Problem 8.2 In the case study we have seen how an electrocyclization reaction of an enediyne in calicheamicin generates a reactive aryl diradical. Suggest a mechanism for the cyclization of a simple enediyne to a benzene diradical using fishhook arrows to keep track of the movement of electrons.

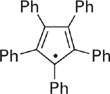
Problem 8.3 Shown below is a radical that exhibits remarkable stability. It is stable in its solid form, even in the presence of oxygen. Provide an explanation for why this particular radical is so unreactive.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


