Environmental
Hospital system
Postoperative care
Home situation
City vs. remote residence
Personal
Social
Smoking
Behavioral
Occupational
Socioeconomic
Cultural
Psychological state
Genetic
Anomalies
Hyper-coagulation states
Anticoagulation states
Trauma
Direct contusion/laceration
Joint strain/fracture
Immobility
Injury mechanism
Pressure effects
Positional
Constriction
Thermal
Hypothermia
Hyperthermia
Burns
Electric shock
Respiratory
Hypoxia
Atelectasis
Respiratory depression/compromise
Aspiration pneumonitis
Pneumonia
Pneumothorax
Hemothorax
Cardiac
Cardiac failure
Arrhythmias
Hypotension
Anemia
Hemolysis
Blood loss
Dilutional
Neurological and psychiatric
Organic brain syndrome
Acute confusional state
Chronic issues
Pain
Fluid and electrolyte imbalance
Fluid overload
Dehydration
Hypokalemia
Hyperkalemia
Hyponatremia
Hypocalcemia
Allergy
Rash
Anaphylaxis
Immunological
Immunosuppression
Immune/vaccinated state
Renal
Acute renal failure
Chronic renal impairment
Hepatic
Hepatic failure
Cholestasis
Jaundice
Hematological
Deep venous thrombosis (DVT)/pulmonary embolism (PE)
Myelosuppression
Bleeding
Excessive anticoagulation
Drug effects
Infective
Viral – shingles, MRSA, HIV and Hep B and C
Cellulitis – IV and CVC line
Urinary
Endocarditis and myocarditis
Antibiotic-related complications
Pseudomembranous colitis
Diarrhea
Nausea
Rash
Anaphylaxis
Candidiasis and fungal infections
Resistance
Gastroenterological
Pancreatitis
Peptic ulcer
Nausea
Vomiting
Constipation
Metabolic
Hyperglycemia and diabetes
Hypoglycemia
Sheehan’s
Cushing’s
Hypothyroidism
Hyperthyroidism
Hypoadrenalism
Pheochromocytoma
Addison’s disease
Drug effects
Direct effects, e.g., narcotic, diuretic, and amphetamine
Interactions
Allergy/hypersensitivity
Drug withdrawal, e.g., alcohol and benzodiazepines
Idiosyncratic
Nutritional
Malnutrition
Obesity
Deficiency
Inability to eat
“The key is to be observant towards preexisting preoperative factors or medical problems that if averted, lessened, taken into account, countered, or avoided might lead to a better postoperative clinical outcome.”
Exacerbation of Medical Conditions as Perioperative Complications
Although not often regarded as strictly complications of surgery, preexisting medical conditions may become unstable or worsen in association with surgery or become evident. Such instability may, however, be every bit as important as direct surgical complications. The condition for which surgery is required (e.g., a perforated peptic ulcer) may place additional “stress” on the patient’s preexisting medical problem(s) prior to surgery. This is especially true for elderly patients with multiple comorbidities who require surgery and for those patients with a substantial delay to access to medical services, delayed diagnosis, or delayed intervention. When surgery is performed on a medically unfit patient, the additional strain of a general anesthetic and a surgical procedure may directly worsen the preexisting medical condition (e.g., asthma, COPD, diabetes, cardiac failure, renal failure, or psychiatric disorders). Preoperative optimization and planning may avert or improve the situation and reduce risk. Medical/anesthetic clinics to optimize the patient early, prior to surgery, and “plan” for the best operative state and best intra- and postoperative care are ideal for such situations and patients.
Postoperative Pain
Certain signs or symptoms are not detailed above, but are sometimes considered to be “complications” or possibly regarded as “associated consequences” of surgical intervention. These include pain, which may be considered as either a natural consequence or a pathological entity, depending on the severity, duration, response to therapy, and the perceived degree of harm to the patient. This is dealt with in Chap. 7.
Postoperative Temperature
Raised temperature usually signifies underlying pathology. This might depend on timing of the development of elevated temperature. Depending on the anesthesia used and surgery type, for example:
A raised temperature in the acute postoperative phase – respiratory, urinary, or I/V drip site infection are relatively common causes.
A raised temperature 48–72 h after surgery – respiratory, wound infection, bowel leakage, and deep infection are possible causes.
A raised temperature 3–14 days after surgery – late respiratory, delayed wound infection, deep infection, abscess formation, and DVT are not uncommon causes.
A raised temperature >14 days after surgery – abscess after antibiotic therapy, infection related to foreign material, infected hematoma, and separate unrelated infection.
Normal body temperature is between 36.1 and 37.2 °C (97–99 °F) measured orally. Elevated temperature is usually above 37.5 °C (about 99.5 °F). High fevers above 38 °C (100.4 °F), especially with rigors, usually signify septicemia (blood sepsis), typically caused by biliary, urinary, or respiratory infection. Swinging fevers often signify collections of pus or infected fluid (abscess formation). Night fevers and sweats frequently indicate deep sepsis (abscess) or occasionally lymphoma (“B” symptoms).
Complications: Predisposing and Perpetuating Factors
A range of predisposing factors may influence wound healing and overall health, and these may influence the risk of an individual developing a complication. Some of these factors may act as perpetuating factors for persistence of a particular complication, which may make the complication chronic, rather than acute or subacute.
It is worth noting that some of these predisposing or perpetuating factors may not only affect the development of a particular complication, but, when that complication develops, the initial predisposing or perpetuating factors can be made worse by the complication. For example, malnutrition may predispose an individual to poor wound healing, fistula, or sinus formation, and the consequent open wound, fistula, or sinus may cause further malnutrition, so perpetuating the problem. Malnutrition in this case represents both a predisposing and perpetuating factor.
The situation often arises where more perpetuating factors develop as a complication worsens, and this in turn, leads to further worsening of the complication. Additionally, other new complications can develop as the patient’s condition worsens, leading to a “snowballing effect” where multiple complications develop from the presence of the initial complication. In its worst form, multisystem organ failure is where multiple body systems fail, following initial failure of one system. For example, initial respiratory failure may produce hypoxia, resulting in cardiac insufficiency, ischemia, and failure, and leading to poor circulation with renal failure. The patient may have coped adequately with failure of one system, but not two or three.
The presence of underlying comorbidities (diseases), such as mild cardiac ischemia, diabetes, mild renal impairment, or respiratory disease from smoking, may increase the susceptibility and risk of an individual to complications and the development of multiple complications from one initial severe complication.
Analogous to “the straw breaking the camel’s back” – where function remains observably, outwardly normal until a relatively “minor event” disturbs the equilibrium and a cascade of complications results in failure of multiple systems, each contingent upon the other.
Examples of predisposing or perpetuating factors include:
Diabetes
Immunosuppression
Renal failure
Advancing age
Malnutrition
Jaundice
Steroid therapy
Preexisting infection
Cardiac disease
Respiratory disease
Complications of Wound Healing
In most patients, wound healing is a rapid and predictable process, with the external skin wound healing over 5–14 days: 5–8 days on the face, neck, and scalp and 10–14 days on the remainder of the body limbs, with the possible exception of the distal lower limbs, where healing may be slower at 10–18 days, especially in the elderly. Some patients consistently heal completely and well with little scarring, while others heal more slowly, with some people forming hypertrophic or keloidal scars, which can be unpredictable and unsightly. Curiously, hypertrophic or keloidal scars may form in only part of the, rather than along the full length (Fig. 2.1). The internal wounds of mucosa and bowel generally heal more rapidly than the respective skin wounds, such that bowel anastomoses are often almost completely healed in 3–4 days, provided deleterious factors are not present. Wounds of muscle, fat, ligament, and other soft tissues usually heal slightly slower than skin wounds; however, insufficient rest, ischemia, or repetitive trauma may significantly delay healing of these tissues, as is sometimes seen with “sporting” type or other traumatic injuries.

Fig. 2.1
Keloid scar. Note that only part of the scar appears involved, which is not an uncommon phenomenon
Factors Inhibiting Wound Healing
Many of the factors that are known to be associated with delayed or failed wound healing are “local” because they affect the wound healing process locally. Even if factors are systemic in nature, the environment of the wound is influenced at a local tissue level. Examples are malnutrition and diabetes.
Making a distinction between systemic and local is therefore not always particularly helpful, except possibly as predictor of potential problems or as an “aide-memoire.” However, some local factors are truly localized to the region of the wound, such as wound closure tension, foreign material, devitalized tissue, local vascular compromise, localized tissue ischemia, and direct involvement of the wound with neoplasia.
The factors that can inhibit wound healing include those shown in Table 2.2.
Table 2.2
The factors that can inhibit wound healing
General factors |
Diabetes |
Immunosuppression |
Renal failure |
Extremes of age [very young or very old] |
Liver failure, jaundice |
Malnutrition (including protein deficiency) |
Steroids |
Carcinoma (systemic effects and/or local involvement) |
Radiotherapy |
Chronic infection |
Chemotherapy |
Local factors |
Infection (local) |
Hematoma formation |
Wound separation, dehiscence |
Foreign body (sutures, mesh, tubes, extraneous material) |
Ischemia (vascular insufficiency, hypoxia, devitalized tissues) |
Wound closure tension (partly ischemic, partly mechanical) |
Factors Sustaining a Fistula or Sinus
A fistula is defined as a communicating conduit, often epithelialized, between two epithelial-lined surfaces or cavities (e.g., between the bowel and skin). A sinus is defined as a communication between one or more non-epithelial-lined surfaces and another surface or cavity (e.g., between an abscess cavity and skin).
Both have similar effects and similar properties that can lead to their persistence shown in Table 2.3. Note the similarity in the factors causing poor wound healing and fistula persistence.
Table 2.3
Factors perpetuating or sustaining a fistula or sinus
Local factors |
Distal obstruction |
High drainage volume |
Relative ease of fistula/sinus drainage, compared to normal pathway |
Infection (local or systemic) |
Hematoma formation |
Wound separation, dehiscence |
Foreign body (sutures, mesh, extraneous material) |
Ischemia (vascular insufficiency, hypoxia) |
General factors |
Diabetes |
Immunosuppression |
Renal failure |
Extremes of age (very young or very old) |
Liver failure, jaundice |
Malnutrition |
Steroids |
Carcinoma (systemic effects or local invasion) |
Radiotherapy |
Chemotherapy |
Anesthesia-Related Complications
Postoperative complications, by definition, are those that are evident after the operation; however, as may be obvious, many of these complications have their origin either intraoperatively or even preoperatively. For example, DVT or pulmonary embolism is commonly recognized as being associated with the period of immobility, during which venous stasis occurs. This may have occurred pre- or intraoperatively and been contributed to by postoperative immobility. Frequently, the exact timing or relative contribution is unclear, and in any case this may not be clinically relevant. Although, it may be vital, because institution of any prophylactic or therapeutic interventions that may be required to reduce any specific risk will often depend on where and when the initiating factors are operational, in order to adequately understand the timing of the origin of the complication (if this is possible).
General anesthesia is therefore often considered as a potential contributor to many “general complications” – for example, DVT, respiratory infection, cardiac failure, arrhythmias, postoperative pain, and pressure injuries. The distinction as to how much is related to the “anesthetic” and how much is related to the “surgery” or postoperative care may be relevant to “root cause analysis” of adverse events, but the exact causation is often unable to be separately defined, with any real science. Instead, an analysis investigating multiple potential contributing factors is often applied and more helpful in a practical sense. If all of the possible risk factors are reduced to as low as possible, then the overall risk should usually also be reduced significantly. System factors might also be important here, for example, if there is an effective system in place to ensure that the correct prophylactic antibiotic is given at the correct time in relation to surgery, then the risk of missing correct antibiotic dosing and the risk of postoperative wound infection should be reduced. Similarly, if hypothermia is reduced intraoperatively, then the risk of wound infection may be reduced also. Incorporation of these relatively simple measures as “standard practices” has the capacity to reduce error of omission and to reduce poor clinical outcomes.
“Incorporation of the relatively simple measures, such as prophylactic anticoagulants, antibiotics, and patient warming, as ‘standard practices’ has the capacity to reduce error of omission and to reduce poor clinical outcomes.”
Complications of general anesthesia are discussed in more detail in Chap. 5.
Complications related to general anesthesia, in brief, include:
Thermal
Respiratory
Cardiac
Hematological/anemia
Neurological
Fluid and electrolyte imbalance
Allergic
Pressure effects
Gastroenterological
Iatrogenic or Hospital-Acquired Injury
Considerations of “iatrogenic” or “doctor-induced” injuries are difficult topics to quantify or encapsulate sufficiently in a meaningful way. Many of these are related closely to healthcare system problems that are accompanied by human factors leading to errors, omissions, or adverse events. Many of the considerations relating to these are detailed in Chap. 8. Iatrogenic complications are often multifactorial and frequently relate to environmental factors within the heath system, patient factors, and nursing staff factors. The attribution of “blame” for iatrogenic injury is often unhelpful and more damaging than constructive, since many factors leading to adverse events can be effectively “designed out” and avoided, which is a far smarter, more cost-effective, and positive approach. It is disturbing that a patient might leave a health system in a worse state than they entered a health system. Indeed, the notion of a “hospital-acquired” infection is likewise disturbing.
Examples of complications where there may be an iatrogenic component are included in Table 2.4.
Table 2.4
Examples of complications where an iatrogenic component may exist
Trauma |
Direct contusion/laceration |
Joint strain/fracture |
Falls |
Pressure effects |
Positional |
Constriction |
Ulceration |
Thermal |
Hypothermia |
Hyperthermia |
Burns |
Electric shock |
Respiratory |
Hypoxia |
Atelectasis |
Respiratory depression |
Aspiration pneumonitis |
Pneumonia |
Pneumothorax |
Hemothorax |
Cardiac |
Cardiac failure |
Arrhythmias |
Hypotension |
Intravenous access |
Site infections |
Dislodgement |
Catheter breakages/embolization |
Thrombosis |
Stenosis |
Venous obliteration |
Drug |
Dosage errors |
Drug-type errors |
Combination errors |
Communication errors |
Psychological |
Poor communication |
Patient demoralization |
Failed explanation |
Surgeon or staff error |
Dosage errors |
Wrong site or side |
Wrong patient |
Wrong operation |
Retained body (e.g., instrument or swab) |
Intraoperative injury (e.g., trocar injury to bowel or vessel) |
“The attribution of “blame” for iatrogenic injury is often unhelpful and more damaging than constructive, since many factors leading to adverse events can be effectively “designed out” and avoided, which is a far smarter, more cost-effective, and positive approach.”
Hematological Complications
A homeostatic balance usually exists between thrombosis (coagulation) and thrombolysis (anticoagulation) mechanisms within the body. This balance is delicate and determines the appropriate processes of clot formation to stop bleeding following traumatic transection of a vessel or the lack of clotting to ensure adequate blood flow for perfusion of tissues to supply nutrition and removal of waste products, for example, in wound healing. When the balance is disturbed, either failure to control bleeding or intravascular clotting can occur, sometimes inappropriately.
A series of concurrent events occurs in the process of coagulation and clot formation that are listed in Table 2.5. These include:
Table 2.5
Sequence of thrombosis and hemostasis
Tissue trauma |
Vascular spasm |
Ligature application/diathermy |
Platelet plug |
Clotting factor cascade |
Fibrin cross-linking/stabilization |
Hemostasis |
Re-endothelialization |
Revascularization |
Vasospasm: Laceration of a vessel causing incomplete transection often results in more bleeding than complete division of the vessel. This is because with complete transection, concentric vasospasm from smooth muscle in the vessel wall typically causes significant narrowing or closure of the small vessel ends allowing platelet plug formation. In larger vessels over about 4–5 mm in size, vasospasm may be unable to narrow the lumen sufficiently to permit adequate clot formation to stop bleeding completely. For larger vessels >5 mm diameter, a ligature or diathermy is usually required to stop bleeding. Cut vessels may also retract, which can make locating the vessel difficult in the surrounding fat and soft tissues. The use of adrenaline (epinephrine) usually within the local anesthetic solution, injected into the site of the wound edges, can induce vasospasm to assist the process.
Platelet Adhesion and Plug Formation: In brief, platelet adhesion is activated by chemokines and cytokines released by endothelial cells during vessel injury, to form a platelet plug.
Coagulation Cascade and Fibrin Formation: The platelet plug activates a cascade of coagulation (clotting) factors in the blood via a variety of mechanisms, and fibrin is formed to cross-link and stabilize the platelet plug, forming a more permanent seal over the bleeding point(s). Defects in the coagulation pathway may lead to failure of the clotting process and continued bleeding.
Re–endothelialization: Side holes in the vessel wall(s) are repaired by stabilized clot and then incorporation into the vascular wall and followed by clot dissolution, retraction, and resolution, with eventual re-endothelialization and reconstitution of the vessel lumen(s).
Revascularization: Reestablishment of flow in the damaged vessel then occurs, due to recanalization, to reperfuse distal tissues. Formation of new vessels (angiogenesis) commonly occurs through scar tissue or to replace an obstructed vessel, sometimes with opening and expansion of collateral vessels around the obstruction.
These processes commence at the time of injury and continue for weeks to months after surgery or trauma, with rapid revascularization across the wound with capillaries and lymphatics within several days. Scar maturation takes 6 months to several years to complete. However, the use of tape over the scar can be useful in reducing scarring and improving the final cosmetic appearance, presumably by reducing skin tension and placing local pressure onto the wound (Fig. 2.2 and Tables 2.5 and 2.6).
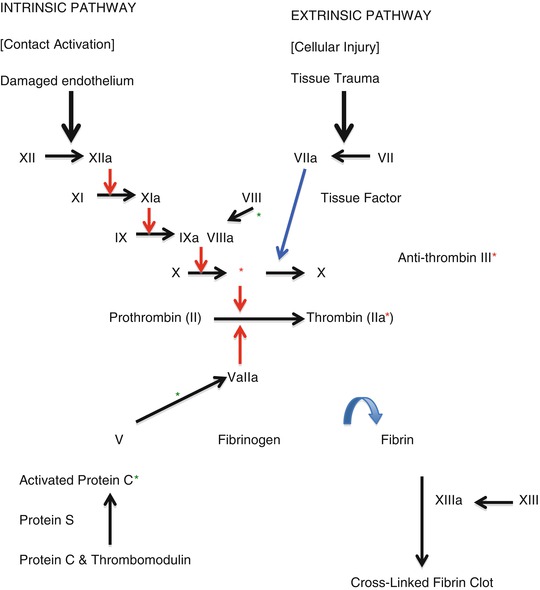

Fig. 2.2
Coagulation factors and the coagulation cascade pathway. Note: The actions of protein C and antithrombin III occur at the sites marked by the respective colored asterisks
Table 2.6
Common causes of bleeding
Release of vascular spasm |
Slipped or loose ligature |
Diathermy seal leakage |
Antiplatelet agents |
Coagulation factor deficiencies |
Oral anticoagulants |
Bleeding tendency |
Blood pressure rises (sneezing, vomiting, coughing) |
Repetitive trauma |
Thrombosis and Thromboembolism
Deep Venous Thrombosis (DVT)
Thrombosis within the deep veins can occur at any site in the body although it is most common within the deep calf veins of the lower limb, partly due to their structure and the effects of gravity. Thrombosis in the upper limb, neck, subclavian veins, or vena cava most frequently occurs when an intravascular device is inserted (e.g., a subclavian or internal jugular venous access line) resulting in venous obstruction or following infusion of fluids irritant to the vein walls. In these situations, the risk of thrombosis is more common with the increased duration of placement of the device or infusion.
Deep venous thrombosis (DVT) usually refers to thrombosis of the deep calf, thigh, and iliac veins, and these can extend to the inferior vena cava. In general, the incidence of DVT is of the calf > thigh > iliac; however, the risk of pulmonary thromboembolism (PTE), and hence the relative danger of serious sequelae including death, is the reverse with iliac > thigh > calf. However, calf DVT may extend to the thigh, and then to iliac veins, forming a large clot throughout part or all of the lower limb venous system. The risk of PTE increases as the DVT extends more proximally into larger vessels. Pelvic surgery, in particular hip and gynecological surgery, is associated with an increased risk of iliac DVT and PTE. DVT restricted to the calf of itself carries a low risk of PTE; however, risk of extension to involve the thigh vessels is the main indication for treatment.
DVT has an annual incidence of about 160 per 100,000 population, and many of these are below the knee; however, when the thigh veins are involved, risk of PTE increases. One of the main sequelae of DVT is chronic venous congestion or insufficiency. This arises from damage to the valves within the veins and development of superficial varicose veins, chronic venous congestion, and chronic skin changes, including recurrent ulceration chronic edema, poor healing after injury, chronic pain, and recurrent cellulitis. These sequelae are often very significant economically and socially, causing readmission to hospital with thousands of bed-days. The cost of DVT is appreciable for most western countries and also for many less developed regions. In Australia, for example, it is estimated to cost over A$200 M annually or about A$10 M (US$12 M) per person. Effective prophylaxis is one method of reducing this enormous cost burden.
Factors associated with increased risk of DVT are listed in Tables 2.7 and 2.8. These are not mutually exclusive or independent factors, and they frequently coexist in a particular patient situation (e.g., trauma – bed rest, clotting tendency, vessel injury).
Table 2.7
Broad risk factors for clotting (Virchow’s triad)
Stasis |
Hypercoagulability (obesity, cancer, GA, trauma) |
Vessel trauma |
Table 2.8
Specific risk factors for DVTa
Age > 40 years (annual incidence of VTE rises with each decade >40) |
Obesity |
Smoking |
Pregnancy/postpartum |
Cancer (active or occult malignancy) |
GA or spinal/epidural anesthesia |
Laparoscopic surgery |
Thrombophilia (hypercoagulability; including inherited deficiency of antithrombin, protein C or protein S, homozygosity, or double heterozygosity for factor V Leiden) |
Estrogen therapy (also OCP*) |
Major trauma |
Fracture |
Pelvic, knee, or hip surgery |
Plaster or other cast or boot |
Immobility and ICU care |
Previous prolonged travel or bed rest |
Previous DVT or PE |
Family history of DVT/PE |
Varicose veins |
Risks related to an acute medical illness: |
Acute or acute on chronic, chest infection, heart failure, myocardial infarction, stroke with immobility, some forms of cancer chemotherapy, and acute inflammatory bowel disease |
Risks related to an injury or surgical procedure: |
All surgical procedures, especially abdominal, pelvic, thoracic, or orthopedic |
The incidence of DVT (symptomatic and subclinical) without prophylaxis is presented in Table 2.9.
Table 2.9
Incidence of DVT (symptomatic and subclinical) without prophylaxisa
Stroke | 56 % |
Elective hip replacement | 51 % |
Multiple trauma | 50 % |
Total knee replacement | 47 % |
Hip fracture | 45 % |
Spinal cord injury | 35 % |
General surgery | 25 % |
Myocardial infarction | 22 % |
Neurosurgery | 22 % |
Gynecological surgery | 14–22 % |
General medical patients | 17 % |
Pulmonary Thromboembolism
When clot from a DVT breaks off and embolizes via the larger veins and pulmonary circulation, it is termed pulmonary thromboembolism (PE or PTE). The overall incidence of PE is about 70 per 100,000 population, with about 20 per 100,000 being symptomatic nonfatal PE and some 50 per 100,000 being fatal premortem PE detected at autopsy. The number of nonfatal, asymptomatic PE is difficult to determine with certainty, but these subclinical PE do occur and may serve as the forerunner for later, larger symptomatic PE, even with separate subsequent admissions to hospital. Previous episodes of DVT and PE serve as an indicator of a patient at increased risk of subsequent DVT and PE. Both DVT and PTE may be difficult to diagnose and occasionally may go undetected, perhaps being treated as cellulitis, cardiac ankle edema, or chest infection, respectively.
Superficial Thrombophlebitis
This is due to thrombosis within superficial veins, which causes obstruction of the affected vein(s) and associated inflammation. Although this problem is not life-threatening, it can be a significant symptomatic issue for the affected patient. It often presents as painful, hot, red, discolored, and indurated areas typically over the lower limb. A tender cord-like mass may be palpable along the affected vein. Superficial thrombophlebitis usually occurs in patients with preexisting varicose veins who are immobilized for a period of time. It is rarely associated with concurrent deep venous thrombosis or risk of pulmonary embolism. It can be a recurrent, painful, and debilitating problem. It may contribute to superficial chronic venous changes of the skin.
The incidence of thrombotic and thromboembolic events is presented in Table 2.10.
Table 2.10
Incidence of thrombotic and thromboembolic eventsa
Event | Approximate incidence |
|---|---|
DVT | 160 per 100,000 population |
PE | 70 per 100,000 population |
Symptomatic nonfatal PE | 20 per 100,000 population |
Fatal premortem PE | 50 per 100,000 population |
Coagulopathies
Coagulation is reversed intrinsically by thrombolytic mechanisms in the body, including circulating antithrombin III. The concentration and function of platelets and clotting factors are integral to adequate clotting occurring. When platelets are either low in number (e.g., thrombocytopenic purpura or excessive clotting) or dysfunctional (e.g., after aspirin or nonsteroidal anti-inflammatory agent therapy), failure of formation of the platelet plug can lead to excessive oozing, bleeding, and bruising. Similarly, in situations where coagulation factors are reduced in amount or function, failure of the coagulation cascade can lead to excessive bleeding and oozing. Two conditions where aberrant clotting and excessive bleeding tendency can be experienced are outlined below:
Disseminated Intravascular Coagulation (DIC)
Widespread clotting can occur within smaller vessels of the circulation associated with severe sepsis, major trauma, malignancy, and some serious obstetric complications (e.g., placental abruption, placenta previa, intrauterine death, amniotic fluid embolism). It is a relatively poorly understood entity often with an unpredictable appearance and course. It may also be associated with prolonged ICU care, nutritional disturbances, excessive blood loss, and transfusion and in those situations where etiological factors are usually difficult to separate. Hematological testing may be normal or near normal, despite profound uncontrollable bleeding. However, a prolonged INR (international normalized ratio) and APTT (activated partial thromboplastin time) with reduced fibrinogen and platelet count are almost diagnostic for DIC, but not pathognomonic. Measurement of the fibrinogen degradation products (FDP) may also be helpful, as these may be elevated. Correction with platelet transfusions, cryoprecipitate, or fresh frozen plasma may be used to correct the problem, but correction of the underlying pathology is almost always essential for reversal of DIC. Mortality is high in patients who develop DIC that does not respond with correction of hematological factor administration. DIC may be averted in situations of massive bleeding by early correction of blood loss, with early intervention with platelets and clotting factor replacement when a coagulopathy is suspected. An easy test is to place the patient’s blood in a plain test tube (with no additives) and wait to see if it clots over a few minutes. If it remains liquid and fails to clot after 5 min, a coagulopathy should be strongly considered.
Consumption and Transfusion Coagulopathy
This can occur after multiple rapid transfusions of blood usually in a patient with extensive blood loss after acute major trauma. In an attempt to stop the bleeding, coagulation factors are deposited at the site of bleeding or lost from the body as blood clots extracorporeally; clotting factors are therefore “consumed” at a rate greater than production. Following massive transfusion, foreign blood products can sometimes cause activation and deposition of clotting factors intravascularly, and “consumption” similarly occurs. Additionally, with each unit of blood that is transfused, a small amount of acid-citrate buffer anticoagulant is given in the preserved blood. With massive transfusion of blood, anticoagulant levels may become significant and cause bleeding. In these settings, FFP or cryoprecipitate should be given after each 6 units of packed red blood cells are given to avoid the consumption and anticoagulant effects of large blood transfusions.
Liver Failure and Vitamin K Deficiency
The liver produces the vitamin K-dependant clotting factors II, VII, IX, and X, so that either liver dysfunction or vitamin K deficiency can reduce production of these factors. The INR is a good measure of the activity of these liver-derived clotting factors. Warfarin and other coumarins are competitive inhibitors of vitamin K production in the liver. Liver disease reduces factors V and VIII as well, and defective fibrinogen is often produced. Some reversal is common with administration of vitamin K parenterally, even in the presence of moderately severe liver failure.
Platelet Problems
Bleeding may arise due to low numbers of platelets or platelet dysfunction.
Platelet Numbers
Platelets <100,000/mL are associated with an increased risk of bleeding, and platelet transfusion of 4–6 units of fresh platelets is usually required to increase platelet numbers and reduce this risk. Heparin-induced thrombocytopenia (HIT) is not uncommonly associated with heparin therapy, especially unfractionated heparin. Platelets are consumed relative rapidly so transfusion is usually preferable on the operating table, just prior to surgical incision.
Platelet Function
Platelets may be normal in number, but unable to aggregate, typically due to ingestion of antiplatelet agents, such as aspirin (which is irreversible) or nonsteroidal anti-inflammatory agents (e.g., piroxicam, diclofenac, ibuprofen, indomethacin), which are reversible within a few days of cessation and clearance.
Blood Product Transfusion
Transfusion of whole blood or separated blood components is often used where the hemoglobin has fallen below 7 g/L or rapid blood loss has occurred and urgent replacement is needed or where specific replacement of blood components is required.
Blood products include:
Whole blood (RBC, platelets, and plasma proteins)
Packed red cells (RBC)
Platelets
Fresh frozen plasma
Cryoprecipitate (clotting factors)
Specific clotting factors
Albumin
Plasma protein solution
Antibodies
The risks of allogeneic transfusion are detailed with vascular surgery in Volume 6.
Hematological problems often further complicate multisystem organ failure (MSOF) arising from either drug therapy (e.g., heparin-induced thrombocytopenia; HIT), blood product transfusion reactions, disseminated intravascular coagulation (DIC), or clotting factor abnormalities (e.g., due to liver failure). Intractable coagulopathic bleeding is a common sequel of severe MSOF and is often difficult to reverse and a major contributing factor to death. DVT and/or pulmonary embolism are other complications of prolonged immobility and coagulopathies.
Perioperative Thromboprophylaxis
These are principally directed against development of deep venous thrombosis and pulmonary thromboembolism (DVT and PTE).
Measures to reduce risk of deep venous thrombosis (DVT) fall into three broad groups:
Basic measures: Good in hydration, short immobilization, and early mobilization after surgery
Mechanical prophylactic methods
Pharmacological prophylactic agents
The effectiveness can vary dependent on the clinical procedure and health system or specific patient-related risk factors.
Basic Measures
Periods of immobilization of the lower limbs can lead to stasis of blood and loss of function of the calf muscle contractile pump for good circulatory venous return. Confinement to bed; direct trauma; preoperative immobility from pain, plasters casts, traction, external fixation, prolonged travel before or after surgery, and ICU periods; or prolonged surgery and general anesthesia can cause this. Good hydration (prevention of dehydration) lowers the viscosity of blood. Mobility in the perioperative period (where desirable), short bed confinement, and good pain relief to permit early mobilization are all good practice measures, but are difficult to quantify or test.
Mechanical Methods
The mechanical options include:
Knee or thigh length graduated compression stockings (GCS)
Knee or thigh length intermittent pneumatic compression (IPC) devices
Venous foot pumps, e.g., foot impulse technology (FIT) and foot impulse devices (FID)
These reduce venous stasis by reducing venous pooling in the periphery through external compression using graduated compression stockings, intermittent pneumatic compression, or venous foot pumps, used alone or in combination, during surgery and frequently after and/or prior to surgery.
Pharmacological Agents
The main consideration at the outset is to evaluate the relative risk of DVT development and to balance this risk of clotting, against the relative risk of inducing problematic perioperative bleeding. Antithrombotic agents reduce the coagulation tendency and susceptibility to clot formation and subsequent extension. The effectiveness of aspirin appears better for prevention of arterial thrombosis, while venous thromboprophylaxis appears better with agents that affect the coagulation cascade, such as the heparin-like and warfarin-type agents. In many studies, low molecular weight heparin (LMWH; fractionated heparin), unfractionated heparin (heparin), fondaparinux, danaparoid, rivaroxaban, dabigatran etexilate, aspirin, and warfarin have been treated as separate classes of agents, as they have a variety of separate actions. (See Australian Medicines Handbook 2009, or equivalent.)
The pharmacological options currently include:
Subcutaneous unfractionated (UFH) or low molecular weight (LMWH) heparins
Subcutaneous selective inhibitor of activated factor X (Xa), fondaparinux
Subcutaneous heparinoid, danaparoid
Oral direct factor Xa inhibitor, rivaroxaban
Oral direct thrombin inhibitor, dabigatran etexilate
Oral aspirin
Oral vitamin K antagonist, warfarin
However, newer agents are being investigated.
Diagnosis of VTE (DVT and PE)
VTE is a spectrum of disease, from asymptomatic distal DVT to fatal PE, and most DVT (and PE) are initially asymptomatic. The clinical relevance of asymptomatic distal DVT as an indicator of the efficacy of VTE prophylaxis is debated. Confirmation of the clinical diagnosis of DVT is usually by compression duplex ultrasonography, to show venous anatomy and flow patterns. The “gold standard” diagnostic tool used in most historical clinical trials has been ascending venography, considered to be of greater sensitivity than compression ultrasound for distal (below-knee) DVT. Venography is an invasive technique, injecting radiographic contrast agents into the foot vein(s) and viewing the venous drainage patterns using serial radiographs. This has essentially been replaced by compression duplex ultrasonography in clinical practice in most western countries, as the ultrasound technology has advanced in reliability and become readily available. Radionucleotide methods of detection of DVT have been used in many trials but are often considered oversensitive and are overall less reliable and more invasive, with radiation risks. There is also some risk of inducing DVT from venography.
PE is usually diagnosed or excluded by computed tomographic (CT) pulmonary angiography (helical CT), ventilation-perfusion isotope radionucleotide scan, or occasionally magnetic resonance imaging (MRI). PE incidence in randomized controlled trials is probably underestimated because diagnosis was on clinical suspicion, followed by investigation, rather than routine scanning.
The Evidence: Issues to Consider
Between clinical trials, the patient groups, randomization methods, diagnosis, and therapies vary considerably and make comparison and interpretation potentially problematic and imprecise. The time periods over which these studies were performed also vary, with different patient risk profiles, hospitalization periods, mobilization schedules, perioperative management, anesthetic, and surgical types applying during these studies. How these results then apply to contemporary practice remains uncertain. Nevertheless, VTE, DVT, and PE remain as serious complications of hospitalization. Many trials have used preoperative thromboprophylaxis, but current practice in many institutions is to initiate the dosing just after surgery or on the next day. Therefore, significant lack of available evidence in important areas necessarily limits the scope of any evidence-based interpretation or recommendations. However, the relative risks from VTE (death, pulmonary dysfunction, pain) and thromboprophylaxis (bleeding, platelet dysfunction) need to be thoughtfully balanced for each patient. Despite this, the absolute risk of VTE is still not removed entirely even with diligent thromboprophylaxis. In addition, clinicians and patients may perceive the risk and consequences of bleeding significantly differently, with surgeons being very concerned about intraoperative bleeding and operative difficulty, while patients are often primarily concerned about their total operative course. An extremely good evidence-based resource is the National Health and Medical Research Council of Australia (2009) Clinical Practice Guideline document addressing Prevention of Venous Thromboembolism in Patients Admitted to Australian Hospitals.
Risk of Bleeding or Hematoma Formation
Bleeding is a side effect of trauma and surgery from vessel laceration and injury. Hematoma and bruising are consequences of bleeding into tissues or spaces. The risk of bleeding is multifactorial and is increased where particularly vascular regions or organs are operated on, with extensive surgery and where coagulation is impaired. Because pharmacological thromboprophylaxis is a means of altering coagulation to reduce risk of clot formation, especially DVT, there is often an increased risk of operative or postoperative bleeding. This risk, of course, needs to be balanced and interpreted against the risk of VTE and may be contraindicated in certain situations.
Relative or absolute contraindications to pharmacological VTE prophylaxis are as follows1:
Regional central anesthesia or lumbar puncture (planned or recent)
Recent central nervous (intracranial or spinal) system bleeding or lesion
Intracranial or spinal lesion at high risk for bleeding
Current active major bleeding, >2 units of blood or blood products
Thrombocytopenia (platelets <50,000/μL) or severe platelet dysfunction
Coagulopathy or coagulation factor abnormalities
Recent major surgical procedure/trauma at high risk for bleeding
Current anticoagulation
Surgery with high risk of bleeding
Renal impairment
High risk of falls or trauma
General Surgery
General surgery includes a range of procedures from short-duration, superficial procedures perhaps under local anesthesia to longer-duration major surgical procedures requiring long general or regional anesthetic immobilization times, with a range of ages and comorbidities and sometimes with ICU support or limited postoperative mobility. Given the variability of factors for risk of VTE, the results of RCTs need to be balanced for the individual patient. For younger patients with no adverse risk factors and for short procedures under local anesthesia, little or no thromboprophylaxis may be necessary.
For the patient with an assessed risk of DVT, then thromboprophylaxis is wise, and it is in this group where the evidence for use of these measures is progressively strongest.
NHMRC data (2009) showed that low molecular weight heparin or unfractionated heparin were both equally effective in reducing the occurrence of DVT compared with no treatment. Bleeding rates were significantly greater for LMWH, than for unfractionated heparin, and unfractionated heparin had the same bleeding rate as no treatment. Most trials used LMWH or UFH for thromboprophylaxis for a duration of up to 1 week following surgery.
Recommendations2
Thromboprophylaxis is recommended for all patients undergoing major general surgery, until the patient is fully mobile postoperatively (in patients without contraindications), using either:
Low molecular weight heparin
Unfractionated heparin
Graduated compression stockings can significantly reduce the occurrence of DVT in RCTs either as the sole prophylactic agent or in addition to heparin and should be used until the patient is fully mobile.
Foot pump effectiveness in preventing VTE following general surgery (bilaterally) significantly reduced DVT compared with no thromboprophylaxis in one small RCT.
Intermittent pneumatic compression was compared with unfractionated heparin in one study with inconclusive results.
Abdominal Surgery
Within the broader category of general surgery, the subgroup of major abdominal surgery, especially pelvic surgery, increases the risk of VTE, and therefore thromboprophylaxis is recommended for all patients following major abdominal surgery. Evidence from 23 RCTs showed low molecular weight heparin significantly reduced DVT (including symptomatic DVT) compared with unfractionated heparin, but with no difference in adverse events including major hemorrhage or PE. Therefore, low molecular weight heparin is recommended for thromboprophylaxis following abdominal surgery for between 5 and 9 days. Fondaparinux significantly increased major bleeding compared with no treatment, but was an equally effective VTE prophylaxis to low molecular weight heparin. The majority of trials administered either LMWH or UFH preoperatively. If planned to be given before operation, then this should be discussed with the anesthetist.
Recommendations3
Pharmacological low molecular weight heparin thromboprophylaxis should be used for abdominal surgery operations and continued for 1 week (5–9 days) or until the patient is fully mobile postoperatively (in patients without contraindications).
Options are either:
Low molecular weight heparin.
Unfractionated heparin might be considered, although the rate of DVT is higher than LMWH, despite the rate of bleeding or PE being the same.
Graduated compression stockings can significantly reduce the occurrence of DVT in RCTs either as the sole prophylactic agent or in addition to LMWH or UFH and should be until the patient is fully mobile.
Intermittent pneumatic compression was compared with unfractionated heparin in one study with inconclusive results.
Cardiac, Thoracic, and Vascular Surgery
Cardiac, thoracic, or vascular surgery increases the risk of VTE, and four RCTs showed that low molecular weight heparin or unfractionated heparin was equally effective for VTE prophylaxis for this surgery. Adverse events were similar, except for one small trial which showed more wound hematomas in the UFH group. Intermittent pneumatic compression (thigh length) with heparin reduced symptomatic PE, but IPC did not provide any further thromboprophylactic benefit for patients wearing graduated compression stockings and receiving 325 mg/day of aspirin after coronary artery bypass surgery.
Thromboprophylaxis is recommended following cardiac, thoracic, or vascular surgery.
Recommendations4
Pharmacological thromboprophylaxis should be used for cardiac, thoracic, and vascular surgery operations and continued for 1 week or until the patient is fully mobile postoperatively (in patients without contraindications).
Options are either:
Low molecular weight heparin
Unfractionated heparin
Physical thromboprophylaxis should be used after cardiac, thoracic, and vascular surgery +/− pharmacological prophylaxis, until the patient is fully mobile.
Options are either:
Graduated compression stockings
Intermittent pneumatic compression
Anesthesia Type and DVT or PTE Risk
For patients receiving central regional anesthesia (whether regional anesthesia: epidural or spinal) alone compared with general anesthesia alone:
DVT rates were significantly lower in the regional anesthesia group.
Evidence based on one systematic review and four RCTs.
PE rates were significantly lower in the regional anesthesia group.
Evidence based on seven RCTs.
No significant difference in major bleeding rates.
For patients receiving central regional plus general anesthesia compared with general anesthesia alone:
DVT rates showed no significant difference.
Based on two RCTs.
Significantly lower blood loss in patients receiving regional plus general anesthesia compared with GA alone in one RCT.
Recommendation5
Epidural or spinal anesthesia (central neural blockade) should be considered if feasible, because of lower risks of DVT and PE overall compared with general anesthesia. Risk of an epidural hematoma is present with central neural blockade, if used. Thromboprophylaxis needs to be planned and timed to minimize this risk.
Pulmonary Complications
Among the most common postoperative complications following general anesthesia, or even heavy sedation, are continued respiratory depression, airway obstruction, respiratory airway collapse, and pulmonary infections. Under–breathing due to painful respiration or prolonged sedation frequently leads to reduced airway expansion, alveolar collapse, and ventilation-perfusion inequalities. This produces ineffective clearance of airway secretions, stasis of material within airways, especially behind collapsed smaller airways, with consequent consolidation and bacterial growth. “Stasis is the Basis” for most infections. Infection within the airways results in a spectrum of disease from bronchopneumonia (where predominantly bronchial and peribronchial tissues become inflamed) to lobar pneumonia (where larger areas of lung become involved with inflammation and a consolidation process ensues). The processes may occur together and can be localized to a lung region or may be “patchy” across one or both lung fields.
Encouraging deep breathing and coughing at an early stage postoperatively is vital to reverse this process, can assist in expanding airways adequately, and can permit proper airway clearance, which may avert atelectasis from continuing and pneumonia from developing. If stasis and airway obstruction continues, three principal problems typically can develop, sometimes very rapidly over a 12- to 24-h period:
Atelectasis – often involving basal lung regions
Consolidation of lung tissue and pneumonia (broncho or lobar pneumonia)
Hypoxemia, due to shunting of blood, causing ventilation-perfusion inequalities
These are critical problems in patients recovering from surgery and for patients with other comorbidities, especially smoking, intercurrent preoperative chest infection, chronic lung diseases, chest trauma, or asthma.
“Stasis is the Basis” for most infection.
Basal Atelectasis
This is a process where small airways close down principally due to under-breathing with loss of volume of alveolae and eventual collapse of alveolar units. This typically occurs at the lung bases and can often be reversed with good analgesia to permit more effective deep breathing and coughing. Lung expansion opens the airways, expands the alveoli, and restores perfusion to the re-ventilated alveolar units. A common early clinical sign of atelectasis is a tachycardia, usually without a fever, occurring in the early postoperative period. Good pain relief is essential for good lung expansion to avoid under-breathing, permit adequate deep breathing and coughing, and to encourage proper expansion of alveolar lung units to avoid atelectasis.
“Good pain relief is essential for good lung expansion to avoid under-breathing and permit adequate deep breathing and coughing.”
Pneumonia
Once atelectasis is established, if material is then aspirated into the airways or if primary upper airways infection occurs, then established lung infection and inflammation can prevail. The patient usually develops a fever as an early clinical sign.
Where alveolar units are not being ventilated, oxygenation and clearance of carbon dioxide are reduced, so that perfusion of these units is inefficient and does not assist respiration. In this situation, a state of relative ventilation-perfusion (V:Q) inequality exists. In order to compensate for this inequality, blood is reflexly redirected away from non-ventilated alveolar units by selective vasoconstriction in the pulmonary vascular bed. This encourages more blood to reach those alveolar units and regions of the lung that are better ventilated than those that are poorly ventilated. This process more effectively matches ventilation with perfusion, enabling more effective oxygenation and CO2 removal.
Where pneumonia is more widespread, involving a pulmonary lobe or lobes (or indeed a whole lung), the extent of V:Q inequality may be so severe as to produce profound hypoxemia. Because CO2 diffuses much faster than O2 and is breathed-off more rapidly than the O2 can be absorbed, CO2 does not usually accumulate in the blood as fast as O2 levels decrease. Thus, hypoxemia typically occurs before hypercapnia develops.
The risk of atelectasis and pneumonia is associated with a variety of risk factors, which are listed in Table 2.11.
Table 2.11
Risk factors associated with atelectasis and pneumonia
Prolonged general anesthesia and surgery |
Smoking |
Obesity |
Chest injury, including rib fractures |
Thoracotomy |
Abdominal surgery |
Pain |
Preexisting chest infection |
Chronic lung diseases |
A number of risk reduction strategies are useful in patients at higher risk of lung infections, where possible: prior to surgery, during surgery, and after surgery (Table 2.12).
Table 2.12
Strategies associated with reduction in the risk of respiratory infection
Preoperative measures |
Smoking cessation |
Inspired humidified air/bronchodilator |
Optimization of the respiratory status |
Weight reduction |
Pain relief |
Intraoperative measures |
Regional instead of general anesthesia |
Minimizing the length of the surgical procedure |
Local anesthesia/block to the wound |
Postoperative measures |
Good pain control |
Adequate reversal of sedation and respiratory depression |
Deep breathing and coughing |
Breathing humidified inspired air |
Avoiding smoking |
These measures represent key factors in reducing the risk of postoperative chest infection.
Lung Collapse
When airways become occluded either by foreign bodies or secretions, absorption of gases distal to the obstructing point can lead to collapse of alveoli and small airways. The affected section of lung can collapse partially or entirely. This may occur independently or as part of a pneumonic process. Clearance of the airway obstruction is paramount to expansion of the collapsed lung, either by surgical bronchoscopic removal of the foreign body or secretions and/or from antibiotic therapy and chest physiotherapy. The presence of a pleural effusion can be severely detrimental by reducing lung expansion, facilitating lung collapse, and creating poor mucus clearance and sometimes considerable discomfort. Drainage of a large pleural effusion is often essential for proper lung expansion, oxygenation, and avoidance or resolution of pneumonic consolidation.
Pneumothorax
Pneumothorax may arise from pulmonary barotrauma due to ventilation, direct surgical injury, chest tube trauma, or spontaneously, the latter often from apical bullous disease.
Pneumothorax is recognized as loss of chest expansion and air entry on the affected side and may be signified by chest pain and breathlessness in the alert patient, by or poor gas exchange.
Two main types occur:
Tension pneumothorax
Non-tension pneumothorax
Tension pneumothorax is due to progressive buildup of pressure from air leakage into the pleural space without release, as a “flap-valve” effect. The pressure typically displaces the mediastinum towards the opposite side, compressing the heart, great vessels in the chest, and opposite lung, leading to difficulty with respiration or ventilation, worsening gas exchange, and compromised venous return. This is a surgical emergency and needs to be corrected urgently by releasing the pressure in the chest by inserting a large-bore needle through the chest wall on the side of the pneumothorax.
Non–tension pneumothorax is where leakage of gas has occurred around the lung into the pleural space leading to collapse of the lung away from the chest wall; the pressure does not continue to build within the chest and so does not displace mediastinal structures nor cause cardiovascular compromise. Occasionally, a non-tension pneumothorax can evolve into a tension pneumothorax, especially when positive pressure ventilation is used after a (non-tension) pneumothorax has developed.
Surgical Emphysema
When gas leaks from the lung, pleural space, or airways into the tissues, the tissues can swell and develop crepitus (crackling) to palpation. In these cases, gas may often be seen tracking along between tissue planes on plain x-ray or CT scans. Occasionally this phenomenon can occur after laparotomy, but is not uncommon after laparoscopy or thoracoscopy, where CO2 is insufflated. It is usually benign and resolves spontaneously over a few days. Surgical emphysema is an important sign to note and may be a serious concern for the patient if it is palpable, audible, extensive, or associated with a fever. Rarely, it can signify gas gangrene, superficial infections, rupture of the esophagus, an anastomotic leak, or result from peritoneal gas following viscus perforation; in these situations early diagnosis and treatment is paramount.
Aspiration Pneumonitis
The lower airways and lung are usually virtually sterile containing few, if any, bacteria. When foreign material reaches the larynx or enters the airways, in an alert patient it typically causes extreme irritation and activation of the coughing reflex. Aspiration of food, other material, or regurgitated gastric contents can occur in the awake patient with an active gag reflex, but the airways are usually protected by reflex coughing and laryngeal spasm. In the sedated or unconscious patient, or in those with neurological problems, the risk of aspiration of material into the airways is much higher, due principally to depression of the protective reflex mechanisms.
Aspiration pneumonitis is the term usually given to inhalation of regurgitated gastric contents into the airways. Gastric contents are extremely irritant, and inflammation occurs rapidly, leading to bronchospasm with obstruction of smaller affected airways. An initial chemical pneumonitis occurs and may subsequently proceed to an infective pneumonia. Typical early chest x-ray appearances can be unremarkable; with progression, a focal right middle or lower lobe pneumonic picture may be seen or a patchy multifocal appearance evident. The left lung may be affected if the patient is lying on their left side.
Risk factors for aspiration pneumonitis:
Pregnancy
Non-fasted state
Gastroesophageal reflux
Non-alert patient
Neurologically impaired patient
Obesity
Raised abdominal pressure (e.g., laparoscopy)
Neck Swelling
Edema of the neck tissues for any reason may cause respiratory compromise either through direct pressure on the airways causing closure or from laryngeal spasm or both. Bleeding into the neck tissues (postoperative or otherwise) and neck trauma are the most common causes; however, allergic reactions with profound neck swelling, gross bloating from hypoproteinemia, burns involving the upper respiratory tract, and even severe surgical emphysema can produce breathing difficulties. Anaphylaxis is a rare event in most patients, but some patients are particularly prone, and it is typically associated with laryngospasm and neck swelling, in addition to bronchospasm.
Tracheal or Bronchial Obstruction
Perhaps the commonest cause of tracheal or bronchial obstruction intraoperatively is from a misplaced endotracheal tube with obstruction of a main stem bronchus. Postoperatively, mucus plugging of the airway with thick inspissated secretions is most commonly observed.
Inhaled foreign bodies are the commonest preoperative cause in children and adults, and aspiration of solid gastric contents in trauma patients is not uncommon. Inhalation of a dislodged tooth either before or during laryngoscopy and intubation for anesthesia is well reported, although uncommon. Bronchoscopy may be required to remove the foreign material.
Cardiovascular Complications
The major cause of morbidity and death after surgery are cardiovascular events or complications, leading to reduced blood pressure and under-perfusion of the brain, inducing coma or neurological injury. Under-perfusion of other organs causes renal, hepatic, gastrointestinal, and peripheral limb injury, ischemia, and failure.
Blood Pressure
As mentioned previously, blood pressure is determined to three main factors:
Heart function
Blood vessels
Blood volume
Changes in blood pressure can be evaluated by consideration of these three factors in turn, and the cause(s) determined as being due to one or a combination of these.
If under-perfusion is significant enough, a state of “shock” may result causing hypoxemic organ injury or death.
Cardiac Function
Cardiac function and output is determined by heart rate and stroke volume. Stroke volume, in turn, is dependent on wall integrity, wall size, cardiac valve function, and cardiac muscle contractile function. It is often useful to analyze the potential cause(s) of cardiac failure by considering these factors.
Heart rate is a function of the electrical rhythmicity of the sinoatrial (SA) node which typically fires at 70–100 beats per minute, being conducted to the atrioventricular (AV) node and conducting system to cause cardiac muscle contractions. Disturbances in the rate or rhythm can cause abnormal filling of the heart and alter cardiac output. Sinus rhythm is regular in nature and varies with respiration due principally to differences in intrathoracic pressure and the amount of venous return, and thereby right heart filling. Heart rate is increased by sympathetic stimulation and decreased by parasympathetic stimulation. Drugs can influence heart rate by primarily mimicking these pathways and their receptors, for example, atropine can quicken (by removing vagal inhibitory tone) and beta-blockers may slow (by removing noradrenergic stimulatory effects) cardiac rate. If the SA node fails as a pacemaker, the next most active part of the atrium or ventricle takes over as the pacemaker; arrhythmias may ensue, for example, atrial fibrillation (AF), heart block, ventricular ectopic beats, or ventricular fibrillation. During myocardial ischemia and infarction, abnormal rhythm circuits can occur which can effectively take over cardiac rhythm and cause bizarre ventricular rhythms, most of which result in less effective cardiac output than normal.
Arrhythmias arise when normal sinus rhythm is disturbed by alteration of function of the normal pattern of conduction. These principally occur when the sinoatrial node fails to function (e.g., sinus arrest, sinus block, sinus delay, sinus arrhythmia) or another part of the atrium (or ventricle) takes over the pacemaker function. Other causes include septal infarction, which can involve the AV node or septal conducting system, or ventricular circus movements where part(s) of the ventricle causes cardiac contraction overriding the normal pathway and conduction pattern. Serious arrhythmias are the most common cardiac events responsible for major morbidity and mortality associated with surgery and the perioperative period. However, as discussed later, arrhythmias are often the consequence of myocardial ischemia or cardiac failure, with each entity continuously feeding back on the other to worsen the situation (i.e., more arrhythmia, causing more ischemia, causing more arrhythmia), unless the deleterious “cycle” can be broken by correcting the arrhythmia or ischemia or both. Common recognized arrhythmias are listed below in Table 2.13.
Table 2.13
More commonly recognized arrhythmias encountered during surgery
Atrial |
Sinus tachycardia |
Sinus bradycardia |
Supraventricular tachycardia |
Atrial flutter |
Atrial fibrillation |
Ventricular |
Ventricular ectopic beats |
Heart blocks (first and second degree) |
Ventricular fibrillation |
Asystole |
Stroke volume is influenced by factors presented in Table 2.14.
Table 2.14
Cardiac function factors associated with stroke volume
Wall integrity |
Cardiac rupture |
Direct puncture |
Wall size |
Cardiac dilatation |
Ventricular hypertrophy |
Atrophy |
Cardiac valve function |
Regurgitation |
Stenosis |
Chordae tendineae rupture |
Valve rupture |
Cardiac muscle contractile function |
Myocardial ischemia |
Amyloidosis |
Infarction |
Cardiac aneurysm |
Electrical disturbances |
These factors are of course not mutually exclusive, most affect each other in some way, for example, ventricular hypertrophy influences both wall size and contractility.
Myocardial Ischemia, Infarction, and Cardiac Failure
These are the most common cardiac complications associated with the perioperative period. Infarction just prior to surgery substantially worsens the risk of mortality. The risk of perioperative mortality progressively reduces over a 3-month period after a myocardial infarction, then stabilizes; if the surgery can be delayed for 3 months, then the surgery-associated risk might be decreased. This is, of course, a highly patient- and procedure-dependent matter for individual consideration. The indication for surgery, on balance, may predicate early intervention. In some cases, coronary revascularization may be more important to perform before elective surgery. A history of myocardial ischemia, previous infarction, cardiac failure, or previous vascular compromise or surgery should alert the anesthetist and surgeon to the increased risk of a perioperative cardiac event. However, ischemia may be asymptomatic and therefore not evident before surgery, so a careful history is useful to detect any hint of possible cardiac disease. Optimization of the patient with known cardiac disease is discussed in Chap. 4.
Blood Vessels
The integrity of the blood vessels is paramount for containing the blood within a closed system to maintain blood pressure. Blood vessels may dilate and constrict in response to sympathetic and parasympathetic nervous system, physiological, and pathological factors. Peripheral vasodilatation can occur in shock, especially septic shock, where the vascular system expands in its capacity to cause a fall in blood pressure, although the cardiac function is often hyperdynamic and the blood volume may have increased in an attempt to compensate. Vasoconstriction may also occur in response to pain, hypothermia, injury, and cardiogenic shock. Some of these responses are “physiological,” although in the setting in which they occur they become “pathological,” in terms of the survival advantage to the patient. A normal response to hemorrhage would be generalized vasoconstriction of the gut and limbs, to redirect blood to the brain, lungs, and heart (physiological). However, when vascular shutdown is too intense and for too long in duration, organs may become ischemic and infarction may occur (pathological).
Blood Volume
Blood volume is relative to the capacity of the vascular system as pointed out above. An important factor implicit in this consideration is the time frame over which any change in blood volume or vascular system capacity occurs. Rapid bleeding can result in a serious acute decline in blood volume such that vasoconstriction alone may be insufficient to compensate. Heart rate and stroke volume will usually rise to increase cardiac output, but this may not fully compensate for the loss of blood volume. Judicious and appropriate blood, blood product, or other fluid replacement may correct the blood volume deficit sufficiently to normalize blood pressure and organ perfusion. Slower bleeding or oozing can go undetected for some time, especially if clinically concealed, and may only become evident when clinical anemia develops or if acute bleeding supervenes. The time course of slower bleeding may permit adequate compensation of blood volume loss through vasoconstriction and by absorption of fluid from the gut to replace losses.
Blood Pressure Complications
Although hypotension is perhaps the predominant complication that complicates surgical perioperative care, hypertension is not uncommonly encountered. Hypertension is usually silent and asymptomatic until a major cerebral or cardiac event supervenes, such as a hemorrhage or an infarct. Uncontrolled hypertension should be managed urgently to avoid these complications. Serial monitoring over 1 or more days may be appropriate to establish that persistent elevation is present, as opposed to a sporadic isolated reading. Intermittent high blood pressure may rarely herald the presence of a pheochromocytoma, which should be diagnosed and managed pharmacologically prior to any general anesthesia and surgery to avoid a malignant hypertensive crisis. With the exception of the anxiety of hospitalization for surgery, the commonest cause of hypertension in the perioperative period is failure of the patient to take their usual antihypertensive medications due to fasting, which when recommenced usually corrects the problem. Most anesthetists and physicians recommend taking the usual antihypertensives with a sip of fluid by the patient, well prior to surgery.
Shock
Shock is broadly defined as the “failure of oxygen demand to be met by oxygen supply” and can arise from many divergent causes. Blood pressure is a central factor in shock evolution and is determined, as noted, by three key elements: heart (stroke volume and heart rate), blood volume, and blood vessels. Blood pressure largely determines perfusion of tissues, and, together with local autoregulatory mechanisms, the overall vital function of organ systems is also dictated. When one system fails, inevitably effects occur on other organ systems. Classically, respiratory, cardiac, and renal failure are the key system failures that may require ventilatory, inotropic, and dialysis support, respectively, typically in an intensive care setting. Hypotension can lead to local and/or systemic under-perfusion of tissues and has myriad of causes.
Cardiogenic shock results from pump failure, due to disturbance of the cardiac action or rhythm. Often the antecedent factor is underlying cardiac disease, either myocardial, electrical conductive, or valvular.
Septic shock arises from by-products (e.g., cytokines, endotoxins) of infectious processes, causing peripheral vasodilatation and direct toxicity to the heart and other organs. The principal defect is the apparent loss of autoregulation in vascular beds and the resultant inability to regulate the peripheral capacity of the system for the circulatory blood volume present.
Hypovolemic shock results from acute blood loss or severe dehydration causing inadequate blood volume for the circulatory requirements.
When shock is established, a composite situation can develop where several mechanisms of shock are operating simultaneously. For example, “septic” shock often results in cardiac compromise and an element of “cardiogenic” shock, and severe dehydration may also cause a contribution from “hypovolemic” shock.
“Shock broadly is defined as the
‘failure of oxygen demand to be met by oxygen supply’.”
Multisystem Organ Failure (MSOF)
The definitions used to define MSOF have been variable; however, failure of one organ system typically places additional significant strain on other organ systems due to their interdependent nature. Therefore, even if a “single” system fails, many organs may be affected concomitantly. Mortality has been reported to be 40 % for single-organ failure lasting more than 1 day, 60 % mortality with failure of 2 organ systems, and 84 % for failure of three or more organ systems after 3 days (Zimmerman 1996a, b). There was also some evidence of improvement in the effectiveness of intensive care management with a significant reduction in mortality from 98 to 84 % in this latter group between 1982 and 1990, despite the relative incidence of MSOF remaining stable at 14 % of ICU admissions overall. Notably, the respective mortalities of both single- and dual-organ system failure patients remained unchanged over this period. Despite the usefulness of these large multi-institutional longitudinal studies, the specific single, dual, and multisystem organ failure mortalities for “surgical” patients were not separated out from “medical and other” causes of intensive care admission.
Fluid and Electrolyte Complications
Fluids are required for adequate hydration of the tissues and for adequate circulation of blood for perfusion, nutritional supply, and oxygen carriage to the tissues, as well as removal of waste products. Dehydration, including acute blood loss from the circulation, causes reduced blood pressure and tissue perfusion and, if sufficiently severe, results in tissue ischemia, infarction, and necrosis and may manifest as “circulatory shock.” A range of disturbances in organ physiology usually precedes the development of shock; these are typically reversible provided that rehydration and restoration of blood pressure is achieved. Organ dysfunction is usually caused by redistribution of blood to “essential organs” such as the brain and heart, with reduced supplies to the muscles, gut, and kidneys manifested by tiredness and weakness and reduced urine output. The kidneys are among the first organs to suffer injury from low systemic blood pressure and under-perfusion. The “obligatory” urine output is 0.5 to 1 mL/kg of body weight/hour, which represents the basal levels of urine production to maintain renal filtration function. When the urine output falls below this level, the kidneys have usually become severely under-perfused, “shutdown,” and ischemic, being unable to produce adequate urine. Acute renal injury may result in acute renal tubular necrosis, which is usually reversible if blood pressure can be restored; if not, serious cortical necrosis may result, which may not be fully reversible.
Water disperses across most membranes in the body and distributes according to “Starling’s law” reflecting the balance between the hydrostatic (blood) pressures and the oncotic (protein) pressures in the intravascular and tissue “compartments.” Serum proteins largely remain intravascularly and hold fluid inside the vessels except at the arterial end of the capillaries where the hydrostatic pressure exceeds the oncotic pressure, causing fluid to move out of the capillaries into the tissues. At the venous end of the capillary bed, the hydrostatic pressure has typically fallen much lower, and the relatively greater oncotic pressure pulls fluid back into the intravascular compartment. Lack of water, blood pressure (hydrostatic pressure), or tissue proteins (oncotic pressure) can profoundly disturb this fine balance. Edema can result from low intravascular protein levels or from increased hydrostatic venous pressure from poor venous return due to cardiac failure or venous obstruction.
Two basic types of fluid are used for replacement, especially for more urgent fluid replacement; these are crystalloids (saline and balanced salt solutions) and colloids (proteins and artificial sugars). Sodium chloride solution (salt; saline; NaCl) diffuses much more than protein with about 25 % remaining intravascularly after infusion under usual circumstances. Protein diffuses very slowly or minimally out of the blood vessels with about 75 % remaining intravascularly ½ to 1 h after infusion. Five percent dextrose is essentially water, made isotonic with glucose, which diffuses across all compartments rapidly to equilibrate according to the concentration of solutes (proteins and salts) in those compartments.
Saline is often used for most fluid replacement to rapidly replace fluid and sodium and is often satisfactory in many situations. Plasma expanders, such as protein or protein substitute (gelatin-like agents) fluids, are used for acute replacement of fluid in the urgent setting where blood pressure requires more rapid elevation. These latter fluids “hold” fluid in the intravascular compartment better and for longer than saline and can additionally draw fluid in from the tissues to expand the intravascular fluid volume. However, recent evidence in acute trauma resuscitation settings has shown that vigorous and rapid fluid replacement, especially with albumin, may cause relative overhydration and lead to increased morbidity and mortality, especially in elderly patients. Acutely, colloid agents may also cause appreciable fluid shifts and relative dehydration of the tissues as fluid is rapidly drawn into the vessels (SAFE 2004; Perel and Roberts 2007, updated 2009).
Colloids Compared to Crystalloids
The Cochrane Collaboration Review (2007, updated 2009) concluded that there was no evidence that the risk of death was reduced using colloids vs. crystalloids, after resuscitation in patients requiring volume replacement, in patients with trauma, burns, or following surgery. Data was derived from 55 RCTs, which presented mortality data. The use of colloids was more expensive than crystalloids, and since the efficacy was comparable, use of colloids outside the context of RCTs was difficult to justify. Pregnant women and neonates were excluded.
Albumin or Plasma Protein Fraction – Twenty-three trials with 7,754 patients reported data on mortality. The relative risk (RR) from these trials was 1.01 (95 % confidence interval [95 % CI] 0.92 to 1.10). When a trial with poor quality allocation concealment was excluded, pooled RR was 1.00 (95 % CI 0.91 to 1.09).
Hydroxyethyl Starch – Sixteen trials with 637 patients compared hydroxyethyl starch with crystalloids. The RR was 1.05 (95 % CI 0.63 to 1.75).
Modified Gelatin – Eleven trials with 506 patients compared modified gelatin with crystalloid. The RR was 0.91 (95 % CI 0.49 to 1.72).
Dextran – Nine trials with 834 patients compared dextran with a crystalloid. The RR was 1.24 (95 % CI 0.94 to 1.65).
Colloids in Hypertonic Crystalloid Compared to Isotonic Crystalloid – Eight trials with 1,283 patients compared dextran in hypertonic crystalloid with isotonic crystalloid including. The RR was 0.88 (95 % CI 0.74 to 1.05).
Side effects of gels may be unanticipated and occasionally severe with development of allergic reactions, including anaphylaxis. These problems may complicate an already problematic clinical situation, which can be averted to some degree by very careful consideration of when and in whom to use these products.
Fluids used for replacement include:
Saline (0.9 % NaCl; normal saline; NNaCl)
Dextrose (5 %)
4 % Dextrose and 1/5 normal saline
Balanced salt solutions (Ringer’s lactate or Hartmann’s solutions)
Plasma expanders (dextrans, gelatins, starches)
Albumin (5 % albumin; normal serum albumin, NSA)
Stable plasma protein solution (SPPS)
Whole blood (RBC, platelets, and plasma proteins)
Packed red cells (RBC)
Normal intake of fluid per day is of the order of 1.5 to 2 L per day, assuming fluid losses are normal and stable. Higher fluid losses may occur in certain situations such as high temperature environments where dehydration occurs and in disease states such as diarrhea and vomiting. After surgery, “third-space” fluid losses can be appreciable (up to 2–4 L) as fluid collects in swollen tissues as a result of the surgical tissue trauma. These situations often demand more fluid than the usual 2 L/day.
The required fluid input can be determined by urine output, provided renal function is normal, so that the amount of fluid infused per hour can usually be titrated against the urine output over the previous hour(s).
The amount of fluid and rate of infusion that needs to be used for resuscitation are determined by clinical judgement, relating to factors that include known or predicted blood or fluid loss, blood pressure level, degree of trauma or shock, urine output, central venous pressure, known comorbidities, and the age of the patient.
The main complications of fluid infusion are:
Overload or overhydration
Underhydration or dehydration
Overload is caused by infusion of too much fluid such that the vascular capacity is exceeded, with respect to the ability of the heart to adequately pump the fluid to clear the peripheral circulation (resulting in peripheral edema) and/or the ability to clear the lungs (resulting in pulmonary edema). These two aspects essentially reflect the functional capacity of the right and left heart to pump blood, respectively.
Underhydration is the failure to adequately replace fluid or maintain the sufficient fluid volume to keep up with fluid losses, to ensure adequate cardiac filling, circulation, and tissue perfusion. Both of these may precede surgery and complicate anesthesia and perioperative care.
In the intensive care or surgical high-dependency settings, and occasionally in the surgical wards, central venous pressure monitoring can provide a very useful tool to evaluate the amount of venous filling and circulatory load and to enable balancing of fluids more accurately.
Metabolic and Endocrine Complications
Metabolic complications include glucose intolerance, ketoacidosis, metabolic acidosis, hypermetabolic states, thyroid insufficiency, and adrenal impairment. In fact, almost any medical metabolic problem can arise after surgery, although it may not be related to the anesthesia or surgery per se, but is frequently included as a “complication of surgery.” These instances are often indistinguishable from “true” complications following surgery. After surgery on the thyroid or parathyroid, hypocalcemia may develop, either related to injury or removal of the parathyroid glands. The hypocalcemia may be transient, if the stress or injury recovers, and may need oral or rarely intravenous calcium replacement in the shorter term. If the hypocalcemia is slight, no treatment is typically required. However, if hypocalcemia is permanent, continued calcium and vitamin D replacement may be required. Nutritional and trace mineral deficiencies are common, especially in chronically unwell patients, and require careful balancing and supplementation to correct. Magnesium and manganese, in addition to the classical electrolytes sodium and potassium, are essential to monitor. Nutritional complications are dealt with in a separate section later. The endocrine and metabolic changes can sometimes be surprisingly subtle and go undetected for long periods, unless actively sought. Recent reliance on gastrointestinal hyperalimentation, where possible, has helped correct some of the absorptive and metabolic problems encountered previously with long-term parenteral nutrition.
Perhaps the commonest problem after surgery is disturbance of blood glucose. The loss of diabetic control is very common, either due to induction of glucose intolerance, feeding glucose rich foods, withholding usual medications, or the stress of surgery. Preoperative medical and anesthetic consultation is advisable and wise in many situations to plan for surgery and avert or reduce any problems. While fasting for surgery or postoperatively, a 5 % glucose intravenous fluid infusion is worthwhile to provide fluid and small amounts of glucose to reduce the risk of a hypoglycemic episode.
Some metabolic problems relevant to surgery include:
Metabolic acidosis
Ketoacidosis
Hyperglycemia
Hypoglycemia
Hypocalcemia
Hypercalcemia
Cushing’s syndrome
Sheehan’s syndrome
Hypothyroidism
Hyperthyroidism
Hypoadrenalism
Pheochromocytoma
Addison’s disease
Nutritional Complications
Many surgical conditions are associated with a decline in nutritional status, particularly when diagnosis is later in the course of a disease. This is especially true for advanced malignancies and for gastrointestinal problems that cause anorexia, difficulties with food intake, vomiting, or hypermetabolic/catabolic states. Where surgery is performed when nutritional status is poor and malnutrition is present, even mild forms, the risks of poor healing, infection, and wound complications are increased. One of the main difficulties arising from poor nutritional status is poor immune function, which is vital for almost every aspect of the healing response and surgical care. Immune function is notoriously difficult to monitor or augment, apart from ensuring global nutritional improvement, where possible.
Nutritional deficiencies, although often global, with deficits in many areas of the metabolic pathways essential for good health and surgical recovery, some deficiencies may be very specific and may not be clinically obvious or easily detectable. Examples of subclinical deficiencies include deficits in the trace elements magnesium, manganese, zinc, molybdenum, copper, cobalt, chromium, selenium, and iron, which were first recognized in animals grazing on or crops growing for long periods in deficient soils, and subsequently similar elemental deficiencies were recognized in long-term intensive care patients. Nutritional deficiencies fall into several groups:
Protein and essential amino acids
Fats
Carbohydrates
Vitamins (A, B group, C, D, E, and K)
Trace elements (including Mg, Mn, Zn, Mo, Cu, Co, Cr, Se, Fe)
Nutritional Support
In general terms, when nutritional intake cannot be restored by the usual oral feeding route within 7–10 days after surgery, a state of severe catabolism has commenced and malnutrition will usually result, unless some alternative form of nutritional maintenance is provided, whether by enteral or parenteral means.
Enteral Feeding
The most effective form of nutrition is via the gut with nutrient absorption in the usual manner. This means that enteral nutrition provides the most preferable and predictable form of administering nutrition to patients, where at all possible.
Forms of enteral feeding include:
Standard oral feeding (with or without supplements)
Nasogastric tube
Nasoenteric tube
Percutaneous gastroenterostomy tube
Feeding enterostomy tube
Enteral nutrition may be used preoperatively, where possible, to improve nutritional status as much as possible before surgery and to reduce the risks and complications from malnutrition after surgery. This may reverse any catabolic effects existing before surgery, depending on the clinical problem, to place the patient in a more favorable situation to be able to cope with the catabolic state that usually ensues, for a variable period, after surgery.
If a period of total parenteral nutrition (TPN) is required, either before and/or after surgery, then it is wise to move as soon as practicable to enteral forms of nutrition to provide better and more cost-efficient administration of nutrition.
Complications and problems associated with enteral feeding:
Vomiting or regurgitation
Aspiration pneumonitis
Tube dislodgement
Abdominal pain
Diarrhea or constipation
Electrolyte disturbances
Over- or dehydration
Parenteral Feeding
When enteral administration of nutrition is not possible, or inadvisable, then total parenteral nutrition (TPN) is able to be used to provide an intravenous form of balanced nutritional support. TPN requires administration of high osmolarity concentrated fluids via a large diameter vein, usually using a central venous catheter, because TPN fluids often require relatively rapid infusion and, due to their concentration, are extremely irritant to smaller peripheral veins. TPN is used where gut function is not possible or the gut route needs to be rested, either following gastrointestinal or other surgery, for example, where fistulae are trying to be closed or ischemic gut is present. The use of preoperative TPN represents a “two-edged sword” to some degree, because there is the benefit of reversal of the catabolic state with improvement of nutrition and immune function, but there is also a complication rate (up to 10 %) associated with central venous line insertion, maintenance, and TPN administration over time.
Sometimes, placement of a percutaneous gastroenterostomy tube or feeding enterostomy tube is useful during surgery to permit rapid transfer to enteral feeding after a period of TPN.
Components usually used in TPN include:
Fatty acids
Glucose
Insulin
Amino acids
Vitamins
Trace elements
In many institutions, these are mixed to order in the hospital pharmacy under sterile conditions and delivered in bag(s) to the surgical high dependency, ICU, or ward as needed. Between bags of TPN, IV administration of 10 % glucose is advisable, as this will usually reduce the risk of hypoglycemia from the late effects of insulin contained in the TPN. When TPN is being ceased, then a scaling-down process, with gradual reduction of the rate of TPN administration is also a useful technique to avoid rapid and potentially dangerous changes in glucose or other levels in the blood.
Lipids are usually administered as 10, 20, or 30 % concentration emulsions of long-chain fatty acids, containing essential fatty acids (linoleic and linolenic acid).
Complications and problems associated with TPN:
Hyperglycemia
Hypoglycemia (insulin induced)
Hyperlipidemia
Hypercapnia and respiratory decompensation
Hyponatremia
Hypokalemia
Hypophosphatemia
Hypomagnesemia
Vitamin deficiencies (especially thiamine)
Fluid balance problems (over- or dehydration)
Cholestasis and gallstones
Acalculous cholecystitis
Transient liver dysfunction
Acid-based disturbances
Complications of central venous catheter lines
Local infection
Systemic sepsis
Thrombosis
Embolism
Catheter dislodgement
Pneumothorax
Cardiac arrhythmias
Catheter obstruction
Hypokalemia, hypophosphatemia, hypomagnesemia, and glucose intolerance are not uncommonly experienced when a severely malnourished patient is fed using TPN – the so-called refeeding syndrome.
Drug-Related Complications
Among the most important and prolific complications that are identified in hospitals and healthcare facilities and in community settings are complications related to drugs. These primarily arise from:
Consumption of the wrong drug or dose
Consumption of the wrong combination of drugs
Side effects or reactions from drugs
Noncompliance with drug taking
Failure to prescribe or dispense properly
Failure of communication
These may arise from disabilities such as poor vision, memory, tactile ability, or from simple time constraints, supervision, lapses of thought, knowledge, or process – either on the patient’s, relatives/carers or even on the practitioner’s (medical or nursing) part.
A myriad of drug-related complications exist, and all of these cannot possibly be dealt with here. The less common interactions and side effects may never reach description in the now voluminous literature. Individual drugs must be evaluated for their potential risks and side effects and for the potential interactions between different drugs and possible interactions with other agents used, for example, the type(s) of fluids being administered or nonprescription items.
Overall, however, in a population sense, one of the rather amazing realizations concerning drugs (prescription, pharmacy only, nonprescription, foods, and herbal) is that the range and severity of drug interactions and side effects which are clinically detectable and evident is so incredibly relatively low! Most people must be on vast numbers of mixed and multi-mixed pharmacologically active agents and foods, and yet no or few adverse effects become evident, and the patient is not perturbed in any definable way.
These problems and potential for side effects, interactions, and adverse events would be expected to only increase over time as the number of agents and their application become greater.
Despite this point, drug interactions among patients receiving healthcare and especially surgical and medical inpatient interventions remains one of the most common and costly adverse events for healthcare systems and governments. This is primarily because in the healthcare setting, although we are usually endeavoring to control the situations as much as we can, we are within a relatively complex high-risk environment where serious problems are being dealt with, often with multiple staff, interventions, protocols, time pressures, urgencies, and comorbidities, which all tend to magnify the risks of errors arising.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


