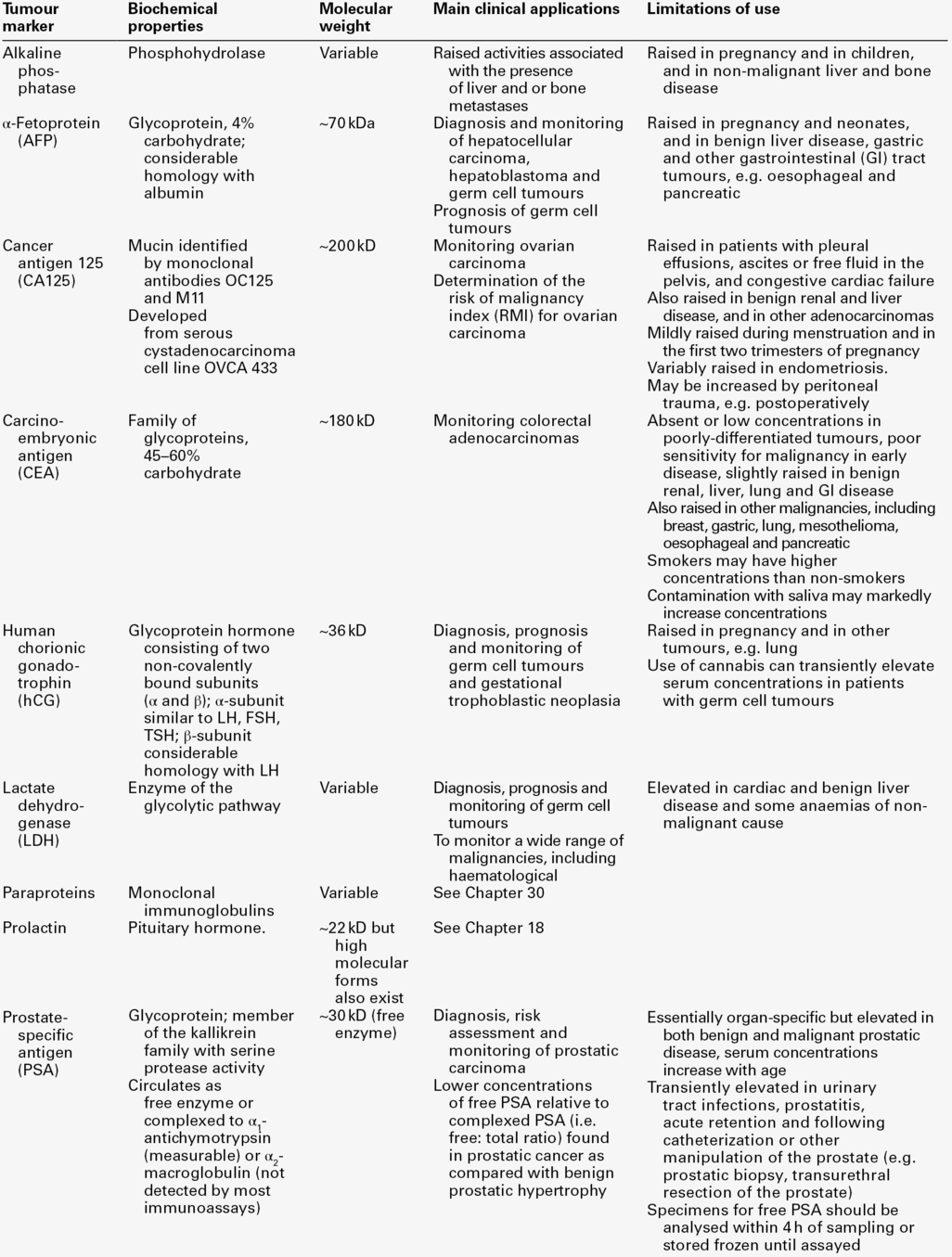
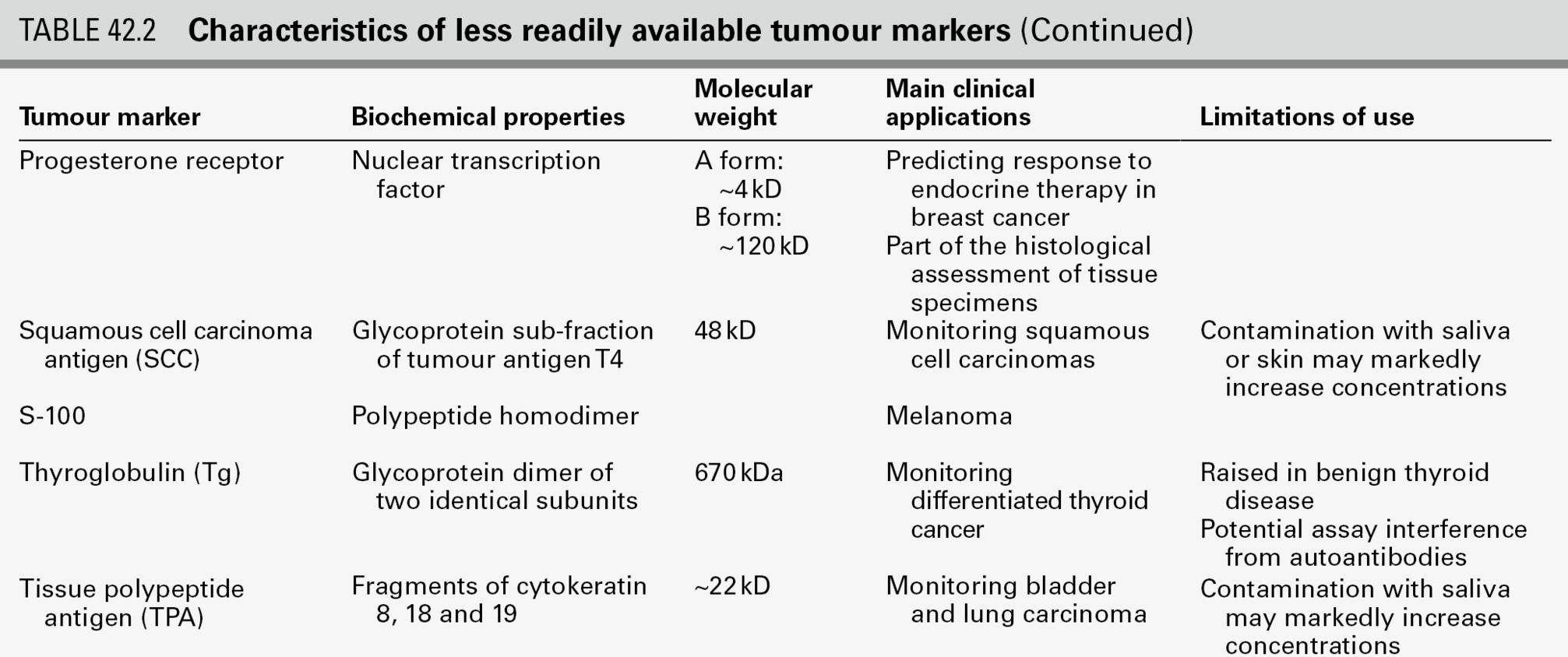
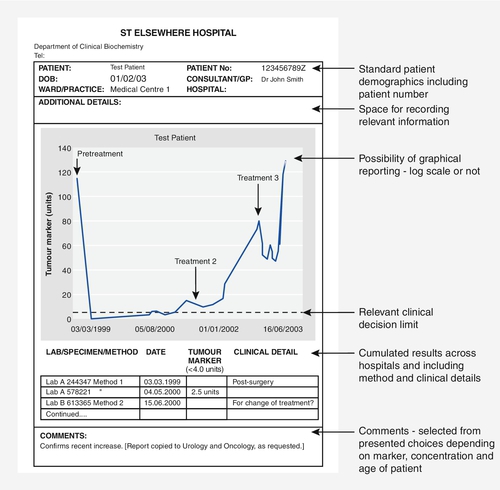
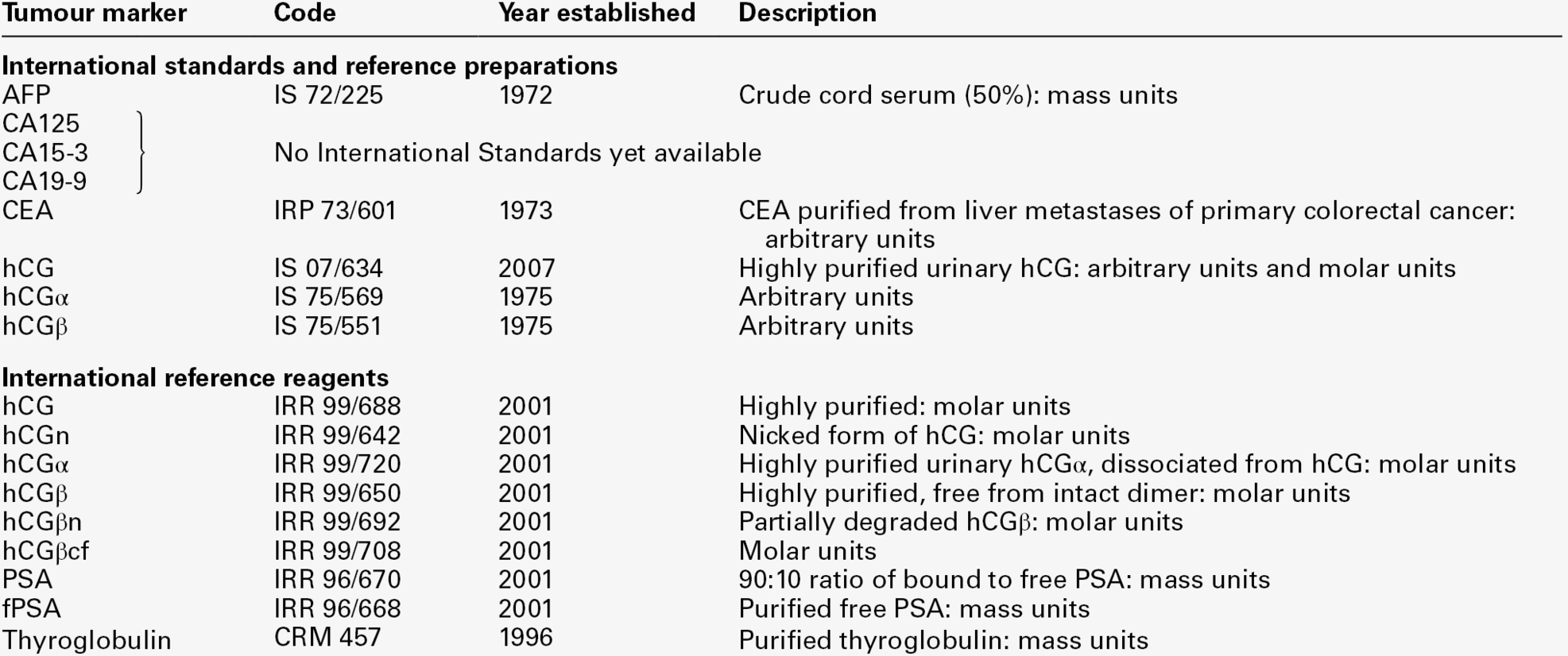
CHAPTER 42 CHAPTER OUTLINE Evaluation of the clinical utility of tumour markers Tumour marker requests and the responsibilities of the clinical laboratory TUMOUR MARKERS IN THE MANAGEMENT OF SPECIFIC CANCERS Gastrointestinal stromal tumours (GIST) Gestational trophoblastic neoplasia Hepatocellular carcinoma (primary liver cancer) Neonatal and paediatric tumours Tumour markers are substances (often proteins, enzymes or hormones) that are present in body fluids or tissues and whose measurement provides information about the presence, progression or remission of tumours. They may be tumour-derived (produced within the tumour by malignant or stromal cells) or tumour-associated (produced by non-malignant cells as a metabolic consequence of tumour presence). Some are tumour-specific (produced by cancerous but not normal tissue), while others are present in normal tissue but are produced at higher concentrations in body fluids or malignant tissue from cancer patients. A few are organ-specific but many are produced by a variety of different cancers. Histopathological identification of tumour markers expressed at the tumour cell surface and detectable in biopsy specimens can provide both diagnostic and prognostic information, with genetic tests becoming increasingly important in predicting which patients are most likely to respond to costly new therapies. Secreted tumour markers, present in blood or other body fluids, are measured quantitatively, often by immunoassay. While knowledge of circulating marker concentrations may assist in diagnosis and prognosis, their most important clinical use is in monitoring the success of treatment. This chapter focuses primarily on the properties and clinical applications of the tumour marker tests that usually fall within the remit of clinical biochemistry laboratories (Tables 42.1 and 42.2), with the aim of enabling readers to provide advice about which tumour marker tests are most likely to be helpful, to recognize which results require immediate action, and to develop a high-quality interpretative tumour marker service. Before considering these aspects, however, it is useful to review some recent developments relevant to the introduction of tumour markers into routine clinical practice. Abbreviations used in the chapter for the names of individual tumour markers are explained in Tables 42.1 and 42.2. Although hundreds of potential tumour markers have been investigated, with more than 200 000 papers describing them, the number that contribute significantly to the management of cancer patients is remarkably small, as is evident in Tables 42.1 and 42.2. Historically, over-enthusiastic reporting of small, poorly designed studies of new markers has led to disillusionment when the results have not been confirmed in larger studies or in other centres. Together with the difficulties inherent in comparing different studies reported in non-standard form, this has made objective assessment of the utility of many tumour markers very difficult and has meant that the recommendations made by expert panels about their clinical use tend to be conservative. The three critical factors that should be considered when evaluating a tumour marker are its clinical utility, the magnitude of the benefit of its use and its reliability. Tumour marker measurement can provide information at multiple stages of diagnosis and treatment (Table 42.3) but, until appropriate studies have been performed, it can be difficult to know how best to use a tumour marker in particular circumstances. Having identified a specific use (i.e. utility), the clinical value (i.e. magnitude) of using the tumour marker for that application needs to be assessed, by evaluating the difference in outcome between marker-positive patients and marker-negative patients, ideally in a randomized controlled trial designed for the purpose. Clinical precision and accuracy (i.e. reliability) then need to be established, since the marker will only be useful if results are reproducible. In this context, it is important to note that, although it is necessary to demonstrate a statistically significant difference in tumour marker concentrations between patient groups to show that a marker may have potential utility, this is not in itself sufficient evidence that a marker is of clinical benefit (i.e. that it should be used), but merely suggests that the differences observed are not likely to occur by chance. It is also essential to ensure that biochemical analysis of the marker is reliable and reproducible, that assays are standardized and that their analytical and clinical performance is tested objectively in appropriately designed and well-conducted studies. TABLE 42.3 Examples of applications of tumour marker measurements at different stages of diagnosis and treatment The Tumor Marker Utility Grading System (TMUGS), developed some years ago, provides a useful framework for such evaluation and it is encouraging that the recommendations made therein have been implemented for some analytes, e.g. by improvements in the standardization of immunohistochemical assays for HER-2 and in the analytical accuracy and equimolarity of serum assays for PSA. The TMUGS also describes levels of evidence for grading the clinical utility of tumour markers, an approach that has been adopted by the US National Academy of Clinical Biochemistry (NACB), which has developed Laboratory Medicine Practice Guidelines for the use of tumour markers in the clinic. Other initiatives include development of Reporting Recommendations for Tumor Marker Prognostic Studies (REMARK), which provide guidance about study design, pre-planned hypotheses, patient and specimen characteristics, assay methods and statistical analysis methods and complement the broader statements on Consolidated Standards of Reporting Trials (CONSORT) and Standards for Reporting of Diagnostic Accuracy (STARD). These should help to encourage improved design and publication of tumour marker studies in the future. Knowledge of tumour marker results can contribute useful clinical information relating to different aspects of patient management (Table 42.3). For use in screening and diagnosis, the ‘perfect’ tumour marker would be absent in all healthy subjects (100% specificity), would be raised in all patients with a single tumour type (100% sensitivity) and its serum concentration would accurately reflect tumour size. Unfortunately, the ‘perfect’ marker does not exist and the extent to which currently available markers meet this ideal is very variable. Consequently, the predictive value of a positive or negative result also varies and is highly dependent on the population considered (see Chapter 2 for detailed discussion). There is increasing interest in improving the specificity of some markers (e.g. AFP, CA125 and PSA) by using serial measurements to assess the rate of change in the concentration of the marker with time in individual patients. Such measurements may contribute to diagnosis, but in practice, are more likely to aid post-treatment monitoring, which is the most established clinical role for tumour markers. Whether such monitoring improves patient outcome depends on the availability of further treatment options, should increases in marker concentrations indicate progressive disease. If potentially curative treatment is available, as for example is frequently the case for patients with recurrent germ cell tumours, it is essential to identify patients with rising tumour marker concentrations promptly; failure to do so may constitute a critical clinical error. However, if alternative therapy is not available, knowledge of progressive disease before it is clinically evident may not benefit the patient and can have adverse psychological consequences, in which case it may be advisable to discontinue tumour marker monitoring. Serial monitoring is also desirable for patients with cancers that are unlikely to progress and for which treatment is not necessary or can be delayed: active surveillance (sometimes called ‘watchful waiting’) programmes, including serial PSA measurements, are appropriate for some men with indolent prostate cancers. In practice, the clinical biochemistry laboratory receives three types of tumour marker requests: those for patients with diagnosed malignancy who have already been referred to specialist centres; those for patients being investigated for suspected malignancy in secondary care, and those for patients presenting to their general practitioners. Requests in the first group are the most likely to be appropriate and are usually made to clarify the patient’s diagnosis or to monitor response to treatment and/or detect recurrence. Non-specialist users, whether in hospital or general practice, should be encouraged to consider carefully whether knowledge of a tumour marker result is likely to be helpful before requesting it. Requests such as ‘tumour marker screen’ or ‘?malignancy’, particularly from emergency departments and other receiving units, should be actively discouraged. Since it is not usually practicable for requests to be scrutinized prior to analysis, laboratories should provide comprehensive advice about appropriate choice of tumour markers, as well as reminders of their limitations. Most tumour markers do not have sufficient sensitivity or specificity, particularly for early stage disease, to be regarded as diagnostic tests, although they may contribute to diagnosis. Importantly, whatever the malignancy or tumour marker, a result within the reference interval never excludes malignancy or progressive disease. Requestors should be aware that the converse is also true. Raised tumour marker concentrations do not necessarily indicate malignancy, as they may be increased in a number of benign conditions. Increased concentrations may be associated with more than one tumour type, since, with few exceptions, tumour markers are not organ-specific. Clinical biochemists themselves should also think carefully before requesting any additional testing which may lead to a diagnosis of malignancy and before doing so should seek the agreement of the doctor managing the patient. General advice that can be readily disseminated to non-specialist users has recently been prepared by Pathology Harmony UK. Although the timing of blood sampling is not usually critical, a pre-treatment specimen is helpful when interpreting subsequent results. Specimens should always be taken before any investigative procedure, since some of these may cause transient releases of tumour markers into the circulation (e.g. increases of PSA following insertion of a urinary catheter or prostatic biopsy, of CA125 following abdominal surgery, and of CEA following colonoscopy). Possible conditions, which can transiently affect tumour marker concentrations (e.g. marked increases of PSA in men with active urinary tract infections or of CA19.9 in patients with cholestasis) should be excluded. Failure to recognize that misleadingly high results can be obtained if sampling times are inappropriate may cause undue distress to the patient, as well as decreasing confidence in laboratory testing. Some additional examples are listed in Tables 42.1 and 42.2. Manufacturers’ instructions should always be followed when performing tumour marker measurements, with carefully defined internal quality control (IQC) and external quality assessment (EQA) procedures in place to monitor performance. Internal quality control and EQA specimens should closely resemble patient sera and be of clinically relevant concentrations, including those near important decision limits. Regular assessment of reproducibility and stability of results with time is particularly important for all tumour markers, as these are often monitored over long periods. Ensuring good reproducibility at low concentrations is critical where treatment may be instituted solely on the basis of a relatively small increase in tumour marker concentration, as is the case for AFP and hCG in germ cell tumours and for PSA following prostatectomy for prostate cancer. Major international efforts are being directed towards improved comparability of tumour marker methods by encouraging accurate calibration against the relevant international standards where these exist (Table 42.4), by producing reference reagents to enable improved characterization of assays, by organizing collaborative workshops to identify the most clinically appropriate antibody specificities and by encouraging use of equimolar assays where relevant (e.g. PSA assays that recognize free and complexed PSA equally well). Nevertheless, the molecular heterogeneity of most tumour markers (as illustrated by the number of hCG-related molecules in Table 42.4) means that results obtained with different methods are not interchangeable. Clinical biochemists should be aware of the characteristics of the methods used in their own laboratory. They should also be familiar with the vulnerability of their methods to potential interferences such as the high-dose hook effect, specimen carry-over and interference from heterophilic or human anti-mouse antibodies (Box 42.1). TABLE 42.4 International standards (IS), reference preparations (IRP) and reference reagents (IRR) for tumour markers Most currently available immunoassays are calibrated against the relevant standards where they exist. The lack of international standards for the CA antigens is a major hindrance to improved between-method comparability. hCGα, hCG α-subunit; hCGβ, hCG β-subunit; hCGn, nicked hCG; hCGβn, nicked hCG β-subunit, hCGβcf, hCG β-core fragment. International Standards and Reference Reagents are available from the National Institute for Biological Standards and Control, Potters Bar, Herts, UK: http://www.nibsc.ac.uk/catalog/standards/preps/sub_endo.html Cumulative and/or graphical reporting of serial results can identify trends in marker concentrations, which are generally more informative than single values, and may alert the laboratory to unexpected results (e.g. sudden changes) that require further investigation. Recording brief clinical information (e.g. ‘postoperative’), preferably both in the laboratory computer and on any printed reports, can be helpful both in identifying results that are out-of-accord and when interpreting results. In view of method-related differences in tumour marker results, it is recommended that the method used is stated on the clinical report. If there has been an intervening method change, it is highly desirable that the laboratory also indicates whether this is likely to have affected interpretation of the trend in results. Reference intervals specific to the method used should be provided, although for serial monitoring, the patient’s own baseline provides the most important reference point for future results. Laboratory and clinical staff should engage in active dialogue about appropriate clinical decision points (e.g. when using PSA measurements to select patients for biopsy) and should know how these limits were derived. Laboratories can increase the value of their reports by adding short interpretative comments relating to the analytical results and preferably individually tailored to the requestor (e.g. omitting these for specialist users). Tumour marker half-lives, which are defined as the time to 50% reduction of circulating tumour marker concentrations following complete removal of tumour tissue, provide an important measure of the efficacy of therapy, particularly for germ cell tumours, and it may be helpful for the laboratory to calculate these for AFP and hCG (see p. 833 for further information). Providing advice about whether a change in marker concentration is likely to be significant or not is also helpful and should take into account both biological and analytical variation. A confirmed increase of 30%, or two serial increases of 20–25%, are often considered to be of clinical significance. Recommendations about the need for confirmatory specimens and the desirable frequency of tumour marker measurements are also helpful. The clinical biochemist should identify urgent results that may be required for immediate patient management and ensure that these reach the relevant clinician promptly, telephoning results when appropriate. Results in this category include those which can be used to diagnose advanced disease in critically ill but treatable patients (e.g. AFP in hepatoblastoma; hCG in choriocarcinoma; AFP and hCG in non-seminomatous germ cell tumours; PSA in men with advanced prostate cancer that may respond to endocrine therapy). Provision of a proactive and high-quality tumour marker service helps to encourage good communication between laboratory and clinical staff, and is likely to facilitate both appropriate use of tumour marker tests and early identification of any results that are not in accord with the clinical picture. An example of a laboratory report meeting many of these requirements is shown in Figure 42.1. FIGURE 42.1 Possible template for a clinical laboratory report for tumour markers that fulfil current reporting recommendations. Regularly updated national guidelines on the management of the majority of tumours are now widely available, often provided electronically on the worldwide web. Regional cancer networks are also a valuable source of information and have often developed modified versions of national guidelines tailored for local use. The optimal use of tumour markers for assessment of prognosis, monitoring treatment and detecting recurrent disease has been studied in most detail for choriocarcinoma and germ cell tumours, relatively rare diseases for which tumour marker measurements are mandatory for clinical management. Measurements of serum tumour markers also contribute significantly to the management of some of the more common malignancies (e.g. ovarian, colorectal and prostate), while they are less widely used in others (e.g. bladder, breast and lung). In the following sections, the extent to which tumour markers currently contribute to the clinical management of a number of important malignancies is briefly reviewed. The most common symptom of bladder cancer is intermittent haematuria, which is present in 80–85% of patients. The majority of bladder cancers are transitional cell carcinomas but adenocarcinomas, squamous cell carcinomas and sarcomas also occur. In some patients, urine cytology is positive for tumour cells, but the diagnosis is usually established by cystoscopic evaluation. Urine cytology is very effective in detecting high-grade bladder cancers but will miss the majority of papillary urothelial neoplasms of low malignant potential. Commercially available assays for two tumour markers present in urine have been approved by the United States Food and Drug Administration (FDA) for the detection of recurrent bladder cancer. The BTA (bladder tumour associated antigen) -Trak™ and -Stat™ tests detect complement factor H and related proteins, which are involved in the regulation of the alternative pathway of complement activation to prevent complement-mediated damage to healthy cells. A point of care version of this test is also available. The nuclear matrix protein 22 (NMP22™) test is a quantitative measure of the nuclear mitotic apparatus protein, a component of the nuclear matrix which is over-expressed in bladder cancer. Both these tests are more sensitive than cytology in detecting low grade bladder cancers but are less specific, and their high false positive rates limit their clinical application. Better specificity has been achieved with a fluorescence in situ hybridization assay (UroVysion™), which detects bladder cancer-associated aneuploidy of selected chromosomes and which has been approved by the FDA for screening patients for recurrent bladder cancer. Breast cancer is by far the most common cancer affecting women worldwide with approximately one million new cases diagnosed each year. The main presenting features in women with symptomatic breast cancer include a lump in the breast, nipple change or discharge and skin contour changes. Currently available blood-based biomarkers are of no value in the early diagnosis of symptomatic or asymptomatic breast cancer, the latter being addressed by national screening programmes using mammography. Individuals who are at increased risk of breast cancer because they are carriers of the BRCA1, BRCA2 or TP53 genetic mutations and those with a strong family history of breast cancer (e.g. close relatives diagnosed with breast cancer at a young age) may be eligible for screening with magnetic resonance imaging (MRI), as is recommended in the UK by the National Institute for Health and Care Excellence (NICE). Definitive diagnosis requires biopsy and histopathology. Measurement of oestrogen-binding receptors (ER) and progesterone-binding receptors (PR) in tumour biopsy tissue obtained at diagnosis is mandatory in order to determine the likely response to endocrine therapy of both early and advanced (metastatic) breast cancer. Early ligand-binding and enzyme-linked immunosorbent assay methods for measuring ER and PR have been superseded by immunohistochemical assessment, which can be performed on paraffin-fixed tissue sections of the smaller tumours now detected by screening. Immunohistochemical or fluorescence in-situ hybridization tissue measurement of HER-2 (or its gene), a glycoprotein that controls cell growth and is amplified in ~ 30% of early-stage breast cancers, is now essential in all newly diagnosed patients with breast cancer. Patients with tumours that do not produce HER-2 are not likely to benefit from treatment with trastuzumab (Herceptin®), a humanized monoclonal antibody against HER-2, either administered on its own or in addition to other chemotherapy treatment. An assay is available for the measurement of the soluble shed form of HER-2 in serum and has potential value, both for prognosis and for monitoring trastuzumab therapy in patients with advanced breast cancer. However, the serum assay is not widely used. CA15-3, a high molecular weight mucin, and similar MUC-1 based glycoprotein markers (e.g. BR27-29) may be used to monitor response to treatment, rising serum concentrations providing early indication of progression in some patients. Their routine use is not currently recommended, since there is as yet no evidence that therapeutic intervention prior to radiological or clinical detection of recurrent tumour is beneficial. However, in individual patients, measurement of CA15-3 may help to determine whether bone symptoms are due to benign or malignant disease, as high concentrations are frequently associated with metastatic disease. CA15-3 may also be useful in monitoring response to therapy in advanced disease if there are no other indicators of response and if rising concentrations would be an indication to stop all but palliative treatment. As with other tumour markers, the low sensitivity of CA15-3 means that results within the reference interval do not exclude active disease or progression. Worldwide, cervical cancer is the major cause of death from gynaecological cancer, with reported incidence rates in developing countries much higher than those in developed countries. Since cervical cancer progresses slowly from pre-invasive cervical intraepithelial neoplasia to invasive cancer, screening asymptomatic women with regular smears provides an effective means of early detection. The addition of human papilloma virus (HPV) testing appears to improve the effectiveness of screening as certain types of HPV are involved in the development of cervical cancer. Women who have not been screened may present with symptoms of abnormal vaginal bleeding and, in advanced cases, pelvic pain and pressure symptoms relating to the bowel or bladder. Treatment of early stage disease usually requires radical hysterectomy and pelvic lymphadenectomy, with adjuvant radiation therapy if disease has spread to the lymph nodes. Surgery and concomitant chemoradiation or neoadjuvant chemotherapy followed by radical surgery are options for bulky disease. Measurement of plasma CEA and CA125 may have clinical utility in patients with cervical adenocarcinomas, which constitute 10–15% of all cervical cancers. The majority of cervical cancer patients (~ 85%), however, have squamous cell cervical carcinomas, for which the potentially most useful serum tumour marker is squamous cell carcinoma antigen (SCC), a serine protease inhibitor. The low sensitivity and specificity of SCC, particularly for early stage disease, preclude its use in screening or diagnosis, which requires immunohistological evidence. Although 60% of patients with cervical cancer will have elevated concentrations of SCC at diagnosis, raised values are also found in other squamous cell carcinomas (e.g. lung, oesophagus and head and neck) as well as in benign diseases (e.g. psoriasis, eczema, sarcoidosis). Some studies suggest that a raised concentration of SCC is an independent risk factor for poor survival but other studies contradict this. It has also been suggested that pre-treatment SCC concentrations may be helpful in stratifying patients at greater risk of recurrence and therefore requiring more intensive therapy, but formal trials will be necessary to confirm this. Various studies have confirmed that SCC shows a strong correlation with the clinical course of cervical cancer, with a lead time of up to 14 months in detecting progression prior to the onset of clinical symptoms, but whether earlier detection improves treatment outcome is not known. As only 10% of patients with recurrent disease can be cured and most patients with recurrent disease have clinical symptoms, until treatment options improve it is unlikely that SCC measurement will be widely adopted. See Gestational trophoblastic neoplasia, below. Worldwide, colorectal cancer (CRC) is the third most common cancer, with more cases in the developed world than in Africa and Asia. Its incidence increases as populations adopt a western style diet. The risk of recurrence and subsequent death from CRC is closely related to the stage of the disease at the time of the primary operation. Although the treatment of colon and rectal cancer differ, they can be considered together in terms of tumour marker measurement. Individuals at high risk of developing CRC (e.g. those with hereditary non-polyposis colon cancer (HNPCC, Lynch syndrome), familial adenomatous polyposis (FAP) or a strong family history of CRC (e.g. first degree relative with CRC diagnosed before age 45) should be referred to a clinical genetics unit for risk assessment and endoscopic screening. Mismatch repair gene mutations (e.g. MLH1, MSH2) predispose individuals to additional tumours, including those of the endometrium, ovary, genitourinary tract, small bowel and biliary tract. Individuals with FAP are at increased risk of developing duodenal tumours and thyroid tumours. Early detection of CRC using faecal occult blood testing (FOBT) has been shown to reduce mortality in several randomized controlled trials. A number of countries now provide screening programmes for defined age groups (often > 60 years old in the general population and at younger ages in high-risk groups) based on FOBT. Such screening is undertaken biannually in the UK, currently using a guaiac test, which detects the pseudoperoxidase activity of either intact or free haemoglobin. The test has many limitations, including relatively low clinical sensitivity and specificity for CRC; lack of specificity for human haemoglobin; vulnerability to interference from some foodstuffs and medications and difficulties associated with automating it. Screening using guaiac-based methods is therefore gradually being replaced by faecal immunochemical tests, which detect the globin component of haemoglobin, can be used quantitatively with an adjustable cut-off concentration and are generally superior. All FOBTs lack specificity, and positive screens must be followed-up with colonoscopy. Markers based on DNA detection are potentially more specific than FOBT and may ultimately replace them, provided a clinically cost-effective panel of markers can be identified. At present, however, the cost and technical difficulties associated with these assays preclude their adoption. Although CEA is the most frequently used marker for CRC, depending on the cut-off point chosen, serum CEA will be raised in only 30–50% of CRC patients at the time of diagnosis. Its specificity is also low, since CEA concentrations can be raised in benign liver and kidney disease as well as in other malignancies (e.g. breast, gastric, lung, mesothelioma, oesophageal and pancreatic cancers), and may also be raised in smokers. Therefore, CEA cannot be used in isolation to diagnose even advanced CRC. However, raised concentrations can aid in diagnosis in certain clinical circumstances (e.g. indicating a high probability of malignancy in a frail elderly patient who cannot undergo invasive investigations) and confirmed markedly raised concentrations (serum CEA > 40 μg/L) are suggestive of metastatic disease. The strongest predictor of recurrence of CRC is the histopathological stage of disease, as assessed using the Dukes staging system or one of its modified versions (Table 42.5), which take into account local tumour invasion, involvement of regional lymph nodes and presence of distant metastases. The five-year survival for patients with Dukes Stage A disease is > 90% but that for those with Stage D disease is < 10%.
Tumour markers
INTRODUCTION
Evaluation of the clinical utility of tumour markers
Application
Examples
Assessment of risk
PSA: used with or without digital rectal examination to assess risk of prostate cancer and determine the need for biopsy
Screening
HCG: screening of women who have had a previous molar pregnancy and who are at high risk of developing choriocarcinoma
Differential diagnosis
CA125: together with menopausal status and ultrasound findings, contributes to calculation of the risk of malignancy index in the differential diagnosis of women with pelvic masses
Prognosis: prediction of relapse or progression:
in primary disease
in metastatic disease
CEA: measurement at three-monthly intervals following curative surgery in patients to assess need for further treatment
Thyroglobulin: following ablation of the thyroid, increasing serum concentrations suggest an alternative site of production
Prognosis: prediction of response to therapy:
in primary disease
in metastatic disease
Oestrogen and progesterone receptors: their presence or absence in breast cancer tissue determines whether endocrine therapy is likely to be effective
AFP, hCG and LDH: in patients with germ cell tumours, concentrations of these markers are used to assess prognosis
Monitoring course of disease
To detect relapse in patient with no evidence of disease post-therapy
To follow detectable disease
AFP, hCG and LDH used in the follow-up of patients treated for germ cell tumours
CA125 used to monitor ovarian cancer patients
Tumour marker requests and the responsibilities of the clinical laboratory
Reasons for requesting tumour markers
Choice of tumour marker test
Pre-analytical requirements
Analytical requirements
Reporting of tumour marker results
TUMOUR MARKERS IN THE MANAGEMENT OF SPECIFIC CANCERS
Bladder cancer
Breast cancer
Screening and diagnosis
Prognosis
Monitoring
Cervical cancer
Screening and diagnosis
Prognosis
Monitoring
Choriocarcinoma
Colorectal cancer
Screening
Diagnosis
Prognosis and staging
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree