Venous Bypass
Peter Gloviczki
Introduction
Reconstruction of the occluded iliofemoral vein or the inferior vena cava (IVC) may be required in patients with chronic venous insufficiency (CVI) or occasionally in those with acute traumatic or iatrogenic venous injury. Venous reconstruction is also performed in those patients who undergo excision of malignant tumors invading large veins like the IVC or the iliac veins. Chronic iliofemoral venous occlusion or stenosis (May–Thurner syndrome) is responsible for CVI in up to one-third of the patients with CVI. Endovascular treatment for iliocaval obstruction has progressed rapidly, and today venous stenting is the primary choice for treatment of benign iliac, iliofemoral vein, or iliocaval venous occlusions in patients who failed conservative compression therapy. Venous stenting is used for patients with malignancy only in those cases when excision of the tumor is not possible and the goal of therapy is palliation. Hybrid procedures are performed occasionally when endophlebectomy is added to improve inflow for venous stenting. In the last two decades, results of open surgical reconstructions have improved, and symptomatic patients who are not candidates or who failed endovascular reconstructions can be treated with venous bypasses to relieve venous outflow obstruction. A thorough preoperative evaluation to identify significant venous outflow obstruction in these patients is essential.
Etiology
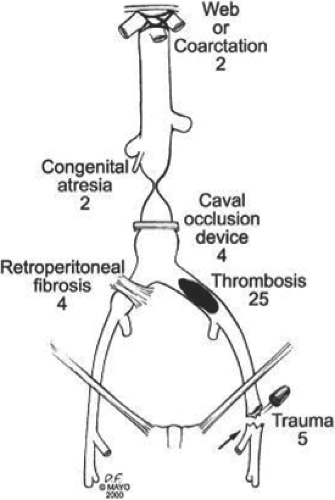
Chronic deep venous obstruction is usually the result of a previous acute deep venous thrombosis (DVT), although it can also be caused by retroperitoneal fibrosis; iatrogenic, blunt, or penetrating trauma; congenital venous anomalies; or benign or malignant tumors (Fig. 1). Compression of the left common iliac vein by the overriding right common iliac artery (May–Thurner syndrome) is a frequently overlooked cause of left iliofemoral venous thrombosis. May and Thurner observed secondary changes, such as an intraluminal web or “spur,” in the proximal left common iliac vein in 20% of 430 autopsies. The most frequent primary malignant tumor originating from large veins is venous leiomyosarcoma; secondary tumors invading the vena cava include adenocarcinoma or liposarcoma. Renal carcinoma may extend into the IVC, and the tumor thrombus in some patients may reach all the way into the right atrium. Congenital suprarenal caval occlusion can occur due to webs or caval coarctation that may also present with associated hepatic vein occlusion (Budd–Chiari syndrome).
Pathophysiology
During the acute phase of DVT, the thrombus in the vein activates the inflammatory cascade, which in turn promotes partial
lysis of the thrombus and leads to recanalization. However, these processes are also responsible for damage to the vein wall and to the venous valves, leading to chronic obstruction and valvular incompetence. If collateral venous circulation in iliofemoral venous occlusion is inadequate, ambulatory venous hypertension develops due to a functional venous outflow obstruction. In postthrombotic syndrome (PTS), deep reflux and obstruction of multiple venous segments often coexists.
lysis of the thrombus and leads to recanalization. However, these processes are also responsible for damage to the vein wall and to the venous valves, leading to chronic obstruction and valvular incompetence. If collateral venous circulation in iliofemoral venous occlusion is inadequate, ambulatory venous hypertension develops due to a functional venous outflow obstruction. In postthrombotic syndrome (PTS), deep reflux and obstruction of multiple venous segments often coexists.
Patients with chronic iliac vein or iliocaval obstruction present with signs and symptoms of CVI. These include pain that frequently develops after exercise (venous claudication), swelling, varicosity, pigmentation, and skin changes (eczema, induration, ulcers) of the affected limb. In the Mayo Clinic experience with 64 venous reconstructions, performed in 60 patients with benign disease, mean duration of symptoms was 6 years; 94% of patients presented with swelling, 90% with venous claudication, and 84% with both. Leg ulcers were present in 12 patients (19%) and 12% had healed ulcerations.
Preoperative evaluation of CVI must focus on two major issues: Confirming the cause of chronic iliofemoral venous occlusion and establishing the presence and significance of poor venous outflow due to venous obstruction. The physician must exclude any abdominal or pelvic malignancy and consider May–Thurner syndrome as the cause of benign left iliac vein thrombosis.
Venous duplex scanning should be performed in all patients with symptoms of CVI to help define the location, cause, and severity of the underlying problem. Duplex scanning will diagnose both valvular incompetence and venous obstruction. Typical appearance of a postthrombotic vein at duplex scanning is that of a thickened, hardly compressible vessel with damaged, incompetent valves and variable degrees of venous flow due to partial recanalization. Obesity and bowel gas may prevent good visualization of the common hepatic veins and the IVC with ultrasound.
Air or strain gauge plethysmography is designed to evaluate the global leg hemodynamics by measuring reflux, obstruction, and calf pump function. Decreased vein wall compliance in patients with PTS may interfere with proper evaluation of calf muscle pump function. Unfortunately, the site and the level of reflux cannot be localized with plethysmography, and this test is absent today from many vascular laboratories. Plethysmography, however, is suitable to confirm functional venous outflow obstruction.
Magnetic resonance (MR) imaging and MR venography and computed tomographic (CT) angiography with venous phase will identify any obstructing mass or tumor, and provides sufficient information in most patients about venous anatomy, obstruction, or stenosis. Contrast venography is still useful in some patients before open surgical deep venous reconstructions, and it is routinely done in those who have endovascular intervention. Ascending venography is useful as a “road map” of the deep veins of the limb; it defines the sites of obstruction, and images the collateral venous circulation and the patterns of preferential flow. Descending venography under fluoroscopy permits evaluation of sites of reflux in the saphenous and deep system. Contrast venography is combined with direct venous pressure measurement to document a pressure difference between the femoral vein and the vena cava. A resting pressure differential of 5 mm Hg or greater is considered evidence for significant obstruction. A lower pressure at rest but an increase to 10 mm Hg after exercise is also a sign of functional obstruction. Exercise consists of 10 dorsiflexions of the ankles or 20 isometric contractions of the calf muscle. Intravascular ultrasound is used frequently today to assess the degree of iliac vein stenosis before stenting, but in patients with venous occlusions intravascular ultrasound has a limited role.
Conservative Management
Symptoms of chronic deep venous obstruction should be first treated with frequent leg elevation, graduated compression stockings (30 to 40 mm Hg), and local wound care of venous ulcerations, if there are any. Compression garments result in variable degrees of success and mandate strict patients’ compliance, which in a hot climate can be both distressing and difficult. The benefits of graduated compression stockings reside in their theoretic effects on venous hemodynamics, skin circulation, and calf muscle pump function. Randomized prospective studies demonstrated a 50% net risk reduction of developing PTS in patients wearing elastic compression stockings after DVT. Patients with persistent disabling symptoms, such as venous claudication, severe swelling, and nonhealing or recurrent ulcers not responding to conservative treatment, should be considered for endovascular or open surgical reconstruction.
Endovascular Treatment
Iliac or iliocaval stenting has become the primary treatment for chronic nonmalignant venous occlusions. Early and midterm results of endovascular techniques, such as angioplasty and stenting, using most frequently self-expandable stents, have been good. In one large study that included 982 patients, venous stenting was performed with no mortality and low morbidity. Five-year secondary patency rate was 100% in nonthrombotic venous disease, while it was 86% in postthrombotic syndrome. Risk factors for restenosis after venous stenting include PTS, positive thrombophilia test results, and long stents extending below the inguinal ligament.
Hybrid Treatment
Patients with common femoral vein and iliac or iliocaval obstruction are candidates. Venous stents are combined with femoral vein endophlebectomy and patch angioplasty to improve inflow to the stents. The stent may be placed only proximal to the patch or it can also be placed into the venous patch. In our practice, we perform these procedures rarely as the last resort.
Open Surgical Treatment
Patients who are not candidates or who failed endovascular reconstructions can be treated with venous bypasses to relieve symptomatic venous outflow obstruction. Venous reconstruction is also performed in those patients who undergo excision of malignant tumors invading the vena cava or iliac veins.
Crossover Saphenous Vein Transposition (Palma Procedure)
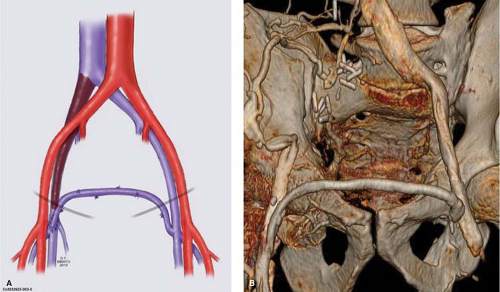
Patients with symptomatic unilateral iliac vein obstruction are candidates for saphenous vein transposition (Palma procedure) (Fig. 2). With this technique, the contralateral saphenous vein is used for a crossover bypass to decompress venous congestion in the affected limb. The common femoral vein on the affected side is exposed first through a 6- to 8-cm long longitudinal groin incision. The collateral veins should be preserved if possible. The great saphenous vein of the contralateral leg is dissected through a 3- to 5-cm incision made in the groin crease, starting just medial to the femoral artery pulse. Tributaries of the saphenous vein are ligated and divided and the saphenous vein is mobilized in a length of about 10 to 12 cm. A short second upper-thigh incision is made
to dissect a 20- to 25-cm long portion of the saphenous vein. Distally the vein is ligated, and proximal to the ligature it is divided and pulled up to the groin incision. Alternatively, endoscopic harvesting of the saphenous vein can also be performed, but the vein proximally should not be divided. It is essential to free up the saphenofemoral junction completely and dissect at least the anterior wall of the common femoral vein around the saphenous vein so that there is no kink or buckle when the saphenous vein is pulled into the suprapubic tunnel. In some patients with a low saphenofemoral junction, a kink is unavoidable. Excision of the saphenous vein with a 2-mm cuff from the common femoral vein and reanastomosis to the femoral vein with running 6-0 polypropylene suture, after turning the junction upward 180 degrees, is the way to solve this problem. Before tunneling, a small Satinsky clamp is placed on the common femoral vein to allow distention of the saphenous vein and the saphenofemoral junction under gentle pressure using heparinized papaverine-saline solution. The vein is then tunneled subcutaneously in the suprapubic space over to the contralateral side using an aortic clamp to ensure a large tunnel without any constriction of the graft whatsoever. Saphenous vein graft in morbidly obese patients is not recommended because of the high chance of external compression of the vein. The common femoral vein is cross-clamped with small vascular clamps or bulldogs and the vein is opened longitudinally in a length of about 2 cm. The anastomosis between the saphenous vein and the femoral vein is performed with running 6-0 polypropylene suture. A vein at least 5 mm in diameter is required to achieve a satisfactory result and provide high venous flow to treat the basic problem in these patients: Poor venous emptying. For a smaller vein, a temporary arteriovenous fistula (AVF) can be added between the superficial femoral artery and the saphenous or common femoral vein using a 4- to 5-mm polytetrafluoroethylene (PTFE) graft or a large tributary of the saphenous vein. This fistula must be taken down at 3 months to enjoy the full benefit of the saphenous bypass. Endovascular occlusion of the fistula with colis or a plug should be considered. The Palma procedure, however, should not be performed with veins 4 mm or smaller.
to dissect a 20- to 25-cm long portion of the saphenous vein. Distally the vein is ligated, and proximal to the ligature it is divided and pulled up to the groin incision. Alternatively, endoscopic harvesting of the saphenous vein can also be performed, but the vein proximally should not be divided. It is essential to free up the saphenofemoral junction completely and dissect at least the anterior wall of the common femoral vein around the saphenous vein so that there is no kink or buckle when the saphenous vein is pulled into the suprapubic tunnel. In some patients with a low saphenofemoral junction, a kink is unavoidable. Excision of the saphenous vein with a 2-mm cuff from the common femoral vein and reanastomosis to the femoral vein with running 6-0 polypropylene suture, after turning the junction upward 180 degrees, is the way to solve this problem. Before tunneling, a small Satinsky clamp is placed on the common femoral vein to allow distention of the saphenous vein and the saphenofemoral junction under gentle pressure using heparinized papaverine-saline solution. The vein is then tunneled subcutaneously in the suprapubic space over to the contralateral side using an aortic clamp to ensure a large tunnel without any constriction of the graft whatsoever. Saphenous vein graft in morbidly obese patients is not recommended because of the high chance of external compression of the vein. The common femoral vein is cross-clamped with small vascular clamps or bulldogs and the vein is opened longitudinally in a length of about 2 cm. The anastomosis between the saphenous vein and the femoral vein is performed with running 6-0 polypropylene suture. A vein at least 5 mm in diameter is required to achieve a satisfactory result and provide high venous flow to treat the basic problem in these patients: Poor venous emptying. For a smaller vein, a temporary arteriovenous fistula (AVF) can be added between the superficial femoral artery and the saphenous or common femoral vein using a 4- to 5-mm polytetrafluoroethylene (PTFE) graft or a large tributary of the saphenous vein. This fistula must be taken down at 3 months to enjoy the full benefit of the saphenous bypass. Endovascular occlusion of the fistula with colis or a plug should be considered. The Palma procedure, however, should not be performed with veins 4 mm or smaller.
Although few large series have been reported, overall patency of Palma grafts in nine series, including 412 operations ranged between 70% and 83% at 3 to 5 years. Results were better in patients who had no or minimal infrainguinal venous disease in those with May–Thurner syndrome without previous deep vein thrombosis. We observed a 70% primary and a 78% secondary patency rate at 5 years in 25 Palma vein grafts. Endoscopic vein harvest was associated with decreased primary but not secondary patency rates.
Crossover Femoral Venous Prosthetic Bypass
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree