1. Explain the routes of transmission for Bartonella infections, and describe the organism’s interaction with the host. 2. Discuss the clinical manifestations of Trench fever, including signs, symptoms, and individuals at risk of acquiring the disease. 3. Explain the criteria used to diagnose Bartonella henselae. 4. Describe the two methods for culturing Bartonella, including growth rates, media, incubation temperature, and other relevant conditions. 5. Explain why the sensitivity and specificity has been questioned with indirect fluorescent antibody and enzyme-linked immunoassay testing. 6. Describe the strategies to prevent exposure and infection by these organisms in immunocompromised individuals. Bartonella spp. were previously grouped with members of the family Rickettsiales. However, because of extensive differences, the family Bartonellaceae was removed from this order. As a result of phylogenetic studies using molecular biologic techniques, the genus Bartonella currently includes 22 species and subspecies, most of which were reclassified from the genus Rochalimeae and from the genus Grahamella. Only five species are currently recognized as major causes of disease in humans (Table 33-1), but other members of the genus have been found in animal reservoirs such as rodents, ruminants, and moles. Bartonella spp. are most closely related to Brucella abortus and Agrobacterium tumefaciens and are short, gram-negative, rod-shaped, facultative intracellular, fastidious organisms that are oxidase negative and grow best on blood-enriched media or cell co-culture systems. TABLE 33-1 Organisms Belonging to the Genus Bartonella and Recognized to Cause Disease in Humans* Note: Other Bartonella species have caused incidental infections in humans, but only one or a few cases have been documented. *Disease confined to a small endemic area in South America; characterized by a septicemic phase with anemia, malaise, fever, and enlarged lymph nodes in the liver and spleen, followed by a cutaneous phase with bright red cutaneous nodules, usually self-limited. Organisms belonging to the genus Bartonella cause numerous infections in humans; most of these infections are thought to be zoonoses. Interest in these organisms has increased because of their recognition as causes of an expanding array of clinical syndromes in immunocompromised and immunocompetent patients. For example, Bartonella species have been recognized with increasing frequency since the early 2000s as a cause of culture-negative endocarditis. Humans acquire infection either naturally (infections caused by Bartonella quintana or Bartonella bacilliformis) or incidentally (other Bartonella species) via arthropod-borne transmission. Nevertheless, questions remain regarding the epidemiology of these infections; some epidemiologic information is summarized in Table 33-1. The diseases caused by Bartonella species are listed in Table 33-1. Because B. quintana and B. henselae are more common causes of infections in humans, these agents are addressed in greater depth. B. henselae is associated with bacteremia, endocarditis, and bacillary angiomatosis. Of note, recent observations indicate that B. henselae infections appear to be subclinical and are markedly underreported, as problems with current diagnostic approaches are recognized (see Laboratory Diagnosis). In addition, B. henselae causes CSD and peliosis hepatitis. About 24,000 cases of CSD occur annually in the United States; about 80% of these occur in children. The infection begins as a papule or pustule at the primary inoculation site; regional tender lymphadenopathy develops in 1 to 7 weeks. The spectrum of disease ranges from chronic, self-limited adenopathy to a severe systemic illness affecting multiple body organs. Although complications such as a suppurative (draining) lymph node or encephalitis are reported, fatalities are rare. Diagnosis of CSD requires three of the four following criteria:
Bartonella and Afipia
Bartonella
General Characteristics
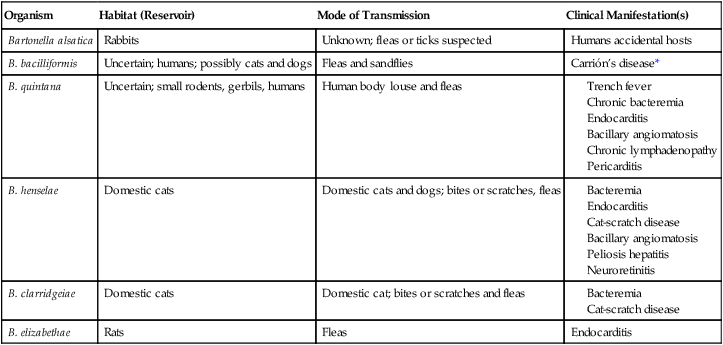
Organism
Habitat (Reservoir)
Mode of Transmission
Clinical Manifestation(s)
Bartonella alsatica
Rabbits
Unknown; fleas or ticks suspected
Humans accidental hosts
B. bacilliformis
Uncertain; humans; possibly cats and dogs
Fleas and sandflies
Carrión’s disease*
B. quintana
Uncertain; small rodents, gerbils, humans
Human body louse and fleas
B. henselae
Domestic cats
Domestic cats and dogs; bites or scratches, fleas
B. clarridgeiae
Domestic cats
Domestic cat; bites or scratches and fleas
B. elizabethae
Rats
Fleas
Endocarditis
Epidemiology and Pathogenesis
Spectrum of Disease
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Bartonella and Afipia
