Langerhans Cell Sarcoma
Sa A. Wang, MD
Key Facts
Terminology
Neoplastic proliferation of Langerhans cells with overtly malignant cytologic features
Clinical Issues
LCS most often presents as de novo mass
Subset of cases can progress from antecedent Langerhans cell histiocytosis (LCH)
Skin & soft tissue are most commonly involved sites
˜ 20% of patients present with lymphadenopathy
Multiorgan involvement is common
Many patients with LCS have clinically aggressive disease
Mortality rate is ˜ 50%
Death often within 2 years
Microscopic Pathology
LCS tends to resemble other types of sarcoma
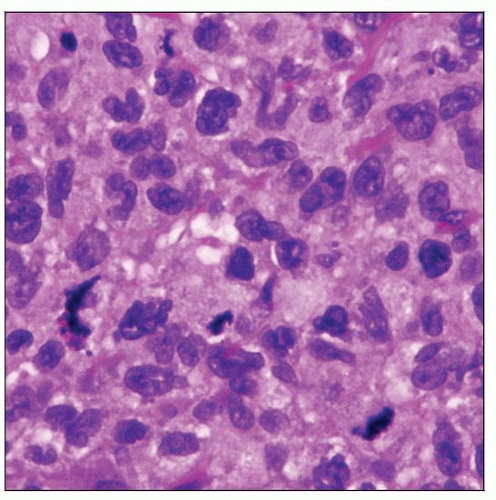
Overtly malignant cytologic features
High mitotic rate (> 50 mitosis/10 HPF)
Large areas of coagulative necrosis
Few neoplastic cells show characteristic cytologic features of Langerhans cells
IHC and ultrastructure similar to LCH
CD1a(+), S100 protein(+), langerin(+)
Birbeck granules
Top Differential Diagnoses
Langerhans cell histiocytosis
Interdigitating dendritic sarcoma
Follicular dendritic cell sarcoma
Histiocytic sarcoma
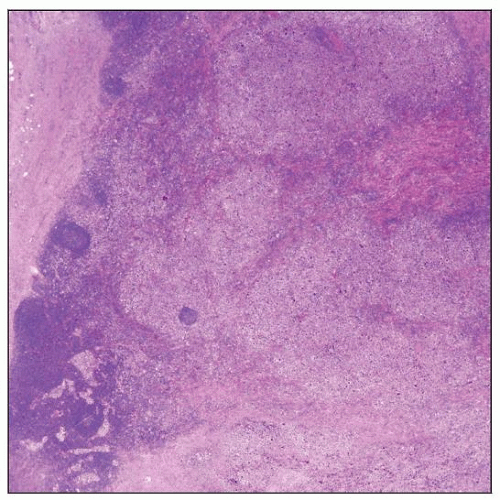 Langerhans cell sarcoma replacing lymph node parenchyma in a 61-year-old man. Uninvolved lymph node is at the bottom left of the field. This neoplasm presented de novo. |
TERMINOLOGY
Abbreviations
Langerhans cell sarcoma (LCS)
Synonyms
Dendritic/histiocytic sarcoma, Langerhans cell type
Malignant histiocytosis X
Malignant Langerhans cell tumor
Definitions
Neoplastic proliferation of overtly malignant Langerhans cells
CLINICAL ISSUES
Epidemiology
Incidence
Rare
Age
Median: 46 years (range: 10-81 years)
Gender
Female predominance; M:F ratio = 1:2
Site
Skin and soft tissue are most commonly involved sites
Multiorgan involvement is common
Lymph nodes, lung, liver, spleen, bone, gallbladder, brain, intestines, pancreas
Primary lymph node involvement in ˜ 20% of patients
Presentation
Langerhans cell sarcoma usually arises de novo
Rare cases of LCS progress from antecedent Langerhans cell histiocytosis (LCH)
Rare case reports
Most patients present with extranodal mass without other symptoms
˜ 40% of patients have stage III or IV disease
˜ 20% have hepatosplenomegaly
˜ 10% have pancytopenia; often associated with bone marrow involvement
Systemic symptoms in subset of patients
Fatigue, night sweats, weight loss
Treatment
Surgical excision, if resectable, is best approach
Chemotherapy and radiation therapy for disseminated disease
Often poor response
Prognosis
Clinically aggressive neoplasm
Many patients show progressive disease
˜ 50% mortality rate; many patients die within 2 years
IMAGE FINDINGS
General Features
CT and MR
Mass lesion
Often show multifocal or disseminated disease
Positron emission tomography (PET)
Abnormal utilization of isotope
MICROSCOPIC PATHOLOGY
Histologic Features
Skin and soft tissue
Resembles other types of sarcoma
Diffuse pattern
Neoplastic cells are overtly malignant
Pleomorphic cells with prominent nucleoli and clumped chromatin
Mitotic rate often high; up to > 50 mitoses/10 high-power fields
Atypical mitotic figures may present
Neoplastic cells can have moderate or abundant cytoplasm, ± foamy or vacuolated
Focal necrosis often present
Eosinophils are few or absent in background
Lymph nodes
LCS may show variety of patterns; often a mixture present
Diffuse effacement
Vaguely nodular
Sinusoidal involvement
Histologic clues to diagnosis of LCS
Neoplastic cells show irregular nuclear contours or nuclear grooves
Usually only small subset of neoplastic cells
Small areas where eosinophils are present in background
Cytologic Features
Very large cells with round or folded nuclei, ± prominent nucleoli, and abundant cytoplasm
Difficult diagnosis to establish without immunohistochemistry
ANCILLARY TESTS
Immunohistochemistry
CD1a(+), S100 protein(+), or langerin/CD207(+)
Expression can be focal or patchy
CD40(+), HLA-DR(+), vimentin(+/-)
CD56/NCAM(+)
CD56 is usually negative in LCH
CD31(+); CD31 is negative in normal Langerhans cells
CD68(+/-), CD45(+/-); when positive, CD68 and CD45 can be weak or variable
CD4(+/-); CD163(-/+)
Lysozyme(-/+); if positive, it is often in only a subset of cells
Fascin(-)
Follicular dendritic cell markers(-)
CD21, CD23, CD35, clusterin, etc.
Molecular Genetics
No evidence of immunoglobulin or T-cell receptor gene rearrangements
Very few cases assessed by human androgen-receptor assay (HUMARA) are reported
“Transdifferentiation” reported
LCS and B-lymphoblastic leukemia
Electron Microscopy
Birbeck granules are present
No evidence of
Desmosomes/junctional specialization
Interdigitating cell processes
DIFFERENTIAL DIAGNOSIS
Langerhans Cell Histiocytosis (LCH)
Langerhans cells have bland cytologic features
“Twisted towel” nuclei with nuclear grooves and open chromatin
Often many eosinophils in background
Some cases of LCH have eosinophilic microabscesses
Necrosis can be present; often associated with eosinophils
Variable number of macrophages and small lymphocytes in background
Multinucleated giant cells can be present
Clinical behavior of LCH can overlap with LCS
Some patients with LCH present with multiorgan involvement
Infants: Letterer-Siwe disease (disseminated)
Young children: Often unisystem multifocal disease
Patients with systemic involvement can have aggressive clinical course
Some cases of LCH can show appreciable mitotic rate
10-20 mitosis/10 HPF, some cases can be up to 30 mitosis/10 HPF
Histologic features that distinguish LCS from LCH
Lesion looks like sarcoma
Immunophenotypic features may distinguish LCS from LCH
CD56 is positive in LCS but negative in LCH
Expression of markers of Langerhans cells is often patchy in LCS
Interdigitating Dendritic Sarcoma
Fascicles, storiform arrays, whorls growth pattern
This pattern may not be obvious in high-grade lesions
Neoplastic cells can be spindled or epithelioid
Immunohistochemistry
S100(+); can be patchy
CD11c(+), HLA-DR(+)
Vimentin(+), fascin(+)
CD4(+/-), CD45/LCA(+/-)
CD68(+/-), CD163(+/-)
CD15(-/+), lysozyme(-/+)
CD1a(-), langerin(-)
Electron microscopy (EM)
No Birbeck granules
Follicular Dendritic Cell Sarcoma
Commonly involves head and neck region
Clinical course can be indolent
Histologic features
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree