Upper Respiratory Tract Infections
KEY CONCEPTS
![]() Most upper respiratory tract infections have a viral etiology and tend to resolve spontaneously without pharmacologic therapy.
Most upper respiratory tract infections have a viral etiology and tend to resolve spontaneously without pharmacologic therapy.
![]() The most common bacterial causes are Streptococcus pneumoniae (acute otitis media and acute rhinosinusitis) and group A β-hemolytic Streptococcus (acute pharyngitis).
The most common bacterial causes are Streptococcus pneumoniae (acute otitis media and acute rhinosinusitis) and group A β-hemolytic Streptococcus (acute pharyngitis).
![]() Vaccination against influenza and pneumococcus may decrease the risk of acute otitis media.
Vaccination against influenza and pneumococcus may decrease the risk of acute otitis media.
![]() Because upper respiratory tract infections are so common, antibiotics used to treat them serve as catalysts for the emergence and spread of antibiotic resistance, thereby making prudent antibiotic use critically important.
Because upper respiratory tract infections are so common, antibiotics used to treat them serve as catalysts for the emergence and spread of antibiotic resistance, thereby making prudent antibiotic use critically important.
![]() When antibiotics are prescribed, the empirical medications of choice are amoxicillin for acute otitis media, amoxicillin–clavulanate for acute rhinosinusitis, and amoxicillin and penicillin for acute pharyngitis.
When antibiotics are prescribed, the empirical medications of choice are amoxicillin for acute otitis media, amoxicillin–clavulanate for acute rhinosinusitis, and amoxicillin and penicillin for acute pharyngitis.
![]() For otitis media, high-dose amoxicillin (80–90 mg/kg/day) is recommended if the patient is at high risk for a penicillin-resistant pneumococcal infection.
For otitis media, high-dose amoxicillin (80–90 mg/kg/day) is recommended if the patient is at high risk for a penicillin-resistant pneumococcal infection.
More patients present to physicians’ offices and emergency departments for upper respiratory tract infections than any other infectious disease.1,2 Otitis media, rhinosinusitis, and pharyngitis are the three most common upper respiratory tract infections. Because they are so common, community and emergency health care workers must be familiar with the diagnosis, assessment, and management of patients with these infections. Furthermore, antibiotics used for the treatment of upper respiratory tract infections serve as catalysts for the emergence and spread of antibiotic resistance, thereby making prudent antibiotic use critically important.
ACUTE OTITIS MEDIA
The term otitis media comes from the Latin oto– for “ear,” itis for “inflammation,” and medi– for “middle”; otitis media, then, is an inflammation of the middle ear. There are three subtypes of otitis media: acute otitis media, otitis media with effusion, and chronic otitis media. The three are differentiated by (a) acute signs of infection, (b) evidence of middle ear inflammation, and (c) presence of fluid in the middle ear.3 Acute otitis media is the subtype with the greatest role for antibiotics and will be discussed in detail.
Epidemiology
Otitis media is one of the leading reasons for physicians’ office visits and emergency department visits in the United States, accounting for more than 16 million clinic and emergency department visits annually.1,2 There are more than 709 million cases of otitis media worldwide each year; half of these cases occur in children under 5 years of age.4 Many patients with acute otitis media will receive a prescription, and the costs associated with managing otitis media are almost $3 billion annually in the United States.5
Etiology
![]() Approximately 40% to 75% of acute otitis media cases are caused by viral pathogens.6
Approximately 40% to 75% of acute otitis media cases are caused by viral pathogens.6 ![]() Common bacterial pathogens include Streptococcus pneumoniae (35% to 40%), nontypeable Haemophilus influenzae (30% to 35%), and Moraxella catarrhalis (15% to 18%).7 The microbial etiology has changed as a result of the introduction and widespread use of the seven-valent pneumococcal conjugate vaccine (PCV7). Specifically, the proportion of S. pneumoniae cases has declined, and the proportion of H. influenzae cases has risen.3 A new S. pneumoniae serotype (19A) has begun to emerge.8
Common bacterial pathogens include Streptococcus pneumoniae (35% to 40%), nontypeable Haemophilus influenzae (30% to 35%), and Moraxella catarrhalis (15% to 18%).7 The microbial etiology has changed as a result of the introduction and widespread use of the seven-valent pneumococcal conjugate vaccine (PCV7). Specifically, the proportion of S. pneumoniae cases has declined, and the proportion of H. influenzae cases has risen.3 A new S. pneumoniae serotype (19A) has begun to emerge.8
S. pneumoniae, H. influenzae, and M. catarrhalis can all possess resistance to β-lactams. S. pneumoniae develops resistance through alteration of penicillin-binding proteins, whereas H. influenzae and M. catarrhalis produce β-lactamases. Up to 40% of S. pneumoniae isolates in the United States are penicillin nonsusceptible, and up to half of these have high-level penicillin resistance.9 Approximately 30% to 40% of H. influenzae and greater than 90% of M. catarrhalis isolates from the upper respiratory tract produce β-lactamases.10–12 Risk factors have been identified for amoxicillin-resistant bacteria. These include attendance at child care centers, recent receipt of antibiotic treatment (within the past 30 days), and age younger than 2 years.6
Pathophysiology
Acute otitis media usually follows a viral upper respiratory tract infection that impairs the mucociliary apparatus and causes Eustachian tube dysfunction in the middle ear.7,13 The middle ear is the space behind the tympanic membrane, or eardrum. A noninfected ear has a thin, clear tympanic membrane. In otitis media, this space becomes blocked with fluid, resulting in a bulging and erythematous tympanic membrane. Bacteria that colonize the nasopharynx enter the middle ear and are not cleared properly by the mucociliary system. The bacteria proliferate and cause infection. Children tend to be more susceptible to otitis media than adults because the anatomy of their Eustachian tube is shorter and more horizontal, facilitating bacterial entry into the middle ear.
Clinical Presentation
Patients or caregivers frequently characterize acute otitis media as having an acute onset of otalgia (ear pain). For parents of young children, irritability and tugging on the ear are often the first clues that a child has acute otitis media.
CLINICAL PRESENTATION Acute Otitis Media
The diagnoses of acute otitis media and otitis media with effusion are easily confused, and careful attention to history, signs, and symptoms is important. Otitis media with effusion is characterized by fluid in the middle ear without signs and symptoms of acute ear infection, such as pain and a bulging eardrum.3,7 A diagnosis of acute otitis media requires that three criteria be satisfied: (a) acute signs of infection, (b) evidence of middle ear inflammation, and (c) presence of fluid in the middle ear.3 Middle ear effusion is indicated by any of the following: bulging of the tympanic membrane, limited or absent mobility of the tympanic membrane, or otorrhea.6 Signs and symptoms of middle ear inflammation include either distinct erythema of the tympanic membrane or distinct otalgia.6 A diagnosis is considered to be “uncertain” if the patient does not have all three of these diagnostic criteria.
TREATMENT
Desired Outcome
![]() Treatment goals include pain management and prudent antibiotic use. These will be discussed in detail, but, first, it is important to consider primary prevention of acute otitis media through the use of bacterial and viral vaccines.
Treatment goals include pain management and prudent antibiotic use. These will be discussed in detail, but, first, it is important to consider primary prevention of acute otitis media through the use of bacterial and viral vaccines.
![]() A systematic review demonstrated that the PCV7 reduced the occurrence of acute otitis media episodes by 6% to 7% when the vaccine was administered during infancy.14 Children with a history of acute otitis media did not benefit when the PCV7 was administered at an older age.14 In June 2012, a new 13-valent pneumococcal conjugate vaccine was approved for use in adults 19 years of age or older.
A systematic review demonstrated that the PCV7 reduced the occurrence of acute otitis media episodes by 6% to 7% when the vaccine was administered during infancy.14 Children with a history of acute otitis media did not benefit when the PCV7 was administered at an older age.14 In June 2012, a new 13-valent pneumococcal conjugate vaccine was approved for use in adults 19 years of age or older.
The H. influenzae type b (Hib) vaccine has been available for two decades and is thought to be responsible for a significant reduction in invasive Hib disease; now it is the nontypeable H. influenzae that is of greatest concern in acute otitis media.15 Other vaccines, including nontypeable H. influenzae and M. catarrhalis vaccines, are in development.
Finally, because acute otitis media cases often follow influenza cases, influenza vaccination should be considered as a possible means to prevent acute otitis media. Refer to the U.S. Centers for Disease Control and Prevention website (www.cdc.gov) and statements from the Advisory Committee on Immunization Practices for the most up-to-date information regarding recommended immunization practices.
General Approach to Treatment
The first step is to differentiate acute otitis media from otitis media with effusion or chronic otitis media, as the latter two types do not benefit substantially from antibiotic therapy. If the patient has acute otitis media, then consider if the disease severity warrants antibiotic therapy. Recognize that amoxicillin is the mainstay of therapy and that penicillin resistance can be overcome, in many cases, with higher doses of amoxicillin. Address the patient’s pain as described below. The therapeutic strategy should be changed if complications develop or if symptoms fail to resolve within 3 days.
Nonpharmacologic Therapy
Regardless of the decision to administer antibiotics, acetaminophen or a nonsteroidal antiinflammatory drug, such as ibuprofen, should be offered early to relieve pain in acute otitis media.6 In addition, eardrops with a local anesthetic, such as amethocaine, benzocaine, or lidocaine, provide pain relief when administered with oral pain medication to children aged 3 to 18 years.16 Because of minimal benefit and increased side effects, neither decongestants nor antihistamines should be routinely recommended in cases of acute otitis media or otitis media with effusion.6,17,18
Pharmacologic Therapy
National clinical practice guidelines for appropriate diagnosis and treatment of acute otitis media were first published in 2004 by the American Academy of Pediatrics (AAP) and the American Academy of Family Physicians (AAFP).6 These guidelines are focused on children 2 months to 12 years of age with uncomplicated cases. These guidelines do not pertain to children with systemic illness or with underlying conditions that may alter the course of acute otitis media (e.g., anatomic abnormalities, genetic conditions such as Down’s syndrome, immunodeficiencies, and cochlear implants).
Systematic reviews and randomized controlled trials have more closely examined the value of antibiotics for acute otitis media.3,19–22 Taken together, these studies suggest a moderate benefit of antibiotics for the treatment of acute otitis media, particularly in patients with severe symptoms.
![]() Antibiotic therapy for upper respiratory diseases must be balanced with possible increases in adverse drug events and increased antibiotic pressure. One strategy to reduce antibiotic use in this setting is “delayed therapy.”23 Delayed therapy most often means that a healthcare worker provides the patient with a prescription, but encourages the patient to wait to use the medication for 48 to 72 hours to see if the symptoms will resolve on their own. Candidates for delayed therapy include (a) children 6 months to 2 years of age without severe symptoms plus uncertain diagnosis, (b) children 2 years and older without severe symptoms, and (c) children 2 years and older with an uncertain diagnosis.6 Delayed therapy decreases antibiotic use but also decreases patient satisfaction and may harm patients.3 A Cochrane review concluded that this strategy is no better than avoiding antibiotics altogether.23
Antibiotic therapy for upper respiratory diseases must be balanced with possible increases in adverse drug events and increased antibiotic pressure. One strategy to reduce antibiotic use in this setting is “delayed therapy.”23 Delayed therapy most often means that a healthcare worker provides the patient with a prescription, but encourages the patient to wait to use the medication for 48 to 72 hours to see if the symptoms will resolve on their own. Candidates for delayed therapy include (a) children 6 months to 2 years of age without severe symptoms plus uncertain diagnosis, (b) children 2 years and older without severe symptoms, and (c) children 2 years and older with an uncertain diagnosis.6 Delayed therapy decreases antibiotic use but also decreases patient satisfaction and may harm patients.3 A Cochrane review concluded that this strategy is no better than avoiding antibiotics altogether.23
![]() If antibiotics are to be administered, then amoxicillin should be given to most children (80 to 90 mg/kg/day in two divided doses).6 S. pneumoniae resistance to penicillin can be overcome with this amoxicillin dose. If pathogens that produce β-lactamase are known or suspected, then amoxicillin should be given in combination with a β-lactamase inhibitor: amoxicillin–clavulanate (90 mg/kg/day of amoxicillin with 6.4 mg/kg/day of clavulanate in two divided doses).6 In patients with moderate to severe illness (temperature greater than 39°C [102°F] and/or severe otalgia), amoxicillin–clavulanate is recommended. Table 86–1 lists antibiotic recommendations for acute otitis media.
If antibiotics are to be administered, then amoxicillin should be given to most children (80 to 90 mg/kg/day in two divided doses).6 S. pneumoniae resistance to penicillin can be overcome with this amoxicillin dose. If pathogens that produce β-lactamase are known or suspected, then amoxicillin should be given in combination with a β-lactamase inhibitor: amoxicillin–clavulanate (90 mg/kg/day of amoxicillin with 6.4 mg/kg/day of clavulanate in two divided doses).6 In patients with moderate to severe illness (temperature greater than 39°C [102°F] and/or severe otalgia), amoxicillin–clavulanate is recommended. Table 86–1 lists antibiotic recommendations for acute otitis media.
TABLE 86-1 Antibiotics and Doses for Acute Otitis Media
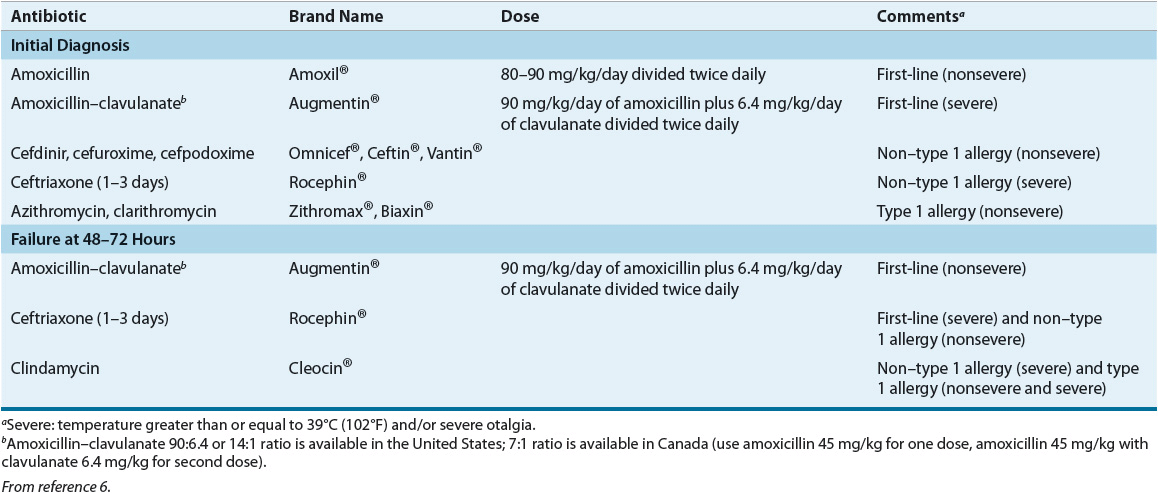
Clinical trials have not provided a clear answer as to which antibiotics are most efficacious3; therefore, the choice of amoxicillin is largely based on microbiology and pharmacokinetic–pharmacodynamic studies. Amoxicillin has the best phar-macodynamic profile against drug-resistant S. pneumoniae of all available oral antibiotics. In addition, amoxicillin has a long record of safety, possesses a narrow spectrum of activity, and is inexpensive. Higher middle ear fluid concentrations of amoxicillin as a result of higher dosing overcome most drug-resistant S. pneumoniae even with its increased minimum inhibitory concentration (MIC).6 Its excellent efficacy against S. pneumoniae outweighs the issue of β-lactamase-producing H. influenzae and M. catarrhalis, against which amoxicillin may not be effective. This is because both H. influenzae and M. catarrhalis are more likely than S. pneumoniae to lead to a spontaneous resolution of the infection.
If treatment failure occurs with amoxicillin, an antibiotic should be chosen with activity against β-lactamase-producing H. influenzae and M. catarrhalis, as well as drug-resistant S. pneumoniae.6 Amoxicillin–clavulanate is recommended. Other choices are cefuroxime, cefdinir, cefpodoxime, and intramuscular ceftriaxone.6 Second-generation cephalosporins, though β-lactamase stable, are expensive, have an increased incidence of side effects, and may increase selective pressure for resistant bacteria. Furthermore, most cephalosporins do not achieve adequate middle ear fluid concentrations against drug-resistant S. pneumoniae for the desired duration of the dosing interval. Use of trimethoprim–sulfamethoxazole and erythromycin–sulfisoxazole is discouraged because of high rates of resistance.6 Intramuscular ceftriaxone is the only antibiotic other than amoxicillin that achieves middle ear fluid concentrations above the MIC for greater than 40% of the dosing interval.6 Although single doses of ceftriaxone have been used, daily doses for 3 days are recommended to optimize clinical outcomes.6 Ceftriaxone should be reserved for severe and unresponsive infections or for patients for whom oral medication is inappropriate because of vomiting, diarrhea, or possible nonadherence. It is an expensive antibiotic, and the intramuscular injections are painful. The drug can be given IV, but the risk-to-benefit ratio of starting an IV line must also be examined. Tympanocentesis can also be considered for treatment failure or persistent acute otitis media. It has a therapeutic effect of relieving pain and pressure and can be used to collect fluid to identify the causative agent. Clindamycin may also be considered at this point for coverage of documented penicillin-resistant S. pneumoniae.6 Patients with a penicillin allergy can be treated with several alternative antibiotics. If the reaction is not type I hypersensitivity, cefdinir, cefpodoxime, or cefuroxime can be used. If the reaction is type I, a macrolide such as azithromycin or clarithromycin may be used. If S. pneumoniae is documented, clindamycin is an alternative. However, the incidence of resistance is much higher with these antibiotics, and of these antibiotics, only clindamycin is recommended by the AAP/AAFP guidelines.6
There is ongoing debate regarding the optimal duration of therapy for acute otitis media. Traditional recommendations call for 10 days of antibiotic therapy; however, some experts have speculated that patients can be treated for as little as 3 to 5 days. Unfortunately, the data to support this theory are inconclusive, with some studies demonstrating similar outcomes and others demonstrating worse outcomes with short-course therapy.3 The advantages of short-course therapy are an increased likelihood that the patient will adhere to the full course of treatment, decreased side effects and cost, and decreased bacterial-selective pressure for both the individual and the community. Short-course treatment is not recommended in children younger than 2 years of age. In children at least 6 years old who have mild to moderate acute otitis media, a 5- to 7-day course may be used.
Clinical Controversy…
Recurrent acute otitis media is defined as at least three episodes in 6 months or at least four episodes in 12 months. Recurrent infections are of concern because patients younger than 3 years are at high risk for hearing loss and language and learning disabilities. Clinical studies generally do not favor prophylaxis. Treatment can be delayed until the onset of symptoms of an upper respiratory tract infection or antibiotic prophylaxis can be limited to 6 months’ duration during the winter months. Surgical insertion of tympanostomy tubes (T tubes) is an effective method for the prevention of recurrent otitis media. These small tubes are placed through the inferior portion of the tympanic membrane under general anesthesia and aerate the middle ear. Children with recurrent acute otitis media should be considered for T-tube placement.
Personalized Pharmacotherapy
One of the most exciting developments in the world of infectious diseases has to do with clinical biomarkers. Procalcitonin increases in response to bacterial infection and declines as the infection resolves. Clinicians are starting to use procalcitonin blood levels to decide when to initiate and discontinue antibiotics in patients with acute upper respiratory infections (URIs).25 A Cochrane systematic review of 14 trials with 4,221 participants found that procalcitonin protocols significantly reduced antibiotic consumption without negatively impacting patient survival or treatment failure.26 The finding was driven by lower prescription rates in primary care and shorter durations of antibiotic therapy in emergency departments and intensive care units.26 Despite the enthusiasm for this approach, there are still several aspects of procalcitonin monitoring that need to be resolved, including the timing of levels, the procalcitonin cutoff values for different clinical decision points, and the cost-effectiveness of this technology.27
Evaluation of Therapeutic Outcomes
Patients with acute otitis media should be reassessed after 3 days. By this time, there should be clinical improvement in the signs and symptoms of infection, including pain, fever, and erythema/bulging of the tympanic membrane. If the patient has not responded and antibiotics were withheld initially, they should be instituted now. If the patient initially received an antibiotic, then the antibiotic should be changed (Table 86–1). Most children will become asymptomatic at 7 days.
Early reevaluation of the eardrum when signs and symptoms are improving can be misleading because effusions persist. Over a period of 1 week, changes in the eardrum normalize, and the pus becomes serous fluid. Air–fluid levels are apparent behind the eardrum, at which point the stage is now referred to as otitis media with effusion. This does not represent ongoing infection, nor are additional antibiotics required. Two weeks after an acute otitis media episode, 60% to 70% of children still have a middle ear effusion—40% at 1 month and 10% to 25% at 3 months.6 Younger children and those with a history of recurrent infections have a further delay in resolution.
Immediate reevaluation is appropriate if hearing loss results from persistent middle ear effusions following infection. Complications of otitis media are infrequent but include mastoiditis, bacteremia, meningitis, and auditory sequelae with the potential for speech and language impairment.6
ACUTE BACTERIAL RHINOSINUSITIS
Sinusitis is an inflammation and/or infection of the paranasal sinuses, or membrane-lined air spaces, around the nose.28 The term rhinosinusitis is now preferred because sinusitis typically also involves the nasal mucosa.28 Even though the majority of rhinosinusitis infections are viral in origin, antibiotics are frequently prescribed. It is thus important to differentiate between viral and bacterial rhinosinusitis to avoid antibiotic overuse.
A new set of clinical practice guidelines for acute bacterial rhinosinusitis was published in 2012.28 These guidelines provide “a systematic weighting of the strength of recommendation (e.g., ‘high, moderate, low, very low’) and quality of evidence (e.g., ‘strong, weak’)” using a well-known rating system.29 Several of the recommendations in these new guidelines differ substantially from prior guidelines.
Epidemiology
![]() Nearly 30 million cases of rhinosinusitis are diagnosed annually in the United States.30 Acute bacterial rhinosinusitis is overdiagnosed; thus, antibiotics are overprescribed. Most rhinosinusitis infections have a viral etiology; nevertheless, one in five antibiotics prescribed for adults in the United States is for rhinosinusitis.31,32 Adults with rhinosinusitis miss an average of 6 workdays/y with these infections.33 Patients with rhinosinusitis are significantly more likely to use the emergency room, spend more than $500/y on medical care, and see a medical specialist.33
Nearly 30 million cases of rhinosinusitis are diagnosed annually in the United States.30 Acute bacterial rhinosinusitis is overdiagnosed; thus, antibiotics are overprescribed. Most rhinosinusitis infections have a viral etiology; nevertheless, one in five antibiotics prescribed for adults in the United States is for rhinosinusitis.31,32 Adults with rhinosinusitis miss an average of 6 workdays/y with these infections.33 Patients with rhinosinusitis are significantly more likely to use the emergency room, spend more than $500/y on medical care, and see a medical specialist.33
Etiology
![]() Acute bacterial rhinosinusitis is caused, most often, by the same bacteria implicated in acute otitis media: S. pneumoniae and H. influenzae. These organisms are responsible for ~50% to 70% of bacterial causes of acute bacterial rhinosinusitis in both adults and children.28 M. catarrhalis is also frequently implicated in adults and children (~8% to 16%).28 Streptococcus pyogenes, Staphylococcus aureus, gram-negative bacilli, and anaerobes are associated less frequently with acute bacterial rhinosinusitis.28 Issues of bacterial resistance are similar to those found with acute otitis media.
Acute bacterial rhinosinusitis is caused, most often, by the same bacteria implicated in acute otitis media: S. pneumoniae and H. influenzae. These organisms are responsible for ~50% to 70% of bacterial causes of acute bacterial rhinosinusitis in both adults and children.28 M. catarrhalis is also frequently implicated in adults and children (~8% to 16%).28 Streptococcus pyogenes, Staphylococcus aureus, gram-negative bacilli, and anaerobes are associated less frequently with acute bacterial rhinosinusitis.28 Issues of bacterial resistance are similar to those found with acute otitis media.



