CHAPTER 35 Intermediate Filaments
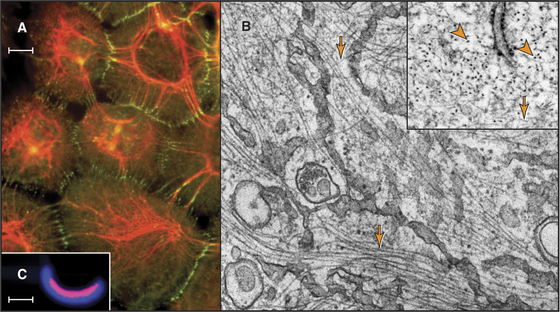
Intermediate filaments (Fig. 35-1) are strong but flexible polymers that provide mechanical support for cells ranging from bacteria to human tissues. These filaments were named intermediate because their diameter of about 10 nm is intermediate between the diameters of the thick and thin filaments in striated muscles (see Figs. 39-3 and 39-8). Cytoplasmic intermediate filaments tend to cluster into wavy bundles that vary in compactness, forming a branching network between the plasma membrane and the nucleus. Desmosomes anchor intermediate filaments to the plasma membrane (see Fig. 31-7) and transmit mechanical forces between adjacent cells. Hemidesmosomes connect intermediate filaments across the plasma membrane to the extracellular matrix.
The continuum of intermediate filaments and junctions prevents excessive stretching of the cells and gives tissues such as epithelia their mechanical integrity. Hair, which is built from cross-linked intermediate filaments, nicely illustrates their flexibility and high tensile strength. Molecular defects in cytoplasmic intermediate filaments or junctions associated with intermediate filaments result in rupture of skin cells and blistering diseases. Defeats in lamins cause a bewildering array of diseases (see Fig. 14-9).
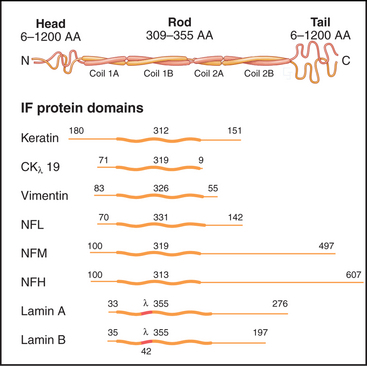
The nearly 70 human genes for intermediate filament proteins are expressed selectively in various cell types. All intermediate filament proteins possess an α-helical coiled-coil domain that forms the core of the filaments, while the flanking N- and C-terminal domains vary considerably in size (Fig. 35-2). These variable domains give each type of intermediate filament distinctive features but made it difficult to appreciate the general features of intermediate filament structure and function until the sequences of the proteins were determined in the 1980s.
Structure of Intermediate Filament Subunits
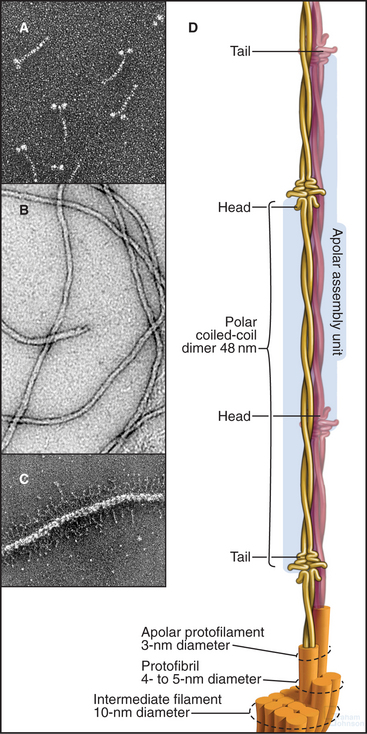
Each intermediate filament protein isoform has a unique amino acid sequence, but all have a rod-like domain between head and tail domains of variable length at the two ends. The rod is a parallel coiled-coil of two α-helices, usually about 47 nm long (Fig. 35-3A). Analysis of sequences, spectroscopic data, and X-ray fiber diffraction from materials composed of intermediate filaments, like wool, established this coiled-coil structure of the rod. Like other coiled-coils (see Fig. 3-10), rod domains of intermediate filament proteins have a heptad repeat of amino acids, the first and fourth residues providing a continuous row of hydrophobic interactions along the interface of the two helices. Zones of positive and negative charge alternate along the rod. When staggered appropriately, these zones provide complementary electrostatic bonds for assembly of filaments. About 20 highly conserved residues at each end of the rod are essential for filament elongation through head-to-tail interactions between dimeric molecules. Assembly studies with mutant proteins suggest that these parts of the rod contribute to lateral associations within filaments. Amino acid sequences indicate three interruptions in the coiled-coil (Fig. 35-2).
Less is known about the structures and functions of the nonhelical N- and C-terminal domains. They can influence assembly and in some cases project from the filament surface (Fig. 35-3C) to interact with other cellular components.
Intermediate filament protein molecules are commonly referred to as dimers, as they consist of two polypeptide chains. Some intermediate filament molecules are homodimers; others are heterodimers. Lamins and class III intermediate filament molecules are parallel dimers of identical polypeptides (Fig. 35-3A), whereas keratins are obligate heterodimers of one acidic (class I) and one basic (class II) keratin polypeptide. Many intermediate filament molecules form stable, partially overlapping, antiparallel molecular dimers (referred to as tetramers; Fig. 35-3D) that are believed to be intermediates in polymer assembly.
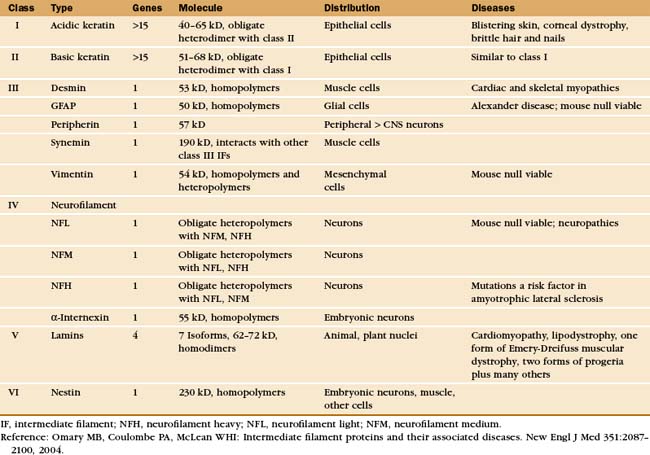
In Bacteria, the intermediate filaments are required for the asymmetrical shape of Caulobacter crescentus (Fig. 35-1C). Genes for animal intermediate filament proteins arose in early metazoan cells, encoding nuclear lamins (see Fig. 14-7). Most metazoans including chordates, mollusks, insects, and nematodes (see Fig. 2-9) retain genes for lamins. An invertebrate organism in the lineage leading to chordates duplicated a lamin gene, which was subsequently modified by deletion of the nuclear localization sequence and the CAAX box (a C-terminal prenylation site, see Fig. 7-9) giving rise to cytoplasmic intermediate filaments. (Cytoplasmic intermediate filaments of mollusks and nematodes retain more features of lamins, so they seem to have evolved separately.) After deletion of the codons for 42 residues of the coiled-coil in early chordates, further gene duplication and divergence produced the four families of genes for cytoplasmic intermediate filaments of vertebrates (Table 35-1).
Polymer Structure
Intermediate filaments are about 10 nm in diameter with wavy profiles in electron micrographs of thin sections of cells (Fig. 35-1B) or after negative staining isolated filaments (Fig. 35-3B). In some cases, the N- or C-terminal domains project from the surface (Fig. 35-3C). The most carefully studied intermediate filaments are built from four-chain, antiparallel molecular dimers that associate end to end in protofibrils like the strands of a rope (Fig. 35-3D). A cross section has up to 16 coiled-coils, but their internal arrangement is not known. Since tetramers lack polarity, intermediate filaments are considered to be apolar (i.e., both ends of the filament are equivalent; Fig. 35-3D). This is a striking difference from actin filaments (see Fig. 33-8) and microtubules (see Fig. 34-5), which depend on their polarity for many functions, including the unidirectional motion of motor proteins.