causing hospital-acquired and healthcare-associated pneumonia are Staphylococcus aureus and gram-negative aerobic bacilli. Anaerobic bacteria are the most common etiologic agents in pneumonia that follows aspiration of gastric or oropharyngeal contents.
The most prominent pathogen causing community-acquired pneumonia in otherwise healthy adults is S. pneumoniae, whereas the most common pathogens causing hospital-acquired and healthcare-associated pneumonia are Staphylococcus aureus and gram-negative aerobic bacilli. Anaerobic bacteria are the most common etiologic agents in pneumonia that follows aspiration of gastric or oropharyngeal contents.
![]() Treatment of community-acquired pneumonia may consist of humidified oxygen for hypoxemia, bronchodilators (albuterol) when bronchospasm is present, rehydration fluids, and chest physiotherapy for marked accumulation of retained respiratory secretions. Antibiotic regimens should be selected based on presumed causative pathogens and pulmonary distribution characteristics and should be adjusted to provide optimal activity against pathogens identified by culture (sputum or blood).
Treatment of community-acquired pneumonia may consist of humidified oxygen for hypoxemia, bronchodilators (albuterol) when bronchospasm is present, rehydration fluids, and chest physiotherapy for marked accumulation of retained respiratory secretions. Antibiotic regimens should be selected based on presumed causative pathogens and pulmonary distribution characteristics and should be adjusted to provide optimal activity against pathogens identified by culture (sputum or blood).
![]() Treatment of nosocomial pneumonia requires aggressive therapy with careful consideration of the dominance and susceptibility patterns of the pathogens present within the institution.
Treatment of nosocomial pneumonia requires aggressive therapy with careful consideration of the dominance and susceptibility patterns of the pathogens present within the institution.
![]() Respiratory tract infections remain the major cause of morbidity from acute illness in the United States and most likely represent the single most common reason patients seek medical attention. This chapter focuses on bacterial and viral infections involving the lower respiratory tract, which includes the tracheobronchial tree and lung parenchyma.
Respiratory tract infections remain the major cause of morbidity from acute illness in the United States and most likely represent the single most common reason patients seek medical attention. This chapter focuses on bacterial and viral infections involving the lower respiratory tract, which includes the tracheobronchial tree and lung parenchyma.
![]() The respiratory tract has an elaborate system of host defenses, including humoral immunity, cellular immunity, and anatomic mechanisms.1 When functioning properly, the host defenses of the respiratory tract are markedly effective in protecting against pathogen invasion and removing potentially infectious agents from the lungs. For the most part, infections in the lower respiratory tract occur only when these defense mechanisms are impaired, as in cases of dysgammaglobulinemia or compromised ciliary function, such as that caused by the chronic inflammation accompanying cigarette smoking. In addition, local defenses may be overwhelmed when a particularly virulent microorganism or excessive inoculum invades lung parenchyma. The majority of pulmonary infections follow colonization of the upper respiratory tract with potential pathogens, which, after achieving sufficiently high concentrations, gain access to the lung via aspiration of oropharyngeal secretions. Less commonly, microbes enter the lung via the blood from an extrapulmonary source or by inhalation of infected aerosolized particles. The specific type of pulmonary infection caused by an invading microorganism is determined by a variety of host factors, including age, anatomic features of the airway, and specific characteristics of the infecting agent.
The respiratory tract has an elaborate system of host defenses, including humoral immunity, cellular immunity, and anatomic mechanisms.1 When functioning properly, the host defenses of the respiratory tract are markedly effective in protecting against pathogen invasion and removing potentially infectious agents from the lungs. For the most part, infections in the lower respiratory tract occur only when these defense mechanisms are impaired, as in cases of dysgammaglobulinemia or compromised ciliary function, such as that caused by the chronic inflammation accompanying cigarette smoking. In addition, local defenses may be overwhelmed when a particularly virulent microorganism or excessive inoculum invades lung parenchyma. The majority of pulmonary infections follow colonization of the upper respiratory tract with potential pathogens, which, after achieving sufficiently high concentrations, gain access to the lung via aspiration of oropharyngeal secretions. Less commonly, microbes enter the lung via the blood from an extrapulmonary source or by inhalation of infected aerosolized particles. The specific type of pulmonary infection caused by an invading microorganism is determined by a variety of host factors, including age, anatomic features of the airway, and specific characteristics of the infecting agent.
The most common infections involving the lower respiratory tract are bronchitis, bronchiolitis, and pneumonia. Lower respiratory tract infections in children and adults most commonly result from either viral or bacterial invasion of lung parenchyma. The diagnosis of viral infections rests primarily on the recognition of a characteristic constellation of clinical signs and symptoms. Because treatment is largely supportive, only occasionally does the diagnosis require laboratory confirmation; this is achieved through serologic tests or identification of the organism by culture or antigen detection in respiratory secretions.2 Laboratory techniques using polymerase chain reaction (PCR), real-time PCR, microarrays, and multiplex ligation-dependent probe amplification, to name a few, have emerged as a means to identify specific pathogens rapidly and accurately.3
In contrast, because bacterial pneumonia usually necessitates expedient, effective, and specific antibiotic therapy, its management depends, in large part, on an understanding of the risk factors for acquiring pneumonia, predominant pathogens within the community, and, if necessary, isolation of the etiologic agent by culture from lung tissue or secretions.4–7 The pharynx is colonized with many organisms that can cause pneumonia; therefore, culture of expectorated sputum can be misleading unless the specimen is examined to ensure that it has originated from the lower respiratory tract. The Gram stain provides the easiest method for distinguishing lower from upper respiratory tract secretions; moreover, through determination of the shape and color of the bacteria, the Gram stain frequently narrows the microbiologic differential diagnosis sufficiently to allow accurate initial therapy. Scanned under low-power microscopy, Gram-stained expectorated upper respiratory tract secretions contain many irregularly shaped epithelial cells with little evidence of inflammation and may not reflect the pathogen. In contrast, a lower-tract specimen from a patient with bacterial pneumonia usually contains multiple neutrophils per high-powered field and a single or predominant bacterial species. Culture of specimens confirmed to originate from the lower tract by Gram stain provides valuable diagnostic information for the majority of patients with bacterial pneumonia. In addition, pneumonia promotes the release of inflammatory mediators and acute-phase proteins such as C-reactive protein, which is significantly elevated in serum in the presence of respiratory tract infections.8
![]() An appropriate treatment regimen for the patient with an uncomplicated lower respiratory tract infection usually can be established by history, physical examination, chest radiograph, and properly collected sputum cultures interpreted in light of the most common lung pathogens and their antibiotic susceptibility patterns within the community.2,4,6,9 More sophisticated or invasive diagnostic methods (e.g., computed tomography, bronchoscopy, and lung biopsy)2 are reserved for severely ill patients who are unable to expectorate sputum or who are not responding to empirical therapy or for pulmonary infections occurring in immunocompromised patients.
An appropriate treatment regimen for the patient with an uncomplicated lower respiratory tract infection usually can be established by history, physical examination, chest radiograph, and properly collected sputum cultures interpreted in light of the most common lung pathogens and their antibiotic susceptibility patterns within the community.2,4,6,9 More sophisticated or invasive diagnostic methods (e.g., computed tomography, bronchoscopy, and lung biopsy)2 are reserved for severely ill patients who are unable to expectorate sputum or who are not responding to empirical therapy or for pulmonary infections occurring in immunocompromised patients.
BRONCHITIS
Bronchitis and bronchiolitis are inflammatory conditions of the large and small elements, respectively, of the tracheobronchial tree. The inflammatory process does not extend to the alveoli. Bronchitis frequently is classified as acute or chronic. Acute bronchitis occurs in individuals of all ages, whereas chronic bronchitis primarily affects adults. Bronchiolitis is a disease of infancy.
Acute Bronchitis
Epidemiology and Etiology
Acute bronchitis occurs most commonly during the winter months, following a pattern similar to those of other acute respiratory tract infections. Cold, damp climates and the presence of high concentrations of irritating substances (e.g., air pollution, cigarette smoke) may precipitate attacks.
![]() Respiratory viruses are by far the most common infectious agents associated with acute bronchitis.10 The common cold viruses (rhinovirus and coronavirus) and lower respiratory tract pathogens (influenza virus and adenovirus) account for the majority of cases. Wider use of reverse transcriptase PCR diagnostic evaluations has identified respiratory viral pathogens not previously described as etiologic agents in acute bronchitis and bronchiolitis including the human metapneumovirus and bocavirus.11 In children, similar pathogens are observed, with the addition of the parainfluenza viruses. Although the true incidence remains to be defined, Mycoplasma pneumoniae appears to be a frequent cause of acute bronchitis. Additionally, Chlamydophila pneumoniae (also referred to as Chlamydophila)12 and B. pertussis13 (agent responsible for whooping cough) have been associated with acute respiratory tract infections. Although a variety of bacteria, including S. pneumoniae, Streptococcus species, Staphylococcus species, and Haemophilus species, may be isolated from throat or sputum culture, these organisms probably represent contamination by normal flora of the upper respiratory tract rather than true pathogens. Although a primary bacterial etiology for acute bronchitis appears rare, secondary bacterial infection may be involved.
Respiratory viruses are by far the most common infectious agents associated with acute bronchitis.10 The common cold viruses (rhinovirus and coronavirus) and lower respiratory tract pathogens (influenza virus and adenovirus) account for the majority of cases. Wider use of reverse transcriptase PCR diagnostic evaluations has identified respiratory viral pathogens not previously described as etiologic agents in acute bronchitis and bronchiolitis including the human metapneumovirus and bocavirus.11 In children, similar pathogens are observed, with the addition of the parainfluenza viruses. Although the true incidence remains to be defined, Mycoplasma pneumoniae appears to be a frequent cause of acute bronchitis. Additionally, Chlamydophila pneumoniae (also referred to as Chlamydophila)12 and B. pertussis13 (agent responsible for whooping cough) have been associated with acute respiratory tract infections. Although a variety of bacteria, including S. pneumoniae, Streptococcus species, Staphylococcus species, and Haemophilus species, may be isolated from throat or sputum culture, these organisms probably represent contamination by normal flora of the upper respiratory tract rather than true pathogens. Although a primary bacterial etiology for acute bronchitis appears rare, secondary bacterial infection may be involved.
Pathogenesis
![]() Because acute bronchitis is primarily a self-limiting illness and rarely a cause of death, few data describing the pathology are available. In general, infection of the trachea and bronchi yields hyperemic and edematous mucous membranes with an increase in bronchial secretions. Destruction of respiratory epithelium can range from mild to extensive and may affect bronchial mucociliary function. In addition, the increase in desquamated epithelial cells and bronchial secretions, which can become thick and tenacious, further impairs mucociliary activity. The probability of permanent damage to the airways as a result of acute bronchitis remains unclear but appears unlikely. However, epidemiologic evaluations support the belief that recurrent acute respiratory infections may be associated with increased airway hyperreactivity and possibly the pathogenesis of asthma or chronic obstructive pulmonary disease (COPD).
Because acute bronchitis is primarily a self-limiting illness and rarely a cause of death, few data describing the pathology are available. In general, infection of the trachea and bronchi yields hyperemic and edematous mucous membranes with an increase in bronchial secretions. Destruction of respiratory epithelium can range from mild to extensive and may affect bronchial mucociliary function. In addition, the increase in desquamated epithelial cells and bronchial secretions, which can become thick and tenacious, further impairs mucociliary activity. The probability of permanent damage to the airways as a result of acute bronchitis remains unclear but appears unlikely. However, epidemiologic evaluations support the belief that recurrent acute respiratory infections may be associated with increased airway hyperreactivity and possibly the pathogenesis of asthma or chronic obstructive pulmonary disease (COPD).
Clinical Presentation
Acute bronchitis usually begins as an upper respiratory infection with nonspecific complaints.10,14 Cough is the hallmark of acute bronchitis and occurs early. The onset of cough may be insidious or abrupt, and the symptoms persist despite resolution of nasal or nasopharyngeal complaints; cough may persist for up to 3 or more weeks. Frequently, the cough initially is nonproductive but then progresses, yielding mucopurulent sputum. In older children and adults, the sputum is raised and expectorated; in the young child, sputum often is swallowed and can result in gagging and vomiting. Substantial discomfort may result from the coughing. Dyspnea, cyanosis, or signs of airway obstruction are observed rarely unless the patient has underlying pulmonary disease, such as emphysema or COPD. Fever, when present, rarely exceeds 39°C (102.2°F) and appears most commonly with adenovirus, influenza virus, and M. pneumoniae infections. The diagnosis typically is made on the basis of a characteristic history and physical examination. Bacterial cultures of expectorated sputum generally are of limited use because of the inability to avoid normal nasopharyngeal flora by the sampling technique. In routine cases, viral cultures are unnecessary and frequently unavailable. Viral antigen detection tests, developed to identify respiratory viral antigens from nasal secretions rapidly, can be obtained in many hospital laboratories and in some practice settings when a specific diagnosis is necessary for clinical or epidemiologic reasons. Cultures, serologic or PCR diagnosis of M. pneumoniae, and culture, direct fluorescent antibody detection, or PCR for B. pertussis should be obtained in prolonged or severe cases when epidemiologic considerations would suggest their involvement.
TREATMENT
Desired Outcome
In the absence of a complicating bacterial superinfection, acute bronchitis almost always is self-limiting. The goals of therapy are to provide comfort to the patient and, in the unusually severe case, to treat associated dehydration and respiratory compromise.10
General Approach to Treatment
![]() Treatment of acute bronchitis is symptomatic and supportive in nature. Reassurance and antipyretics frequently are all that are needed. Bedrest for comfort may be instituted as desired. Patients should be encouraged to drink fluids to prevent dehydration and possibly to decrease the viscosity of respiratory secretions. Mist therapy (use of a vaporizer) may promote the thinning and loosening of respiratory secretions.
Treatment of acute bronchitis is symptomatic and supportive in nature. Reassurance and antipyretics frequently are all that are needed. Bedrest for comfort may be instituted as desired. Patients should be encouraged to drink fluids to prevent dehydration and possibly to decrease the viscosity of respiratory secretions. Mist therapy (use of a vaporizer) may promote the thinning and loosening of respiratory secretions.
Pharmacologic Therapy
Mild analgesic–antipyretic therapy often is helpful in relieving the associated lethargy, malaise, and fever. Aspirin or acetaminophen (650 mg in adults or 10 to 15 mg/kg per dose in children; maximum daily adult dose <4 g and pediatric dose 60 mg/kg) or ibuprofen (200 to 800 mg in adults or 10 mg/kg per dose in children; maximum daily adult dose 3.2 g and pediatric dose 40 mg/kg) should be administered every 4 to 6 hours. In children, aspirin should be avoided and acetaminophen used as the preferred agent because of the possible association between aspirin use and the development of Reye’s syndrome.15
Use of ibuprofen as an antipyretic has increased. The drug’s antipyretic efficacy appears identical to that of aspirin or acetaminophen, although its duration of antipyretic effect may be slightly longer (e.g., 3 to 4 hours for aspirin and acetaminophen vs. 5 to 6 hours for ibuprofen). Caution should be exercised in the administration of ibuprofen for patients younger than 6 months, elderly patients, and individuals with poor renal function. Aspirin and ibuprofen inhibit prostaglandin synthesis and may adversely influence renal function in these predisposed patient populations.
Patients may present with mild to moderate wheezing. In otherwise healthy patients, no meaningful benefits have been described with the use of oral or aerosolized β2-receptor agonists and/or oral or aerosolized corticosteroids. A Cochrane review showed limited benefit of β2-receptor agonists in both pediatric and adult patients compared with the side effects; however, in adults with airflow obstruction there was a trend toward improvement.16 Some clinicians, despite no data, may initiate a brief trial (e.g., ~5 to 7 days) of oral or inhaled corticosteroid for patients with persistent (>14 to 20 days), troublesome cough. Despite several studies, none support the use of mucolytic agents.
Patients suffering from acute bronchitis frequently medicate themselves with nonprescription cough and cold remedies containing various combinations of antihistamines, sympathomimetics, and antitussives despite the lack of definitive evidence supporting their effectiveness. In fact, the tendency of these agents to dehydrate bronchial secretions could aggravate and prolong the recovery process. Although not recommended for routine use, persistent, mild cough, which may be bothersome, can be treated with dextromethorphan; more severe coughs may require intermittent codeine or other similar agents.10,14 In severe cases, the cough may be persistent enough to disrupt sleep, and use of a mild sedative–hypnotic, concomitantly with a cough suppressant (e.g., codeine), may be desirable. However, antitussives should be used cautiously when the cough is productive. The primary or supplemental use of expectorants is questionable because their clinical effectiveness has not been well established.
Routine use of antibiotics for treatment of acute bronchitis should be discouraged due to limited benefit.10,17 In previously healthy patients who exhibit persistent fever or respiratory symptoms for more than 4 to 6 days or for predisposed patients (e.g., elderly, immunocompromised), the possibility of a concurrent bacterial infection should be suspected. When possible, antibiotic therapy should be directed toward anticipated respiratory pathogen(s) (i.e., S. pneumoniae). M. pneumoniae, if suspected by history or if confirmed by culture serology or PCR, can be treated with azithromycin. Alternatively and empirically, a fluoroquinolone antibiotic with activity against these suspected pathogens (e.g., levofloxacin) can be used. During known epidemics involving the influenza A virus, amantadine or rimantadine may be effective in minimizing associated symptoms if administered early in the course of the disease, although treatment with adamantanes is no longer recommended by the Centers for Disease Control and Prevention (CDC) due to increasing influenza resistance to these two agents.18 The neuraminidase inhibitors (e.g., zanamivir and oseltamivir) are active against both influenza A and B viral infections and may reduce the severity and duration of the influenza episode if administered promptly during the onset of the viral infection and are the preferred treatment (see Chap. 87).19,20 Unfortunately, the incidence of influenza virus resistance to available antiviral drugs is increasing.18,21
Chronic Bronchitis
Epidemiology and Etiology
Chronic bronchitis, a component of COPD, is a clinical diagnosis for a nonspecific disease that primarily affects adults. An in-depth presentation of the spectrum and management of COPD is given in Chapter 16. This section will focus solely on chronic bronchitis, a disease that affects most patients with COPD but can develop in patients with normal spirometry.22 In developed countries the prevalence of chronic bronchitis is slightly higher in men than in women and possibly more common in whites.22,23
![]() Chronic bronchitis is defined clinically as the presence of a chronic cough productive of sputum lasting more than 3 consecutive months of the year for 2 consecutive years without an underlying etiology of bronchiectasis or tuberculosis. The disease is a result of several contributing factors; the most prominent include cigarette smoking, exposure to occupational dusts, fumes, and environmental pollution, and host factors (e.g., genetic factors and bacterial [and possibly viral] infections). The contribution of each of these factors and of others (either alone or in combination) to chronic bronchitis is unknown. Cigarette smoke is a well-known airway irritant and is believed to be the predominant factor in the etiology of chronic bronchitis. Although previously assumed the most common etiologic cause of chronic bronchitis, more strict prohibition of public smoking and the resultant decrease in chronic tobacco smokers, particularly in developed countries, underscores the importance of other factors as causes of this chronic disease. Additional airway irritants including occupational dust, chemicals, or air pollution, either alone or more probably in combination, are also responsible for the pathogenesis of chronic bronchitis. Furthermore, genome-wide association studies have begun to identify single-nucleotide polymorphisms that may have clinical relevance in COPD; see Chapter 16.24 Lastly, the influence of recurrent respiratory tract infections during childhood or young adult life on the later development of chronic bronchitis remains obscure, but recurrent respiratory infections may predispose individuals to the development of chronic bronchitis.23 Whether these recurrent respiratory tract infections are a result of unrecognized anatomic abnormalities of the airways or impaired pulmonary defense mechanisms is unclear.
Chronic bronchitis is defined clinically as the presence of a chronic cough productive of sputum lasting more than 3 consecutive months of the year for 2 consecutive years without an underlying etiology of bronchiectasis or tuberculosis. The disease is a result of several contributing factors; the most prominent include cigarette smoking, exposure to occupational dusts, fumes, and environmental pollution, and host factors (e.g., genetic factors and bacterial [and possibly viral] infections). The contribution of each of these factors and of others (either alone or in combination) to chronic bronchitis is unknown. Cigarette smoke is a well-known airway irritant and is believed to be the predominant factor in the etiology of chronic bronchitis. Although previously assumed the most common etiologic cause of chronic bronchitis, more strict prohibition of public smoking and the resultant decrease in chronic tobacco smokers, particularly in developed countries, underscores the importance of other factors as causes of this chronic disease. Additional airway irritants including occupational dust, chemicals, or air pollution, either alone or more probably in combination, are also responsible for the pathogenesis of chronic bronchitis. Furthermore, genome-wide association studies have begun to identify single-nucleotide polymorphisms that may have clinical relevance in COPD; see Chapter 16.24 Lastly, the influence of recurrent respiratory tract infections during childhood or young adult life on the later development of chronic bronchitis remains obscure, but recurrent respiratory infections may predispose individuals to the development of chronic bronchitis.23 Whether these recurrent respiratory tract infections are a result of unrecognized anatomic abnormalities of the airways or impaired pulmonary defense mechanisms is unclear.
Numerous consensus statements and published authoritative guidelines define chronic bronchitis and emphysema as the two main components of COPD/chronic obstructive lung disease (COLD).22,23,25 The Global Initiative for Chronic Obstruction Lung Disease (GOLD)22 guidelines document does not distinguish these two diagnoses (e.g., emphysema or chronic bronchitis) in the definition of COPD, but it does define COPD as a disease characterized by airflow obstruction that is not fully reversible and progressive. The GOLD guidelines provide a COPD classification scoring system according to severity that can be helpful in staging patients for intensity of therapy and prognosis.22 Unfortunately, differences in definitions between authoritative organizations22,23,25 may cause confusion in the assignment of patients in clinical trials and thus to assessment and applications of study results to clinical care.
Pathogenesis
Chronic inhalation of an irritating noxious substance compromises the normal secretory and mucociliary function of bronchial mucosa.23 Bronchial biopsy specimens in bronchitic patients underscore the importance of proinflammatory cytokines (e.g., interleukins 1β, 6, and 8, transforming growth factor-β, leukotriene B4, and tumor necrosis factor-α) in the pathogenesis and propagation of the observed inflammatory changes. In chronic bronchitis, the bronchial wall is thickened, and the number of mucus-secreting goblet cells on the surface epithelium of both larger and smaller bronchi is increased markedly. In contrast, goblet cells generally are absent from the smaller bronchi of normal individuals. In addition to the increased number of goblet cells, hypertrophy of the mucous glands and dilation of the mucous gland ducts are observed. As a result of these changes, chronic bronchitics have substantially more mucus in their peripheral airways, further impairing normal lung defenses. This increased quantity of tenacious secretions within the bronchial tree frequently causes mucous plugging of the smaller airways. Accompanying these changes are squamous cell metaplasia of the surface epithelium, edema, and increased vascularity of the basement membrane of larger airways and variable chronic inflammatory cell infiltration. In addition, the amounts of several proteases derived from inflammatory cells are increased and due to COPD-induced defective antiproteases lead to continued destruction of connective tissue. Continued progression of this pathology can result in residual scarring of small bronchi and peribronchial fibrosis augmenting airway obstruction and weakening of bronchial walls.
Clinical Presentation
![]() The hallmark of chronic bronchitis is a cough that may range from a mild to a severe, incessant coughing productive of purulent sputum.22,23 Coughing may be precipitated by multiple stimuli, including simple, normal conversation. Expectoration of the largest quantity of sputum usually occurs on arising in the morning, although many patients expectorate sputum throughout the day. The expectorated sputum usually is tenacious and can vary in color from white to yellow-green. Patients with chronic bronchitis often expectorate as much as 100 mL/day more than normal. As a result, many patients complain of a frequent bad taste in their mouth and of halitosis.
The hallmark of chronic bronchitis is a cough that may range from a mild to a severe, incessant coughing productive of purulent sputum.22,23 Coughing may be precipitated by multiple stimuli, including simple, normal conversation. Expectoration of the largest quantity of sputum usually occurs on arising in the morning, although many patients expectorate sputum throughout the day. The expectorated sputum usually is tenacious and can vary in color from white to yellow-green. Patients with chronic bronchitis often expectorate as much as 100 mL/day more than normal. As a result, many patients complain of a frequent bad taste in their mouth and of halitosis.
The diagnosis of chronic bronchitis is based primarily on clinical assessment and history. Any patient who reports coughing sputum on most days for at least 3 consecutive months each year for 2 consecutive years presumptively has chronic bronchitis.23 The diagnosis of chronic bronchitis is made only when the possibilities of bronchiectasis, cardiac failure, cystic fibrosis, and lung carcinoma have been effectively excluded. In an attempt to be more specific in the diagnosis, some investigators have added the criteria of lost wages for 3 or more weeks. In addition, many clinicians attempt to subdivide their patients based on severity of disease to guide therapeutic interventions. A useful diagnostic/clinical severity-based classification system is often used to categorize patients to assist in defining an acute therapeutic strategy. The classification system used most often utilizes three descriptive categories: (a) simple chronic bronchitis best describes patients with no major risk factors and sputum flora reflects the common associated pathogens where the patient usually responds well to first-line oral antibiotic therapy. (b) Complicated chronic bronchitis refers to those patients with what would be considered a “simple chronic bronchitis” exacerbation, but the patients have two or more disease-associated risk factors such as forced expiratory volume in the first second of expiration (FEV1) <50% (<0.50) predicted, age >64 years, >4 exacerbations per year, home oxygen use, underlying cardiac disease, use of immunosuppressants, or use of antibiotics for an exacerbation within the past 3 months. These group II patients may also harbor drug-resistant pathogens. (c) Severe complicated chronic bronchitis refers to those patients with group II symptoms but clinically are much worse, for example, FEV1 <35% (<0.35) predicted, >4 acute exacerbations per year, increased risk for infection with P. aeruginosa, and presence of pathogens that are multidrug resistant (MDR). The latter patients often require hospitalization and aggressive parenteral antibiotics including combination therapy. A clinical algorithm for the diagnosis and treatment of chronic bronchitic patients with an acute exacerbation incorporating the principles of the clinical classification system is shown in Figure 85–1. The importance of accurate classification for grouping patients of similar disease involvement cannot be overemphasized with respect to assessing publications outlining treatment strategies for these patients.26 Although gross, these classifications attempt to capture specific phenotypes of chronic bronchitis patients. It is hoped that within the next 2 to 4 years, pharmacogenomic advances will provide a more sophisticated tool for defining specific phenotypes linked to specific, optimal therapies.27 The typical clinical presentation of chronic bronchitis is listed in Table 85–1. Comparison of the trends in changes in patient’s physical activity, symptoms, and clinical/physical findings from the patient’s “routine” is extremely helpful in determining the presence and severity of an acute exacerbation. In general, a good clinical relationship exists between the purulence of the sputum and the bacterial load (>90% of cases) and for sputum color, for example, the greener the color, the greater the amount of leukocyte myeloperoxidase (indicating that more inflammatory cells are present).
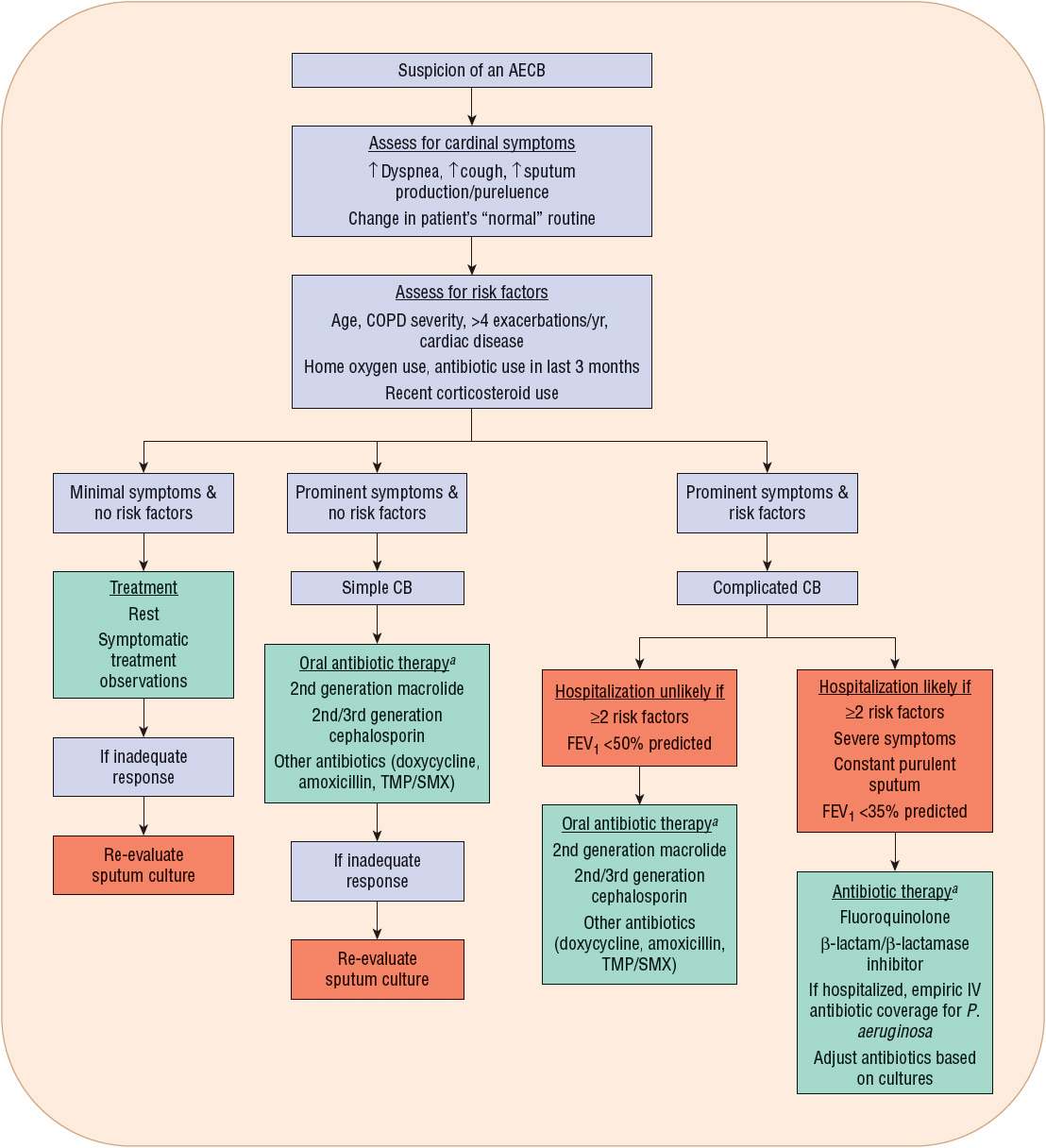
FIGURE 85-1 Clinical algorithm for the diagnosis and treatment of chronic bronchitic patients with an acute exacerbation incorporating the principles of the clinical classification system. (AECB, acute exacerbation of chronic bronchitis; COPD, chronic obstructive pulmonary disease; CB, chronic bronchitis; TMP/SMX, trimethoprim/sulfamethoxazole.) aSee Table 85–3 for commonly used antibiotics and doses. (Adapted from reference 34.)
TABLE 85-1 Clinical Presentation of Chronic Bronchitis
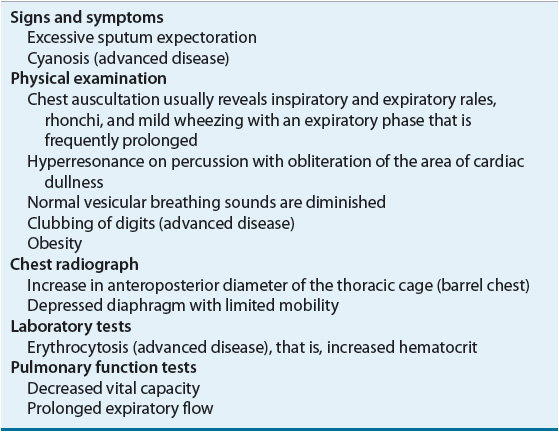
In more advanced stages of chronic bronchitis, physical findings associated with cor pulmonale, including cardiac enlargement, hepatomegaly, and edema of the lower extremities, are observed. In general, chronic bronchitics tend to maintain at least normal body weight and commonly are obese. Radiographic studies are of limited value in either the diagnosis or followup of a patient. The microscopic and laboratory assessments of sputum are considered important components in the overall evaluation of patients with chronic bronchitis. A fresh sputum specimen obtained as an early morning sample is preferred. Comparison of the cellular constituents of chronic bronchitic sputum with those of normal sputum can provide insight into the degree of activity of the disease processes. An increased number of polymorphonuclear granulocytes often suggests continual bronchial irritation, whereas an increased number of eosinophils suggests an allergic component that should be further investigated. Gram staining of the sputum often reveals a mixture of both gram-positive and gram-negative bacteria, reflecting normal oropharyngeal flora and chronic tracheal colonization (in order of frequency) by nontypeable H. influenzae, S. pneumoniae, and M. catarrhalis. Table 85–2 lists the most common bacterial isolates identified from sputum culture for patients experiencing an acute exacerbation of chronic bronchitis (AECB). For patients with more severe airflow disease (e.g., FEV1 <40% [<0.40]), enteric gram-negative bacilli, E. coli, Klebsiella species, Enterobacter species, and P. aeruginosa may be significant pathogens during acute exacerbations.28
TABLE 85-2 Common Bacterial Pathogens Isolated from Sputum of Patients with Acute Exacerbation of Chronic Bronchitis

TREATMENT
Desired Outcome
The goals of therapy for chronic bronchitis are twofold: to reduce the severity of chronic symptoms and to ameliorate acute exacerbations and achieve prolonged infection-free intervals.
General Approach to Treatment
The approach to treatment of chronic bronchitis is multifactorial.23 First and foremost, attempts must be made to reduce the patient’s exposure to known bronchial irritants (e.g., smoking, workplace pollution). A complete occupational and environmental history for determination of exposure to noxious, irritating gases, as well as preference toward cigarette smoking, must be assessed. Often easier discussed than accomplished, honest, yet reasonable attempts should be made with the patient to reduce or eliminate the number of cigarettes smoked daily and to reduce exposure to secondhand smoke. In an organized, coordinated, smoking cessation program, including counseling and hypnotherapy, the adjunctive use of nicotine substitutes (e.g., nicotine gum or patch) or other pharmacotherapy (e.g., bupropion, varenicline) may promote the reduction or complete withdrawal from cigarette smoking. Often just as difficult is modification of exposure to irritating substances within the home and workplace.
![]() Measures to provide pulmonary toilet can be instituted. During acute pulmonary exacerbations of the disease, the patient’s ability to mobilize and expectorate sputum may be reduced dramatically. In these instances, attempts at postural drainage techniques, with instruction, active participation, or both from a respiratory therapist, may assist in promoting clearance of pulmonary secretions. In addition, humidification of inspired air may promote the hydration (liquefaction) of tenacious secretions, allowing for removal that is more productive. Use of mucolytic aerosols, such as N-acetylcysteine and DNAse, is of questionable therapeutic value, particularly considering their propensity to induce bronchospasm (N-acetylcysteine) and their excessive cost. A Cochrane meta-analysis of mucolytic therapy in subjects with chronic bronchitis or COPD found that treatment with mucolytics was associated with a small reduction in acute exacerbations and did not cause any harm, improve quality of life, or slow the decline of lung function.29 The clinical benefit may be greater for chronic bronchitics/COPD patients who have frequent or prolonged exacerbations and are unable to utilize inhaled corticosteroids or long-acting β2-agonists.29 Although limited data are available, chronic use of oral or aerosolized bronchodilators may be of benefit by increasing mucociliary and cough clearance. For patients with moderate to severe COPD, combination therapy with a long-acting β2-agonist and inhaled corticosteroid led to decreased exacerbations and rescue medication use while it also improved quality of life, lung function, and symptom scores compared with long-acting β2-agonist monotherapy.30 Furthermore, patients may benefit from inhaled corticosteroids; patients with severe disease (FEV1 <50% [<0.50]) with a history of frequent exacerbations should receive chronic inhaled corticosteroid therapy. Phosphodiesterase 4 (PDE4) inhibitors are a new class of drug approved for use in chronic bronchitis as add-on therapy for Group C and D COPD patients22,31,32 (see Chap. 16 for a full description of COPD classifications); use of systemic corticosteroid therapy (oral or IV) for patients with an acute exacerbation significantly reduces treatment failures and the need for additional medical treatment.22 Finally, in the face of an acute exacerbation, a trial of antibiotics directed against the most likely underlying pathogens should be initiated.
Measures to provide pulmonary toilet can be instituted. During acute pulmonary exacerbations of the disease, the patient’s ability to mobilize and expectorate sputum may be reduced dramatically. In these instances, attempts at postural drainage techniques, with instruction, active participation, or both from a respiratory therapist, may assist in promoting clearance of pulmonary secretions. In addition, humidification of inspired air may promote the hydration (liquefaction) of tenacious secretions, allowing for removal that is more productive. Use of mucolytic aerosols, such as N-acetylcysteine and DNAse, is of questionable therapeutic value, particularly considering their propensity to induce bronchospasm (N-acetylcysteine) and their excessive cost. A Cochrane meta-analysis of mucolytic therapy in subjects with chronic bronchitis or COPD found that treatment with mucolytics was associated with a small reduction in acute exacerbations and did not cause any harm, improve quality of life, or slow the decline of lung function.29 The clinical benefit may be greater for chronic bronchitics/COPD patients who have frequent or prolonged exacerbations and are unable to utilize inhaled corticosteroids or long-acting β2-agonists.29 Although limited data are available, chronic use of oral or aerosolized bronchodilators may be of benefit by increasing mucociliary and cough clearance. For patients with moderate to severe COPD, combination therapy with a long-acting β2-agonist and inhaled corticosteroid led to decreased exacerbations and rescue medication use while it also improved quality of life, lung function, and symptom scores compared with long-acting β2-agonist monotherapy.30 Furthermore, patients may benefit from inhaled corticosteroids; patients with severe disease (FEV1 <50% [<0.50]) with a history of frequent exacerbations should receive chronic inhaled corticosteroid therapy. Phosphodiesterase 4 (PDE4) inhibitors are a new class of drug approved for use in chronic bronchitis as add-on therapy for Group C and D COPD patients22,31,32 (see Chap. 16 for a full description of COPD classifications); use of systemic corticosteroid therapy (oral or IV) for patients with an acute exacerbation significantly reduces treatment failures and the need for additional medical treatment.22 Finally, in the face of an acute exacerbation, a trial of antibiotics directed against the most likely underlying pathogens should be initiated.
Pharmacologic Therapy
For patients who consistently demonstrate clinical limitation in airflow, a therapeutic challenge of a short-acting β2-agonist bronchodilator (e.g., as albuterol aerosol) should be considered. Pulmonary function tests should be performed before and after β2-agonist aerosol administration for more objective determination of a patient’s propensity to benefit from supplemental aerosol therapy. Sufficient published experience supports the use of inhalation therapy with a β2-agonist for patients with chronic bronchitis (COPD) to improve pulmonary function and exercise tolerance and to reduce the sense of breathlessness.23 Regular use of a long-acting β-receptor agonist aerosol (e.g., salmeterol, formoterol) in responsive patients may be more effective and probably more convenient than short-acting β2-receptor agonists. The aerosol route for β2-receptor agonist and/or corticosteroid administration is favored over systemic formulations for improved patient acceptance and compliance and to minimize the number and magnitude of associated adverse effects.22 Chronic inhalation of the salmeterol/fluticasone combination has been associated with improved pulmonary function and quality of life.
Published experience with inhaled anticholinergic drugs, including ipratropium and tiotropium, is limited. In stable patients, long-term inhalation of ipratropium has been associated with a decreased frequency of cough, less severe coughing, and a decrease in the volume of expectorated sputum. Once-daily tiotropium inhalation was associated with significant bronchodilation and dyspnea relief compared with placebo but had no significant effect on the incidence or severity of cough.22,23 Although chronic theophylline administration has been used extensively in the past, this therapy is used with decreasing frequency in favor of aerosolized β2-receptor agonists. A salmeterol/fluticasone combination markedly reduced the number of chronic bronchitis–associated emergency room visits and hospitalizations compared with an ipratropium-based regimen.33
PDE4 inhibitors, compared with the nonselective phosphodiesterase inhibitor theophylline, only affect phosphodiesterase in the airway smooth muscle, immune (eosinophils, monocytes, and neutrophils), and proinflammatory cells. Roflumilast, the only available PDE4 inhibitor, was approved by the U.S. FDA in 2011 for use in patients with severe COPD with chronic bronchitis and a history of exacerbations. Moderate to severe exacerbations treated with steroids were reduced by 15% to 20% in patients receiving roflumilast and add-on therapy with long-acting β2-agonists had effects on lung function.22 GOLD guidelines suggest the use of PDE4 inhibitors as an alternative therapy for COPD Group C patients with chronic bronchitis and as add-on therapy for COPD Group D patients with chronic bronchitis.22 The side effect profile for PDE4 inhibitor differs from inhaled COPD medications; patients may experience nausea, vomiting, decreased appetite, and sleep disturbances.22,32 Intolerable nausea and vomiting had occurred with cilomilast, a PDE4 inhibitor primarily targeting isozyme PDE4D located in the CNS compared with isozyme PDE4B, which is located in immune and proinflammatory cells.31 Roflumilast is nonselective for the PDE4 isozymes and is less likely to cause nausea and vomiting. Unexplained weight loss has also been reported in studies; therefore, a patient’s weight should be taken into consideration prior to treatment and monitored during therapy.
Use of antimicrobials for treatment of chronic bronchitis has been controversial but is becoming more accepted. Numerous comparative evaluations, including placebo-controlled studies of antibiotic administration with acute and chronic treatment of chronic bronchitics, have suggested definite clinical benefit, whereas other similar studies have not.22,25,26,28,34 The antibiotics selected most frequently possess variable in vitro activity against the common sputum isolates H. influenzae, S. pneumoniae, M. catarrhalis, and M. pneumoniae. Conflicting published results appear independent of the antibiotic used or the regimen compared. The wide disparity that exists in the results from these studies, combined with the difficulties in recognition and lack of standardized diagnostic criteria for acute exacerbations of chronic bronchitis, serves as the basis for the enormous controversy surrounding the use of antibiotics in this condition.23 A review of 14 double-blinded, randomized clinical trials compared fluoroquinolones with more standard antibiotic regimens (e.g., macrolides, azalides, oral cephalosporins, and the combination drug amoxicillin/clavulanate).28 As expected, no significant differences were observed between treatment arms. However, in a small subset of studies (n = 4), the sputum culture became negative in a significantly higher number of fluoroquinolone-treated patients. Other studies showed an increase in the interval between acute exacerbations for patients who received fluoroquinolone therapy. An additional advantage of fluoroquinolone therapy is the short course (e.g., 5 days) and once-daily dosing compared with other antibiotic regimens.
A useful paradigm for the assessment and treatment of acute exacerbations of chronic bronchitis and antibiotic decision making is shown in Figure 85–1. Furthermore, many clinicians will use the so-called Anthonisen criteria to determine if antibiotic therapy is indicated.35 With the Anthonisen criteria if a patient exhibits two of the following three criteria during an AECB, the patient will most likely benefit from antibiotic therapy and, thus, should receive a treatment course: (a) increase in shortness of breath; (b) increase in sputum volume; (c) production of purulent sputum. There are greater healthcare costs for patients who are noncompliant with their antibiotic regimen for their AECB.36
The increasing resistance of the common bacterial pathogens to first-line agents further complicates antibiotic selection. As many as 30% to 40% of H. influenzae and 95% to 100% of M. catarrhalis isolates produce β-lactamases. Moreover, up to 40% of S. pneumoniae isolates demonstrate resistance to penicillin (minimum inhibitory concentration [MIC] = 0.1 to 2 mg/L), with approximately 20% of isolates being highly resistant (MIC >2 mg/L). Concern regarding S. pneumoniae resistance is increasing, now ≥30% for macrolides. Despite these changes in bacterial susceptibility, the current recommendation is to initiate therapy with first-line agents in less severely affected patients (see Fig. 85–1). Trimethoprim/sulfamethoxazole has been extremely useful for patients with less severe disease.37 However, the public campaign in the United Kingdom by the Committee of Safety of Medicines to discourage the use of trimethoprim/sulfamethoxazole based on rare but possibly life-threatening cases of Stevens-Johnson syndrome has markedly reduced the use of this agent worldwide. For patients with more moderate to severe disease, many clinicians will begin antibiotic therapy with the second-line agents, amoxicillin/clavulanate, a macrolide (such as azithromycin or clarithromycin, although they are being used less frequently), and more frequently with a fluoroquinolone, such as levofloxacin (see Fig. 85–1).22,28,34,38
Regardless of the antibiotic selected, predetermined outcome measures should be monitored closely for each patient to determine the success or failure of the therapeutic intervention. Oral antibiotics with broader antibacterial spectra (e.g., amoxicillin/clavulanate, fluoroquinolones, or azalides) that possess potent in vitro activity against sputum isolates are increasingly becoming first-line antibiotics as initial therapy for treatment of acute exacerbations of chronic bronchitis.22,28,33,34,37
An important clinical outcome variable directing drug selection and criteria for beginning antibiotics in individual patients is the infection-free period when chronic bronchitics are off antibiotics. The actual length of the infection-free time period and the change in the number of physician office visits and hospital admissions with a particular antibiotic regimen are extremely important to identify, whenever possible, for each patient. The antibiotic regimen that results in the longest infection-free period defines the “regimen of choice” for specific patients for future acute exacerbations of their disease. Trials of prophylactic antibiotic use may provide a slight benefit in exacerbation rates; however, GOLD guidelines do not currently support this indication.22
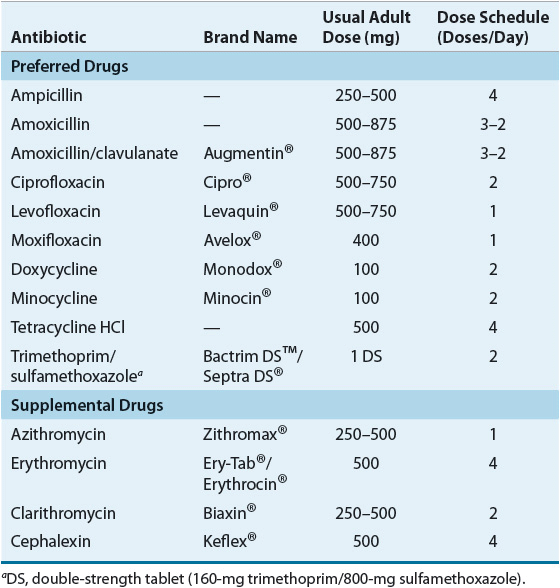
Antibiotics should be selected that are effective against responsible pathogens, demonstrate the least risk of drug interactions, and can be administered in a manner that promotes compliance. Antibiotics commonly used for treatment of these patients and their respective adult starting doses are listed in Table 85–3. Doses of antibiotics should be adjusted as needed to the desired clinical effect and the lowest incidence of acceptable side effects. A frequently used clinical strategy to enhance the duration of symptom-free periods incorporates higher-dose antibiotic regimens using the upper limit of the recommended daily antibiotic dose for a period of 5 to 7 days. More clinicians are electing to limit their antibiotic treatment regimen to 5 days as compelling data continue to support equal efficacy and possibly less side effects with short-duration antibiotic therapy versus longer treatment regimens (>7 days).39
TABLE 85-3 Oral Antibiotics Commonly Used for the Treatment of Acute Respiratory Exacerbations in Chronic Bronchitis
BRONCHIOLITIS
Epidemiology and Etiology
![]() Bronchiolitis is an acute viral infection of the lower respiratory tract that affects approximately 50% of children during the first year of life and 100% by age 2 years. The occurrence of bronchiolitis peaks during the winter months and persists through early spring. Bronchiolitis remains the major reason for hospital admission during the first year of life. The incidence of bronchiolitis appears to be more common in males than in females.40,41
Bronchiolitis is an acute viral infection of the lower respiratory tract that affects approximately 50% of children during the first year of life and 100% by age 2 years. The occurrence of bronchiolitis peaks during the winter months and persists through early spring. Bronchiolitis remains the major reason for hospital admission during the first year of life. The incidence of bronchiolitis appears to be more common in males than in females.40,41
Respiratory syncytial virus (RSV) is the most common cause of bronchiolitis, accounting for up to 75% of all cases. During epidemic periods, the incidence of RSV-induced bronchiolitis may approach 90% of cases. Other detectable viruses include parainfluenza, adenovirus, and influenza. Bacteria serve as secondary pathogens in a minority of cases.40,42
Clinical Presentation
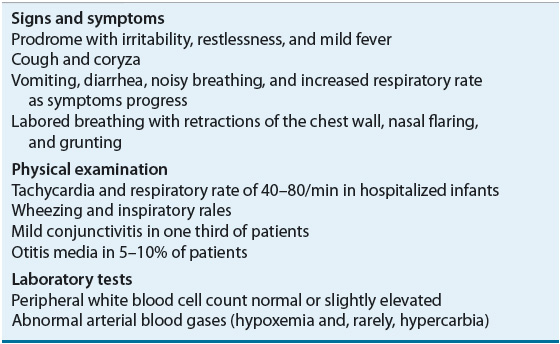
A prodrome suggesting an upper respiratory tract infection, usually lasting from 2 to 8 days, precedes the onset of clinical symptoms (Table 85–4). Due to limited oral intake because of coughing combined with fever, vomiting, and diarrhea, infants frequently are dehydrated. The increased work of breathing and tachypnea most likely further increase fluid loss. In most cases, this clinical picture persists between 3 and 7 days. Although the hospital course of bronchiolitic children often is variable, substantial clinical improvement usually is observed within the first 2 days, with gradual improvement and complete resolution sometimes requiring 4 to 8 weeks.
TABLE 85-4 Clinical Presentation of Bronchiolitis