GMP regulations place responsibility for ensuring the quality of the materials used to manufacture drug products squarely on the shoulders of the pharmaceutical company. The globalization of the pharmaceutical supply chain, and the increasing use of materials manufactured in emerging countries (from a pharmaceutical perspective) such as India and China, has made this responsibility increasingly difficult to achieve.
Over the last few years, much debate and discussion has occurred regarding the establishment of a drug pedigree to ensure the integrity and authenticity of each and every drug product that enters the pharmaceutical supply chain. Much of this discussion has arisen as a result of the increase in counterfeit pharmaceutical products entering the supply chain and putting consumers and patients at significant risk. It is imperative that every organization involved in the pharmaceutical supply chain does their part to minimize or eliminate the introduction of counterfeit materials or products.
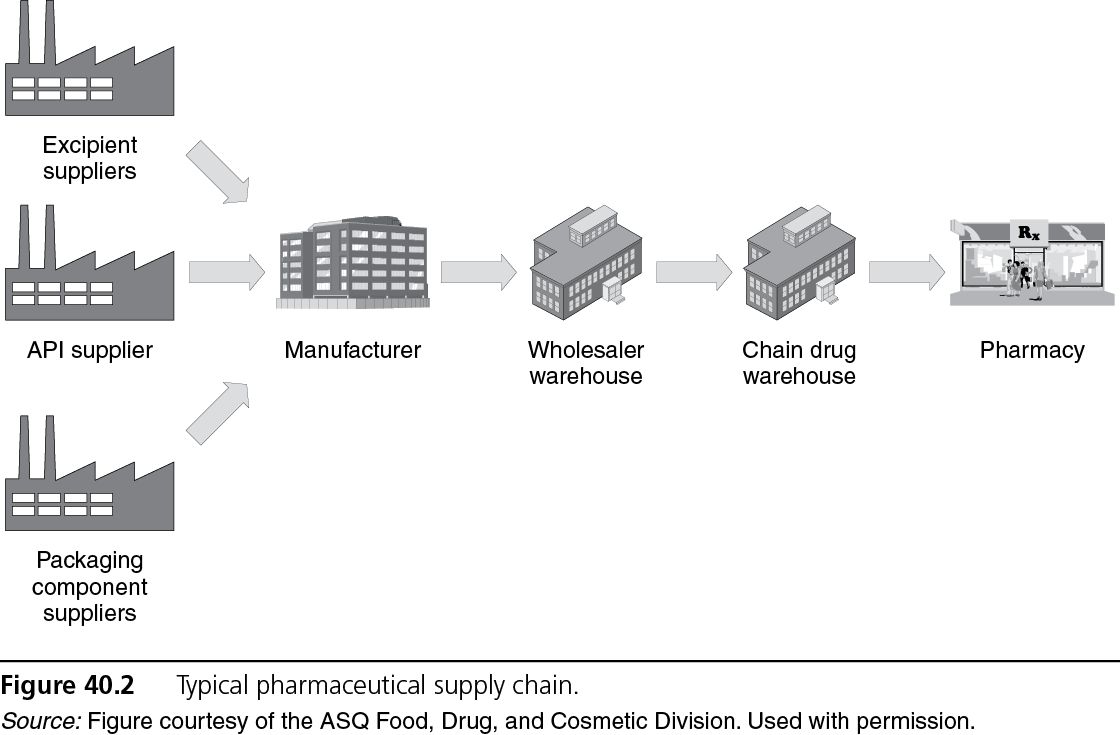
Pharmaceutical Supply Chain
A typical supply chain for a pharmaceutical company is shown in Figure 40.2. Inputs (for example, raw materials, packaging components) into the finished dosage form manufacturing process typically are purchased from outside suppliers. Once the finished dosage form is manufactured, packaged, tested, and released, it is shipped to a wholesaler who may store it for a period of time. The wholesaler may sell the product to a chain drug store (which requires the product to pass through the chain drug store warehouse before reaching the pharmacy) or directly to a pharmacy customer. The finished product typically passes through a number of hands before it reaches the end customer or patient.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


