Food Additives, Contaminants, and Natural Toxicants: Maintaining A Safe Food Supply1
Steve L. Taylor
Joseph L. Baumert
1Abbreviations: BHT, butylated hydroxytoluene; BPA, bisphenol A; DDT, dichlorodiphenyltrichloroethane; FDA, Food and Drug Administration; FD&C, Food, Drug, & Cosmetic Act; FEMA, Flavor and Extract Manufacturers Association; GRAS, generally recognized as safe; HCN, hydrocyanic acid; MSG, monosodium glutamate; PBB, polybrominated biphenyl; PCB, polychlorinated biphenyl; RDA, recommended dietary allowance.
Foods can be viewed as complex mixtures of chemicals. As noted throughout this book, many of those chemicals are nutrients needed to sustain life. However, many nonnutrients exist in foods also. Some of these nonnutrient components—food additives, food contaminants, processing-induced toxicants, and naturally occurring toxicants—may be toxic under certain circumstances of exposure but are not hazardous under typical circumstances. Food contaminants and naturally occurring toxicants are examples of chemical components that are more likely to be hazardous. Processing-induced toxicants form under certain processing conditions, especially heat treatments. Because processing treatments prolong the shelf life of foods and guard against microbial hazards, the development of processing-induced toxicants is a classic risk-versus-benefit puzzle. Among food additives, certain nutrient components of food also can be potentially toxic under certain circumstances, and several of these situations will serve as examples in this chapter. However, this occurs primarily when the nutrients are used as additives or supplements rather than as a consequence of the typical levels ingested with traditional dietary practices. In recent decades, the science of food toxicology has emerged as an approach toward understanding and assessing the risks posed by the various chemicals in food.
Food toxicology could be defined as the science that establishes the basis for judgments about the safety of foodborne chemicals (1). The central axiom of toxicology, as set forth by Paracelsus in the 1500s, states, “Everything is poison. Only the dose makes a thing not a poison.” Thus, all chemicals in foods—whether natural or synthetic, inherent, adventitious, or added—including nutrients, are potentially toxic. Most foodborne chemicals are not hazardous under typical circumstances of exposure, because the doses ingested with traditional diets are insufficient to cause injury. The degree of risk posed by exposure to any specific foodborne chemical is determined by the dose, duration, and frequency of exposure or, in the case of allergies, the degree of sensitivity of the individual. The age-old wisdom about the benefits of eating moderate amounts of a varied diet protects most consumers from any harm. Unusual diets can sometimes result in toxic responses from chemicals that normally would be considered safe under more typical circumstances of exposure.
Acute adverse reactions to foods are those adverse reactions occurring within hours to a few days following exposure to the particular foods/foodborne chemicals. Acute foodborne intoxications usually result in the rapid onset of symptoms. However, the diagnosis of chronic foodborne intoxications is complicated because symptoms typically develop more slowly with prolonged exposure. Food-associated intoxications are caused by chemicals in food, although the chemical constituents of foods vary widely in toxicity. All consumers are susceptible to most food intoxications. This chapter focuses on the various categories of chemical components that occur in foods (food additives including nutrient additives, food contaminants both manufactured and naturally occurring,
processing-induced toxicants, and nonnutrient components that can be classified as naturally occurring toxicants). In each case, illustrative examples are presented.
processing-induced toxicants, and nonnutrient components that can be classified as naturally occurring toxicants). In each case, illustrative examples are presented.
FOOD ADDITIVES
Numerous chemical substances are added knowingly to foods to provide a wide variety of technical benefits, including nutrients (Table 101.1). Several thousand food additives exist, but many are used in very small amounts. Food additives can be classified on the basis of their regulatory standing in the United States: (a) generally recognized as safe (GRAS) substances, (b) flavors and extracts, (c) direct additives, and (d) color additives. Food additives are added to foods intentionally and carefully evaluated for safety. Food additives are generally not hazardous under normal circumstances of exposure.
In the United States, GRAS substances are those food ingredients that were in common use before the latest version of the Food, Drug, and Cosmetic (FD&C) Act that was enacted in 1958. The 1958 FD&C Act required approval by the US Food and Drug Administration (FDA) of any newly developed food additives, but recognized the long history of safe use of many additives. More than 600 chemicals are on the FDA GRAS list, including sucrose, salt, butylated hydroxytoluene (BHT), and spices. Many nutrient ingredients are on the GRAS list also. Most of the common food ingredients on the GRAS list were in common use before 1958. From a legal standpoint, GRAS substances are not actually additives, but that distinction is seldom made by the consumer. Although GRAS substances were in common use before 1958, it is not necessarily true that substantial toxicologic data exist for all these substances. For example, the concerns around sodium levels in the diet and hypertension ensued long after 1958, and salt and other sodium-containing ingredients such as monosodium glutamate (MSG) were placed on the GRAS list before this scientific information became available. Reviews of the safety of the GRAS substances have been conducted since 1958, and deficiencies in our information on their toxicity have been identified and corrected in some cases. Substances, or certain uses of substances, can be removed from the GRAS list if the FDA acquires evidence of hazard to consumers.
TABLE 101.1 CATEGORIES OF FOOD ADDITIVES | ||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
Flavors and extracts represent a large percentage of the total number of food additives. The Flavor and Extract Manufacturers Association (FEMA) keeps a list of accepted flavors and extracts. FEMA is also responsible for evaluating the safety of the chemicals on the list. In essence, the FEMA list is a GRAS list for flavors and extracts. More than 1000 chemicals are on the FEMA list, although some of these chemicals and extracts are no longer used.
Direct food additives are the category for those new food additives that have been approved by the FDA since 1958. In reality, few approvals for new food additives have been granted in recent years. Extensive and costly safety data are required to gain FDA approval for a new food additive. Aspartame, a nonnutritive sweetener, is one of the most notable direct food additives in use in the United States (and many other countries). Aspartame was approved as a nonnutritive sweetener for certain types of uses some years ago, and the consumption of this new additive has become substantial. More recently, Olestra, a noncaloric fat replacer, was approved for certain uses such as the deep fat frying of chips. Aspartame and Olestra are examples of the types of substances for which potential usage levels are high enough to warrant the cost of acquiring the toxicologic information needed to gain food additive approval.
Color additives are regulated in a separate part of the FD&C Act. New artificial color additives must be approved by the FDA in much the same way that new food additives are approved. Some color additives have been banned since 1958 because of concerns about their possible chronic toxicity. A good example is FD&C Red #2, which is banned in the United States, although it is allowed in Canada and other countries. Conversely, FD&C Red #40 is allowed for use in the United States but banned in Canada.
The degree of hazard associated with the presence of additives in our foods is quite low for several reasons. First, the level of exposure to most food additives, especially flavoring ingredients, is generally low. Furthermore, the oral toxicity of food additives tends to be extremely low, especially for acute toxicity. Still, some concerns have arisen about the chronic toxicity of a few food additives, including saccharin, cyclamate, and others. Yet another reason for the low hazard associated with food additives is the established safety of many additives. Many food additives have been subjected to safety evaluations in laboratory animals. In these cases, the toxicity of these food additives is well known, and exposure can be limited to levels far below any dose that would be hazardous. By contrast, the toxicity of naturally occurring chemicals
in foods is often not known, and we cannot be certain that hazardous circumstances will not exist under certain conditions of exposure. Other food ingredients, particularly the GRAS substances, have long histories of safe use even if classical toxicologic evaluations in laboratory animals have not always been exhaustively performed.
in foods is often not known, and we cannot be certain that hazardous circumstances will not exist under certain conditions of exposure. Other food ingredients, particularly the GRAS substances, have long histories of safe use even if classical toxicologic evaluations in laboratory animals have not always been exhaustively performed.
The safety of some food additives has been called into question. In some cases, the questions have revolved around evidence for weak carcinogenic activity in laboratory animals. Some additives, such as FD&C Red #2 and cyclamate, have been banned as a result of such evidence. Warning labels are required for saccharin. In addition to carcinogenicity, other concerns have arisen, such as the role of sugar in dental caries and abnormal behavioral reactions, the role of MSG in asthma, the role of aspartame in headaches and other behavior and neurologic reactions, and the role of Olestra in gastrointestinal complaints. The concerns range from acute toxicity such as aspartame and headaches or MSG in asthma to chronic toxicity such as saccharin and bladder cancer or sugar in dental caries. Although a detailed discussion of all of these issues is beyond the scope of this chapter, many of these assertions have been questioned and remain controversial.
A few illustrative examples are discussed. The examples include several food additives that have caused acute illness under certain conditions of exposure. These intoxications are usually the result of either excessive consumption of the additive or ingestion by an individual who has an abnormal degree of sensitivity to the additive. Misuse of food additives by consumers, or food processors, has also created hazardous situations on occasion. Saccharin will be discussed briefly as an example of an additive where chronic toxicity concerns exist. Many other examples could be cited for chronic toxicity concerns, including cyclamate and FD&C Red #2.
Sorbitol and Hexitols
Sorbitol and various other hexitols are commonly used alternative sweeteners. Dietetic food diarrhea associated with these polyol food additives is a good example of an intoxication resulting from the excessive consumption of a food additive. The hexitols and sorbitol are widely used sweeteners in dietetic foods. The hexitols and sorbitol are especially common in noncariogenic candies and chewing gum. Although these sugar alcohols are not as easily absorbed as sugar, they are equally as caloric as sugar once absorbed. Because of their slow absorption, these sweeteners can cause an osmotic-type diarrhea if excessive amounts happen to be consumed. Several cases have been reported in which consumers were ingesting more than 20 g of these sweeteners per day (2, 3). The levels of hexitols and sorbitol used in foods vary, but in one case the ingestion of 12 pieces of hard candy over a short period of time provided 36 g of sorbitol and resulted in diarrhea (2).
Sulfites
Sulfites (sodium and potassium metabisulfite, sodium and potassium bisulfite, sodium sulfite, sulfur dioxide) have been widely used as food additives for many years. Sulfites serve several important technologic functions, including as antimicrobial agent, inhibitor of enzymatic and nonenzymatic browning, and bleaching agent. Sulfite-induced asthma is well established as an example of sensitivity to a food additive that afflicts only a small percentage of the population.
Tartrazine (FD&C Yellow #5)
Tartrazine, also known as FD&C Yellow #5, is an approved artificial food color. This colorant has been widely used in foods and pharmaceuticals for many years. Tartrazine is a color additive that is associated with adverse reactions (asthma and chronic hives) in a sensitive subpopulation of consumers (4). However, unlike the situation with sulfite-induced asthma, the association of tartrazine in the provocation of asthma and chronic urticaria is controversial. Some studies have revealed a cause-and-effect relationship, whereas other studies have not (5). Both asthma and chronic urticaria are chronic illnesses whose symptoms tend to flare up at unpredictable times. In some of the clinical trials on tartrazine, key pharmaceutical agents have been withdrawn from the human subjects prior to the tartrazine challenges. If the study is not designed carefully, the flare-up of the asthma or urticaria in such a trial could be caused by either the administration of tartrazine or the withdrawal of the medication. The plethora of poorly designed clinical trials on tartrazine has led several groups to conclude that tartrazine may not actually provoke asthma or chronic urticaria (5, 6).
Olestra
Olestra received food additive approval more recently and can be used as a fat replacer. Because Olestra is poorly absorbed and is not metabolized, it does not provide the calories that would be obtained with fat in similar products. However, the use of Olestra has been associated with acute gastrointestinal complaints, including anal leakage (7). Only certain consumers seem to be affected, although the prevalence and seriousness of such complaints is a matter of some controversy (8). Obviously, many consumers occasionally experience postprandial gastrointestinal maladies, which makes the association of such complaints with a particular ingredient rather challenging.
Saccharin
Saccharin was one of the first nonnutritive sweeteners approved for food use in the United States. High doses of saccharin have been shown to cause bladder cancer in laboratory animals (9). However, the extrapolation of these results to humans, who typically ingest much lower
levels of the ingredient, has stimulated controversy. Thus, the carcinogenicity of saccharin to humans at typical levels of intake is, at best, uncertain, and saccharin remains on the market in the United States. Despite the uncertain relevance of the toxicologic data, warning labels remain on saccharin products indicating that saccharin is known to cause cancer in laboratory animals.
levels of the ingredient, has stimulated controversy. Thus, the carcinogenicity of saccharin to humans at typical levels of intake is, at best, uncertain, and saccharin remains on the market in the United States. Despite the uncertain relevance of the toxicologic data, warning labels remain on saccharin products indicating that saccharin is known to cause cancer in laboratory animals.
NUTRITIONAL FOOD ADDITIVES
Many food additives, including vitamins and minerals, serve nutritional functions. Although these functions are widely and appropriately viewed as beneficial, nutrients can occasionally be associated with adverse reactions. Because nutrients are chemicals and all chemicals are toxic at some dose, nutrients can exert toxic effects under some conditions of exposure. In fact, in the latest version of dietary reference intakes developed by the US National Academy of Sciences, tolerable upper intake levels are provided for many of the common micronutrients (8). Although data on micronutrient toxicity are often scant, elevated levels of nutrients are clearly hazardous in some situations. For example, the intake of large amounts of vitamin A was hazardous to polar explorers consuming large amounts of polar bear liver (10). However, this would be one of the very few examples where the ingestion of food (albeit atypical “food”) was responsible for nutrient toxicity. More commonly, tolerable upper intake levels can only be exceeded by use of supplements or inappropriate addition of elevated levels when used as additives. Several examples are discussed as follows.
Niacin
Excessive consumption of the B vitamin, niacin, can cause an acute onset of flushing, pruritus, rash, and burning or warmth in the skin especially on the face and upper trunk (11); and gastrointestinal discomfort has been noted by some patients (12). Outbreaks have occurred from the excessive enrichment of flour used to make pumpernickel bagels (12) or corn meal (13). These episodes occurred as a result of inaccurate or inadequate labeling of food ingredient containers. The amount of niacin required to elicit such adverse reactions is at least 50 times the recommended dietary allowance (RDA) (12, 13). The symptoms of niacin intoxication are acute, self-limited, and without sequelae.
Vitamin A
In addition to the example with polar bear liver, the toxicity of vitamin A can be illustrated by other more conventional, but also unwise, dietary choices. Vitamin A intoxication was reported in twin infants who were provided a diet consisting largely of pureed chicken livers, pureed carrots, milk, and vitamin supplements (2). Apparently, the mother of these infants initiated this diet because she did not trust commercial baby foods. After several weeks on this diet, the infants began to vomit and developed a skin rash. The symptoms disappeared when a more normal diet was instituted. The estimated intake of vitamin A and carotene was 44,000 IU/day compared with the RDA of 1500 to 4500 IU/day for infants.
INTENTIONAL FOOD ADULTERANTS
The preceding discussion centered on the use of ingredients that had been approved for addition to foods by some regulatory agency. Of course, the illegal and unscrupulous addition of chemical adulterants that are not approved for addition to foods and have not been tested for safety or had a long history of safe use could be quite hazardous. Several noteworthy situations can be cited including the toxic oil syndrome that occurred in Spain several decades ago and the more recent melamine adulteration of infant formula and pet foods in China.
Toxic Oil Syndrome
In the early 1980s, an epidemic involving more than 10,000 cases and more than 300 deaths occurred in Spain that was ultimately linked to the ingestion of unlabeled, illegally marketed cooking oils (14, 15). The illicit cooking oil contained oils that were denatured and intended for industrial rather than food use. The causative toxin in the oils remains unknown, although fatty acid anilides resulting from the denaturation process are suspected to be at least partially responsible (16).
Multiple organ systems were involved in the clinical manifestations of this illness (16). Initially, affected individuals experienced fever, chills, headache, tachycardia, cough, chest pain, and pruritus. Physical examinations revealed various skin exanthema, splenomegaly, and generalized adenopathy. Pulmonary infiltrates were noted in 84% of the affected individuals, probably as the result of increased capillary permeability. The intermediate phase of the illness persisted from the second week after onset through the eighth week after ingestion. Gastrointestinal symptoms, primarily abdominal pain, nausea, and diarrhea, predominated during this intermediate phase. At this stage, clinical examination revealed marked eosinophilia in 42% of patients, high immunoglobulin E levels, thrombocytopenia, abnormal coagulation patterns, and evidence of hepatic dysfunction with abnormal enzymes. Some individuals became jaundiced, and many had hepatomegaly. The late phase of the illness, characterized initially by neuromuscular and joint involvement, developed in 23% of cases and began after 2 months of illness. Later, individuals in this late phase of the illness developed vasculitis and a scleroderma-like syndrome. Affected individuals complained of intense muscular pain, edema, and progressive muscular weakness. Muscular atrophy was apparent in some individuals. Neurologic involvement included depressed deep tendon reflexes, anesthesia, and dysesthesia. Respiratory problems
developed because of neuromuscular weakness and progressed to pulmonary hypertension and thromboembolic phenomena. The scleroderma-like symptoms included Raynaud phenomenon, sicca syndrome, dysphagia, and contractures caused by thickening skin collagen. Vascular lesions were noted in all organs, apparently resulting from endothelial proliferation and thrombosis. All patients in the late group had antinuclear antibody and many had antibodies against smooth muscle and skeletal muscle (17). These pathologic and clinical features are consistent with an autoimmune mechanism for the illness. Because the precise causative agent and its mechanism have not been delineated, this serious epidemic might occur again if similar circumstances exist. Also, it is unknown whether the toxin might be present in small amounts in other foods, and thus might be producing or aggravating other clinical conditions.
developed because of neuromuscular weakness and progressed to pulmonary hypertension and thromboembolic phenomena. The scleroderma-like symptoms included Raynaud phenomenon, sicca syndrome, dysphagia, and contractures caused by thickening skin collagen. Vascular lesions were noted in all organs, apparently resulting from endothelial proliferation and thrombosis. All patients in the late group had antinuclear antibody and many had antibodies against smooth muscle and skeletal muscle (17). These pathologic and clinical features are consistent with an autoimmune mechanism for the illness. Because the precise causative agent and its mechanism have not been delineated, this serious epidemic might occur again if similar circumstances exist. Also, it is unknown whether the toxin might be present in small amounts in other foods, and thus might be producing or aggravating other clinical conditions.
Melamine
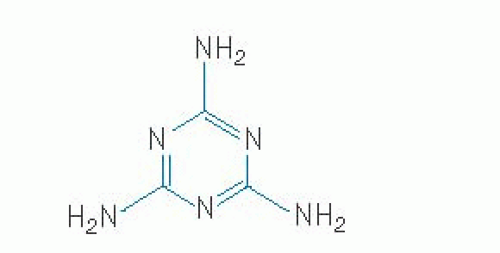
Melamine (2,4,6-triamino-1,3,5-triazine [Fig. 101.1]) is an industrial chemical used for manufacturing of plastics, adhesive, fabrics, and flame retardants. Several years ago, melamine was intentionally added to pet foods and dairy products including infant formula in China to increase the “apparent” protein content of these products (18, 19). Melamine gives a strong positive result in the Kjedahl and Dumas methods for nitrogen determination, which often are used to assess the protein content of foods. In 2007, wheat gluten from China was adulterated with melamine and that wheat gluten was subsequently used to manufacture pet foods. Many pets in North America suffered renal toxicity as a result of consuming the pet food contaminated with melamine and the related substance, cyanuric acid (18). When melamine and cyanuric acid are consumed together, crystals of melamine cyanurate form readily in the kidney tubules leading to acute renal failure. An unknown number of pet cats and dogs died as a result of kidney failure. Subsequently, in 2008, infant formula and other milk products in China were found to be adulterated with melamine; cyanuric acid was not detected in this episode. As many as 294,000 infants and young children in China were affected by the contaminated foods (19) leading to 50,000 hospitalizations and at least 6 fatalities. As in the pet food episode, renal toxicity was the primary manifestation. Apparently, melamine alone under some conditions can lead also to the formation of crystals in the renal tubules leading to nephrolithiasis.
FOOD CONTAMINANTS
Potentially hazardous chemicals may contaminate foods from a variety of sources, including natural contaminants, agricultural chemicals, industrial contaminants, and processing-induced contaminants. The natural contaminants can include mycotoxins from molds, phycotoxins from marine algae, and bacterial toxins. Other contami nants are manufactured and have useful purposes although not intended to occur in foods.
Agricultural Chemicals
Many different chemicals are used in modern agricultural practices. Residues of these agrichemicals can occur in raw and processed foods. Federal regulatory agencies evaluate the safety of such chemicals and regulate and monitor their use on plant food products and in food-producing animals (2). The major categories of agricultural chemicals include insecticides, herbicides, fungicides, fertilizers, and veterinary drugs including antibiotics.
Insecticides
Insecticides are added to foods to control insect pests. Insecticides fall into several major categories including organochlorine compounds (dichlorodiphenyltrichloroethane [DDT], chlordane, and others, many of which are now banned), organophosphate compounds (e.g., parathion and malathion), carbamate compounds (e.g., carbaryl and aldicarb), botanical compounds (e.g., nicotine and pyrethrum), and inorganic compounds (e.g., arsenicals).
Insecticide residues in foods are not particularly hazardous, especially on an acute basis, because of the exceedingly low residue levels of insecticides found in most foods. Certainly, large doses of insecticides can be toxic to humans. Some are neurotoxins such as the organophosphates and carbamates, which are cholinesterase inhibitors and act by blocking synaptic nerve transmission. Insecticide residues in foods pose a low degree of hazard for several reasons: (a) the level of exposure is very low; (b) some insecticides are not very toxic to humans; (c) some insecticides decompose rapidly in the environment; and (d) many different insecticides are used, which limits exposure to any one particular insecticide (1).
Acute food poisoning incidents attributable to the proper use of insecticides on foods are exceedingly rare. Most episodes of pesticide intoxications have resulted from the misuse of pesticides, including contamination of foods during storage and transport, the use of pesticides in food preparation because of their mistaken identity as common food ingredients such as sugar and salt, and their misuse in agricultural practice on crops for which they are not intended to be used (20).
Aldicarb poisonings are one of the best examples of acute food poisoning episodes associated with pesticides. In one noteworthy episode, an outbreak of aldicarb intoxication from watermelons occurred on the West Coast in 1985 (21). The use of aldicarb on watermelons is illegal
because excessive levels of aldicarb become concentrated in the edible portion of the melon. In this incident, several farmers used aldicarb illegally, resulting in consumer illnesses and the recall and destruction of thousands of watermelons. The outbreak involved a total of 1373 illness reports with 78% classified as probable or possible aldicarb poisoning cases (21, 22). Thus, this episode is the largest known outbreak of pesticide poisoning in North America (21, 22). Aldicarb has also been involved in several other food poisoning outbreaks. These incidents were associated with ingestion of hydroponically grown cucumbers (22, 23). The symptoms of aldicarb intoxication include nausea, vomiting, diarrhea, and mild neurologic manifestations such as dizziness, headache, blurred vision, and loss of balance (21, 22, 23).
because excessive levels of aldicarb become concentrated in the edible portion of the melon. In this incident, several farmers used aldicarb illegally, resulting in consumer illnesses and the recall and destruction of thousands of watermelons. The outbreak involved a total of 1373 illness reports with 78% classified as probable or possible aldicarb poisoning cases (21, 22). Thus, this episode is the largest known outbreak of pesticide poisoning in North America (21, 22). Aldicarb has also been involved in several other food poisoning outbreaks. These incidents were associated with ingestion of hydroponically grown cucumbers (22, 23). The symptoms of aldicarb intoxication include nausea, vomiting, diarrhea, and mild neurologic manifestations such as dizziness, headache, blurred vision, and loss of balance (21, 22, 23).
Chronic intoxications resulting from pesticide residues in foods have been a long-standing concern (24). For example, DDT is a known animal carcinogen. Because of these concerns for human health and additional concerns resulting from their potential for accumulation in the environment, many organochlorine pesticides have been banned or allowed only under very restricted use conditions. Despite the concerns, the evidence that the low residual amounts of insecticides on foods present a carcinogenic risk to human consumers is not particularly strong. Several important crops including corn and potatoes have been genetically engineered for insecticide resistance. These genetically engineered, insect-resistant crops contain a novel gene that produces a naturally occurring insecticidal protein toxin from Bacillus thuringiensis (Bt). Bt toxins have been used for decades in organic agriculture. The Bt proteins produced by the genetically engineered crops have been thoroughly examined and appear to be quite safe for human consumption (25).
Herbicides
Herbicides are applied to agricultural crops to control the growth of weeds. Several classes of herbicides include chlorophenoxy compounds (e.g., 2,4-D), dinitrophenols (e.g., dinitroorthocresol), bipyridyl compounds (e.g., paraquat), substituted ureas (e.g., monuron), carbamates (e.g., propham), and triazines (e.g., simazine).
In most circumstances, herbicide residues in foods do not present any hazard to consumers. Food poisoning incidents have never resulted from the proper use of herbicides on food crops. The lack of hazard from herbicide residues is associated with the low level of exposure, their low degree of toxicity to humans and selective toxicity toward plants, and the use of many different herbicides, which limits exposure to any particular herbicide (2). Most herbicides pose little hazard to humans simply because they are selectively toxic to plants. The bipyridyl compounds are an exception. These herbicides, including diquat and paraquat, are nonselective and are toxic to humans, with effects on the lung (26). However, no food poisoning incidents have ever been attributed to inappropriate use of the bipyridyl compounds.
Fungicides
Fungicides are used to curtail the growth of molds on food crops. Important fungicide categories include captan, folpet, dithiocarbamates, pentachlorophenol, and the mercurials. The hazards from foodborne fungicides are miniscule because exposure is quite low, most fungicides do not accumulate in the environment, and fungicides typically are not very toxic (2). Several exceptions exist, including the mercurial compounds and hexachlorobenzene. Mercurial fungicides often are used to treat seed grains to prevent mold growth during storage. These seed grains are typically colored pink and are intended for planting rather than consumption. However, especially in times of famine, consumers are tempted to eat the seed grain. On several occasions, consumers have eaten these treated seed grains and developed mercury poisoning (20). Deaths have resulted in several severe episodes. More commonly, mild cases occur. Mild cases of mercury intoxication can be manifested in gastrointestinal symptoms such as abdominal cramps, nausea, vomiting, and diarrhea and dermal symptoms such as acrodynia and itching (20).
Hexachlorobenzene caused one of the most massive outbreaks of pesticide poisoning in recorded history. More than 3000 individuals were affected in this incident in Turkey from 1955 through 1959 in which seed grain was consumed rather than planted (27). Hexachlorobenzene had been used to treat the seed grain. The symptoms were severe with a 10% mortality rate, porphyria cutanea tarda, ulcerated skin lesions, alopecia, porphyrinuria, hepatomegaly, and thyroid enlargement (27).
Fertilizers
Fertilizers are typically combinations of nitrogen and phosphorus compounds. Nitrogen fertilizers are oxidized to nitrate and nitrite in the soil. Both nitrate and nitrite are hazardous to humans if ingested in large amounts (2). Infants are particularly susceptible to nitrate and nitrite intoxication (2). Fertilizers present little, if any, risk to consumers in most typical situations. However, some plants, such as spinach, can accumulate nitrate to hazardous levels if allowed to grow on overly fertilized fields (28, 29). Because nitrite is more toxic than nitrate, the situation can be worsened if nitrate-reducing bacteria are allowed to proliferate on these foods. As another example, improper storage of carrot juice has allowed the proliferation of nitrate-reducing bacteria, resulting in the accumulation of hazardous levels of nitrite in the product (30). After ingestion of low doses, the symptoms include flushing of the face and extremities, gastrointestinal discomfort, and headache; in larger doses, cyanosis, methemoglobinemia, nausea, vomiting, abdominal pain, collapse, and death can occur (26). The lethal dose of nitrite is estimated at approximately 1 g in adults (28).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree