Effects of Therapeutic Procedures on the Epithelia of the Female Genital Tract
HEAT, COLD, AND LASER TREATMENT
Heat, in the form of cautery, is an ancient remedy for local treatment of various lesions. In its more modern form, as the electrocautery, it has enjoyed great popularity in the treatment of various benign disorders of the female genital tract, such as chronic cervicitis. Large loop electrosurgical excision procedure (LEEP) of precancerous lesions of the cervix is another application of electrocautery. Other forms of locally destructive therapy include: cold, in the form of cryosurgery, and energy, transmitted in the form of a laser beam, used in the treatment of intraepithelial neoplastic lesions of the uterine cervix and of the vagina (see Chaps. 11 and 14). Because cytology, and particularly the cervicovaginal preparations, are extensively used as a follow-up measure after treatment, it is important to distinguish cell changes caused by therapy from evidence of recurrent cancer.
All these forms of therapy have in common cell changes that are of two types:
Initial changes, caused by tissue and cell necrosis under the impact of treatment
Secondary changes, caused by epithelial regeneration following the injury
Initial Changes
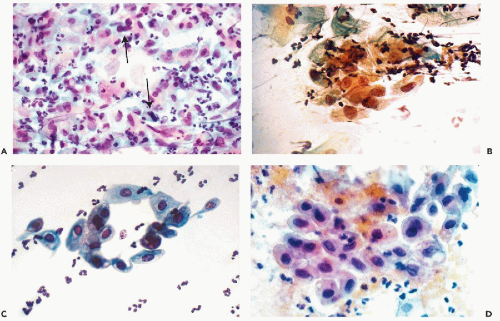
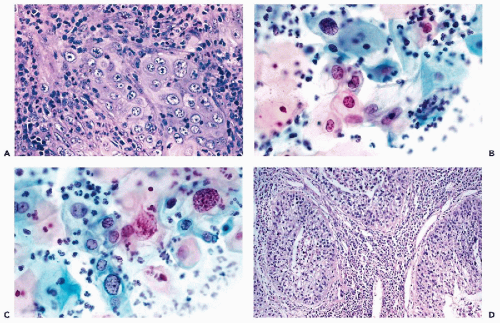
In principle, cervicovaginal samples should not be obtained for about 6 weeks following treatment. However, ever so often, smears are obtained sooner and the cell changes seen in such material are described here. Immediately after, and for about 7 days following treatment, tissue and cell necrosis are the predominant features observed in cytologic and histologic material (Fig. 18-1A). The necrosis is of the coagulative type and, hence, the affected epithelia
may retain their overall structure, even though their component cells may be severely injured.
may retain their overall structure, even though their component cells may be severely injured.
In cervicovaginal smears, the background usually contains cell debris and evidence of acute inflammation in the form of polymorphonuclear leukocytes. The resilient squamous cells may become enlarged because of cytoplasmic vacuolization but often retain their cytoplasmic silhouette. Their nuclei are either “empty” or smudged, without any internal structure, or show nuclear pyknosis and karyorrhexis (Fig. 18-1B). The more fragile endocervical cells may be enlarged and vacuolated, sometimes misshapen, with opaque or fragmented nuclei. For the most part, however, the endocervical cells rarely survive intact and usually are fragmented. Holmquist et al (1976) emphasized “distortion” or odd shapes of endocervical cells after carbon-dioxide laser treatment. Similar observations were reported after cryosurgery (Hasegawa et al, 1975).
Thomas (1997) described an unusual procedure in the form of immediate post-LEEP endocervical brush to determine the presence of residual disease or lesions located beyond the reach of the loop. The endocervical cells were often elongated and showed distortion of nuclear configuration with oddly shaped, often “smudgy” nuclei. Thomas (1997) stressed the difficulties in the interpretation of such smears, compounded by the presence of blood and necrosis. The value of this procedure has not been ascertained.
Secondary Changes
The secondary changes are more common because they may persist for several weeks, when most of the follow-up smears are obtained. Starting on or about 8 days after treatment, the smear background usually shows evidence of inflammation and sometimes persisting necrosis. Lymphocytes and macrophages, the latter sometimes multinucleated, are the dominant inflammatory cells. In the epithelial cells, cytoplasmic vacuolization may persist for as long as 6 weeks after cautery and for several months after cryosurgery (Gondos et al, 1970). Another persisting change is slight nuclear enlargement, hyperchromasia, and the appearance of nuclear “folds” or lines. Within 1 week after treatment, sheets of parabasal squamous cells of various sizes, with well-preserved, dark nuclei with stippled chromatin granules and basophilic cytoplasm, may be observed, signaling the beginning of regeneration or “repair” of the squamous epithelium (Fig. 18-1C). Sheets of smooth muscle
cells may be observed in endocervical brush samples, particularly if the sampling was obtained before epithelial regeneration has been completed or if the brushing was very vigorous.
cells may be observed in endocervical brush samples, particularly if the sampling was obtained before epithelial regeneration has been completed or if the brushing was very vigorous.
In somewhat later stages of epithelial regeneration, cell changes of florid squamous metaplasia or “repair,” as described in detail in Chapter 10, may be noted. Sheets or clusters of parabasal squamous cells with basophilic cytoplasm and relatively large, often granular nuclei with prominent, large nucleoli may be observed. Nucleolar prominence has been emphasized by Hasegawa et al (1975) in patients after cryosurgery. Mitotic figures can occur in such epithelial fragments. These changes may persist for about six weeks after treatment.
The duration of the therapy-induced cell changes is variable and depends on the anatomic extent and mode of treatment. The procedures used are not standardized and, consequently, significant differences occur among practitioners and institutions. If the treatment is confined to a small area of the cervix, its effects will be less noticeable and of shorter duration than if much of the epithelium of the exo- and endocervix has been treated or removed together with the underlying connective tissue and muscle.
The most important practical point is the determination of whether intraepithelial neoplasia has been destroyed by treatment. In our experience, the diagnosis of residual disease should not be made until at least six weeks have elapsed after treatment, or until complete healing of the therapy-induced changes has taken place. Prior to that time, cancer cells derived from the original, adequately treated lesion may still occur in smears, even in patients with a favorable response. Past the 6 week deadline, the presence of cancer or dyskaryotic (dysplastic) cells may be interpreted in the customary fashion, described in previous chapters, and their presence indicates persisting disease. However, the post-treatment cytologic examination to detect persisting lesions is not fully reliable and has its difficulties and failures. An example of this problem is shown in Figure 18-1D wherein the differentiation between atypical repair and recurrent lesion proved to be difficult until further smears revealed a low-grade squamous intraepithelial lesion (LGSIL). Thus, it is advisable to combine the cytologic follow-up with colposcopy and testing for high-risk human papillomavirus (HPV). Chua and Hjerpe (1997) reported that the presence of high risk human papillomavirus, determined by PCR, was an important indicator of recurrent high-grade precancerous lesions. There is no information on the value of this procedure in patients treated by laser or cryotherapy.
TOPICAL ANTIBIOTICS
A topical application of broad-spectrum antibiotics may result in massive desquamation of the squamous epithelium of the cervix and the vagina, as discussed in Chapter 11. Sometimes cancer cells may be concealed by sheets of benign epithelial cells. In some cases of carcinoma in situ, we have observed sloughing of the cancerous epithelium, with resulting disappearance of the lesion. Before pronouncing an in situ carcinoma as “cured” by this method, it is essential to follow the patient for at least 3 years, since cancer cells may reappear in smears at a time when least expected. It would not be wise to rely on this chance action of antibiotics for treatment of precancerous lesions. These observations are reported here as a matter of scientific interest only.
RADIOTHERAPY
Nearly all of the information on the effects of radiotherapy on the organs of the female genital tract, be it as external irradiation, radium or implanted radioactive seeds, pertains to treatment of carcinoma of the uterine cervix or vagina. Although the changes in the benign epithelia of the female genital tract are the same for all forms of radiotherapy and all tumors, regardless of location, the changes observed in cancer cells are limited to cervical carcinoma, because there is very little reliable information on cancers in other component organs of the female genital tract. The effects of radiotherapy may be described as acute and chronic.
Acute Effect on Benign Epithelia
Graham (1947) studied extensively the immediate effect of radiation on benign squamous epithelium of the cervix and the vagina. She noted and described the following cellular changes:
Marked cellular enlargement accompanied by a proportional nuclear enlargement
A peculiar “wrinkling” of the nuclei
Vacuolization of the cytoplasm or, occasionally, of the nucleus
Multinucleation
Appearance of bizarre cell forms
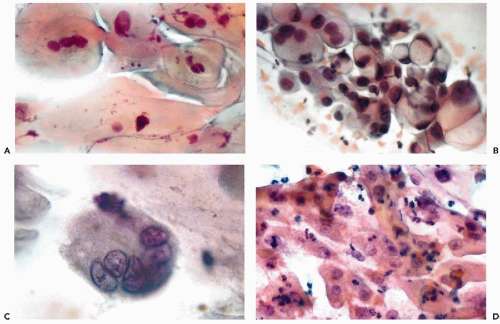
The changes represent the damaging effect of radiation on individual cells and various stages of cell death (Fig. 18-2A). The most striking change in such smears is generalized cellular enlargement, usually affecting the cytoplasm and the nucleus, without a change in the nucleocytoplasmic ratio. For the most part, the enlarged nuclei are homogeneous and pale, easily recognized as benign. The “wrinkling” of the nucleus, described by Graham, occurs rather rarely. Unfortunately, in squamous cells, radiation may also produce nuclear hyperchromasia, multinucleation, and bizarre forms (Fig. 18-2C) and may render the differential diagnosis from cancer cells very difficult.
The endocervical cells are well preserved, but there is a marked vacuolization and enlargement of both the cytoplasm and the nucleus (Fig. 18-2B). Within the nucleus, granules of chromatin stand out against the pale background. Corresponding changes may be noted in histologic sections. Bizarre nuclear abnormalities, common in squamous cells, are less frequent in endocervical cells. Similar observations were reported by Little (1968), Boschann (1981), and Shield et al (1992).
The acute changes in the squamous and endocervical cells usually recede a few weeks after completion of radiotherapy
in favor of the chronic changes, described below. In some patients, however, these changes may persist for several months after completion of radiation therapy.
in favor of the chronic changes, described below. In some patients, however, these changes may persist for several months after completion of radiation therapy.
Persistent Effect on Benign Epithelia
In some patients who have undergone radiation therapy to the pelvic area, there may be persistence of radiation effect upon the benign squamous and the endocervical epithelia, stretching over a period of many years. We have observed such changes 28 years after the completion of radiotherapy. The biologic phenomena that account for this effect are unknown. It may be speculated that, in susceptible patients, the genetic make-up of the irradiated epithelium has been altered. The occurrence of post-radiation carcinoma in situ (see below) and of cancer in organs within the field of radiation support this hypothesis. Neither the amount of radiation nor the manner of application appears to play a role; it is rather a matter of individual response to radiation injury.
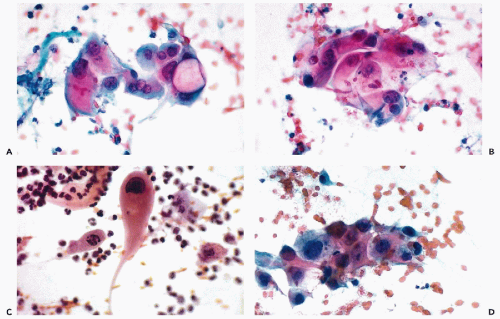
The cytologic manifestations of a late radiation effect differ from the acute radiation effect. The phenomena of acute injury to the cell, such as nuclear and cytoplasmic vacuolization and nuclear necrosis are absent. Commonly, there still is a persisting slight enlargement of cells and their nuclei. The squamous cells often desquamate in cohesive sheets of elongated cells, sometimes mimicking smooth muscle cells, with elongation of the rather homogeneous nuclei (Fig. 18-2D). Among the elongated nuclei, a few are often hyperchromatic. Multinucleation is less frequent than in the acute radiation response. The nuclei of endocervical cells may also show persisting enlargement and some hyperchromasia (Fig. 18-3A). The changes are sufficiently characteristic for an experienced and knowledgeable observer to diagnose late radiation effects in cervicovaginal smears.
Effect on Cancer Cells
During radiotherapy, the cancer cells, regardless of type, undergo essentially the same changes as the benign epithelial cells, that is, cellular and nuclear ballooning and extensive vacuolization of both the cytoplasm and the nucleus. Occasionally, extensive fragmentation of the nuclei may be observed, most likely a form of cell death or apoptosis (see Chap. 6). Squamous cancer cells usually retain some of their cytoplasmic characteristics and can be recognized; however, poorly differentiated cancer cells and cells of adenocarcinomas usually cannot be specifically classified. Marked radiation
effect may enhance or obliterate some of the features of malignant cells, such as abnormal structure of the nuclear chromatin and the presence of large nucleoli (Fig. 18-3B). However, some measure of hyperchromasia usually persists, as does an abnormal nucleocytoplasmic ratio.
effect may enhance or obliterate some of the features of malignant cells, such as abnormal structure of the nuclear chromatin and the presence of large nucleoli (Fig. 18-3B). However, some measure of hyperchromasia usually persists, as does an abnormal nucleocytoplasmic ratio.
Differential Diagnosis Between Radiated Benign and Malignant Cells
The question of differentiation between irradiated benign and malignant cells is of academic interest only. If the malignant cells display radiation effect that obliterates their characteristic features, they are not capable of reproduction; therefore, they provide no information on the presence or the absence of viable tumor. Only those cancer cells that are either unaffected or only slightly affected by radiation are of concern in the diagnosis of persistent or recurrent tumor.
In reference to cancer of the uterine cervix, persistence of unaffected cancer cells in smears during and after treatment suggests that a tumor is not responding to radiation. However, as reported by Zimmer (1959), cancer cells may persist for as long as 3 weeks after completion of treatment, and yet the patients appeared to be cured and did not show tumor recurrence for several years. Such cases are exceptional. It must be stressed that, in spite of apparent favorable cytologic response of the tumor to treatment, the tumor may persist within the subepithelial stroma or other areas not accessible to cytologic sampling. In such situations, the absence of cytologic evidence of persisting carcinoma is of no clinical value whatever.
Recurrent cancer of the uterine cervix, after successful initial treatment, may be recognized in cervicovaginal smears and its manifestations are identical to those of primary cancer, sometimes in the background of smears showing slight persisting radiation effect (Fig. 18-3C,D).
Postradiation Carcinoma In Situ in the Cervix and Vagina (Post-Irradiation Dysplasia)
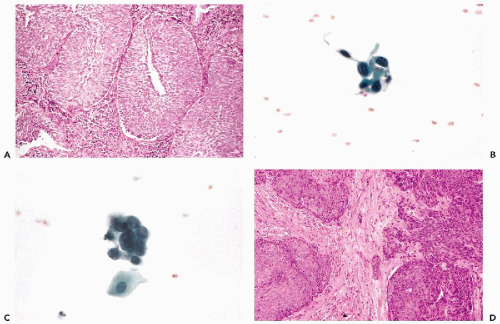
In 1961, we reported on a group of patients who, after a disease-free time interval ranging from 1.5 to 17 years following successful radiotherapy for invasive squamous cancer of the cervix, developed cytologic abnormalitiesconsistent with carcinoma in situ or closely
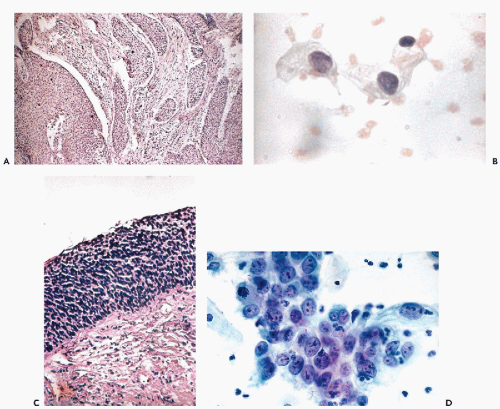
related forms of cervical intraepithelial neoplasia. The term “post-irradiation carcinoma in situ” was proposed by Koss et al (1961). The term “postradiation dysplasia” was subsequently used by Patten et al (1963) in describing this lesion. The abnormal epithelium, located on either the irradiated cervix or vagina, often could not be visualized on inspection or colposcopy and was exceedingly difficult to localize within the scarred genital tract. In some cases, numerous biopsies of the cervix and the vaginal mucosa were required to confirm the presence of postradiation carcinoma in situ (Fig. 18-4). In one of the patients of the original series who was treated by hysterectomy for the postradiation carcinoma in situ, there was associated residual metastatic carcinoma in an obturator lymph node that would not have been discovered and removed were it not for the vaginal lesion. It is of note that Fujimura et al (1991), Holloway et al (1991), and Longatto Filho et al (1997) observed the presence of human papillomavirus (HPV) in cervicovaginal smears of 18 women after completion of radiotherapy for invasive cancer of the uterine cervix. Holloway et al (1991) observed HPV type 16 in cancer of the cervix recurring after therapy.
related forms of cervical intraepithelial neoplasia. The term “post-irradiation carcinoma in situ” was proposed by Koss et al (1961). The term “postradiation dysplasia” was subsequently used by Patten et al (1963) in describing this lesion. The abnormal epithelium, located on either the irradiated cervix or vagina, often could not be visualized on inspection or colposcopy and was exceedingly difficult to localize within the scarred genital tract. In some cases, numerous biopsies of the cervix and the vaginal mucosa were required to confirm the presence of postradiation carcinoma in situ (Fig. 18-4). In one of the patients of the original series who was treated by hysterectomy for the postradiation carcinoma in situ, there was associated residual metastatic carcinoma in an obturator lymph node that would not have been discovered and removed were it not for the vaginal lesion. It is of note that Fujimura et al (1991), Holloway et al (1991), and Longatto Filho et al (1997) observed the presence of human papillomavirus (HPV) in cervicovaginal smears of 18 women after completion of radiotherapy for invasive cancer of the uterine cervix. Holloway et al (1991) observed HPV type 16 in cancer of the cervix recurring after therapy.
Subsequently, in a number of personally observed cases, the ominous significance of these lesions became apparent. Several patients, with postradiation carcinoma in situ (or dysplasia) who were followed conservatively, developed invasive carcinomas of the cervix or of the vagina, sometimes after many years of follow-up. In yet other patients, disseminated metastatic carcinoma developed within a short period of time (Figs. 18-5 and 18-6).
The cytologic presentation of these lesions failed as a means of prognostication. Some cases with a cytologic presentation akin to classic squamous carcinoma required many years to progress to invasive carcinoma (see Fig. 18-6); others, with a cytologic presentation dominated by dyskaryotic
(dysplastic) superficial and intermediate squamous cells, hence resembling a low-grade lesion, were followed by rapid progression and dissemination of the tumor (Fig. 18-5).
(dysplastic) superficial and intermediate squamous cells, hence resembling a low-grade lesion, were followed by rapid progression and dissemination of the tumor (Fig. 18-5).
Patten et al (1963) reported on a group of 28 patients with similar cytologic and histologic patterns, and elected to call the lesion “post-irradiation dysplasia.” One of his patients developed invasive squamous carcinoma after 19 months of follow-up. Wentz and Reagan (1970) subsequently reported on 84 patients with “post-irradiation dysplasia.” Seventy-one of these patients developed the lesion within 3 years or less after completion of radiotherapy for invasive carcinoma of the cervix, whereas 13 patients developed the lesion 3 to 12 years after completion of therapy. Forty-seven (56%) of the 84 patients developed recurrent carcinoma and the majority of them died of disease. The probability of developing recurrent cancer was much higher for patients who developed the post-irradiation change within 3 years or less than for the patients with a delayed onset. The overall 5-year survival rate for the 84 patients was only 44%, although most of them initially had carcinomas of stage I (30 patients) and stage II (44 patients), wherein a much better survival rate could be expected for these stages of disease. This study fully confirmed the serious prognostic significance of the post-irradiation intraepithelial lesion, regardless of the name attached to it.
Okagaki et al (1974) studied 60 patients who received radiotherapy for carcinoma of the cervix of various stages. Twenty-three patients (38.5%) showed evidence of postirradiation lesions. The study of DNA content of the abnormal cells by destaining the slides and re-staining with Feulgen stain showed diploid, polypoid, or aneuploid patterns. Twenty-seven of the 60 patients died; 13 of these had “post-irradiation dysplasias,” 6 of which were aneuploid. Two of the 33 surviving patients also had aneuploid dysplasia. The conclusions of this paper, suggesting that DNA measurements are of prognostic value, have been confirmed by Davey et al (1992, 1998).
Regardless of the controversy over the name of the cytologic and histologic lesions observed following completion of radiotherapy for invasive cancer of the uterine cervix, it may be unequivocally stated that the presence of post-irradiation intraepithelial neoplasia carries with it a very serious prognostic connotation. The majority of these patients will die of invasive and metastatic carcinoma, unless rapidly treated.
The use of periodic cytologic examinations is mandatory following irradiation treatment of cervical cancer to detect local recurrences promptly and to treat them without delay. The early identification of post-irradiation intraepithelial neoplasia should lead to vigorous treatment of patients at risk.