Cardiovascular Disease
The clinical biochemistry laboratory often plays a central role in the diagnosis and management of acute myocardial infarction. A number of cardiovascular risk factors have been described, some of which can be assayed in the biochemistry laboratory, and these are discussed in this chapter.
MYOCARDIAL INFARCTION
One of the largest killers in Western urbanized societies is acute myocardial infarction. Its diagnosis is usually made on the clinical presentation and electrocardiographic findings and confirmed by the characteristic changes in plasma enzyme activities or troponin levels. Myocardial infarction can be defined as a typical rise and/or fall of cardiac biomarkers (preferably troponin) with at least one value above the 99th percentile of the upper reference limit together with evidence of ischaemia with at least one of the following: symptoms of ischaemia, electrocardiogram (ECG) changes (new ST changes or new left bundle branch block), development of ECG pathological Q waves or imaging evidence of myocardial ischaemia. (Fig. 22.1). This classification
also recognizes that patients previously classified as having unstable angina or minor myocardial injury are now reclassified as having non-ST segment elevation myocardial infarction (NSTEMI).
also recognizes that patients previously classified as having unstable angina or minor myocardial injury are now reclassified as having non-ST segment elevation myocardial infarction (NSTEMI).
CASE 1
A 46-year-old man was admitted late at night (23.30 h) to casualty by ambulance because of a tight central chest pain that had started at 03.00 h that morning. He had delayed calling for medical assistance because he thought it was only indigestion. In the ambulance, the paramedics gave him an intramuscular injection of diamorphine for pain relief. The ECG was normal.
Plasma
Creatine kinase (CK) 565 U/L (< 250)
Troponin T 1.0 ng/L (< 10)
DISCUSSION
The normal plasma troponin T makes a diagnosis of acute myocardial infarction unlikely, as this would be expected to be elevated in view of when the chest pain started. The elevated plasma CK concentration is not specific for cardiac damage and may have been raised as a result of muscle damage secondary to the intramuscular injection: beware of interpreting elevated plasma CK concentrations in the presence of intramuscular injections. A CK-MB concentration would be less likely than total CK concentration to be elevated in skeletal muscle damage, as CK-MB is predominantly cardiac in origin.
CASE 2
A 66-year-old man was sent by his general practitioner to casualty because of tight chest pain that had occurred 3 days previously. The pain had largely resolved after 6 h, but he was left feeling weak and breathless, which worsened over a few days, causing him eventually to seek medical attention. The following laboratory test results were found:
Plasma
Creatine kinase (CK) 235 U/L (< 250)
Troponin T 13.0 ng/L (< 10)
An electrocardiogram showed changes suggestive of a myocardial infarction in the lateral leads V4-V6.
DISCUSSION
The plasma CK activity has returned to normal because of the time delay since myocardial infarction, while the plasma troponin T concentration still remains elevated. Plasma CK usually starts to rise 4-6 h after a myocardial infarction and to normalize after a couple of days. Conversely, plasma troponin T starts to rise at 4-6 h post infarct and remains elevated for as long as about 10 days. Troponins are thus useful cardiac markers in both the early hours and a few days later.
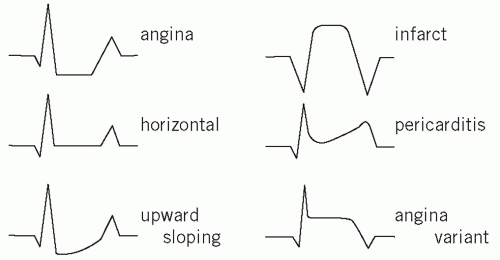 Figure 22.1 ECG changes; showing ST depression as in ischaemic angina pectoris and ST elevation after a myocardial infarction. Adapted with kind permission from Kinirons M and Ellis H. French’s Index of Differential Diagnosis, 15th edition. London: Hodder Arnold, 2011. |
It is useful to note that acute myocardial infarction and NSTEMI have a common pathophysiological pathway, namely acute coronary artery plaque rupture, followed by a series of events resulting in thrombosis formation. The outcome is dependent on the state of the collateral circulation and the severity of artery occlusion (Fig. 22.2). In other words, clinically we no longer think in terms of the presence or absence of an acute myocardial infarction, but instead of a spectrum of disease ranging from angina pectoris through to acute myocardial infarction; this stratification is based upon cardiac markers reflecting ischaemic damage. Acute coronary syndrome is the term used to embrace ST segment elevation myocardial infarction (STEMI), as well as NSTEMI and unstable angina pectoris
It is also particularly important to diagnose acute myocardial infarction promptly, as thrombolytic therapy may be indicated to try to dissolve the thrombus in situ or angioplasty. The earlier this therapy is given, the better the prognosis, as ‘minutes are myocardium’. Thus, biochemical markers need to be specific, sensitive and change rapidly to optimize the diagnosis and treatment of acute myocardial infarction.
Troponins
Troponins are muscle-regulatory proteins present in skeletal and cardiac muscle. Three troponins have been reported, namely troponin C (TnC), troponin I (TnI) and troponin T (TnT). There are no structural differences between cardiac and skeletal muscle TnC. However, the cardiac and skeletal forms of TnI and TnT are structurally different and can be distinguished by immunological assays.
Troponin I and TnT appear in the plasma 4-8 h or earlier with high-sensitivity troponin assays after symptoms of acute myocardial infarction, and are best measured 12 h after the start of chest pain. They are therefore not early markers of acute myocardial infarction, but they do stay elevated for about 7-10 days in plasma, which makes them useful in the late presentation of chest pain.
Troponin T may be elevated in patients with chronic kidney disease and thus may not be so cardiospecific. However, TnI can be extensively modified by proteolysis after release into the plasma. This means that measurements may give varying results as different antigenic epitopes are revealed, depending upon the commercial supplier of the assay. There is no universal agreement as to which troponin assay, i.e. TnI or TnT, is best.
An increased TnI or TnT concentration is a sensitive marker of occult myocardial damage even in nonischaemic conditions. Plasma troponin concentrations are increased in subarachnoid haemorrhage (due to vasoactive peptide release affecting the myocardium),
hypertension, tachyarrhythmias, cardiac surgery, sepsis, congestive cardiac failure, pulmonary embolism and hypothyroidism. It should be remembered that the universal definition of an acute myocardial infarction requires not only a change in troponin but also clinical or ECG evidence.
hypertension, tachyarrhythmias, cardiac surgery, sepsis, congestive cardiac failure, pulmonary embolism and hypothyroidism. It should be remembered that the universal definition of an acute myocardial infarction requires not only a change in troponin but also clinical or ECG evidence.
Research has shown that troponins allow a subset of patients with rest-unstable angina to be classified despite normal plasma CK-MB concentrations. Those with raised plasma cardiac troponin concentrations were found to be more likely to have an acute myocardial infarction or die within 6 months of the event. In other words, troponins allow risk stratification of patients with acute coronary syndrome. In another study it was found that, unlike CK-MB, cardiac troponins have prognostic value after coronary thrombolysis therapy, with troponin concentrations on admission correlating with 6-week mortality. Low-molecular-weight heparin or plateletreceptor blockers (glycoprotein GIIB/IIIA inhibitors) reduce cardiac events in non-Q-wave coronary syndromes with raised concentrations of cardiac troponins compared with those without troponin concentration elevation. There are also new sensitivity troponin assays which may allow earlier diagnosis and better risk stratification.
In view of these findings, cardiac troponins have been recommended as the biochemical cardiac marker of choice. However, sometimes a panel of cardiac markers may be useful in making the diagnosis of acute myocardial infarction, for example myoglobin rises early, thus alerting the clinician to possible cardiac problems, while the later rise of troponin allows greater specificity. Other cardiac markers that are being studied include ischaemia-modified albumin, pro-brain natriuretic peptide (pro-BNP) and glycogen phosphorylase isoenzymes BB.
Myoglobin
As mentioned in Chapter 21, myoglobin is a lowmolecular- weight haem-containing protein found in both skeletal and cardiac muscle. Because of its low molecular weight, it is rapidly released from the myocardium upon damage, and a typical rise occurs within 2-4 h after the onset of acute myocardial infarction. This is useful for the early diagnosis of acute myocardial infarction, as this rise is generally earlier than that of the other currently used cardiac markers. Unfortunately, myoglobin is not cardiac specific, being also found in skeletal muscle, and thus is less useful in the diagnosis of acute myocardial infarction unless used in conjunction with other markers.
Cardiac enzymes
The plasma enzyme estimation of greatest value is that of CK (see also enzymology, Chapter 18), although this has been largely superseded by the assay of cardiac troponins. At one time, the enzymes lactate dehydrogenase (LDH) or hydroxybutyrate dehydrogenase (HBD) and aspartate aminotransferase (AST) were used diagnostically, although they are now rarely assayed in the diagnosis of acute myocardial infarction or NSTEMI.
An approximate guide to the sequence of changes after acute myocardial infarction is given in Table 22.1 (see also Fig. 22.3).
All plasma enzyme activities (including that of CKMB) may be normal until at least 4 h after the onset of chest pain due to a myocardial infarction; blood should not be taken for enzyme assay until this time has elapsed. If the initial plasma CK activity is normal, a second sample should be taken about 4-6 h later. A rise in the plasma CK activity supports the diagnosis of an infarction. The simultaneous measurement of plasma CK-MB activity, which is shown to exceed about 5 per cent of the total CK activity, may occasionally help in early diagnosis; a raised plasma CK-MB activity or concentration alone is not diagnostic of an infarction.
Most of the CK released after a myocardial infarction is, in fact, the MM isoenzyme (CK-MM), which is found in both skeletal and myocardial muscle and has a longer half-life than the MB fraction. After about 24 h, the finding of a high MM and undetectable MB does not exclude myocardial damage as a cause of high total CK activities; by this time the plasma HBD activity is usually raised.
Table 22.1 The time sequence of changes in plasma cardiac markers after acute myocardial infarction | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree