Fallopian Tube
Anatomy, Histology, and Function of the Fallopian Tube
Approach to Examining Tubal Specimens
Bilateral Tubal Ligation for Sterilization
Salpingectomy for Tubal Ectopic Gestation
Salpingectomy (with or without Oophorectomy and/or Hysterectomy)
Introduction
As the fallopian tube is the intermediary between the ovary and the uterus, it is the seat of various interactions that culminate in a normally implanted pregnancy. Its multiple functions include conditioning of both gametes before fertilization, guiding their journeys before encounter, providing an appropriate chemical environment for fertilization, supplying nutriment to the fertilized ovum for its first few hours of life, and delivering it to the uterine cavity at the proper time for nidation. The exact processes by which these various mechanisms are accomplished are still rather poorly understood, partly because many of them vary significantly from species to species, so that the study of experimental animals does not always help to understand the situation in women. Recently, the fallopian tube has been implicated for its potential role in a more sinister pathologic process. A growing body of evidence strongly suggests that the distal fallopian tube is the site of origin for some proportion of high-grade pelvic serous cancers previously classified as primary ovarian or peritoneal malignancies. For a detailed discussion of this concept, see Chapter 25.
Anatomy, Histology, and Function of the Fallopian Tube
Anatomy
The fallopian tube is divided into four zones, which, extending distally to proximally, are the infundibulum, the ampulla, the isthmus, and the interstitial (or intramural) portions. The infundibulum is the distal end of the tube and forms the funnel-like expansion opening onto the peritoneal cavity, about 1 cm in length and diameter that ends in a variable number of irregular, fringe-like extensions, the fimbriae. Proximal to the infundibulum and making up about half of the length of the tube is the ampullary portion. The ampulla is narrower than the infundibulum and runs a tortuous course. The isthmus, which is 2–3 cm in length, has a narrower lumen and more muscular wall than the ampulla. The interstitial portion of the tube has a lumen with a simple, stellate, or almost circular cross section. In this segment of the tube, the muscle of the tubal wall merges with that of the myometrium.
Histology
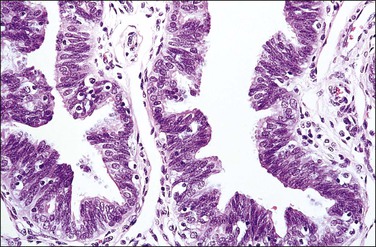
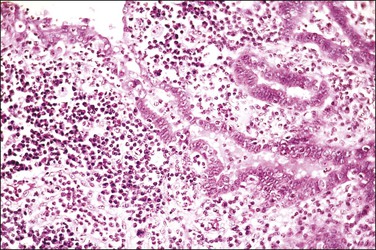
The fallopian tube is composed of a mucosal lining, a muscular layer, and an outer serosa. The mucosa consists of nonstratified epithelium and a sparse underlying fibrovascular lamina propria. The epithelium (Figure 21.1) comprises three cell types: ciliated cells, secretory cells, and intercalated (or peg) cells. The ciliated cells have a centrally placed nucleus with a perinuclear halo, a prominent terminal bar, and definable surface cilia. The nuclei of the secretory cells vary in position, depending on the stage in the menstrual cycle. The intercalated cells are secretory cells that have discharged their secretions with the result that the cell walls are collapsed around the nucleus. The ciliated component is the most prominent near the fimbrial end of the tube and the secretory cells are more numerous in the isthmus than they are in the ampulla.
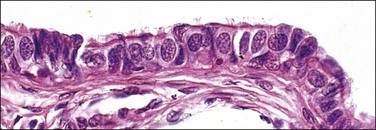
Figure 21.1 Normal tubal epithelium An admixture of secretory and ciliated cells constitute the majority of the cells.
The tubal epithelium shows well-defined histologic alterations in response to cyclic hormonal variations, which affect the height of the epithelial cells, rather than the number of cilia, as happens in other primates. In the proliferative phase of the cycle the estrogen predominance results in epithelial cells with increasing height whereas in the progesterone-dominated secretory phase, the cell height may be as little as half that seen in the first half of the cycle. Similarly, the height of both epithelial cell types is low in pregnancy. Oral contraceptives produce a similar appearance to those of pregnancy; the epithelial cells are relatively flat and show a lack of secretory activity, features that doubtless play some part in the effectiveness of the medication. In the late postmenopausal state, the epithelium becomes thin and atrophic (Figure 21.2).
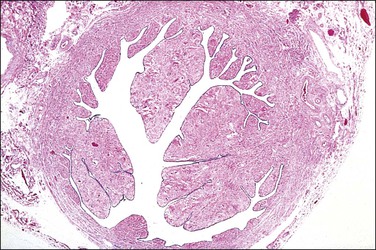
Figure 21.2 Atrophic tubal mucosa. The epithelium is thin and atrophic and the plical lamina propria is fibrotic.
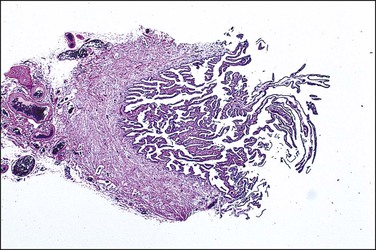
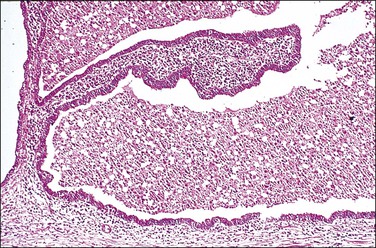
In cross section, at low-power magnification, the mucosa of the tubal ampulla (Figure 21.3) forms a complicated maze-like pattern of folds (the plicae) that branch but do not join. The epithelial surfaces of the plicae are apposed to one another so that even in the widest part of the tube the traversing ovum is not floating in a spacious lumen but is at all times nurtured by the ciliated and secretory epithelium with which it is in contact on all sides. These folds become less complex as the medial end of the tube is approached, becoming stellate in the isthmus (Figure 21.4) and forming an irregular, almost rounded, outline to the lumen in the interstitial part. In postmenopausal women, the plical stroma becomes fibrous and the plicae themselves club shaped, an appearance that may be mistaken for one sequela of infection, and the epithelium flattened.
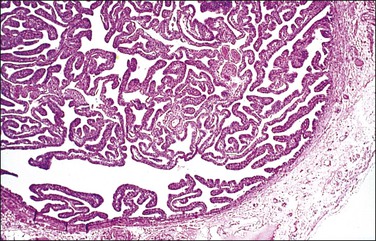
Figure 21.3 Normal ampulla. The mucosa forms a complex maze-like pattern of folds that branch but do not join.
Mucosal Epithelial Alterations
Secretory Cell Outgrowths and p53 Signatures
Expansions of morphologically unremarkable secretory cells, uninterrupted by ciliated cells, can occasionally be identified as discrete, linear foci in a background of the usual mixed ciliated and secretory cells populating the fallopian tube epithelium. Such expansions have been designated as secretory cell outgrowths (SCOUTs) when they comprise at least 30 or more secretory cells. The majority are associated with loss of expression of PAX2, a member of the pair box gene family, which is normally expressed in structures derived from the müllerian duct.1 The related p53 signature is also composed of secretory cells, but with the incremental changes of acquired p53 mutation, evidence of DNA damage, and relative predominance in the distal portion of the fallopian tube (Figure 21.5A and B).2 Neither SCOUTs nor p53 signatures exhibit significant proliferative activity (Figure 21.5C). Both are currently interpreted as early clonal expansions short of neoplastic proliferation.
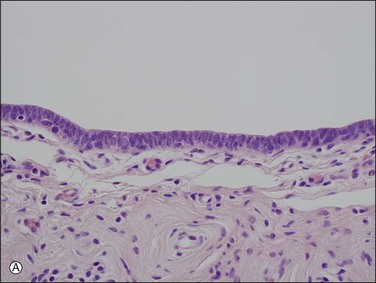
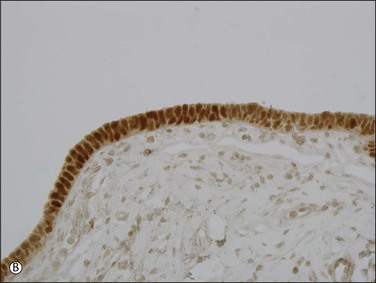

Figure 21.5 (A–C) The p53 signature is a histologically unremarkable stretch of tubal epithelium (A) that is associated with p53 mutations, exhibiting strong nuclear staining for p53 (B) and has a low MIB1 index (C).
Both SCOUTs and p53 signatures are candidate early steps toward high grade serous tubal carcinoma, as discussed in Chapter 25. The reader is cautioned, however, that the prospective cancer risk of women with isolated SCOUTs and/or p53 signature is unknown, so neither is a diagnostic entity that should be invoked in clinical practice. Fallopian tubes from women with high-grade pelvic serous carcinoma harbor a significantly higher frequency of SCOUTs than those from women whose tubes were removed for a non-malignant condition.1 Tubes in which serous tubal intraepithelial carcinoma is present are more likely to contain p53 signatures than those without; in some cases, a p53 signature may be found in direct continuity with intraepithelial carcinoma. In a given case of pelvic serous carcinoma, shared p53 mutations may be identified among p53 signatures, serous tubal intraepithelial carcinoma, and invasive tumor elsewhere in the pelvis.2–5
Other Mucosal Epithelial Proliferations
Another notable pattern of epithelial growth in the fallopian tube, which does not appear to be associated with malignancy, either in the tube or elsewhere, is characterized by an apparent ‘hyperplasia’ of the normal mucosal epithelial cells. Such areas are populated by a mix of ciliated and secretory cells, which appear stratified or are arranged as small tufts. These areas may be focal or more diffuse. The cells themselves may exhibit some nuclear atypia, but not to the degree found in intraepithelial carcinoma. Mitotic activity is generally minimal (Figure 21.6). This mucosal alteration may be seen in the setting of pregnancy, but can be found in the tubes of non-pregnant women as well.
Function
Ovum Transport
The ciliary action in the tube is toward the uterus and is under the influence of many mediators. Physiologic levels of prostaglandins F2α, E1, and E2 stimulate the ciliary activity as do β-adrenergic agonists; the latter effect is potentiated by estrogen and progesterone. Recent data have shown that progesterone affects the tubal ciliary beat frequency.6 Incubation with progesterone suppresses the beat frequency by 40–50% but estradiol has no effect. Cilia from the tubal ampulla beat significantly faster than those from fimbrial segments.
Approach to Examining Tubal Specimens
Bilateral Tubal Ligation for Sterilization
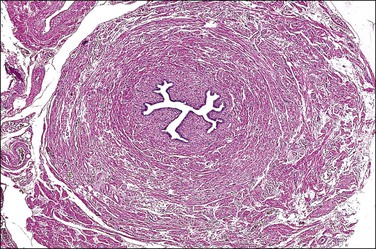
The most important aspect in the sectioning and preparation of the excised specimen for microscopic examination is that sufficient sections are made to show the complete cross section of the tube (Figure 21.7). An efficient method is to cut the fallopian tube into sections no more than 1–2 mm thick so that a single slide contains up to six sections. In this manner, even if some sections are embedded improperly or cut on a bias, at least a few should show the complete cross section, if in fact it is present. Any case where none of the pieces shows a complete cross section should be reported as incomplete (Figure 21.8). This can occur when the surgeon has removed arteries and fascia or only the tubal fimbriae. In these cases, it commonly happens that the clinician had difficulty definitively identifying the fallopian tube, even though this information was not transmitted to the pathologist. It is most important that an incomplete ligation be clearly reported in part for medicolegal consideration. The pathologist must be careful not to interpret a paratubal cyst as fallopian tube lumen; the latter has a much thicker wall with ample smooth muscle.
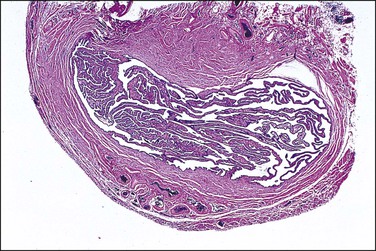
Figure 21.7 Sterilization procedure, complete transection. This section of the tube shows the complete circumference of muscular wall surrounding the mucosa, confirming complete transection of the tube.
Salpingectomy (with or Without Oophorectomy And/or Hysterectomy)
The SEE-FIM Protocol
The majority of early serous carcinomas in the fallopian tube are detected microscopically rather than by gross examination, in the distal portion of the fallopian tube, in the fimbriae. Salpingectomy performed in high-risk patients, such as those undertaken prophylactically in BRCA mutant women, are a common setting for clinically and grossly occult disease. In an effort to facilitate a thorough examination of the distal fallopian tube a protocol for sectioning and extensively examining the fimbriated end (SEE-FIM) was developed (see also Chapter 35).6 According to this protocol, the entire fallopian tube is first well fixed to prevent exfoliation of the mucosal epithelial cells. The fimbriae are then amputated from the proximal tube and sectioned longitudinally into multiple sections to permit maximum exposure of the fimbrial mucosa. The remainder of the tube is cross sectioned at 2−3 mm intervals and the entire tube is submitted for histopathologic review (Figure 21.9).
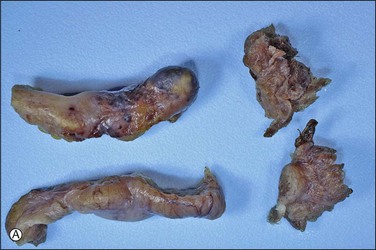
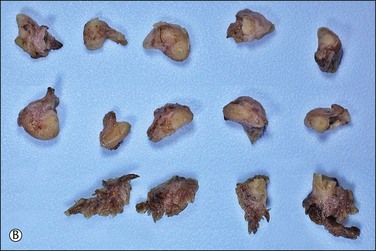
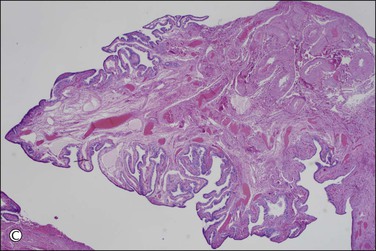
Figure 21.9 (A-C) The protocol for sectioning and extensively examining the fimbriated end (SEE-FIM protocol). Tubes are first fixed. The fimbriae are then amputated from the rest of the tube (A), sectioned longitudinally into multiple sections (B), and then submitted with the remainder of the tube, which has been cross sectioned at 2–3 mm intervals. Longitudinal sectioning permits maximum evaluation of the fimbrial mucosal epithelium (C, low-power magnification).
Indications for extensive tubal sampling are not yet clearly defined by clinical necessity, but should be considered in cases where it is desirable to maximize the likelihood of detecting early tubal cancer. Appropriate specimen types might include risk-reducing (prophylactic) salpingectomies; salpingectomies from women with a history of breast cancer; and salpingectomies in the setting of uterine, ovarian, or peritoneal disease. Some consensus is emerging from within the pathology community, with, for example, the Association of Directors of Anatomic and Surgical Pathology recommending an approach similar to SEE-FIM in prophylactically removed specimens, with an emphasis on longitudinal sectioning of the fimbriae.7 This group also recommends sampling fimbriae as part of processing of routine tubal specimens.
Given that the distal fallopian tube is the site most likely to harbor occult tubal malignancy, the suggestion has been made by some authors to submit the entire fimbriae for histopathologic review even in cases in which tubes are removed for benign disease.8 Currently, the incidence of occult carcinoma in the fallopian tube in the general population is unknown. However, with increasing attention focused on the distal tube, incidental tubal cancers are being discovered in cases otherwise unsuspected of harboring a malignancy.8 A standard clinical response to incidentally discovered occult in situ tubal cancer has not yet been formulated, but there is a possibility for identification of individuals who may benefit from increased surveillance, or even early disease intervention.
Non-Neoplastic Lesions
Inflammation of the Fallopian Tubes
Inflammatory disease resulting from infection of the fallopian tubes and adjacent ovary is an increasing problem. The investigation and treatment of women with the disease is demanding more and more in the way of time and other resources. Identification of the disease early in its natural history is important to enable treatment to be effected before the damage becomes extensive and the consequent surgery destructive.
Non-Granulomatous Salpingitis
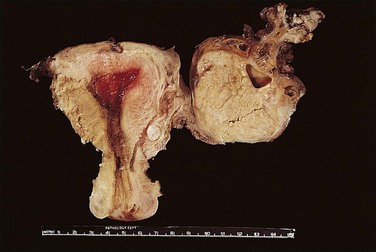
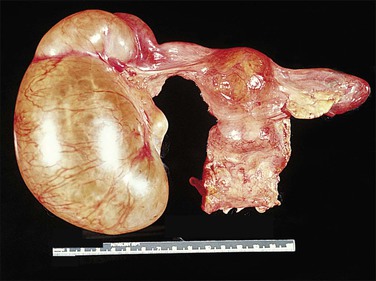
Non-granulomatous salpingitis is predominantly a disease of young, sexually active women, and 70% of those with the disease are under the age of 25. Other factors that have an influence on the development of salpingitis include the method of contraception, induced abortion, and instrumentation of the cervix. Among the infectious organisms responsible for salpingitis, Chlamydia trachomatis and Neisseria gonorrhoeae remain of paramount importance and are responsible for ascending salpingitis.9 Other causative microbial agents are the anaerobic bacteria (bacteroides, clostridia, and streptococci), Mycoplasma hominis, and Ureaplasma urealyticum, and miscellaneous organisms such as Haemophilus influenzae and group A streptococci. Bacterial vaginosis is a common concurrent disorder of women with acute salpingitis, and bacterial vaginosis microorganisms are commonly isolated from the upper genital tracts of patients with pelvic inflammatory disease.9 Salpingitis caused by Actinomyces israelii is associated with the presence of an intrauterine contraceptive device, and may result in the development of a tubo-ovarian abscess (Figure 21.10). Actinomyces-like organisms can be identified in the pus (Figure 21.11). In practice, however, microbiologic investigations often show that the cultured material is already sterile by the time of the investigation, or else there is a combination of organisms.
The spread of etiologic organisms from the lower to the upper genital tract is canalicular, through the cervical canal and endometrial cavity and then into the fallopian tubes. Blockage of this route, either by cornual resection of the fallopian tubes or by sterilization, reduces the risk of salpingitis.10 Salpingitis begins as a mucosal rather than a serosal infection.
Microscopic Features
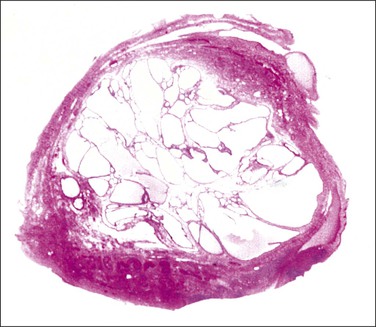
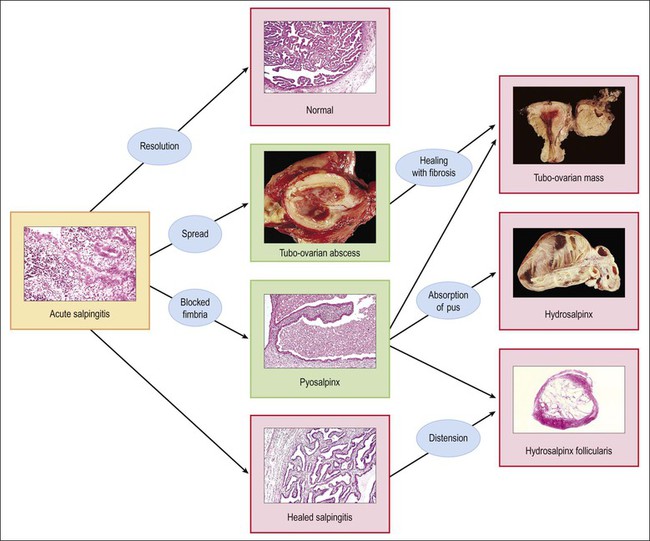
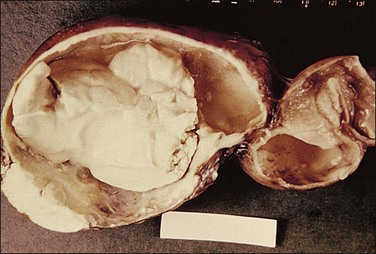
The plicae are greatly swollen and densely infiltrated by neutrophils (Figure 21.12). The epithelial cells soon lose their cilia and the epithelium is shed in severe disease. The lumen contains pus (Figure 21.13). As the disease progresses to chronic salpingitis (Figures 21.14 and 21.15), the inflammatory infiltrate consists predominantly of plasma cells and then lymphocytes. The progression from acute to chronic non-granulomatous salpingitis may take several courses. If the fimbrial end of the tube remains patent, a chronic interstitial salpingitis may ensue, in which case the tube is thickened and the plicae fuse to form epithelial-lined cysts, which are the hallmark of chronic salpingitis. ‘Follicular salpingitis’ (Figure 21.16) rather confusingly refers to the epithelium-lined spaces rather than lymphoid collections. The inflammatory process may eventually become quiescent, leaving only the architectural sequelae of chronic salpingitis (Figure 21.17). Severe inflammation in the tube may spread to the adjacent ovary, resulting in a tubo-ovarian abscess (Figure 21.18). Occlusion of the fimbrial end of the tube prevents release of the tubal contents, so that a pyosalpinx may result (Figure 21.19). In pyosalpinx, the lumen is filled with pus (Figure 21.20) and the wall is thinned, although the attenuation is commonly less marked than in hydrosalpinx. Acute and chronic inflammatory cells infiltrate the plicae and wall. As the exudate is reabsorbed into the tubal wall concurrently with the quiescence of the inflammatory process, clear fluid replaces this pus and a hydrosalpinx results (Figures 21.21 and 21.22). In hydrosalpinx, the epithelium is generally thin and non-ciliated, although a few areas of morphologically normal cells may be seen. The wall of a hydrosalpinx is markedly thinned, with attenuation of smooth muscle and plicae. Usually, by this advanced stage in the disease process, there is little, if any, residual inflammatory infiltrate in the tissues of the wall. From a practical point of view, hydrosalpinx with tubo-ovarian adhesions is commonly misdiagnosed as ovarian ‘serous cystadenoma’ since it commonly presents as an adnexal mass. A careful microscopic examination identifying the muscular layer is diagnostic for hydrosalpinx. A small hydrosalpinx in which there is plical conglutination will have a ‘honeycomb’ cut surface and is termed a ‘hydrosalpinx follicularis’ (Figure 21.23). The relations among these inflammatory changes are shown in Figure 21.24.
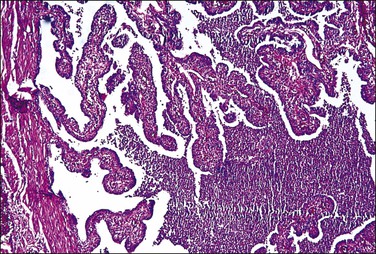
Figure 21.13 Acute salpingitis. The lumen is filled with pus and the plicae are engorged and inflamed.
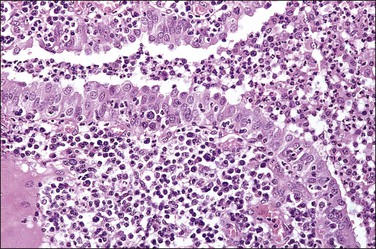
Figure 21.14 Chronic salpingitis. In this early stage of the disease, the plicae are infiltrated by plasma cells and lymphocytes and there is pus in the lumen. The epithelium is intact, although containing inflammatory cells.
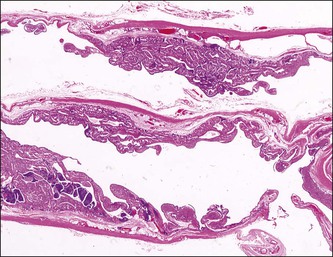
Figure 21.15 Chronic salpingitis. The infiltrate is now predominantly lymphocytic, with germinal centers.
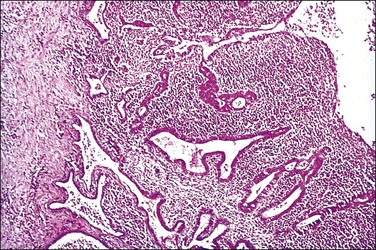
Figure 21.16 Chronic salpingitis. The plicae are partly fused, forming separate epithelium-lined spaces or ‘follicles’ separate from the central lumen (‘follicular salpingitis’). Inflammation is still active.
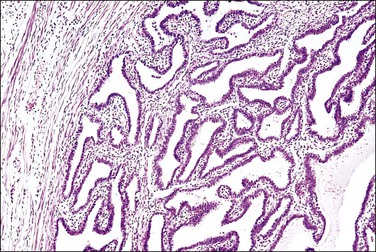
Figure 21.17 Chronic salpingitis, healed. The inflammation has subsided and is near absent, but the plicae remain fused by fibrosis.
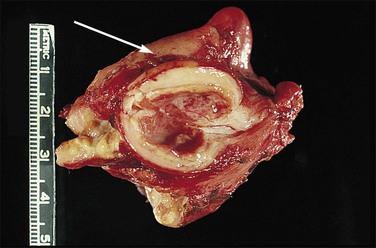
Figure 21.18 Tubo-ovarian abscess. The central mass is the ovary, which contains an abscess. Its cavity communicates with pus in the lumen of the tube (arrow).
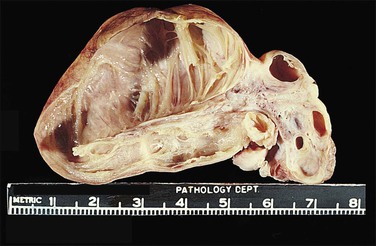
Figure 21.22 Hydrosalpinx. The convoluted shape (often called ‘retort shaped’) of the tube is seen on section.
Granulomatous Salpingitis
Granulomatous salpingitis is nearly always tuberculous in origin. All age groups may be affected but the pattern of the disease has changed over the last few decades. Until the 1970s, tuberculosis of the female genital tract in the developed world affected mainly women of childbearing age but more recently the majority of cases are in postmenopausal women.11 Whereas previously the main complaint of these women was infertility, tuberculosis accounting for about 40% of all cases of infertility,12 the common symptoms are now pain and bleeding.11 When tuberculosis affects the female genital tract, the tube is affected in nearly all cases and involvement of the endometrium is always secondary to it. The pelvic disease is, in turn, secondary to primary disease in the lungs or bowel, spreading to the tubes by hematogenous and lymphatic routes, respectively.
Gross Features
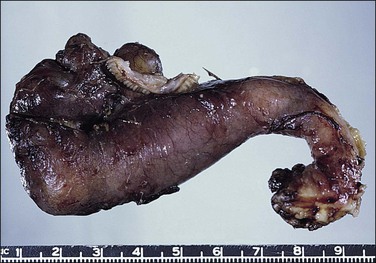
Tuberculous salpingitis is nearly always bilateral. The tube is thickened and congested and there are serosal adhesions. The fimbrial end of the tube is usually patent and the lumen may contain caseous debris (Figure 21.25). The wall is thickened and foci of caseation may be recognized within the tissue of the wall.
Microscopic Features
The hallmark of tuberculous salpingitis histologically is the epithelioid cell granuloma that is situated in the lamina propria of the plicae (Figures 21.26 and 21.27) and, rarely, within the muscular wall. Caseation may or may not be present, but is more often seen in older women. A surrounding, dense lymphocytic infiltrate is found, both in the plical lamina propria and in the muscle, the latter is usually more conspicuous. A frequent finding is the presence of striking epithelial proliferation of the endosalpinx, a feature that may cause confusion with carcinoma (see later). Schaumann bodies are occasionally seen in tuberculous salpingitis (Figure 21.26). Although typically associated with sarcoidosis, these rounded, concentrically laminated calcified bodies may be seen in most forms of epithelioid cell granuloma. The histologic suggestion of tubal tuberculosis is confirmed if acid-fast bacilli are found in the sections, but this is achieved in only 1% of cases in which the culture proves positive. On a practical basis, staining is not useful. With the development of molecular pathology, polymerase chain reaction-based tuberculosis-specific bacilli DNA can be detected in 48 hours.13 The differential diagnosis of tuberculous salpingitis in Europe is sarcoidosis, Crohn disease, and foreign-body granuloma. Worldwide, other conditions include schistosomiasis, blastomycosis, coccidioidomycosis, histoplasmosis, and enterobiasis. A positive tissue diagnosis of tuberculosis is often not possible and the decision to treat the patient must rest on the degree of clinical suspicion.
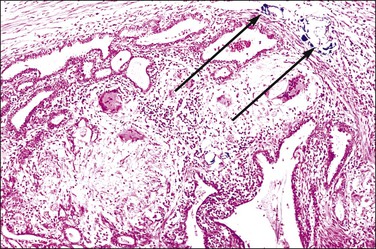
Figure 21.26 Tuberculous salpingitis. Epithelioid cell granulomas are present with giant cells. Schaumann bodies are also seen (arrows).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree