Endometrial Stromal Tumors, Mixed Müllerian Tumors, Adenomyosis, Adenomyomas and Rare Sarcomas
Endometrial Stromal Tumors
Introduction
Even though endometrial stromal tumors represent the second most common category of mesenchymal tumors of the uterus, they are rare and account for less than 1% of all uterine tumors.1 Endometrial stromal and related tumors are defined by the latest classification into the following four main categories based on resemblance to (or lack of) proliferative-type endometrial stroma: (1) endometrial stromal nodule, (2) low-grade endometrial stromal sarcoma, (3) high-grade endometrial stromal sarcoma and (4) undifferentiated endometrial sarcoma.2
Endometrial Stromal Nodule
Definition
This is a benign endometrial stromal tumor characterized by a well-delineated, expansile margin and composed of neoplastic cells that resemble proliferative phase endometrial stroma supported by a large number of small, thin-walled arteriolar vessels. Small irregularities in the form of finger-like projections or satellite islands ≤3 mm in maximum dimension and <3 in number are allowed.2
Clinical Features
Endometrial stromal nodule is the least common of all endometrial stromal tumors. Patients range widely in age (23–86 years) and they often present with abnormal vaginal/postmenopausal bleeding or enlarged uterus.3,4 However, not infrequently, it is an incidental finding in patients who had a hysterectomy for ‘leiomyomas.’
Gross Features
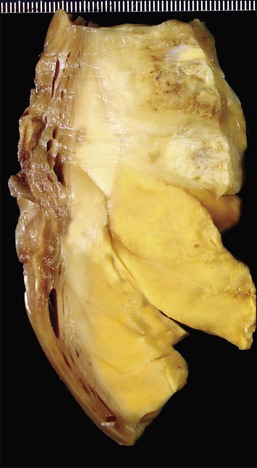
On gross examination, these tumors are centered in the endometrium forming polypoid masses that fill the uterine cavity but almost as frequently they are centered in the myometrium. They range in size from <1 to 22 cm; are well demarcated from the surrounding myometrium; and typically display a fleshy, homogeneous, yellow to tan cut surface (Figure 20.1). Cyst formation (more commonly centrally located) as well as areas of necrosis and/or hemorrhage can be encountered (Figure 20.1).3,4 Rarely they may be predominantly cystic.5
Microscopic Features
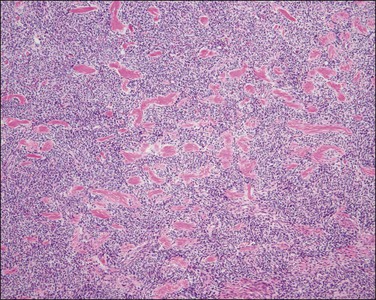
By definition these tumors should have a well-demarcated non-infiltrative margin with the surrounding myometrium (Figure 20.2). However, small irregularities in the form of finger-like projections or satellite islands ≤3 mm in maximum dimension and <3 in number are allowed.3,6 The presence of lymphovascular invasion in a well-circumscribed endometrial stromal tumor excludes the diagnosis of endometrial stromal nodule.2
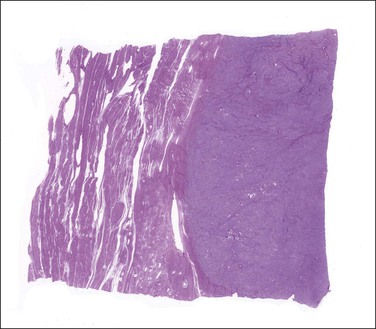
Figure 20.2 Endometrial stromal nodule. A smooth and well-circumscribed border with the surrounding myometrium is characteristic of this tumor.
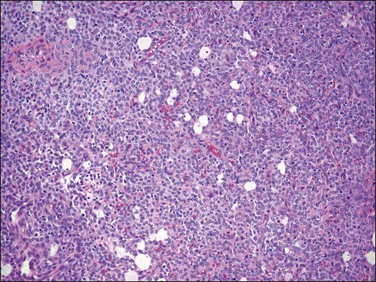
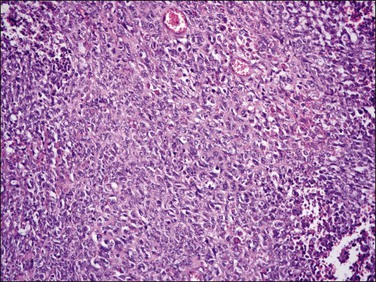
The tumor is composed of a diffuse proliferation of monotonous ‘blue’ cells that resemble proliferative phase endometrial stroma (Figure 20.3). Most of these tumors (if conventional morphology) are densely cellular. The neoplastic cells are small with scant cytoplasm and oval to round nuclei with small nucleoli (minimal cytologic atypia) and variable (sometimes brisk, up to 24/10 HPF) mitotic activity. No atypical mitoses are identified. They have a characteristic network of ‘arteriole-like’ vessels around which neoplastic cells often whorl, but this feature is only striking in less than one-third of tumors (Figure 20.3). Prominent hyalinization of the vessels as well as thin ectatic blood vessels can be seen. In contrast, large thick blood vessels are uncommon; and when present are typically seen close to the tumor–myometrium interface, probably representing entrapped pre-existent vessels. Another characteristic feature is the finding of collagen bands or plaques distributed randomly within the tumor (Figure 20.4). Foamy histiocytes single or in clusters may be present throughout. Infarct-type necrosis can be seen and may result in confluent hyalinization. Cysts, if present, often form secondary to necrosis or hemorrhage. They are lined by stromal cells that are often admixed with histiocytes and may be associated with cholesterol clefts.3
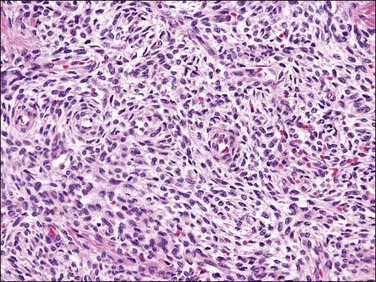
Figure 20.3 Endometrial stromal nodule. The tumor cells are small (‘blue’) with scant cytoplasm and oval to round nuclei that sometimes whorl around arterioles. The microscopic appearance overlaps with that of low-grade endometrial stromal sarcoma.
Low-Grade Endometrial Stromal Sarcoma
Definition
Tumor composed of cells resembling those of proliferative phase endometrial stroma often associated with numerous arteriolar-like vessels that shows a permeative infiltrative growth (‘tongue-like’) into the myometrium with or without associated lymphovascular invasion.2
Clinical Features
Among uterine sarcomas, which constitute between 2% and 5% of all uterine malignancies, low-grade endometrial stromal sarcomas account for approximately 0.2–1% of all uterine malignancies and 6–20% of all uterine sarcomas, representing the second most common uterine sarcoma.1 These tumors tend to occur in premenopausal and perimenopausal women with a median age of 52 years (wide range) and do not have race predilection.5,7 Most patients present with nonspecific signs or symptoms, including vaginal bleeding (most common), pelvic pain, and/or dysmenorrhea.5
Gross Features
Low-grade endometrial stromal sarcomas are typically poorly defined intracavitary and/or intramyometrial often confluent multinodular masses with a homogeneous soft tan to yellow or less commonly white cut surface. As occurs with endometrial stromal nodules, cyst formation as well as areas of necrosis and/or hemorrhage can be seen. However, in contrast to endometrial stromal nodules, intravascular (typically intravenous) worm-like plugs of tumor are variably present within the myometrium and para-uterine soft tissues (the latter easier to identify).5,8–10 Of note, some low-grade endometrial stromal sarcomas display deceptively well-circumscribed borders due to very limited myometrial invasion that cannot be noticed on gross examination, closely mimicking the appearance of an endometrial stromal nodule. Thus, careful sampling of the tumor–myometrium interface is necessary in order to identify these areas and to establish a diagnosis of low-grade endometrial stromal sarcoma.3
Microscopic Features
Low-grade endometrial stromal sarcomas show an irregular interface with the myometrium with a tongue-like pattern of infiltration where irregularly shaped islands of tumor cells are unassociated with a stromal response (Figures 20.5 and 20.6). On high-power examination, the cytologic features are identical to those encountered in endometrial stromal nodules. Tumors are very cellular (‘blue’) and cells typically grow in sheets or vaguely storiform pattern; they have scant cytoplasm and uniform, oval to spindle-shaped nuclei with very small nucleolus (Figure 20.3). The neoplastic cells may focally whorl around small vessels reminiscent of the arterioles seen in proliferative phase endometrium. By definition, significant degrees of nuclear atypia are absent and mitotic rate is usually <5/10 HPF but higher mitotic activity does not exclude this diagnosis. It is important to remember that tumors with typical morphology and characteristic tongue-like growth should be classified as low-grade endometrial stromal sarcoma regardless of their mitotic counts as the latter does not impact prognosis in this category of tumors. Arteriolar walls may be thickened due to hyalinization. Sometimes vessels may be thin and elongated, with a hemangiopericytoma-like morphology; however, thick blood vessels are uncommon and when present are located at the periphery of the tumor islands (Figure 20.7), likely being entrapped. Histiocytes (Figure 20.8), hyaline bands or plaques, and cholesterol clefts often next to areas of necrosis or cyst formation, as seen in endometrial stromal nodules, can also be noticed.5,8–10
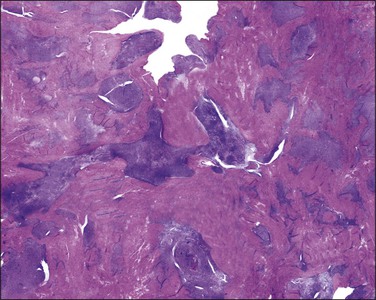
Figure 20.5 Low-grade endometrial stromal sarcoma. The tumor shows a diffuse permeative infiltrative growth.
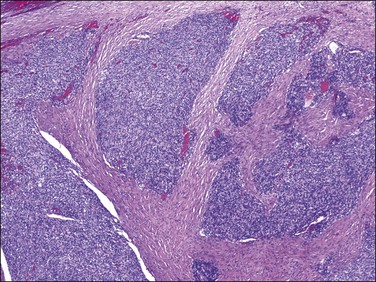
Figure 20.6 Low-grade endometrial stromal sarcoma. The tumor cells form irregular islands that infiltrate the myometrium without an associated desmoplastic response.
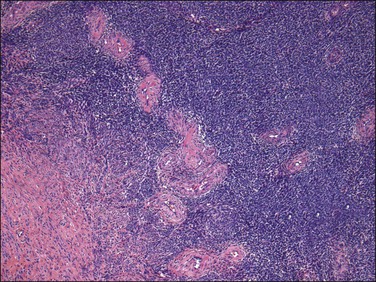
Figure 20.7 Low-grade endometrial stromal sarcoma. Thick blood vessels, most likely entrapped, are seen at the periphery of the tumor islands.
Endometrial Stromal Variants
Both endometrial stromal nodule and endometrial stromal sarcoma display the following types of differentiation (Table 20.1):
1. Smooth muscle: this type of differentiation has been reported in the literature based on morphologic, immunohistochemical, and ultrastructural studies.5,11–16 As prognosis of these tumors relates to margin status of the endometrial stromal component, currently these tumors are defined as endometrial stromal neoplasms (nodule or sarcoma) in which the smooth muscle component accounts for >30% on H&E evaluation. On gross examination, the smooth muscle component may be seen as whitish and firm areas (if prominent) in contrast to the endometrial stromal component (soft and yellow-tan to light brown). On microscopic examination, smooth muscle differentiation can be seen as pale to ‘pink’ irregular islands of slightly epithelioid cells or much more characteristically as the so-called ‘starburst’ pattern present in a background of endometrial stromal neoplasia (Figure 20.9). The latter is characterized by a central area of hyalinization from which collagen bands radiate toward the periphery and cells with an ‘epithelioid’ appearance are embedded in collagen fibers. These epithelioid cells form short and nonorganized fascicles of smooth muscle that in turn may transition into long and well-formed fascicles.16 These starburst areas are often seen at the periphery of the tumor and they may also be present next to areas of ‘sex cord-like’ differentiation.17 The smooth muscle cells are typically cytologically benign but rarely a malignant smooth muscle component has been reported.16
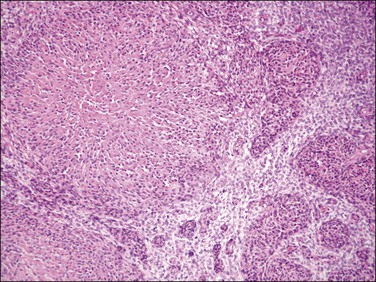
Figure 20.9 Low-grade endometrial stromal sarcoma with smooth muscle differentiation. A nodule of smooth muscle differentiation shows a central area of hyalinization with bands of collagen radiating toward the periphery where ‘plump’ cells are embedded (starburst pattern), which in turn form small fascicles of smooth muscle in a background of endometrial stromal neoplasia.
2. Myxoid and fibroblastic: these unusual types of differentiation are characterized either by prominent myxoid or by fibroblastic (loose collagen) background (Figures 20.10A and B, and 20.11) imparting a hypocellular appearance to the tumors in contrast to their typical hypercellular nature.18,19 On gross examination, they can have a gelatinous (if myxoid) or firm and white (if fibroblastic) cut surface. However, other characteristic morphologic features of endometrial stromal neoplasia are present including the typical ‘tongue’ pattern of infiltration if low-grade sarcoma and arteriole-like vessels (Figure 20.10A and B). Cells are small with scant cytoplasm and oval to spindle shaped with minimal cytologic atypia and mitotic activity. Areas of conventional endometrial stromal neoplasm may be seen. Noteworthy, a fibroblastic component has been reported in approximately 50% of endometrial stromal tumors with t(10;17), which are known to have a predominant high-grade round cell component and aggressive behavior (Figures 20.12 and 20.13).20
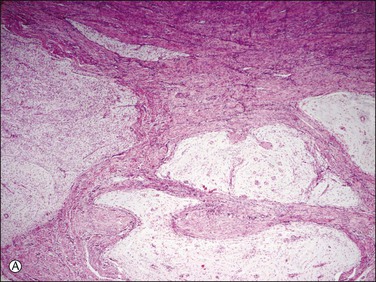
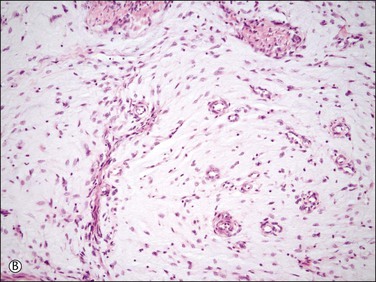
Figure 20.10 Low-grade endometrial stromal sarcoma with myxoid change. The tumor is strikingly hypocellular in contrast to conventional endometrial stromal sarcoma. However, it shows the typical ‘tongue-like’ pattern of infiltration (A) as well as the characteristic arterioles (B).
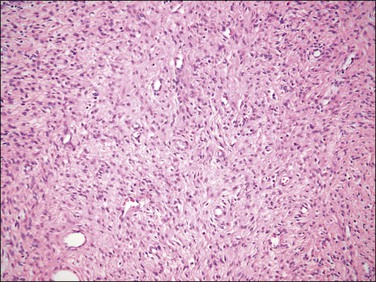
Figure 20.11 Endometrial stromal nodule with fibroblastic differentation. The tumor shows a prominent loose collagenous background.
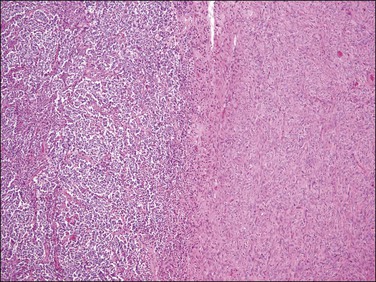
Figure 20.12 t(10;17) high-grade endometrial stromal sarcoma. Tumor cells with an epithelioid morphology (left) are juxtaposed to areas of endometrial stromal neoplasia with a fibroblastic appearance.
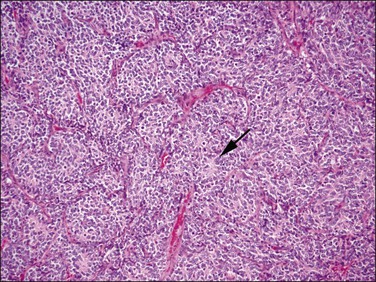
Figure 20.13 t(10;17) high-grade endometrial stromal sarcoma. The epithelioid areas are composed of cells with high-grade cytologic features and brisk mitotic activity. Pseudorosettes may be seen (arrow).
3. Sex cord-like elements have been reported with a variable frequency and extent in endometrial stromal tumors (up to 60%).3,5,13,21–27 They consist of anastomosing cords, trabeculae, islands, small nests, tubules that may be retiform, or sheets of cells reminiscent of the patterns seen in granulosa and/or Sertoli cell tumors of the ovary (Figure 20.14). Cells typically show scant to abundant cytoplasm and round to oval nuclei, sometimes with grooves, indistinct or tiny nucleoli, and minimal mitotic activity. These sex cord-like elements are present within a background of typical endometrial stromal neoplasia or in a loose hypocellular background. Not infrequently they coexist with areas of smooth muscle differentiation, and in these instances both elements often merge imperceptibly with one another.17
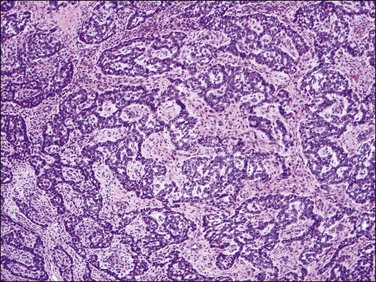
Figure 20.14 Low-grade endometrial stromal sarcoma with sex cord-like elements. Inter-anastomosing cords and trabeculae of ‘epithelial-like’ cells with architecture reminiscent of that of a granulosa cell tumor of the ovary are present in a background of neoplastic endometrial stromal cells.
4. Glandular elements may vary in number, but can be quite extensive. They can be seen in endometrial stromal nodules4 but have been reported more frequently in endometrial stromal sarcomas.15,28,29 Glands range from small and round (proliferative) to slightly irregular or cystically dilated (inactive). They have an endometrioid morphology, lined by cuboidal to columnar cells with eosinophilic and rarely clear cytoplasm and pseudostratified nuclei, which display a minimal degree of cytologic atypia in most cases (Figure 20.15).4,15,28,29 Foci of grade I endometrioid carcinoma have been occasionally reported.28
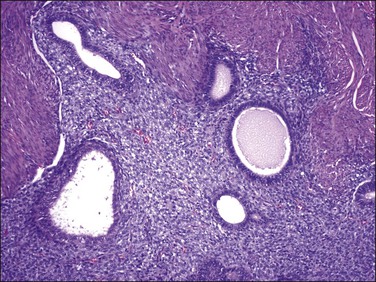
Figure 20.15 Low-grade endometrial stromal sarcoma with müllerian glandular elements. Benign endometrioid-type glands, with a slightly proliferative appearance, are present in an island of tumor.
5. Skeletal muscle may be seen as large cells with copious bright eosinophilic cytoplasm and abundant filament deposits typically wrapping around nuclei as well as cells having a strap-shaped morphology with cross striations. These cells are positive for myoglobin, myo-D1, and myogenin.14,30
6. Epithelioid appearance is defined as cells having abundant eosinophilic cytoplasm that confers an oval to polygonal appearance to the cells. This morphologic feature may be extensive within a given endometrial stromal tumor (up to 90% of cells).3,31 Some of these cells may also have a granular quality of the cytoplasm (Figure 20.16).31 Cells have bland cytologic features in contrast to the epithelioid cells seen in t(10;17) endometrial stromal tumors. This appearance has to be also distinguished from pseudodecidualization of neoplastic stromal cells that can occur in these tumors.
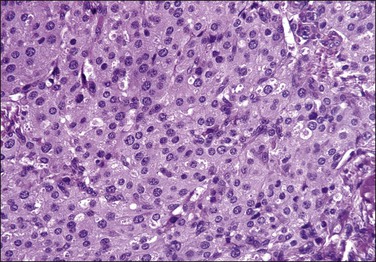
Figure 20.16 Low-grade endometrial stromal sarcoma with epithelioid cells. In contrast to the t(10;17) high-grade endometrial stromal sarcoma, the tumor cells have abundant eosinophilic granular cytoplasm and uniform round nuclei with no cytologic atypia.
7. Rhabdoid morphology can be seen in endometrial stromal tumors more frequently in areas displaying sex cord-like differentiation. Cells characteristically have large eosinophilic hyaline cytoplasmic inclusions and eccentric vesicular nuclei often with prominent nucleoli.25,26 By electron microscopy, abundant paranuclear deposits of intermediate filaments have been noted.25
8. Cells with abundant clear cytoplasm may rarely be seen,32 adding to the differential diagnosis of uterine mesenchymal tumors with clear cells.
9. True papillae and pseudopapillae have been reported in endometrial stromal tumors, mostly endometrial stromal sarcomas, either primary or metastatic, and may be a focal or diffuse finding. Papillae or pseudopapillae are typically small with angulated outlines and may have a glomeruloid appearance. They are lined by stromal cells, which are also present within the vascular cores associated with small vessels (Figure 20.17).33
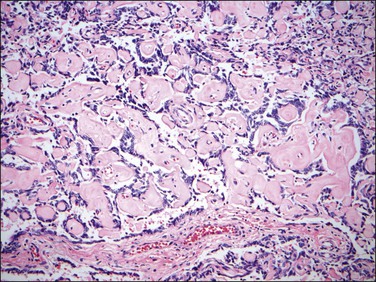
Figure 20.17 Low-grade endometrial stromal sarcoma with pseudopapillae. The tumor shows non-cohesive neoplastic endometrial stromal cells associated with abundant collagen imparting a pseudopapillary architecture.
10. Mature adipose tissue can occasionally be seen admixed with the neoplastic endometrial stromal cells.30
11. Cells with bizarre nuclei as noted more commonly in benign smooth muscle tumors (leiomyomas with bizarre nuclei) can rarely occur. They may be mononucleated or multinucleated and typically have nuclei with smudged chromatin and occasional intranuclear cytoplasmic inclusions.30,31,34
12. Osteoclast-type cells have also been reported in one endometrial stromal sarcoma.35
Immunohistochemical Features
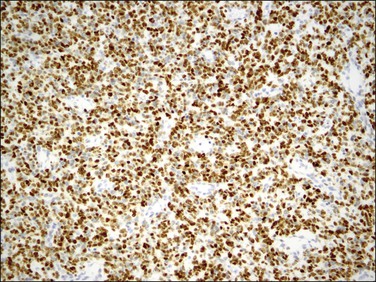
Endometrial stromal tumors similar to normal endometrial stroma are surrounded by a network of reticulin fibers that can be highlighted by a reticulin stain. These tumors are also diffusely positive for vimentin, muscle-specific and smooth muscle actin, and often for keratins.36,37 Areas of conventional endometrial stromal neoplasia show variable positivity (up to 30%) for desmin. Smooth muscle differentiation and sex cord-like elements are extensively positive for desmin and often for h-caldesmon, histone deacetylase (HDAC-8), and smooth muscle myosin (Figure 20.18).11,13,18,37–40 CD10 is a very helpful marker in the diagnosis of endometrial stromal tumors as they typically display diffuse and strong immunoreactivity (Figure 20.19).40,41 However, some tumors may only be weakly and focally positive or completely negative as typically occurs in t(10;17) endometrial stromal tumors20 (Figure 20.20) and undifferentiated endometrial sarcomas of pleomorphic type.42 CD10 can also be positive in smooth muscle and sex cord-like areas.40
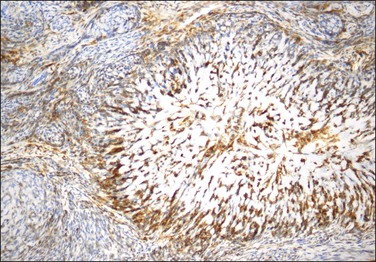
Figure 20.18 Low-grade endometrial stromal sarcoma with smooth muscle differentiation. Desmin immunoreaction highlights the smooth muscle component (‘starburst’) with minimal reaction of the background endometrial stroma.
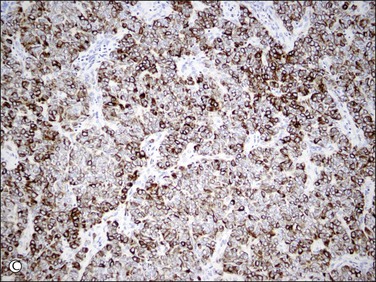
Figure 20.19 Low-grade endometrial stromal sarcoma. The infiltrating islands of tumor display diffuse and strong immunoreaction for CD10.
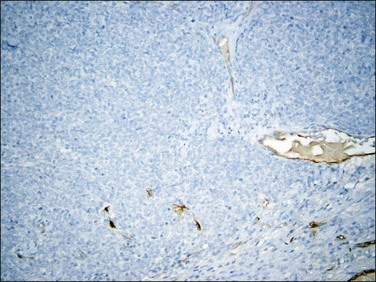
Figure 20.20 t(10;17) high-grade endometrial stromal sarcoma. CD10 staining is completely negative in the cytologically high-grade epithelioid component.
Endometrial stromal tumors express estrogen (ER) and progesterone (PR) receptors.43,44 In contrast to normal endometrial stroma (ER-α and ER-β positive), endometrial stromal sarcoma cells typically express ER-α (involved in cellular proliferation) but not ER-β (involved in apoptosis). PR-A is overexpressed over PR-B in neoplastic endometrial stromal cells except in recurrences where PR-B expression is higher than PR-A.45 The majority of endometrial stromal sarcomas with high-grade cytologic features, including endometrial stromal sarcomas with t(10;17) and undifferentiated endometrial sarcomas (especially if pleomorphic), are ER and PR negative (Figure 20.21).20,45–48 Low-grade endometrial stromal sarcomas may also express androgen receptors with rare undifferentiated endometrial sarcomas being also positive.49 Aromatase, which participates in extraovarian estrogen production, is expressed in endometrial stromal sarcomas, most frequently if high stage.50
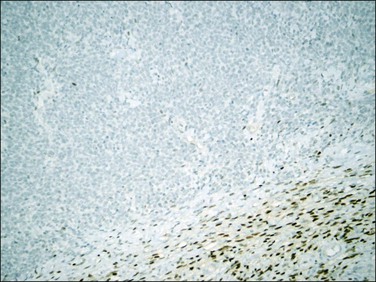
Figure 20.21 t(10;17) high-grade endometrial stromal sarcoma. The tumor cells are negative for ER in contrast to the adjacent fibroblastic component (ER positive).
WT1 is positive in non-neoplastic endometrial stroma as well as in endometrial stromal tumors.51 Sex cord–stromal markers including inhibin, calretinin, CD99, and Melan-A (also known as Mart-1) can be positive in areas of epithelial (sex cord-like) differentiation.21,24
c-Kit has been reported to be positive in a small percentage of endometrial stromal tumors, but no mutation has been found.52 It appears that c-kit may be more often positive in endometrial stromal sarcomas with high-grade cytologic features (especially if t(10;17)).
Nuclear β-catenin expression has been shown in up to 40% of low-grade endometrial stromal sarcomas42 as well as in uniform undifferentiated endometrial sarcomas, the latter frequently associated with cyclin D1 expression and thought to be part of t(10;17) group of tumors.42 p53 expression is typically absent in low-grade endometrial stromal sarcomas, but it has been reported in high-grade/undifferentiated endometrial sarcomas (more often if pleomorphic subtype, as expected).48,53 Cyclin D1 is mostly expressed in endometrial stromal sarcomas with t(10;17)54 (Figure 20.22), although rarely it has been detected in endometrial stromal nodules and low-grade endometrial stromal sarcomas, as well as uniform-type undifferentiated endometrial sarcomas.42
Differential Diagnosis
Among non-neoplastic processes, endometrial stromal tumors should be distinguished from endometrial polyps especially in curettage specimens if the polyp is fragmented and cellular and some fragments are only composed of endometrial stroma. In contrast to endometrial stromal tumors, stromal cells have an inactive ‘compacted’ appearance with unassociated mitotic activity. Even though small vessels reminiscent of the arterioles seen in endometrial stromal tumors may be present, large thick blood vessels are also found. Furthermore, some fragments may show inactive stromal cells admixed with endometrial glands.55
Gland-poor adenomyosis or intravascular adenomyosis (Figure 20.23) can be occasionally confused with an endometrial stromal sarcoma. Grossly these lesions do not form a mass but at most an irregular thickening or irregularity of the myometrial wall and typically represent an incidental finding. The islands of endometrial stroma have an inactive appearance without expansile borders in contrast to endometrial stromal sarcomas and areas of conventional adenomyosis are always identified.56 However, endometrial stromal sarcomas may arise from adenomyotic foci as well as endometriosis.
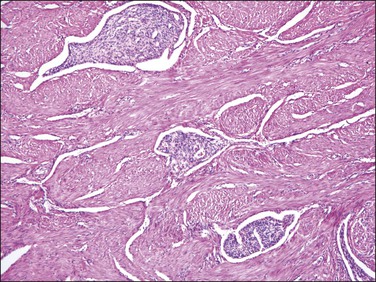
Figure 20.23 Gland-poor adenomyosis. Several islands of adenomyosis composed only of gland-free endometrial stroma are present within the myometrium. The stromal cells have an inactive and solid appearance.
A common pitfall is the misdiagnosis of a highly cellular leiomyoma as an endometrial stromal tumor, more frequently as a low-grade endometrial stromal sarcoma (Table 20.2). Highly cellular leiomyoma shares with the latter the following features: soft consistency and tan to yellow cut surface, dense cellularity (‘blue’), striking vascularity, and irregular margins with the surrounding myometrium.57 However, it also typically forms fascicles (easier to identify at its periphery), cells are spindled with elongated ‘cigar-shaped’ nuclei, and there is merging of the tumor cells with the surrounding myometrium. Small vessels can be found and may be misinterpreted as arterioles, but they do not show the morphology of typical arterioles seen in endometrial stromal tumors. Thick and large blood vessels and cleft-like spaces throughout the tumor are almost constant features of these tumors (Figure 20.24).57 Some smooth muscle tumors may contain highly cellular areas alternating with conventional areas. If the latter are misinterpreted as myometrium, cellular areas may be diagnosed as endometrial stromal sarcoma. Gross evaluation as well as microscopic distinction of myometrium from neoplastic smooth muscle is key in rendering the correct diagnosis. Immunohistochemistry may be helpful in this differential diagnosis. As actins are positive in both categories of tumors they should not be used. CD10, although reported to be specific of endometrial stromal derivation, is often positive in highly cellular leiomyomas and thus should not be used in isolation. Muscle markers, including desmin, h-caldesmon, heavy chain myosin, and HDAC-8 are typically diffusely positive in smooth muscle tumors but can be positive in areas of conventional stromal neoplasia (desmin) and areas with smooth muscle or sex cord-like differentiation (desmin, h-caldesmon, heavy chain myosin).19,38,40,58 Thus, using a panel of antibodies, which includes CD10, desmin, and h-caldesmon (or other specific smooth muscle markers), is most helpful in this differential diagnosis. It is also important to correlate positive areas with the corresponding morphologies of the tumor. Oxytocin has also been used in the differential diagnosis of smooth muscle tumors and endometrial stromal tumors, the latter being typically negative.59
Table 20.2
Contrasting Features of Endometrial Stromal Tumors (ESTs) and Highly Cellular Leiomyomas (HCLs)
| EST | HCL | |
| Yellow | Typical | Less common |
| Gross circumscription | Poor (if sarcoma) | Well demarcated |
| Microscopic circumscription | Poor (if sarcoma) | Limited interdigitation and merging with myometrium |
| Large thick blood vessels | Occasional (mostly at periphery) | Common (random) |
| Arterioles | Usual | Uncommon |
| Clefts | Rare | Common |
| Foamy histiocytes | Frequent | Rare |
| CD10 positivity | Diffuse and strong, in most | Up to 40% positive |
| Desmin positivity | Occasional (usual forms) Expected (if smooth muscle metaplasia) | Typical, diffuse |
| h-Caldesmon positivity | Negative (usual forms) Expected (if smooth muscle metaplasia) | Typical, diffuse |
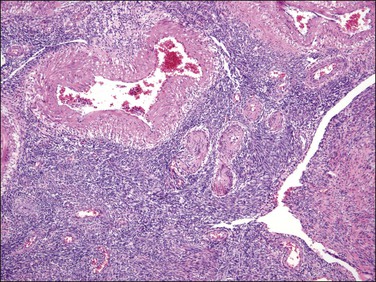
Figure 20.24 Highly cellular leiomyoma. The tumor has a vague fascicular architecture. Large and thick blood vessels as well as cleft-like spaces characteristic of this neoplasm are seen.
Cellular intravenous leiomyomatosis can enter in the differential diagnosis of endometrial stromal sarcomas due to their dense cellularity and intravascular growth especially if associated with a highly cellular leiomyoma. However, in most cases, the microscopic appearance of intravenous leiomyomatosis recapitulates that of typical leiomyomas showing a fascicular growth and large and thick blood vessels, as well as cleft-like spaces60 and not infrequently a subendothelial proliferation of spindle cells may be seen ‘colonizing’ the walls of the veins.
Endometrial stromal tumors with myxoid differentiation should be distinguished from myxoid smooth muscle tumors, often leiomyosarcoma, as both tend to be hypocellular with a similar pattern of myometrial infiltration. However, extensive sampling should evidence a component of spindle or epithelioid leiomyosarcoma with highly atypical cells and brisk mitotic activity. Immunohistochemistry may be of limited utility as some endometrial stromal tumors with fibroblastic and/or myxoid background are positive for desmin18 and positivity for muscle markers may be limited in myxoid smooth muscle tumors. If positive, p53 may favor a diagnosis of myxoid leiomyosarcoma as p53 positivity is restricted to undifferentiated endometrial sarcomas.48,53
Endometrial stromal tumors with sex cord-like differentiation should be distinguished from uterine tumors resembling ovarian sex cord tumors (UTROSCTs), as the latter lack any component of endometrial stromal neoplasia22 (see the section Uterine tumors resembling ovarian sex cord tumors).
Endometrial stromal sarcomas with t(10;17) should be distinguished from epithelioid leiomyosarcoma and epithelioid gastrointestinal stromal tumor secondarily involving the uterus as all have epithelioid cells. Furthermore, t(10;17) endometrial stromal sarcomas are frequently CD10, ER, and PR negative and c-kit positive in the high-grade epithelioid areas,20 creating a diagnostic challenge. As mentioned earlier, this morphologic variant of endometrial stromal sarcoma is associated with a low-grade fibroblastic component in 50% of cases, which, if present, may help to establish the correct diagnosis. These tumors are cyclin D1 positive while epithelioid leiomyosarcomas are typically desmin and caldesmon positive, often keratin and epithelial membrane antigen (EMA) positive,40 and cyclin D1 negative.54 Gastrointestinal stromal tumors present as one or multiple masses typically centered outside the uterus and they are positive for DOG-1 and to a lesser extent CD34 along with c-kit.61
Cytogenetics
t(7;17)(p15;q21) is the most common chromosomal translocation in endometrial stromal tumors resulting in the JAZF1–SUZ12 gene fusion (Figure 20.25). It can be detected by cytogenetics, fluorescence in situ hybridization, or reverse transcriptase polymerase chain reaction (PCR). It has been reported in the majority of endometrial stromal nodules, approximately 50% of low-grade endometrial stromal sarcomas, and a minority of undifferentiated endometrial sarcomas, typically those classified as uniform type.62–67 The presence of the JAZF1–SUZ12 gene fusion in at least 50% of endometrial stromal nodules and in a significant, but smaller, subset of low-grade endometrial stromal sarcomas suggests that JAZF1–SUZ12 fusion represents an early event in the development of endometrial stromal tumors, and that additional events are necessary for tumor progression. The finding of identical translocations in occasional undifferentiated endometrial sarcomas and low-grade endometrial stromal sarcomas may indicate that at least some of the former tumors represent progression from low-grade tumors. The JAZF1–SUZ12 cytogenetic abnormality has also been detected in morphologic variants of endometrial stromal tumors including those with smooth muscle, fibroblastic/myxoid, epithelioid, and sex cord-like differentiation, although to a lesser extent.17,23,48,68,69 t(6;17)(p21;p22) and its variants represent the second most common cytogenetic abnormalities in endometrial stromal tumors.66,70,71 A three-way t(6p10q;10p) has also been reported.71 In general, it appears that there is no correlation between specific variant histologic subtypes and gene fusions among endometrial stromal tumors. However, recently, a t(10;17)(q22;p13) with genetic fusion between YWHAE and FAM22 (Figure 20.26)66,72 has been reported in a subset of high-grade endometrial stromal sarcomas with a concomitant low-grade fibromyxoid component in 50% of cases. Also, PHF1 genetic rearrangement has been found to occur more frequently in endometrial stromal sarcomas with sex cord differentiation than other morphologic variants.73
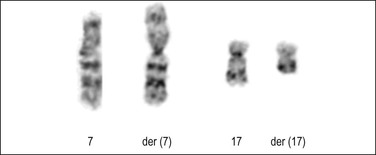
Figure 20.25 Low-grade endometrial stromal sarcoma. Karyotype analysis shows the common translocation involving chromosomes 7 and 17. (Courtesy of Paola Dal Cin.)
Treatment and Prognosis
Low-grade endometrial stromal sarcoma is a relatively indolent mesenchymal tumor with an overall 70–84% and 65–76% 5 and 10 year survival rate, respectively. Recurrences are common (in approximately half of patients), typically occurring years after the initial diagnosis. The most frequent sites include pelvis and abdomen but the tumor can also recur in lung and other locations.10,74 Surgical–pathologic stage is the most powerful prognostic factor. It has been found that patients with tumors confined to the uterus (stage I or II) had an 89% 5 year disease-specific survival compared to 50% in patients with stage III or IV tumors.7 Regarding the impact of stage, cytologic atypia, and mitotic activity in endometrial stromal sarcomas, by univariate analysis, brisk mitotic activity has correlated with poor outcome while cytologic atypia correlated with increased relapse although not with overall survival. However, by multivariate analysis, both parameters lost their predictive impact in stage I tumors in the largest study to date.8 In a recent study, mitotic count and tumor cell necrosis were reported to be important prognostic factors in stage I endometrial stromal sarcomas and in conjunction could be used to stratify patients with low-grade endometrial stromal sarcomas into three prognostic categories.73 Others, however, have found no association of tumor cell necrosis with prognosis.8 Patients with endometrial stromal sarcomas are treated with radical hysterectomy and bilateral salpingo-oophorectomy to avoid secondary hormonal stimulation of the tumor. Younger patients who desire to preserve fertility can be treated with radiation, hormonal therapy, or aromatase inhibitors.50 Gonadotropin-releasing hormone (GnRH) analogs can also be used as an adjunct as they reduce estrogen production in the ovaries.75 Tumors with t(10;17) should be treated aggressively with a combination of radiation and chemotherapy as they do not respond to conventional treatment for low-grade endometrial stromal sarcoma.20
High-Grade Endometrial Stromal Sarcoma
Definition
This is malignant tumor of endometrial stromal derivation with high-grade, round-cell morphology sometimes associated with a low-grade spindle cell component that is most commonly fibromyxoid.42 When unassociated with a low-grade component, these tumors should only be diagnosed after excluding other more common uterine sarcomas, including leiomyosarcoma, sarcomatous overgrowth in a müllerian adenosarcoma, malignant mixed müllerian tumor, or rhabdomyosarcoma.
Microscopic Features
On low-power examination, these tumors may have the typical infiltrative growth and vasculature of its low-grade counterpart; however, there is usually confluent permeative and destructive growth, often with invasion into the outer half of the myometrium. There is a variable mixture of closely juxtaposed high-grade round cell (usually predominant) and low-grade spindle cell component. The round cell areas are hypercellular and the cells are arranged in vague to well defined nests and separated by a delicate capillary network. The round cells have modest amount of eosinophilic to granular cytoplasm, irregular nuclear contours and granular to often vesicular chromatin, with variably distinct nucleoli. Occasionally, the round cells are noncohesive imparting a pseudo-papillary/glandular appearance or have focal rhabdoid morphology. Occasionally, primitive neuroectodermal differentiation in the form of Flexner-Wintersteiner rosettes or Homer-Wright pseudo-rosettes may be seen (Figure 20.13). Mitotic activity is typically >10 per 10 HPF and is very striking. Necrosis is usually present. The spindle cell component often has fibromyxoid features. Lymphovascular invasion is typically present. Rarely, a high-grade sarcoma is seen in association with areas that have the appearance of conventional low-grade endometrial stromal sarcoma.
Immunohistochemistry and Somatic Genetics
The high-grade component of tumors with t(10;17) is CD10, ER and PR negative (Figures 20.20 and 20.21) but shows strong diffuse cyclin D1 positivity (>70% nuclei) (Figure 20.22); the low-grade spindle cell component is typically strongly and diffusely CD10, ER and PR positive and shows variable, heterogeneous cyclin D1 expression (<50%). The high-grade component is also typically c-Kit positive but DOG1 negative. High-grade endometrial stromal sarcoma typically harbours the YWHAE-FAM22 genetic fusion as a result of t(10;17)(q22;p13).
Prognosis
In comparison to low-grade endometrial stromal sarcomas, patients have earlier and more frequent recurrences (often <1 year) and are more likely to die of disease. They appear to have a prognosis that is intermediate, between low-grade endometrial stromal sarcoma and undifferentiated uterine sarcoma.20
Undifferentiated Endometrial Sarcoma
Definition
This is a high-grade uterine sarcoma bearing no resemblance to proliferative-phase endometrium. Typically shows high-grade cytologic features without specific type of differentiation.2
Microscopic Features
On low-power magnification, margins are poorly defined with destructive invasion of the myometrium. The tumor cells are highly atypical, grow in sheets (Figure 20.27) or exhibit a storiform or herringbone patern. The cells can be polygonal or spindle and they show marked nuclear pleomorphism including multinucleated giant cells. Brisk mitotic activity including atypical forms is seen. The characteristic delicate vasculature reminiscent of spiral arterioles as well as the small uniform cells of the proliferative-type stroma are lacking. Lymphovascular invasion, necrosis, and hemorrhage are frequent. Rhabdoid morphology or myxoid background may be seen.
Immunohistochemistry and Somatic Genetics
These tumors are variably CD10 positive and typically ER and PR weakly positive or negative. Cyclin D1 can be diffusely positive but in those cases the tumors are also typically positive for CD10 (which excludes YWHAE-FAM22 sarcomas). Focal smooth muscle actin, desmin, EMA or keratin positivity may be seen.48
Differential Diagnosis
As these tumors are poorly differentiated, extensive sampling is important in order to exclude other high-grade sarcomas (leiomyosarcoma or rhabdomyosarcoma), sarcomatous overgrowth in a müllerian adenosarcoma, or a malignant mixed müllerian tumor where the epithelial component is very limited and has only homologous sarcomatous elements. Leiomyosarcoma may have spindle or epithelioid cells and neoplastic cells with marked cytologic atypia. However, often they display well-developed fascicles and cigar-shaped nuclei. Immunohistochemical stains for smooth muscle markers are positive despite CD10, ER, and PR positivity. Spindle rhabdomyosarcoma is exceedingly rare. Cells are wavy with bright eosinophilic cytoplasm and may show cross striations. Muscle markers including myogenin and myo-D1 are typically positive.76 Müllerian adenosarcoma with sarcomatous overgrowth as well as malignant mixed müllerian tumor may enter in the differential diagnosis if areas of conventional adenosarcoma or the carcinomatous component of a malignant mixed müllerian tumor have not been sampled.77,78
Uterine Tumors Resembling Ovarian Sex Cord Tumors
Definition
These uncommon neoplasms closely recapitulate the morphologic spectrum of sex cord–stromal tumors of the ovary, more frequently granulosa cell tumors.79 Endometrial stromal tumors with any degree of sex cord-like (epithelial) differentiation were initially included under this category, with group A (stromal predominant) composed of tumors with some sex cord elements and group B having exclusively sex cord elements.22 Currently only tumors composed of sex cord elements are diagnosed under this category.55
Clinical Features
These tumors are frequently seen in patients in their fourth to sixth decades who typically present with vaginal/postmenopausal bleeding or nonspecific symptoms and signs related to the growing uterine mass.80
Gross Features
They can be polypoid protruding into the endometrial cavity, intramyometrial, and rarely subserosal,22 or centered in the cervix.81 They range in size from microscopic to 15 (mean 6) cm80 and are typically well circumscribed with a homogeneous yellow to tan or less frequently white to gray cut surface depending on the amount of intervening stroma. Areas of necrosis or hemorrhage are uncommon.
Microscopic Features
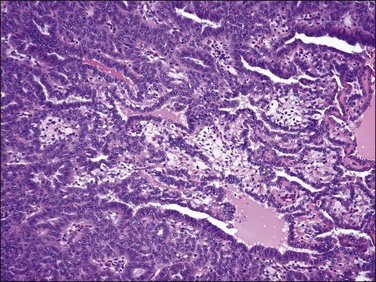
On low-power examination, these tumors are frequently but not always well circumscribed with a pushing border. Some tumors may have an infiltrative border where tumor cells are closely intermixed with fascicles of myometrium that may appear hyperplastic without associated stromal response.55 On closer examination, uterine tumors resembling sex cord–stromal tumors are characterized by morphologic patterns that closely resemble those described in sex cord tumors of the ovary. Tumor cells are frequently arranged in cords (sometimes with plexiform pattern) (Figure 20.28), anastomosing trabeculae (Figure 20.29), nests or irregularly shaped islands, and sheets reminiscent of granulosa cell tumor. However, hollow or solid tubules (Figure 20.30), some with a retiform architecture including small papillae with hyalinized cores reminiscent of Sertoli tubules seen in Sertoli–Leydig or Sertoli cell tumors, are also seen (Figure 20.31).24,55,80,82 Rarely, cystic dilation of tubules, pseudopapillary architecture, Leydig-like or foam cells, and heterologous elements in the form of intestinal epithelium (as reported in ovarian Sertoli–Leydig cell tumor) can occur. Cells are usually small to medium sized with variable amounts of eosinophilic to pale sometimes vacuolated cytoplasm. Rarely rhabdoid morphology has been reported.83 Nuclei are typically uniform with small but visible nucleoli and grooved nuclei may be seen. Mitotic figures are typically scant. The background stroma is frequently scarce with a loose fibroblastic appearance and no endometrial stromal component is present. Lymphovascular invasion is typically absent.22
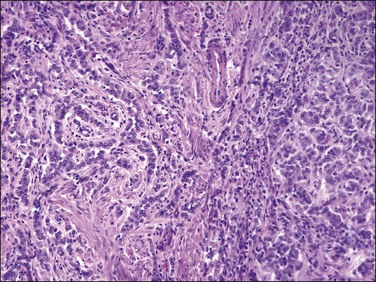
Figure 20.28 Uterine tumor resembling ovarian sex cord tumor. The neoplastic cells form cords, embedded in a collagenous stroma, which are reminiscent of an adult granulosa cell tumor.
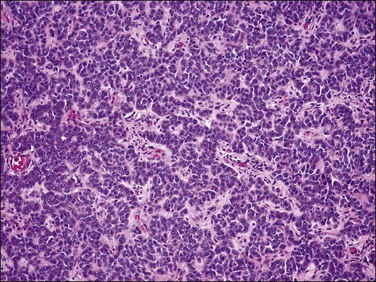
Figure 20.29 Uterine tumor resembling ovarian sex cord tumor. Closely packed interconnecting trabeculae composed of cells with ‘epithelial’ features are seen.
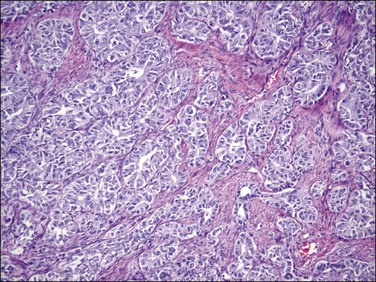
Figure 20.30 Uterine tumor resembling ovarian sex cord tumor. The neoplastic cells form tubular structures reminiscent of Sertoli cell tubules.
Immunohistochemical Features
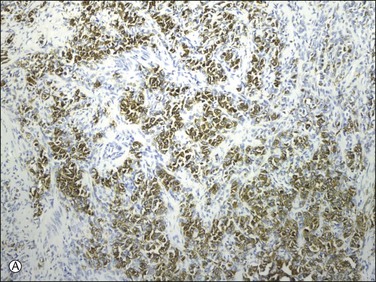
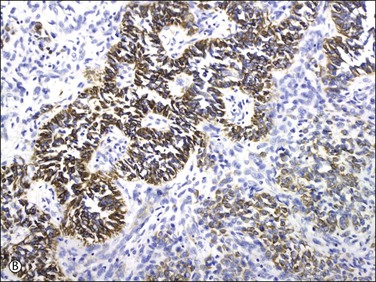
Uterine tumors resembling ovarian sex cord tumors are considered polyphenotypic as they often coexpress epithelial, smooth muscle, and sex cord markers. They are always vimentin positive and often express a variety of keratins (uncommonly CK7) (Figure 20.32A)21,24,80,83,84 and to a much lesser extent EMA.83,85 The tumor cells are often positive for smooth muscle markers including actin, desmin (Figure 20.32B), h-caldesmon, HDAC-8, and smooth muscle myosin heavy chain.21,40,81,83–85 They also express calretinin, inhibin (Figure 20.32C), Melan-A, CD99, and WT1.21,24,40,83,85 Inhibin also stains Leydig-like cells.24 They are also variably positive for ER and PR24,80,83,84,86 and can show focal CD10 and c-kit expression.85
Cytogenetics
Studies have shown that uterine tumors resembling ovarian sex cord–stromal tumors lack the JAZF1–JJAZ1 gene fusion in contrast to endometrial stromal tumors with or without sex cord elements, suggesting an independent origin.88
Differential Diagnosis
The main differential diagnoses include endometrial stromal tumor with sex cord elements, endometrioid carcinoma including ‘sertoliform’ variant, and epithelioid smooth muscle tumors. Obviously, the presence of any endometrial stromal areas is diagnostic of endometrial stromal neoplasia. Furthermore, a diagnosis of uterine tumor resembling a sex cord ovarian tumor can only be made in a hysterectomy specimen where the specimen can be extensively sampled to exclude areas of endometrial stromal neoplasia.55 Low-grade endometrioid carcinomas are by far more common than uterine tumors resembling ovarian sex cord tumors. They can be seen as thickening of the endometrium or irregularly infiltrating the myometrium and exhibit squamous, tubal, or mucinous metaplasia. However, some endometrioid carcinomas may have a sertoliform morphology and others may form cords and trabeculae associated with a hyalinized background,89 while uterine tumors resembling sex cord tumors may have an endometrioid appearance especially if they show a retiform pattern. The finding of well-formed glands with squamous, tubal, or mucinous epithelium and negative staining for sex cord-like and smooth muscle markers supports a diagnosis of carcinoma. Epithelioid smooth muscle tumors may grow in sheets, cords, or nests and stain for smooth muscle markers, often for wide spectrum keratins as well as EMA but often are associated with a conventional spindle component and are negative for sex cord markers.55 Other tumors that infrequently enter in the differential diagnosis include perivascular epithelial cell tumor (PEComa), malignant melanoma, or even metastatic lobular breast carcinoma.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree