Substance-Related Disorders II: Alcohol, Nicotine, and Caffeine
KEY CONCEPTS
![]() Tobacco is the number one preventable cause of death in the United States.
Tobacco is the number one preventable cause of death in the United States.
![]() Nearly 17 million Americans report current heavy alcohol use or alcohol abuse.
Nearly 17 million Americans report current heavy alcohol use or alcohol abuse.
![]() Pharmacogenomic studies have identified genotypic and functional phenotypic variants that either serve to protect patients or predispose them toward alcohol dependence.
Pharmacogenomic studies have identified genotypic and functional phenotypic variants that either serve to protect patients or predispose them toward alcohol dependence.
![]() Alcohol is a CNS depressant that shares many pharmacologic properties with the nonbenzodiazepine sedative–hypnotics.
Alcohol is a CNS depressant that shares many pharmacologic properties with the nonbenzodiazepine sedative–hypnotics.
![]() The metabolism of alcohol is considered to follow zero-order pharmacokinetics, and this has important implications for the time course in which alcohol can exert its effects.
The metabolism of alcohol is considered to follow zero-order pharmacokinetics, and this has important implications for the time course in which alcohol can exert its effects.
![]() Benzodiazepines are the treatment of choice for alcohol withdrawal.
Benzodiazepines are the treatment of choice for alcohol withdrawal.
![]() Disulfiram, naltrexone, and acamprosate are FDA-approved drug therapies for the treatment of alcohol dependence. The clinical utility of these agents to improve sustained abstinence remains controversial. Relapse is common.
Disulfiram, naltrexone, and acamprosate are FDA-approved drug therapies for the treatment of alcohol dependence. The clinical utility of these agents to improve sustained abstinence remains controversial. Relapse is common.
![]() More than three quarters of smokers are nicotine dependent. Tobacco dependence is a chronic condition that requires repeated interventions.
More than three quarters of smokers are nicotine dependent. Tobacco dependence is a chronic condition that requires repeated interventions.
![]() Use of nicotine replacement therapy along with behavioral counseling doubles cessation rates.
Use of nicotine replacement therapy along with behavioral counseling doubles cessation rates.
![]() Bupropion and varenicline are efficacious alone and in combination with nicotine replacement therapy for smoking cessation.
Bupropion and varenicline are efficacious alone and in combination with nicotine replacement therapy for smoking cessation.
![]() Alcohol, nicotine, and caffeine are considered by most to be socially acceptable drugs, yet they impose an enormous social and economic cost on our society. Approximately 443,000 deaths each year are attributable to tobacco use, making tobacco the number one preventable cause of death and disease in this country.1,2 The three leading causes of death attributable to smoking include lung cancer, chronic obstructive pulmonary disease, and ischemic heart disease.3
Alcohol, nicotine, and caffeine are considered by most to be socially acceptable drugs, yet they impose an enormous social and economic cost on our society. Approximately 443,000 deaths each year are attributable to tobacco use, making tobacco the number one preventable cause of death and disease in this country.1,2 The three leading causes of death attributable to smoking include lung cancer, chronic obstructive pulmonary disease, and ischemic heart disease.3
![]() In 2011, heavy drinking was reported by 6.2% of the population aged 12 or older, or 15.9 million people,4 a decrease from the previous year’s data in which 16.9 million people were heavy drinkers. Approximately one quarter (22.6%) of persons aged 12 or older participated in binge drinking at least once in the 30 days prior to the National Survey on Drug Use and Health (NSDUH) in 2011.4
In 2011, heavy drinking was reported by 6.2% of the population aged 12 or older, or 15.9 million people,4 a decrease from the previous year’s data in which 16.9 million people were heavy drinkers. Approximately one quarter (22.6%) of persons aged 12 or older participated in binge drinking at least once in the 30 days prior to the National Survey on Drug Use and Health (NSDUH) in 2011.4
The World Health Organization estimates that there are approximately 2 billion people worldwide who consume alcoholic beverages, and 76.3 million with diagnosable alcohol-use disorders.5 Long-term alcohol abuse often leads to chronic disease. A causal relationship between alcohol abuse and at least 60 types of chronic disease or injury has been established (e.g., esophageal cancer, liver cancer, and cirrhosis of the liver, epileptic seizures, homicide, and motor vehicle accidents) worldwide.5 Nationally, according to the Drug Abuse Warning Network 2010 survey,6 687,574 emergency department visits involved either alcohol in combination with other drugs (for patients of all ages) or alcohol only for patients aged 20 or younger.
![]() Worldwide, alcohol abuse leads to 1.8 million deaths annually.5 Nationally, according to the Alcohol-Attributable Deaths Report, 80,374 U.S. citizens with medium and high average daily alcohol consumption die each year because of alcohol-related causes, including traffic collisions and cirrhosis of the liver.7 Direct and indirect health and social costs of alcoholism to the nation are estimated to be $223.5 billion annually,8 and governments pay more than 60% of their healthcare costs.
Worldwide, alcohol abuse leads to 1.8 million deaths annually.5 Nationally, according to the Alcohol-Attributable Deaths Report, 80,374 U.S. citizens with medium and high average daily alcohol consumption die each year because of alcohol-related causes, including traffic collisions and cirrhosis of the liver.7 Direct and indirect health and social costs of alcoholism to the nation are estimated to be $223.5 billion annually,8 and governments pay more than 60% of their healthcare costs.
Caffeine is currently the most widely used psychoactive substance in the world. In the United States, 80% to 90% of adults regularly consume behaviorally active doses of caffeine.
ALCOHOL
Epidemiology of Alcohol Use
Approximately half of Americans aged 12 or older reported being current drinkers of alcohol according to the NSDUH 2011 (51.8%). This translates to an estimated 133.4 million people, which is similar to the 2010 estimate of 131.3 million people (51.8%).4 In 2011 heavy drinking was reported by 6.2% of the population aged 12 or older, meaning that they drank five or more drinks on the same occasion on at least 5 different days in the past month.4
The Disease Model of Addiction as Applied to Alcoholism
The disease concept of addiction, using alcoholism as a model, states that addiction is a disease, and that individuals who suffer from the disease do not choose to contract the disease any more than someone who suffers from heart disease or diabetes mellitus chooses to contract that illness. A disease is defined as “any deviation from or interruption of the normal structure or function of any part, organ, or system (or combination thereof) of the body that is manifested by a characteristic set of symptoms and signs and whose etiology, pathology, and prognosis may be known or unknown.”9 Diagnostic criteria for alcoholism do not specify frequency of drinking or amount of alcohol consumed. The key determinant is whether drinking is compulsive, out of control, and consequential when one drinks.10
![]() It has long been recognized that alcoholism is heritable, as 50% to 60% of first-degree relatives of alcoholics become alcohol dependent themselves.11 Research has identified several traits (or phenotypes) that attenuate one’s risk of alcohol dependence. Initially based on data from preclinical studies, pharmacogenomic studies have identified genotypic and functional phenotypic variants that either serve to protect patients or predispose them toward alcohol dependence.12 Large-scale pharmacoepidemiologic studies have further elucidated the environmental risk factors that are associated with either protective effects or predisposition toward alcoholism.13 The known susceptibility genes, phenotypic characteristics, and environmental risk factors are summarized in Table 49-1.11–15
It has long been recognized that alcoholism is heritable, as 50% to 60% of first-degree relatives of alcoholics become alcohol dependent themselves.11 Research has identified several traits (or phenotypes) that attenuate one’s risk of alcohol dependence. Initially based on data from preclinical studies, pharmacogenomic studies have identified genotypic and functional phenotypic variants that either serve to protect patients or predispose them toward alcohol dependence.12 Large-scale pharmacoepidemiologic studies have further elucidated the environmental risk factors that are associated with either protective effects or predisposition toward alcoholism.13 The known susceptibility genes, phenotypic characteristics, and environmental risk factors are summarized in Table 49-1.11–15
TABLE 49-1 Genotypic, Phenotypic, and Environmental Factors that Increase Alcohol-Dependence Risk

Pharmacology and Pharmacokinetics of Alcohol
Alcohol as a Drug
![]() Alcohol is a CNS depressant that affects the CNS in a dose-dependent fashion, producing sedation that progresses to sleep, unconsciousness, coma, surgical anesthesia, and finally fatal respiratory depression and cardiovascular collapse. Alcohol affects endogenous opiates and several neurotransmitter systems in the brain, including γ-aminobutyric acid (GABA), glutamine, and dopamine. Alcohol is available in a variety of concentrations in various alcoholic beverages. There is approximately 14 g of alcohol in a 12-oz (355 mL) can of beer (approximately 5%), 4 oz (118 mL) of nonfortified wine (approximately 10% to 14%), or one shot (1.5 oz [44 mL]) of 80-proof whiskey (40%). Full consumption of this amount will cause an increase in blood alcohol level of approximately 20 to 25 mg/dL (4.3 to 5.4 mmol/L) in a healthy 70-kg (154 lb) male, although this varies with the time frame over which the alcohol is consumed, the type of alcoholic beverage, whether food is consumed along with it, and many patient variables. The lethal dose of alcohol in humans is variable, but deaths generally occur when blood alcohol levels are greater than 400 to 500 mg/dL (87 to 109 mmol/L).16
Alcohol is a CNS depressant that affects the CNS in a dose-dependent fashion, producing sedation that progresses to sleep, unconsciousness, coma, surgical anesthesia, and finally fatal respiratory depression and cardiovascular collapse. Alcohol affects endogenous opiates and several neurotransmitter systems in the brain, including γ-aminobutyric acid (GABA), glutamine, and dopamine. Alcohol is available in a variety of concentrations in various alcoholic beverages. There is approximately 14 g of alcohol in a 12-oz (355 mL) can of beer (approximately 5%), 4 oz (118 mL) of nonfortified wine (approximately 10% to 14%), or one shot (1.5 oz [44 mL]) of 80-proof whiskey (40%). Full consumption of this amount will cause an increase in blood alcohol level of approximately 20 to 25 mg/dL (4.3 to 5.4 mmol/L) in a healthy 70-kg (154 lb) male, although this varies with the time frame over which the alcohol is consumed, the type of alcoholic beverage, whether food is consumed along with it, and many patient variables. The lethal dose of alcohol in humans is variable, but deaths generally occur when blood alcohol levels are greater than 400 to 500 mg/dL (87 to 109 mmol/L).16
Pharmacokinetics
Absorption of alcohol begins in the stomach within 5 to 10 minutes of oral ingestion. The onset of clinical effects follows fairly rapidly. Peak serum concentrations of alcohol usually are achieved 30 to 90 minutes after finishing the last drink, although it is variable depending on the type of alcoholic beverage consumed, what and when the person last ate, and other factors.17
More than 90% of alcohol in the plasma is metabolized in the liver by three enzyme systems that operate within the hepatocyte. The remainder is excreted by the lungs and in urine and sweat. Alcohol is metabolized to acetaldehyde by alcohol dehydrogenase in the cell. In turn, acetaldehyde is metabolized to carbon dioxide and water by the enzyme aldehyde dehydrogenase. A second pathway for oxidation of alcohol uses catalase, an enzyme located in the peroxisomes and microsomes. The third enzyme system, the microsomal alcohol oxidase system, has a role in the oxidation of alcohol to acetaldehyde. These last two mechanisms are of lesser importance than the alcohol dehydrogenase–aldehyde dehydrogenase system.17
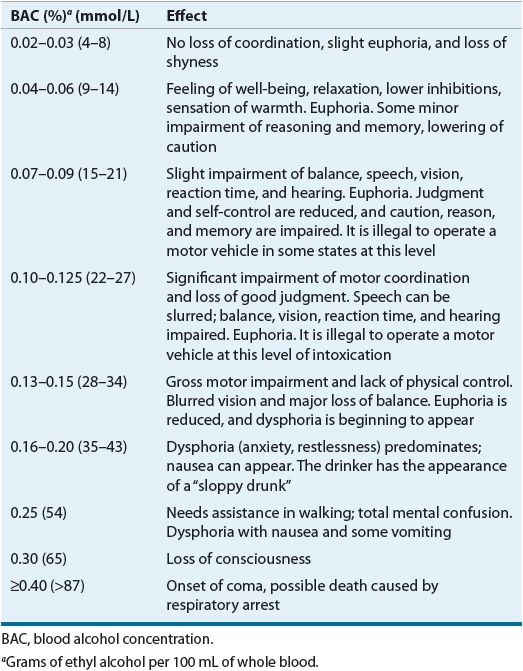
![]() The metabolism of alcohol generally is said to follow zero-order pharmacokinetics.17 This can, in fact, be an oversimplification because at very high or very low concentrations of alcohol the metabolism can follow first-order pharmacokinetics.18 On average, the blood alcohol concentration (BAC) is lowered from 15 to 22.2 mg/dL (3.3 to 4.8 mmol/L) per hour in the nontolerant individual, assuming that the individual is in the postabsorptive state (Table 49-2). Alcohol has a volume of distribution of 0.6 to 0.8 L/kg, representing the total body water.17
The metabolism of alcohol generally is said to follow zero-order pharmacokinetics.17 This can, in fact, be an oversimplification because at very high or very low concentrations of alcohol the metabolism can follow first-order pharmacokinetics.18 On average, the blood alcohol concentration (BAC) is lowered from 15 to 22.2 mg/dL (3.3 to 4.8 mmol/L) per hour in the nontolerant individual, assuming that the individual is in the postabsorptive state (Table 49-2). Alcohol has a volume of distribution of 0.6 to 0.8 L/kg, representing the total body water.17
TABLE 49-2 Specific Effects of Alcohol Related to BAC
Clinical Indicators of Chronic Alcohol Abuse
The CAGE questionnaire is a tool for detecting individuals more likely to be abusing alcohol and therefore at greater risk for alcohol withdrawal. CAGE is a mnemonic for four questions: (a) Do you ever feel the need to cut down on your alcohol use? (b) Have you ever been annoyed by others telling you that you drink too much? (c) Have you ever felt guilty about your drinking or something you did while drinking? (d) Do you ever have an “eye opener”? A positive response to two or more of these four questions suggests an increased likelihood of alcohol abuse with an average sensitivity of 0.71 (71%) and an average specificity of 0.90 (90%).19
Acute Effects of Alcohol
At lower serum concentrations, euphoria and disinhibition may be noted. Slurred speech, altered perception of the environment, impaired judgment, ataxia, incoordination, nystagmus, and hyperreflexia may occur. As plasma levels increase, combative and destructive behavior may occur. With higher levels still, somnolence and respiratory depression may ensue. The typical effects of various BACs are shown in Table 49-2, although effects vary from individual to individual.
Alcohol Poisoning
Acute alcohol poisoning usually occurs with rapid consumption of large quantities of alcoholic beverages. With sustained drinking of moderate amounts of alcohol, the user passes out before a toxic dose of alcohol can be ingested, and/or the person vomits to rid the stomach of its toxic reservoir. With rapid drinking, the person may fall asleep or pass out without vomiting, allowing continued alcohol absorption from the GI tract until fatal BACs are achieved.
Laboratory Studies
In the emergency room, a BAC should be ordered in any patient in whom alcohol ingestion is suspected, regardless of the presenting complaint. For clinical purposes, most laboratories report BAC in units of mg/dL or mmol/L. In legal cases, results are reported in percentage (grams of ethyl alcohol per 100 mL of whole blood). If the diagnosis is unclear, if the intoxication seems atypical, or when there is suspicion of multiple drug ingestions, a complete toxicologic screen to rule out the presence of other substances may be useful.
TREATMENT
Alcohol-Related Disorders
Desired Outcomes
Goals for alcohol-dependent persons trying to decrease or discontinue alcohol intake include: (a) the prevention and treatment of withdrawal symptoms (including seizures and delirium tremens) and medical or psychiatric complications, (b) long-term abstinence after detoxification, and (c) entry into ongoing medical and alcohol-dependence treatment.
Alcohol Withdrawal
Pharmacologic Therapy
![]() Symptom-triggered treatment with a benzodiazepine is the current standard of care in alcohol detoxification to manage and minimize symptoms and avoid progression to the more severe stages of withdrawal. A meta-analysis was performed to provide evidence-based recommendations on the pharmacologic management of alcohol withdrawal.20 A similar study was done to develop treatment strategies for alcohol withdrawal delirium.21 Trials comparing different benzodiazepines demonstrated that all appear similarly efficacious in reducing signs and symptoms of withdrawal.20,21
Symptom-triggered treatment with a benzodiazepine is the current standard of care in alcohol detoxification to manage and minimize symptoms and avoid progression to the more severe stages of withdrawal. A meta-analysis was performed to provide evidence-based recommendations on the pharmacologic management of alcohol withdrawal.20 A similar study was done to develop treatment strategies for alcohol withdrawal delirium.21 Trials comparing different benzodiazepines demonstrated that all appear similarly efficacious in reducing signs and symptoms of withdrawal.20,21
Clinical Controversy
A Cochrane review22 of the effectiveness and safety of benzodiazepines in the treatment of alcohol withdrawal symptoms was published in 2010. According to this report “the available data show that benzodiazepines are effective against alcohol withdrawal seizures when compared to placebo, but data on safety outcomes are sparse and fragmented. There is a need for larger, well-designed studies in this field.”
CLINICAL PRESENTATION Alcohol Intoxication and Withdrawal
Symptom-Triggered Therapy With symptom-triggered therapy, medication is given only when the patient has symptoms. This approach results in treatment that is shorter, potentially avoiding oversedation and allowing the clinician to focus on specific therapy for alcohol dependence.20,21 A typical regimen would include lorazepam 2 mg administered every hour as needed when a structured assessment scale—for example, the Clinical Institute Withdrawal Assessment–Alcohol, Revised (CIWA-AR)—indicates that symptoms are moderate to severe (Table 49-3).23
TABLE 49-3 Dosing and Monitoring of Pharmacologic Agents Used in the Treatment of Alcohol Withdrawal
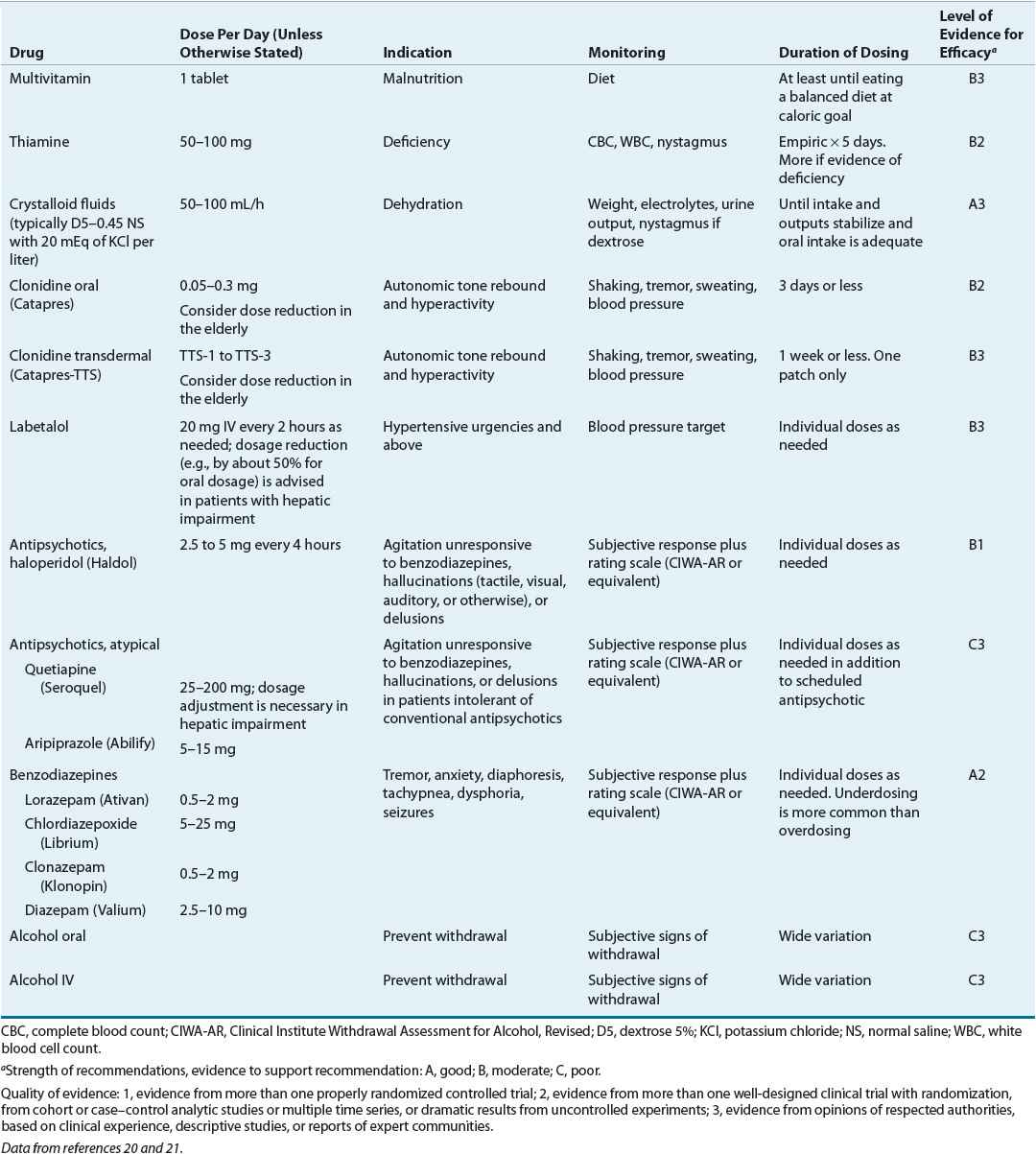
Fixed-Schedule Therapy Over the years, benzodiazepines given regularly at a fixed dosing interval have been used for alcohol withdrawal. The major problem with this approach is underdosing of the benzodiazepine because of cross-tolerance (see Table 49-3). Current guidelines take exception with this rigid approach, urging clinicians to allow for some degree of individualization within fixed-schedule therapy.20,21
Treatment of Alcohol Withdrawal Seizures Alcohol withdrawal seizures do not require treatment with an anticonvulsant drug unless they progress to status epilepticus because seizures usually end before diazepam or another drug can be administered.21 Phenytoin, which is not cross-tolerant to alcohol, does not prevent or treat withdrawal seizures, and without an IV loading dose, therapeutic blood levels of phenytoin are not reached until acute withdrawal is complete. Patients experiencing seizures should be treated supportively. An increase in the dosage and slowing of the tapering schedule of the benzodiazepine used in detoxification or a single injection of a benzodiazepine can be necessary to prevent further seizure activity. Patients with a history of withdrawal seizures can be predicted to experience an especially severe withdrawal syndrome. In such patients, a higher initial dosage of a benzodiazepine and a slower tapering period of 7 to 10 days are advisable.
Treatment of Nutritional Deficits and Electrolyte Abnormalities Fluid status should be carefully assessed, and fluid, electrolyte, and vitamin abnormalities should be corrected. Hydration can be necessary in patients with vomiting, diarrhea, increased body temperature, or severe agitation. Alcoholics often have electrolyte imbalances because of inadequate nutrition and fluid volume related to antidiuretic hormone inhibition. Hypokalemia can be corrected with oral potassium supplementation as long as renal function is adequate. Thiamine (vitamin B1) is often depleted in alcoholics, and supplementation is standard because it can prevent the development of the Wernicke-Korsakoff syndrome (e.g., mental confusion, eye movement disorders, and ataxia [poor motor coordination]). An initial dose of 100 mg IV or IM is commonly used. In practice, thiamine is usually given 100 mg once daily orally, IV, or intramuscularly for 3 to 5 days (see Table 49-3).
Alcohol hypoglycemia usually occurs in the absence of overt liver disease, and it is more likely if the patient is fasting or exercising or is sensitive to alcohol; it is less likely if the patient is obese. The alcohol directly interferes with hepatic gluconeogenesis but not glycogenolysis. The energy required for metabolism of alcohol is diverted away from the energy needed to take up lactate and pyruvate—substrates for gluconeogenesis. So, patients who drink alcohol can become hypoglycemic once glycogen stores are depleted. Neurologic symptoms of hypoglycemia can be confused with alcohol intoxication, and in the inpatient setting, blood glucose should be monitored regularly.
Treatment Settings Alcohol withdrawal treatment can take place in hospitals, inpatient detoxification units, or outpatient settings. Only patients with mild to moderate symptoms should be considered for outpatient treatment, and it is a good idea to have a responsible, sober person available to help the patient monitor symptoms and administer medications. Patients with a strong craving for alcohol, those concurrently using other drugs, and those with a history of seizures or delirium tremens are not good candidates for outpatient treatment. Pharmacologic agents used in the treatment of alcohol withdrawal are summarized in Table 49-3.
Pharmacologic Management of Alcohol Dependence
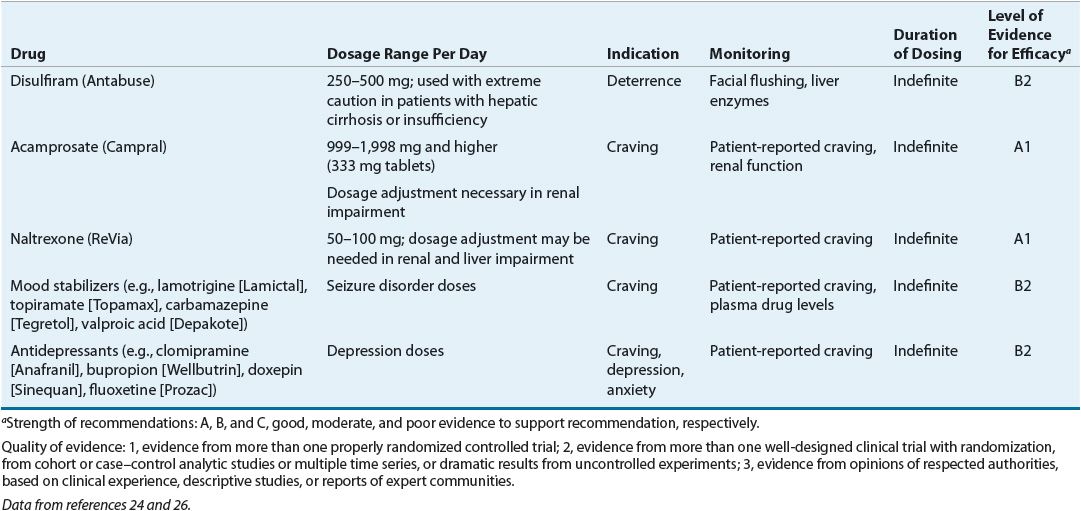
![]() In the United States, disulfiram, naltrexone, once-monthly injectable extended-release naltrexone, and acamprosate are the only four drugs that are FDA-approved for the treatment of alcohol dependence. Disulfiram acts as a deterrent to the resumption of drinking, and naltrexone is a competitive opioid antagonist that has been shown to reduce cravings for alcohol. Acamprosate is a GAB-Aergic agonist that modulates alcohol cravings (Table 49-4). Other drugs, including nalmefene, bupropion, various serotonergic agents (including selective serotonin reuptake inhibitors and vascular serotonin-3 [5-HT3] receptor antagonists), topiramate, and lithium, also have been used either abroad or in the United States off-label for alcohol dependence.
In the United States, disulfiram, naltrexone, once-monthly injectable extended-release naltrexone, and acamprosate are the only four drugs that are FDA-approved for the treatment of alcohol dependence. Disulfiram acts as a deterrent to the resumption of drinking, and naltrexone is a competitive opioid antagonist that has been shown to reduce cravings for alcohol. Acamprosate is a GAB-Aergic agonist that modulates alcohol cravings (Table 49-4). Other drugs, including nalmefene, bupropion, various serotonergic agents (including selective serotonin reuptake inhibitors and vascular serotonin-3 [5-HT3] receptor antagonists), topiramate, and lithium, also have been used either abroad or in the United States off-label for alcohol dependence.
TABLE 49-4 Dosing and Monitoring of Pharmacologic Agents Used in the Treatment of Alcohol Dependence
Disulfiram
Disulfiram deters a patient from drinking by producing an aversive reaction if the patient drinks. In the absence of alcohol, disulfiram has minimal effects. Disulfiram inhibits aldehyde dehydrogenase in the biochemical pathway for alcohol metabolism, allowing acetaldehyde to accumulate. The resulting increase in acetaldehyde causes severe facial flushing, throbbing headache, nausea and vomiting, chest pain, palpitations, tachycardia, weakness, dizziness, blurred vision, confusion, and hypotension. Severe reactions including myocardial infarction, congestive heart failure, cardiac arrhythmia, respiratory depression, convulsions, and death can occur, particularly in vulnerable individuals.24
Naltrexone
Naltrexone, an opiate antagonist available in the United States since 1984 for the treatment of opioid dependence, blocks the effects of exogenous opioids. In 1994, the FDA approved its use in the treatment of alcohol dependence. Naltrexone is thought to attenuate the reinforcing effects of alcohol, and those who consume alcohol while taking naltrexone report feeling less intoxicated and having less craving for alcohol.25 Evidence suggests that genetics plays a role in the clinical response to naltrexone. Carriers of the Asp40 polymorphism in the μ-opioid receptor gene show increased response to naltrexone with lower rates of relapse to heavy drinking.
Naltrexone should not be given to patients currently dependent on opiates because it can precipitate a severe withdrawal syndrome. Naltrexone is associated with dose-related hepatotoxicity, but this generally occurs at doses higher than those recommended for treatment of alcohol dependence. Nevertheless, it is considered contraindicated in patients with hepatitis or liver failure, and liver function tests should be monitored monthly for the first 3 months and every 3 months thereafter.
Nausea is the most common side effect of naltrexone, occurring in approximately 10% of patients. Other side effects are headache, dizziness, nervousness, fatigue, insomnia, vomiting, anxiety, and somnolence. If dosed daily, naltrexone 50 mg is sufficient to effectively block μ-opioid receptors.
In April 2006, the FDA approved Vivitrol, a once-monthly intramuscular naltrexone formulation. The usual effective dose is 380 mg IM each month.26,27 Extended-release formulations reduce the likelihood of forgetting or choosing not to take medication, assuring that once the patient receives an injection, he or she will be “adherent” for the next month.26
Criticism has been leveled at the extended-release dosage form, suggesting that naltrexone’s benefit may be limited to less severe alcohol dependence, and exclusively to reduction in heavy drinking rather than abstinence. Pettinati et al.27 report the results of a study in alcohol-dependent patients who had higher baseline severity, as measured by: (a) the Alcohol Dependence Scale (ADS) or (b) having been medically detoxified in the week before randomization. Higher severity alcohol-dependent patients, when receiving 380 mg (n = 50) of the extended-release compound compared with placebo (n = 47), had significantly fewer heavy-drinking days during the study (hazard ratio = 0.583; P = 0.0049) and showed an average reduction of 37.3% in heavy-drinking days compared with 27.4% for placebo-treated patients (P = 0.039). The authors contend that their data support the efficacy of extended-release naltrexone 380 mg in relatively higher severity alcohol dependence for both reduction in heavy drinking and maintenance of abstinence.
Acamprosate
Acamprosate is a glutamate modulator at the N-methyl-D-aspartate (NMDA) receptor that reduces alcohol craving. Acamprosate, approved in the United States in 2004, had been available in Europe for many years. Patients treated with acamprosate are more successful in maintaining abstinence from alcohol versus placebo. Acamprosate is well tolerated, with GI adverse effects most common.
A Cochrane review of 24 randomized controlled trials (RCTs) with 6,915 participants28 found that, compared with placebo, acamprosate significantly reduced the risk of any drinking (RR 0.86 [95% CI 0.81 to 0.91]; NNT 9.09 [95% CI 6.66 to 14.28]) and significantly increased the cumulative abstinence duration (mean difference 10.94 [95% CI 5.08 to 16.81]), while secondary outcomes did not reach statistical significance. Diarrhea was the only side effect that was more frequently reported with acamprosate than placebo (risk difference 0.11 [95% 0.09 to 0.13]; NNTB 9.09 [95% CI 7.69 to 11.11]). See Table 49-4 for dosing information for this and the other options used in treating alcohol dependence.
NICOTINE
Clinical guidelines for tobacco use and dependence were released in 2000 and updated in 2008.29 Telephone quitlines are available in every state, and more patient are now referred to smoking cessations counseling services. There also is a growing number of Internet and mobile phone text messaging programs that have been developed to reach the teenage and young adult population to promote smoking cessation.30,31 The number of adults who smoke has decreased from 42.4% in 1965 to 19.3% in 2010, and now there are more former smokers than current smokers.
Despite this encouraging news, cigarette smoking continues to be the leading cause of preventable morbidity and mortality in the United States. Data from the 2010 National Health Interview Survey (NHIS)32 found that the overall percent of current smokers from the years 2005 to 2010 in adults ≥18 years old decreased from 20.9 to 19.3. It was determined this represents 3 million fewer smokers in 2010 compared with those in 2005, but this decline has not been uniform across all subsets of the population. The Healthy People 2020 target is currently set for the prevalence of smoking to be less than or equal to 12%, and based on the current rate of decline, this target will not be met. Healthy People 2020 also calls for greater utilization of tobacco use counseling within ambulatory settings to improve smoking cessation rates.33
Epidemiology of Tobacco Use
The NSDUH reported in 2011 that an estimated 26.5% (68.2 million) of the U.S. population 12 years of age and older used a tobacco product at least once in the month prior to being interviewed. In addition, 56.8 million Americans were current cigarette smokers, 12.9 million smoked cigars, 8.2 million used smokeless tobacco, and 2.1 million smoked pipes.4 Comparing age groups, adults between the ages of 18 and 25 years have the highest rate of cigarette use (39.5%), but it is encouraging to see the rates continue to decrease each year since 2002 when 40.8% of young adults were using cigarettes. Within youth aged 12 to 17, use of cigarettes also continued to decline from 15.2% in 2002 to 10% in 2011.4
Data trends from the 2011 NSDUH continue to show smoking prevalence varies based on the level of education. The highest percentage of adults who admitted to smoking were adults who had not completed high school (33.7%). The lowest rate of smoking was seen in adults who graduated from college (11.7%). Results from the NSDUH also showed cigarette smoking was higher in unemployed adults (40.7%) in comparison to adults who were employed full time (23.3%).4
Economic Impact of Smoking
The direct healthcare expenditures associated with smoking total approximately $298 billion a year, which includes factors such as lost productivity, premature death, and direct medical expenditures.34 Medicaid patients’ smoking rates are substantially higher in comparison to the general population. Smoking-attributable medical expenditures are estimated at 11% of Medicaid program expenditures.35
HEALTH RISKS OF SMOKING
Cigarette smoking substantially increases the risk of (a) cardiovascular diseases such as stroke, sudden death, and heart attack; (b) nonmalignant respiratory diseases including emphysema, asthma, chronic bronchitis, and chronic obstructive pulmonary disease; (c) lung cancer; and (d) other cancers.33
Exposure to environmental tobacco smoke (passive exposure) has been cited as the cause of 3,400 lung cancer deaths and 46,000 heart disease–related deaths in the United States every year.36 Children who are exposed to environmental smoke have a higher risk of respiratory infection, asthma, and middle ear infections than those who are not exposed. Sudden infant death syndrome occurs more often in infants whose mothers smoked during pregnancy than in offspring of nonsmoking mothers.36 The harmful effects of smoking on reproduction and pregnancy include reduced fertility and fetal growth, as well as increased risk of ectopic pregnancy and spontaneous abortion.36
PHARMACOLOGY OF NICOTINE
Nicotine is a ganglionic cholinergic agonist with pharmacologic effects that are highly dependent on dose. These effects include central and peripheral nervous system stimulation and depression, respiratory stimulation, skeletal muscle relaxation, catecholamine release by the adrenal medulla, peripheral vasoconstriction, and increased blood pressure, heart rate, cardiac output, and oxygen consumption. Cigarette smoking or low doses of nicotine produce an increased alertness and increased cognitive functioning by stimulating the cerebral cortex. At higher doses, nicotine stimulates the “reward” center in the limbic system of the brain.37
When nicotine is ingested, a feeling of pleasure and relaxation can occur. Repetitive exposure to nicotine leads to neuroadaptation, which builds tolerance to the initial effects. An accumulation of nicotine in the body leads to a more substantial withdrawal reaction if cessation is attempted.38 Common symptoms experienced during withdrawal can include anxiety, difficulties concentrating, irritability, and strong cravings for tobacco. Onset of these withdrawal symptoms usually occurs within 24 hours and can last for days, weeks, or longer.38 This powerful force of nicotine addiction is one reason smokers who attempt to achieve smoking cessation have a high rate of relapse, and only 3% remain abstinent 6 months following the quit date.39
TREATMENT
Desired Outcomes
Ideally, we would hope that more and more people stop smoking altogether and that young people never take up the habit. This is unlikely to happen. The Healthy People 2020 target setting the prevalence of smoking to be less than or equal to 12% discussed above is a realistic and achievable goal.
Nicotine Dependence
Agency for Healthcare Research and Quality Clinical Practice Guideline: Treating Tobacco Use and Dependence
The Agency for Healthcare Research and Quality (AHRQ) periodically convenes expert panels to develop clinical guidelines for healthcare practitioners. Because of the widespread prevalence of smoking-related illnesses, its related morbidity and mortality, and the economic burden imposed, the agency convened a panel of experts in 1994 to develop guidelines on the treatment of tobacco addiction. The resultant guideline for smoking cessation was updated in 2008,29 and no further updates have been released at the time of this writing.



