CHAPTER 40 The eye
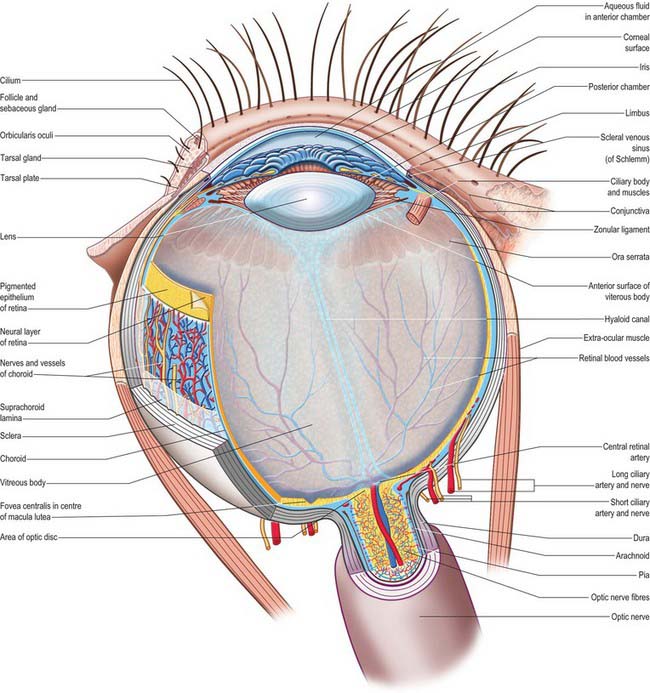
The outer surface of the eye is composed of parts of two spheres with different radii. The anterior segment, part of the smaller sphere, is formed by the transparent cornea and accounts for approximately 7% of the ocular surface. The posterior segment of the eyeball is part of the larger sphere formed by the opaque sclera (Fig. 40.1).
Internal to the sclera is a vascular, pigmented layer consisting of three continuous parts that collectively make up the uveal tract: a thin choroid lying posteriorly, a thicker ciliary body and an anterior iris which is displaced from the outer coat and terminates at the pupillary aperture (Fig. 40.1). The internal surface of the choroid is covered by the photosensory retina, which terminates anteriorly at the ora serrata, which also marks the junction between the ciliary body and choroid. The vasculature of the choroid supplies nutrients to the avascular outer retina.
The ocular lens is located immediately behind the iris and is suspended from the ciliary body via zonular fibres (Fig. 40.1). Smooth muscles within the ciliary body regulate the tension exerted on the elastic lens and hence determine its shape, thereby adjusting the focus of the eye in the process of accommodation. The iris, which does not allow the transmission of light due to a heavily pigmented posterior surface, also contains smooth muscle, allowing it to regulate the size of the pupillary aperture.
The iris and lens separate the eye into three chambers. The largest, the vitreous chamber, is filled with the gel-like vitreous humour, and lies posterior to the lens, comprising about two-thirds of the volume of the eye (Fig. 40.1). The spaces between the lens and iris, and the iris and cornea, are the posterior and anterior chambers respectively of the eye. Both are filled with aqueous humour, which is produced by the epithelium of the ciliary body, travels through the pupil, circulates in the anterior chamber and is drained principally through the canal of Schlemm at the iridocorneal filtration angle. Aqueous humour provides metabolic support to the avascular lens and cornea.
The sole purpose of the eyes and their associated structures within the bony orbit is to form a good image on a healthy retina. The photoreceptors of the retina transduce the optical radiation into neurobiological activity and other cells within the retina then begin to process the image. The retinal signal is transferred via the optic nerve, along the visual pathway (see Fig. 40.33) to various regions within the brain, where further processing results in visual perception.
OUTER COAT
The fibrous outer coat of the eye consists of the opaque posterior sclera and the transparent anterior cornea. Together they form a semi-elastic protective capsule enclosing the eye, which when made turgid by intraocular pressure, determines the optical geometry of the eye and ensures its shape is not distorted when it moves. The sclera also provides attachments for the extraocular muscles, and its smooth external surface rotates easily on the adjacent tissues of the orbit when these muscles contract (see Ch. 39). The opacity of the sclera helps ensure that only light that enters the eye through the pupil reaches the retina. The cornea, on the other hand, not only admits light but its covering tear film (see p. 673) is also the major refractive surface of the eye.
SCLERA
The sclera accounts for approximately 93% of the outer coat of the eye. Anteriorly, it is continuous with the cornea at the corneoscleral junction (limbus) (Fig. 40.1). It is punctured by a number of foramina containing nerves and blood vessels, most notably the optic foramen, which lies 3 mm medial to the midline and 1 mm below the horizontal and houses the optic nerve. Smaller openings contain anterior ciliary arteries which penetrate anteriorly, vortex veins which cross the sclera equatorially, and the long and short ciliary nerves and arteries which enter posteriorly. The sclera is thickest posteriorly (1–1.35 mm), decreasing gradually towards the equator to reach a minimum of 300 μm under the tendons of the recti. Going forwards from the insertions of the recti, it gradually increases in thickness once more to 800 μm at the limbus.
The external surface of the sclera is covered by a delicate episcleral lamina of loose fibro-vascular tissue, which contains sparse blood vessels and is in contact with the inner surface of the fascial sheath of the eyeball (see p. 657). Anteriorly the external scleral surface is covered by conjunctiva which is reflected onto it from the deep surfaces of the eyelids. The scleral internal surface adjacent to the choroid is attached to it by a delicate fibrous layer, the suprachoroid lamina, which contains numerous fibroblasts and melanocytes. Anteriorly, the inner sclera is attached to the ciliary body by the lamina supraciliaris. Posteriorly, the sclera is pierced by the optic nerve. Here the outer half of the sclera turns back to become continuous with the dura mater, while the inner half is modified to form a perforated plate, the lamina cribrosa sclerae. The optic nerve fascicles pass through these minute orifices, while the central retinal artery and vein pass through a larger, central aperture. The lamina cribrosa sclerae is the weakest part of the sclera and bulges outwards (a cupped disc) when intraocular pressure is raised chronically as in glaucoma (see p. 686).
Like the cornea, the scleral stroma is composed mainly of densely packed collagen embedded in a matrix of proteoglycans, which are mixed with occasional elastic fibres and fibroblasts. However, in contrast to the cornea, scleral collagen fibrils show a large variation in diameter and spacing, and the lamellae branch and interlace extensively. This arrangement of fibres results in increased light scatter, which is responsible for the opaque dull white appearance of the sclera, and also imparts a high tensile strength to the sclera to resist the pull of the extraocular muscles and contain the intraocular pressure. Collagen fibre bundles are arranged circumferentially around the optic disc and the orifices of the lamina cribrosa. The fibres of the tendons of the recti intersect scleral fibres at right angles at their attachments, and then interlace deeper in the sclera. Collagen fibres of the scleral spur are orientated circularly, and there is an increased incidence of elastic fibres here. Although the sclera acts as a conduit for blood vessels, scleral vessels are few and mainly disposed in the episcleral lamina, especially close to the limbus. Its nerve supply is surprisingly rich, accounting for the intense pain associated with scleral inflammation (Watson & Young 2004).
Filtration angle and aqueous drainage
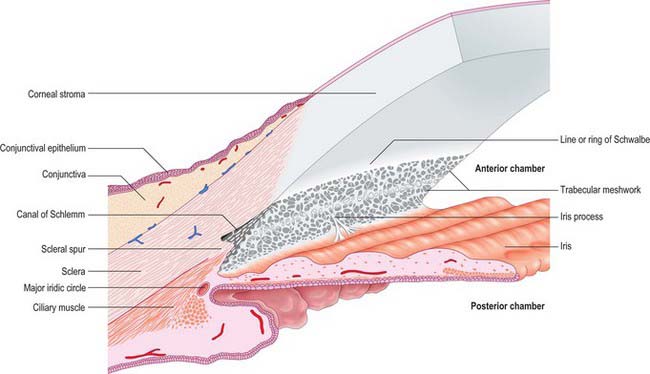
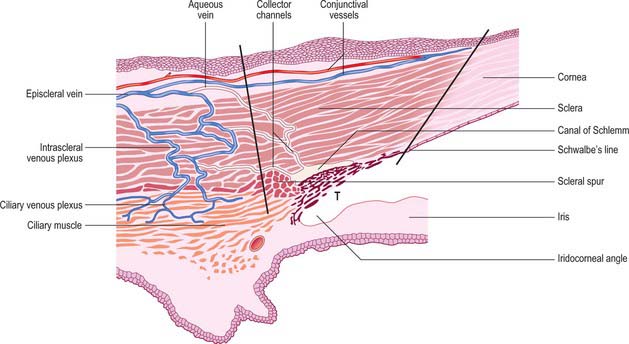
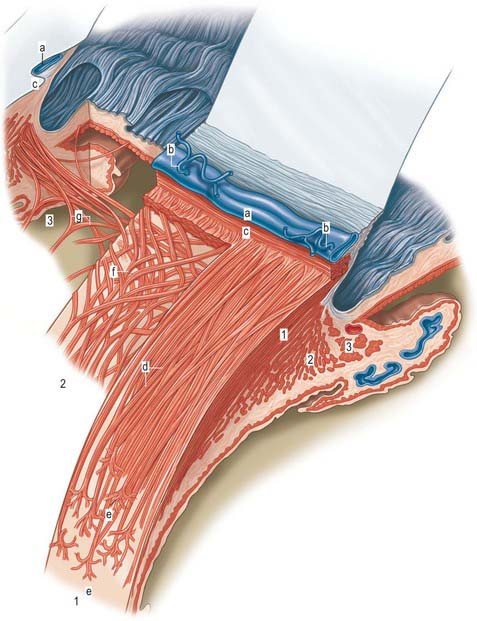
Aqueous humour is produced by the ciliary epithelium, passes through the pupil and circulates within the anterior chamber supplying the avascular cornea and lens with nutrients. It drains from the eye mainly through the trabecular meshwork into the canal of Schlemm at the iridocorneal (filtration) angle (Figs 40.2, 40.3), formed between the posterior aspect of the corneoscleral limbus and the periphery of the iris.
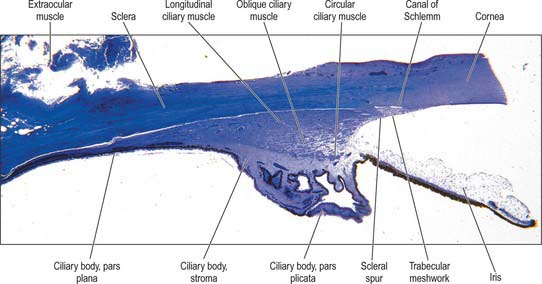
Fig. 40.2 Meridional section through the anterior eye highlighting the ciliary body and drainage apparatus.
The canal of Schlemm (sinus venosus sclerae) is an anular endothelial canal located near the internal surface of the sclera close to the limbus. In section, the canal appears as an oval cleft, with an outer wall that grooves the sclera. Posteriorly, the cleft extends as far as a rim of scleral tissue, the scleral spur, which in section forms a triangle with its apex directed forwards (Figs 40.2, 40.3). The canal of Schlemm may be double or multiple in part of its course, and its walls are constructed of a continuous single thin endothelial layer. Passage of aqueous humour to the canal probably occurs via giant pinocytotic vacuoles, which form on the inner face of the endothelium and discharge into the canal at the outer face, and through intercellular clefts. Aqueous humour passes through a plexus of fine intrascleral vessels which connect the canal of Schlemm with anterior ciliary veins (Fig. 40.4). Normally the canal does not contain blood: pressure gradients prevent the reflux of blood even though the channels between the canal and veins have no valves. However, in venous congestion, blood may enter the canal: the continuous endothelial outer wall of the canal prevents further reflux.
CORNEA
The avascular cornea is the anterior transparent part of outer coat. Convex anteriorly, it projects from the sclera as a dome-shaped elevation with an area of 1.1 cm2, forming approximately 7% of the external tunic area. Since it is more curved (average radius, r = 7.8 mm) than the sclera (r = 11.5 mm), a slight sulcus sclerae marks the corneoscleral junction (limbus). The cornea is approximately 670 μm thick close to the corneoscleral junction, and 520 μm at its centre. At the nasal and temporal limbus the transition from cornea to sclera occurs in a line that is approximately perpendicular to the cornea: this transition occurs more obliquely superiorly and inferiorly, with the sclera overlapping the cornea to a greater extent anteriorly. Consequently, when viewed from in front, the corneal perimeter is slightly elliptical, as its horizontal diameter (11.7 mm) is a little greater than its vertical (10.6 mm). Its posterior perimeter is circular (diameter 11.7 mm).
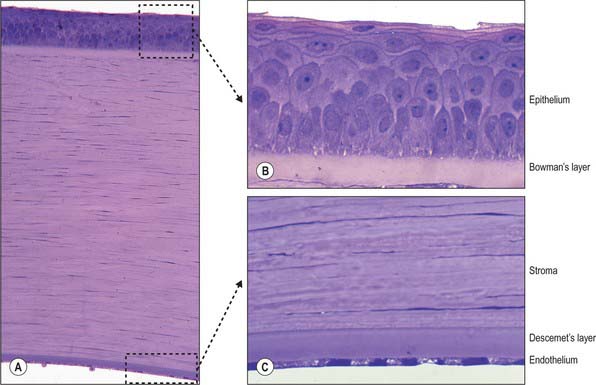
Microscopically, the cornea consists of five layers, namely, corneal epithelium, anterior limiting lamina (Bowman’s layer), substantia propria (stroma), posterior limiting lamina (Descemet’s membrane) and endothelium, arranged anteroposteriorly (Fig. 40.5A).
Corneal epithelium
The corneal epithelium accounts for approximately 10% of the corneal thickness (50 μm). It usually consists of 5–6 layers of cells (Fig. 40.5B) that protect the ocular surface from mechanical abrasion, form a permeability barrier (to small molecules, water and ions) and prevent the entry of pathogens. The deepest cells are columnar with flat bases, rounded apices, and large round or oval nuclei. Outside these are 2–3 layers of polyhedral (often wing-shaped) cells. In the more superficial layers the cells become progressively flatter, and present a smooth, optically perfect surface. Scanning electron microscopy of surface cells reveals extensive finger-like and ridge-like projections (microvilli and microplicae). A complex network of tight junctions links the superficial cells, consistent with their barrier function.
Anterior limiting lamina (Bowman’s layer)
The anterior limiting lamina lies behind the corneal epithelium. It contains a dense mass of collagen fibrils set in a matrix similar to that of the substantia propria. The lamina is 12 μm thick, is readily distinguishable from the substantia propria because it does not contain fibroblasts, and appears amorphous by light microscopy (Fig. 40.5B).
Substantia propria (stroma)
The substantia propria is approximately 500 μm thick and forms the bulk of the cornea. It is a compact and transparent layer, composed of 200–250 sequential lamellae, each made up of fine parallel collagen fibrils mainly of type I collagen. Flat dendritic interconnecting fibroblasts (keratocytes) form a coarse mesh between the lamellae. Alternate lamellae are typically orientated at large angles to each other (Fig. 40.6). X-ray diffraction studies indicate that they run along two preferred directions, superior-inferior and nasal-temporal, to account for the additional tensile stress exerted by the recti along these meridians (Boote et al 2005). Each lamella is approximately 2 μm thick and of variable width (10–250 μm, or, rarely, more). In the central cornea fibrils within the lamellae have a similar diameter of approximately 31 nm. This increases slightly with age and approaching the limbus. The small size of the fibrils (much smaller than the wavelength of light), along with the regularity of their spacing (maintained by collagen: proteoglycan interactions), and the careful control of corneal hydration, are the principal factors which determine stromal transparency. Light scattered by the collagen fibrils is eliminated by destructive interference in all directions other than forwards.
Endothelium
The endothelium covers the posterior surface of the cornea and consists of a single layer of squamous cells (Fig. 40.5C) with prominent interdigitations between adjacent cells, which are also connected by tight and gap junctions. When viewed en face, the endothelium appears as a mosaic of polygonal (typically hexagonal) cells. As these cells have a limited capacity for mitosis, in response to pathology, trauma, age and prolonged contact lens wear, the endothelial mosaic becomes less regular, and shows a greater variation in cell size and shape, as cells spread to fill gaps caused by cell loss. The numerous mitochondria and prominent rough ER within these cells reflects their high metabolic activity. Thus, for example, active pumping mechanisms largely control the degree of corneal hydration.
Corneal innervation
The cornea is innervated by numerous branches of the ophthalmic nerve (see p. 667) which either form an anular plexus around the periphery of the cornea, or pass directly from the sclera and enter the corneal stroma radially as 70–80 small groups of fibres. Upon entering the cornea, the few myelinated nerves lose their myelin sheaths and ramify throughout the corneal matrix in a delicate reticulum, their terminal filaments forming an intricate subepithelial plexus. Axon bundles from this plexus cross the anterior limiting membrane and form a subbasal plexus from which individual beaded axons pass to more superficial epithelial layers, eventually terminating as free nerve endings. Corneal nerves provide the afferent arm of the blink and lacrimal (see p. 673) reflexes and may also have a neurotrophic function.
UVEA
The vascular tunic of the eye consists of the choroid, ciliary body and iris, which collectively form a continuous structure, the uvea (Fig. 40.1). The choroid covers the internal scleral surface, and extends forwards to the ora serrata. The ciliary body continues forward from the choroid to the circumference of the iris, which is a circular diaphragm behind the cornea and in front of the lens, presenting an almost central aperture, the pupil.
CHOROID
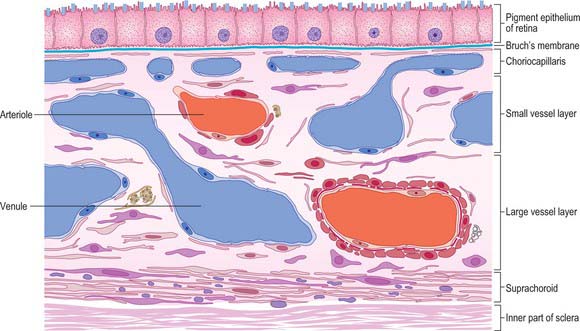
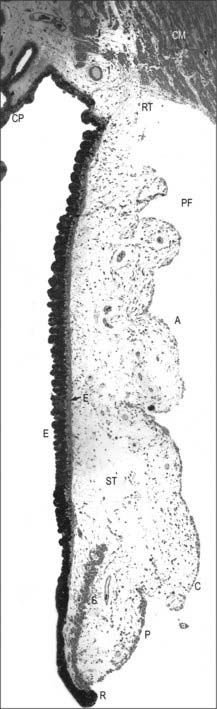
Four layers can be identified in transverse section (Fig. 40.7, see also Fig. 40.23): suprachoroid, vascular stroma, choriocapillaris and lamina vitrea (Bruch’s membrane).
Vessel layer (stroma)
Internal to the suprachoroid lies a layer composed mainly of arteries and veins, but also some loose connective tissue containing scattered pigment cells. These melanocytes limit the passage of light through the sclera to the retina. More importantly, like the retinal pigment epithelium (see p. 689), they also absorb light traversing the retina that is not absorbed by the photoreceptors, so preventing internal reflection.
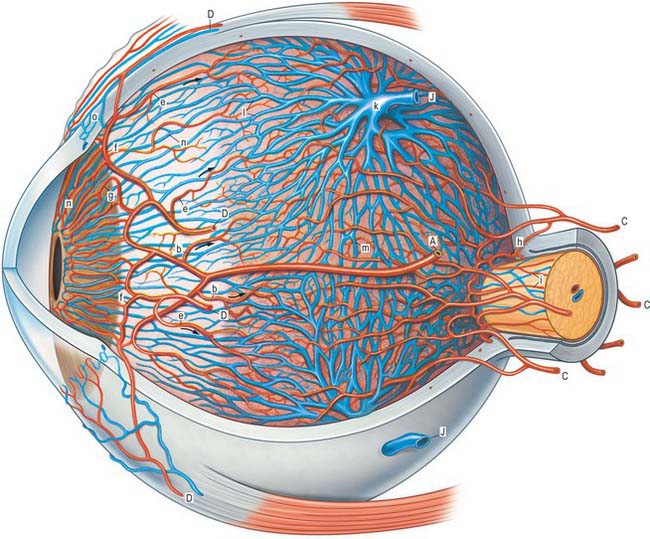
The blood supply of the choroid, as well as the rest of the uvea, is summarized in Figure 40.10. Short posterior ciliary arteries enter the eye through the sclera near the optic disc and supply the posterior choroid. These vessels branch and gradually decrease in size as they approach the retinal border. The choroidal stroma can be divided into layers based on the change in the calibre of these vessels; an outer layer of larger vessels (Haller’s layer), an inner layer of smaller vessels (Sattler’s layer) which eventually give rise to the choriocapillaris (Fig. 40.7). Long posterior ciliary arteries and recurrent branches of anterior ciliary arteries supply the anterior part of the choriocapillaris.
The vessels of the choroid have a rich autonomic vasomotor supply.
CILIARY BODY
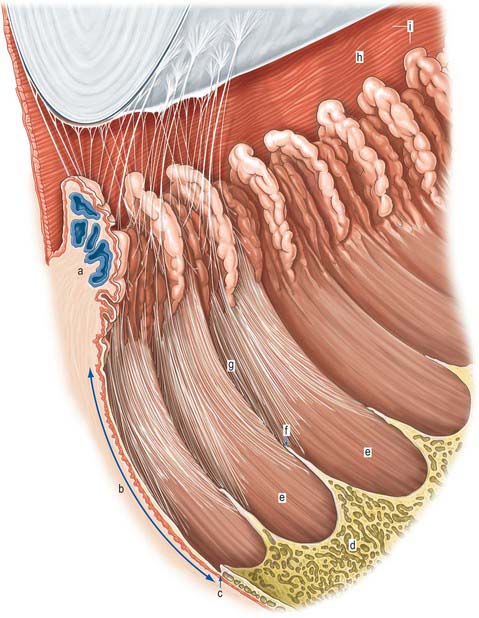
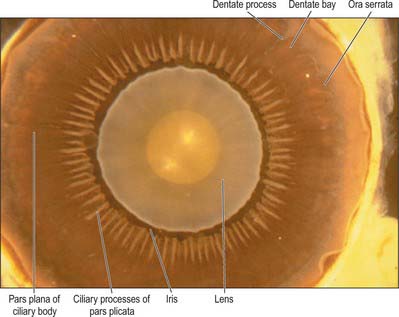
Externally, the ciliary body may be represented by a line which extends from approximately 1.5 mm posterior to the limbus of the cornea (corresponding also to the scleral spur) to a line approximately 7.5–8 mm posterior to this on the temporal side, and 6.5–7 mm on the nasal side. The ciliary body is thus slightly eccentric. It projects posteriorly from the scleral spur, which is its attachment, with a meridional width varying from 5.5 to 6.5 mm. Internally, it exhibits a posteriorly crenated or scalloped periphery, the ora serrata, where it is continuous with the choroid and retina (Figs 40.8, 40.9). Anteriorly, it is confluent with the periphery of the iris, and externally bounds the iridocorneal angle of the anterior chamber.
In cross section the ciliary body is composed of four layers (from internal to external), namely a double layer of epithelial cells, the stroma, ciliary muscle, and a supraciliary layer (Fig. 40.2).
Meridionally, the ciliary body can be divided into two parts (Figs 40.2, 40.9). Anteriorly, the ridged anterior pars plicata (corona ciliaris) surrounds the base of the iris and accounts for about ⅓ rd of the ciliary body. Posteriorly, the relatively smooth and thin pars plana (orbicularis ciliaris) lies adjacent to the ora serrata, and forms a convenient access point for instruments during vitreo-retinal surgery and for intraocular injection. The whole ciliary body is covered by a double epithelium, in which the inner layer is unpigmented, while the outer layer contains melanin. At the ora serrata the retinal pigment epithelium is continuous with the outer pigmented epithelium of the pars plana, while the neural retina is replaced by inner unpigmented ciliary epithelium. Anteriorly, this double epithelium continues over the pars plicata until it merges with the double epithelium on the posterior surface of the iris (where the inner layer of cells also accumulates melanin) (see p. 682). The anterior pars plicata is ridged meridionally by 70–80 ciliary processes radiating from the base of the iris (Figs 40.2, 40.9). In the young eye these process are approximately parallel-sided structures, but in the adult their flanks become less regular and appear thicker. A minor ridge, or ciliary plica, lies in the valley between most of the processes. The crests of the processes are less pigmented, giving them the appearance of white (or light) striae, from which the name ciliary is derived. Fibres of the zonule (the suspensory ligaments of the lens) extend into the valleys. They pass beyond the ciliary processes to fuse with the basal lamina of the superficial epithelial layer of pars plana. Their sites of attachment are marked by striae which pass back from the valleys of the pars plicata, across the pars plana, almost as far as the apices of the dentate processes of the ora serrata (Fig. 40.8).
Ciliary epithelium
The ciliary epithelium is bilaminar, consisting of two layers of simple epithelium which are derived embryonically from the two layers of the optic cup. The superficial layer consists of columnar cells over the pars plana, and cuboidal cells over the ciliary processes of the pars plicata: it becomes irregular and more flattened between the processes. These cells contain little or no pigment. The outer layer of the ciliary epithelium, in contrast contains cuboidal cells which are loaded with melanin. The two layers are normally firmly united, but fluid may separate them pathologically. The pigment layer is attached to the stroma of the ciliary body by its basal lamina, which continues back into the basal lamina of the choroid. As a consequence of the invagination of the optic cup during development, a basal lamina covers the free surface of the bilayer which is continuous with the internal limiting membrane of the retina (see p. 692).
The ciliary epithelium is responsible for the secretion of aqueous humour (see p. 685), while the outer pigmented epithelium additionally contributes to the eye’s ‘black box effect’, like the retinal pigment epithelium, choroidal pigment and posterior iris epithelium, absorbing stray light to enhance image quality.
Ciliary stroma
The ciliary stroma is composed largely of loose bundles of collagen, which form a considerable mass between the ciliary muscle and overlying processes, and extend into both of them. This inner layer of connective tissue contains numerous larger branches of the ciliary vessels. A dense reticulum of large (up to 35 μm diameter) fenestrated capillaries is concentrated in the ciliary processes. This facilitates the passage of substances from the blood plasma during aqueous production (see p. 685). Anteriorly, near the periphery of the iris, the major arterial circle (Fig. 40.10; see also Fig. 40.14) is formed chiefly by long posterior ciliary branches of the ophthalmic artery. These enter the eye some distance behind the ocular equator and pass between the choroid and sclera to the ciliary body. Ciliary veins, also draining the iris, pass posteriorly to join the vortex veins of the choroid.
Ciliary muscle
Ciliary muscle is composed of smooth muscle cells, most of which are attached to the scleral spur and arranged in three different orientations. The outermost fibres are meridional or longitudinal, and pass posteriorly into the stroma of the choroid. The innermost fibres swerve acutely from the spur to run circumferentially as a sphincter near the periphery of the lens. Obliquely interconnecting radial fibres run between these two muscular strata, frequently forming an interweaving lattice (Figs 40.2, 40.11).
Accommodation reflex
At rest distant objects are focused on the retina in an emmetropic eye (see p. 687). In order to focus closer objects the dioptric power of the eye has to be increased, which is achieved by increasing the curvature of the lens. At rest the lens is under tension from the zonular ligaments and hence flattened. On accommodation, the ciliary muscle contracts, moving the ciliary body forwards and inwards towards the optic axis. All parts of the muscle act in concert, and tension on the zonular ligaments is relaxed. As the lens is covered by an elastic capsule (see p. 686), once tension on it is released it assumes a more convex shape suitable for focusing closer objects. The radius of curvature of the anterior lens surface changes most during accommodation.
Information from the retina passing to the visual cortex does not constitute the afferent limb of a simple reflex in the usual sense of the term, but permits the visual areas to assess the clarity of objects in the visual field. Cortical efferent information passes to the pretectal area and thence to the Edinger–Westphal nucleus, which contains preganglionic parasympathetic neurones whose axons travel in the oculomotor nerve (Fig. 40.12). Efferent impulses pass in the oculomotor nerve to the orbit where they synapse in the ciliary ganglion. Postganglionic fibres (short ciliary nerves) innervate the ciliary muscle causing it to contract. There is also a sparse sympathetic innervation of ciliary muscle, which has a very limited capacity to relax the muscle.
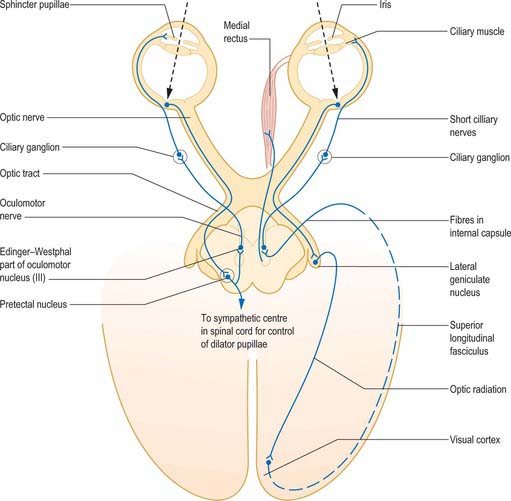
Fig. 40.12 The neural pathways of the pupillary light reflex (left) and the accommodation reflex (right).
(From Oxford Textbook of Functional Anatomy, Vol 3 Head and Neck, MacKinnon P, Morris J (eds) 1990. By permission of Oxford University Press.)
Accommodation is usually accompanied by constriction of the pupil brought about by contraction of the sphincter pupillae (see p. 682) and convergent eye movements caused by contraction of the medial, superior, and inferior recti (all innervated by the oculomotor nerve). This is the ‘near triad’ which may become disrupted in various diseases.
Supraciliary layer
The thin supraciliary layer separates the sclera from the ciliary muscle and is largely composed of collagen fibres derived from the two layers it divides. It also forms an alternative ‘unconventional’ route (other than the canal of Schlemm) for the drainage of aqueous humour (see p. 686).
IRIS
The iris is an adjustable diaphragm around a central aperture (slightly medial to true centre), the pupil. It lies between the cornea and lens and is immersed in aqueous fluid (Fig. 40.1), partially dividing the anterior segment into an anterior chamber, enclosed by the cornea and iris and a posterior chamber, which lies between the iris and the lens anterior to the vitreous. The efficacy of the iris as a light stop is mainly due to a densely pigmented posterior double epithelium. The pupillary aperture is adjusted by the action of two muscles, dilator and sphincter pupillae.
Seen from the front the iris is divided into a large ciliary zone adjacent to the ciliary body and a smaller, inner, pupillary zone (Fig. 40.13
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree